Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (29): 4724-4730.doi: 10.3969/j.issn.2095-4344.2714
Previous Articles Next Articles
Wnt/beta-catenin signaling pathway in the treatment of osteoarthritis: new advances and application prospects
Bei Tao, Liu Junting, Huang Qiulin, Su Wei, Zhao Jinmin
Department of Traumatic Orthopaedics and Hand Surgery, the First Affiliated Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China
-
Received:2019-11-08Revised:2019-11-14Accepted:2020-01-02Online:2020-10-18Published:2020-09-15 -
Contact:Zhao Jinmin, Chief physician, Professor, Doctoral supervisor, Department of Traumatic Orthopaedics and Hand Surgery, the First Affiliated Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
About author:Bei Tao, MD candidate, Attending physician, Department of Traumatic Orthopaedics and Hand Surgery , the First Affiliated Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
Supported by:Guangxi Key Research and Development plan, No. guikeAD17129012; Guangxi University Student Innovation and Entrepreneurship Training Program, No. 201910598010
CLC Number:
Cite this article
Bei Tao, Liu Junting, Huang Qiulin, Su Wei, Zhao Jinmin. Wnt/beta-catenin signaling pathway in the treatment of osteoarthritis: new advances and application prospects[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(29): 4724-4730.
share this article
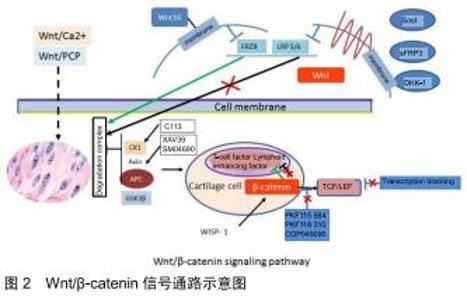
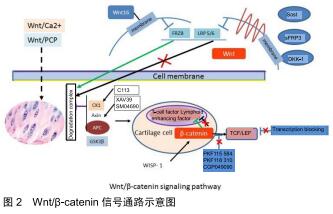
2.1 Wnt/β-catenin信号通路组成及调节 Wnt家族属于一个既高度保守,又相对复杂的信号传导通路,在细胞的分化、胚胎期的骨骼形成、纤维化和关节代谢平衡中,起着重要的作用[22]。Wnt蛋白可激活不同的信号通路,包括经典的Wnt/β-catenin信号通路及非经典的Wnt通路,后者包括Wnt/Ca2+及Wnt/PCP通路。当没有Wnt信号时,β-catenin在胞浆中保持在一个稳定的水平,多余的β-catenin则被降解复合体(蛋白激酶1、轴心蛋白、大肠腺瘤样蛋白及糖原合成酶激酶3β组成)中的糖原合成酶激酶3β磷酸化,然后再通过泛素化及蛋白酶体路径降解。当有Wnt信号时,与其受体结合(主要是卷曲蛋白,Frzb,及低蛋白脂蛋白相关受体蛋白5或6,导致复合体降解和破坏。β-catenin主要在胞浆内聚积,多余的β-catenin进入细胞核内,与T细胞因子/淋巴增强因子结合,进而激活Wnt/β-catenin的转录因子,进而促进相关靶基因的表达,比如c-Myc、细胞周期蛋白D1, 基质金属蛋白酶3和CD44[23]。见图2。 "
|
[1] LOESER RF, GOLDRING SR, SCANZELLO CR, et al. Osteoarthritis: A disease of the joint as an organ. Arthritis & Rheumatism. 2012;64(6):1697-1707.
[2] ZHOU Y, WANG T, HAMILTON JL, et al. Wnt/β-catenin Signaling in Osteoarthritis and in Other Forms of Arthritis. Current Rheumatology Reports. 2017;19(9).
[3] 王超华,朱梅. Wnt/β-catenin信号通路与骨关节炎[J].中华骨质疏松和骨矿盐疾病杂志,2011,4(2):122-126.
[4] WU Q, ZHU M, ROSIER RN, et al. β-catenin, cartilage, and osteoarthritis. Annals of the New York Academy of Sciences. 2010;1192(1):344-350.
[5] 中华医学会骨科学分会关节外科学组.骨关节炎诊疗指南(2018年版)[J].中华骨科杂志,2018,38(12):705-715.
[6] 王华敏,宓轶群,刚嘉鸿.信号通路在膝骨关节炎实验研究中的进展[J].中国组织工程研究,2016,20(2):267-272.
[7] YUASA T, OTANI T, KOIKE T, et al. Wnt/beta-catenin signaling stimulates matrix catabolic genes and activity in articular chondrocytes: its possible role in joint degeneration. Lab Invest.2008;88(3):264-274.
[8] VAN DEN BOSCH MH, BLOM AB, SLOETJES AW, et al. Induction of Canonical Wnt Signaling by Synovial Overexpression of Selected Wnts Leads to Protease Activity and Early Osteoarthritis-Like Cartilage Damage. Am J Pathol. 2015;185(7):1970-1980.
[9] GUO X, DAY TF, JIANG X, et al. Wnt/beta-catenin signaling is sufficient and necessary for synovial joint formation. Genes Dev. 2004;18(19):2404-2417.
[10] SPATER D, HILL TP, GRUBER M, et al. Role of canonical Wnt-signalling in joint formation. Eur Cell Mater. 2006;12: 71-80.
[11] MONTEAGUDO S, LORIES RJ. Cushioning the cartilage: a canonical Wnt restricting matter. Nature Reviews Rheumatology. 2017;13(11):670-681.
[12] JI Q, XU X, KANG L, et al. Hematopoietic PBX-interacting protein mediates cartilage degeneration during the pathogenesis of osteoarthritis. Nat Commun. 2019;10(1):313.
[13] USAMI Y, GUNAWARDENA AT, IWAMOTO M, et al. Wnt signaling in cartilage development and diseases: lessons from animal studies. Lab Invest. 2016;96(2):186-196.
[14] JOHNSON ML, KAMEL MA. The Wnt signaling pathway and bone metabolism. Curr Opin Rheumatol. 2007;19(4):376-382.
[15] WANG Y, FAN X, XING L, et al. Wnt signaling: a promising target for osteoarthritis therapy. Cell Commun Signal. 2019; 17(1):97.
[16] LORIES RJ, CORR M, LANE NE. To Wnt or not to Wnt: the bone and joint health dilemma. Nat Rev Rheumatol. 2013; 9(6):328-39.
[17] BOUAZIZ W, FUNCK-BRENTANO T, LIN H, et al. Loss of sclerostin promotes osteoarthritis in mice via beta-catenin- dependent and -independent Wnt pathways. Arthritis Res Ther. 2015;17:24.
[18] THYSEN S, LUYTEN FP, LORIES RJ. Loss of Frzb and Sfrp1 differentially affects joint homeostasis in instability-induced osteoarthritis. Osteoarthritis Cartilage. 2015;23(2):275-279.
[19] DAY TF, GUO X, GARRETT-BEAL L, et al. Wnt/beta-catenin signaling in mesenchymal progenitors controls osteoblast and chondrocyte differentiation during vertebrate skeletogenesis. Dev Cell. 2005;8(5):739-750.
[20] CORR M. Wnt-β-catenin signaling in the pathogenesis of osteoarthritis. Nature Clinical Practice Rheumatology. 2008; 4(10):550-556.
[21] CLEVERS H, NUSSE R. Wnt/beta-catenin signaling and disease. Cell.2012;149(6):1192-1205.
[22] YANO F, KUGIMIYA F, OHBA S, et al. The canonical Wnt signaling pathway promotes chondrocyte differentiation in a Sox9-dependent manner. Biochem Biophys Res Commun. 2005;333(4):1300-1308.
[23] NG LF, KAUR P, BUNNAG N, et al. WNT Signaling in Disease. Cells. 2019;8(8). pii: E826.
[24] CADIGAN KM, NUSSE R. Wnt signaling: a common theme in animal development. Genes Dev.1997;11(24): 3286-3305.
[25] BLOM AB, BROCKBANK SM, VAN LENT PL, et al. Involvement of the Wnt signaling pathway in experimental and human osteoarthritis: prominent role of Wnt-induced signaling protein 1. Arthritis Rheum. 2009;60(2):501-512.
[26] RYU JH. Regulation of the chondrocyte phenotype by beta-catenin. Development. 2002;129(23):5541-5550.
[27] WANG MN, SAMPSON ER, JIN HT, et al. MMP13 is a critical target gene during the progression of osteoarthritis. Arthritis Research & Therapy. 2013;15(1).
[28] ZHU M, TANG D, WU Q, et al. Activation of β-Catenin Signaling in Articular Chondrocytes Leads to Osteoarthritis- Like Phenotype in Adult β-Catenin Conditional Activation Mice. Journal of Bone and Mineral Research. 2009;24(1):12-21.
[29] ZHU M, CHEN M, ZUSCIK M, et al. Inhibition of beta-catenin signaling in articular chondrocytes results in articular cartilage destruction. Arthritis Rheum. 2008;58(7):2053-264.
[30] 于彩云,刘重慧,魏涛.靶向Wnt/β-catenin信号通路抑制剂研究进展[J]. 河南师范大学学报(自然科学版), 2016;44(2):125-130.
[31] ONUORA S. Wnt inhibitor shows potential as a DMOAD. Nature Reviews Rheumatology. 2017;13(11):634.
[32] HUANG Y, JIANG L, YANG H, et al. Variations of Wnt/beta-catenin pathway-related genes in susceptibility to knee osteoarthritis: A three-centre case-control study. J Cell Mol Med. 2019;23(12):8246-8257.
[33] TONG W, ZENG Y, CHOW DHK, et al. Wnt16 attenuates osteoarthritis progression through a PCP/JNK-mTORC1- PTHrP cascade. Ann Rheum Dis. 2019;78(4):551-561.
[34] CHAN BY, FULLER ES, RUSSELL AK, et al. Increased chondrocyte sclerostin may protect against cartilage degradation in osteoarthritis. Osteoarthritis Cartilage. 2011;19(7):874-885.
[35] WU J, MA L, WU L, et al. Wnt-β-catenin signaling pathway inhibition by sclerostin may protect against degradation in healthy but not osteoarthritic cartilage. Molecular Medicine Reports. 2017;15(5):2423-2432.
[36] BOUAZIZ W, FUNCK-BRENTANO T, LIN H, et al. Loss of sclerostin promotes osteoarthritis in mice via β-catenin- dependent and -independent Wnt pathways. Arthritis Res Ther. 2015;17(1):24.
[37] CHANG JC, CHRISTIANSEN BA, MURUGESH DK, et al. SOST/Sclerostin Improves Posttraumatic Osteoarthritis and Inhibits MMP2/3 Expression After Injury. J Bone Miner Res. 2018;33(6):1105-1113.
[38] DESHMUKH V, O'GREEN AL, BOSSARD C, et al. Modulation of the Wnt pathway through inhibition of CLK2 and DYRK1A by lorecivivint as a novel, potentially disease-modifying approach for knee osteoarthritis treatment. Osteoarthritis and Cartilage. 2019;27(9):1347-1360.
[39] DESHMUKH V, HU H, BARROGA C, et al. A small-molecule inhibitor of the Wnt pathway (SM04690) as a potential disease modifying agent for the treatment of osteoarthritis of the knee. Osteoarthritis and Cartilage. 2018;26(1):18-27.
[40] YAZICI Y, MCALINDON TE, FLEISCHMANN R, et al. A novel Wnt pathway inhibitor, SM04690, for the treatment of moderate to severe osteoarthritis of the knee: results of a 24-week, randomized, controlled, phase 1 study. Osteoarthritis Cartilage. 2017;25(10):1598-1606.
[41] JONES SE, JOMARY C. Secreted Frizzled-related proteins: searching for relationships and patterns. Bioessays. 2002; 24(9):811-820.
[42] LORIES RJU, PEETERS J, BAKKER A, et al. Articular cartilage and biomechanical properties of the long bones inFrzb-knockout mice. Arthritis Rheum. 2007;56(12): 4095-4103.
[43] LIETMAN C, WU B, LECHNER S, et al. Inhibition of Wnt/β-catenin signaling ameliorates osteoarthritis in a murine model of experimental osteoarthritis. JCI Insight. 2018;3(3). pii: 96308.
[44] MIN S, WANG C, LU W, et al. Serum levels of the bone turnover markers dickkopf-1, osteoprotegerin, and TNF-alpha in knee osteoarthritis patients. Clin Rheumatol. 2017;36(10): 2351-2358.
[45] HONSAWEK S, TANAVALEE A, YUKTANANDANA P, et al. Dickkopf-1 (Dkk-1) in plasma and synovial fluid is inversely correlated with radiographic severity of knee osteoarthritis patients. BMC Musculoskelet Disord. 2010;11:257.
[46] OH H, CHUN CH, CHUN JS. Dkk-1 expression in chondrocytes inhibits experimental osteoarthritic cartilage destruction in mice. Arthritis Rheum. 2012;64(8):2568-2578.
[47] FUNCK-BRENTANO T, BOUAZIZ W, MARTY C, et al. Dkk-1-mediated inhibition of Wnt signaling in bone ameliorates osteoarthritis in mice. Arthritis Rheumatol. 2014; 66(11):3028-3039.
[48] 王晓凯,张志成,孙天胜. SIRT1的生理作用及调控机制的研究进展[J].中华临床医师杂志(电子版),2011,5(24):7315-7318.
[49] LI Y, XIAO W, WU P, et al. The expression of SIRT1 in articular cartilage of patients with knee osteoarthritis and its correlation with disease severity. J Orthop Surg Res. 2016; 11(1):144.
[50] HE DS, HU XJ, YAN YQ, et al. Underlying mechanism of Sirt1 on apoptosis and extracellular matrix degradation of osteoarthritis chondrocytes. Mol Med Rep. 2017;16(1): 845-850.
[51] 钟翔,彭婷,涂月菊,等. WISP-1在IgA肾病患者中的表达与肾间质纤维化的关系[J]. 中华肾脏病杂志,2017,33(9):649-655.
[52] VAN DEN BOSCH MHJ, RAMOS YFM, den Hollander W,et al. Increased WISP1 expression in human osteoarthritic articular cartilage is epigenetically regulated and decreases cartilage matrix production. Rheumatology (Oxford). 2019;58(6): 1065-1074.
[53] LANDMAN EB, MICLEA RL, VAN BLITTERSWIJK CA, et al. Small molecule inhibitors of WNT/beta-catenin signaling block IL-1beta- and TNFalpha-induced cartilage degradation. Arthritis Res Ther.2013;15(4):R93.
[54] 鄢德洪,徐美晨,王婧惠,等.青蒿素类药物靶标及其免疫调节抗肿瘤机制研究进展[J].免疫学杂志,2018,34(8):713-21.
[55] ZHONG G, LIANG R, YAO J, et al. Artemisinin Ameliorates Osteoarthritis by Inhibiting the Wnt/β-Catenin Signaling Pathway. Cellular Physiology and Biochemistry. 2018;51(6): 2575-2590.
[56] WENG X, LIN P, LIU F, et al.Achyranthes bidentata polysaccharides activate the Wnt/β-catenin signaling pathway to promote chondrocyte proliferation.Int J Mol Med. 2014; 34(4):1045- 1050.
[57] 马勇,王礼宁,郭杨,等.姜黄素通过调节Wnt/β-catenin信号通路促进软骨细胞增殖的研究[J].广州中医药大学学报,2017,34(1): 90-95.
[58] 陈琼,赵明才,陈悦,等. 姜黄素对骨关节炎软骨细胞增殖及分泌MMP-13,IL-6的影响[J]. 现代中西医结合杂志,2013,22(5): 459-61.
[59] 李洪涛,刘昊,杨方军,等. 针对膝关节骨性关节炎大鼠关节软骨中Wnt3a、β-catenin蛋白表达的影响[J].针灸临床杂志.2017, 33(3):65-68.
[60] 张媛媛,李西海,吴明霞.电针调节Wnt/β-catenin信号通路抑制大鼠膝骨关节炎软骨退变的研究[J].中国针灸,2019,39(10): 1081-1086.
[61] 谢晚晴,郑洪新.基于Wnt/β-catenin信号通路的中医药干预骨关节炎的研究进展[J].中国骨质疏松杂志,2018;24(5):664-670.
[62] 崔建梅,尹大力.药物重新定位策略在新药发现中的应用与进展[J].中国药学杂志,2005,40(20):8-10.
[63] TAKAMATSU A, OHKAWARA B, ITO M, et al. Verapamil protects against cartilage degradation in osteoarthritis by inhibiting Wnt/beta-catenin signaling. PLoS One. 2014;9(3): e92699.
[64] MA L, LIU Y, ZHAO X, et al. Rapamycin attenuates articular cartilage degeneration by inhibiting β-catenin in a murine model of osteoarthritis. Connect Tissue Res.2019;60(5): 452-462.
[65] MIYAMOTO K, OHKAWARA B, ITO M, et al. Fluoxetine ameliorates cartilage degradation in osteoarthritis by inhibiting Wnt/beta-catenin signaling. PLoS One. 2017; 12(9):e0184388.
[66] NAITO M, OHASHI A, TAKAHASHI T. Dexamethasone inhibits chondrocyte differentiation by suppression of Wnt/beta-catenin signaling in the chondrogenic cell line ATDC5. Histochem Cell Biol. 2015;144(3):261-272.
[67] HUANG K, WU LD. Dehydroepiandrosterone: Molecular mechanisms and therapeutic implications in osteoarthritis. J Steroid Biochem Mol Biol. 2018;183:27-38.
[68] BAI M, GE L, CHEN H, et al. Calcitonin protects rat chondrocytes from IL‑1β injury via the Wnt/β‑catenin pathway. Exp Ther Med. 2019;18(3):2079-2085.
[69] MA L, WU J, JIN QH. The association between parathyroid hormone 134 and the Wnt/betacatenin signaling pathway in a rat model of osteoarthritis. Mol Med Rep. 2017;16(6): 8799-8807.
[70] ZHANG Y, HUANG X, YUAN Y. MicroRNA-410 promotes chondrogenic differentiation of human bone marrow mesenchymal stem cells through down-regulating Wnt3a. Am J Transl Res. 2017;9(1):136-145.
[71] BOUAZIZ W, SIGAUX J, MODROWSKI D, et al. Interaction of HIF1alpha and beta-catenin inhibits matrix metalloproteinase 13 expression and prevents cartilage damage in mice. Proc Natl Acad Sci U S A. 2016;113(19):5453-5458.
[72] SUN Y, WANG F, SUN X, et al. CX3CR1 regulates osteoarthrosis chondrocyte proliferation and apoptosis via Wnt/beta-catenin signaling. Biomed Pharmacother. 2017;96: 1317-1323.
[73] ZHONG L, SCHIVO S, HUANG X, et al. Nitric Oxide Mediates Crosstalk between Interleukin 1beta and WNT Signaling in Primary Human Chondrocytes by Reducing DKK1 and FRZB Expression. Int J Mol Sci. 2017;18(11). pii: E2491.
[74] CHEN YY, CHEN Y, WANG WC, et al. Cyclin D1 regulates osteoarthritis chondrocyte apoptosis via WNT3/beta-catenin signalling. Artif Cells Nanomed Biotechnol. 2019;47(1): 1971-1977. |
| [1] | Peng Zhihao, Feng Zongquan, Zou Yonggen, Niu Guoqing, Wu Feng. Relationship of lower limb force line and the progression of lateral compartment arthritis after unicompartmental knee arthroplasty with mobile bearing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1368-1374. |
| [2] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Comparison of the short-term efficacy of extracorporeal shock wave therapy for middle-aged and elderly knee osteoarthritis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1471-1476. |
| [3] | Wu Xun, Meng Juanhong, Zhang Jianyun, Wang Liang. Concentrated growth factors in the repair of a full-thickness condylar cartilage defect in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1166-1171. |
| [4] | Li Jiacheng, Liang Xuezhen, Liu Jinbao, Xu Bo, Li Gang. Differential mRNA expression profile and competitive endogenous RNA regulatory network in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1212-1217. |
| [5] | Geng Qiudong, Ge Haiya, Wang Heming, Li Nan. Role and mechanism of Guilu Erxianjiao in treatment of osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1229-1236. |
| [6] | Liu Xiangxiang, Huang Yunmei, Chen Wenlie, Lin Ruhui, Lu Xiaodong, Li Zuanfang, Xu Yaye, Huang Meiya, Li Xihai. Ultrastructural changes of the white zone cells of the meniscus in a rat model of early osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1237-1242. |
| [7] | Li Shibin, Lai Yu, Zhou Yi, Liao Jianzhao, Zhang Xiaoyun, Zhang Xuan. Pathogenesis of hormonal osteonecrosis of the femoral head and the target effect of related signaling pathways [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 935-941. |
| [8] | He Xiangzhong, Chen Haiyun, Liu Jun, Lü Yang, Pan Jianke, Yang Wenbin, He Jingwen, Huang Junhan. Platelet-rich plasma combined with microfracture versus microfracture in the treatment of knee cartilage lesions: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 964-969. |
| [9] | Liu Xin, Yan Feihua, Hong Kunhao. Delaying cartilage degeneration by regulating the expression of aquaporins in rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 668-673. |
| [10] | Ma Zetao, Zeng Hui, Wang Deli, Weng Jian, Feng Song. MicroRNA-138-5p regulates chondrocyte proliferation and autophagy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 674-678. |
| [11] | Cao Xuhan, Bai Zixing, Sun Chengyi, Yang Yanjun, Sun Weidong. Mechanism of “Ruxiang-Moyao” herbal pair in the treatment of knee osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 746-753. |
| [12] | Li Yonghua, Feng Qiang, Tan Renting, Huang Shifu, Qiu Jinlong, Yin Heng. Molecular mechanism of Eucommia ulmoides active ingredients treating synovitis of knee osteoarthritis: an analysis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 765-771. |
| [13] | Song Shan, Hu Fangyuan, Qiao Jun, Wang Jia, Zhang Shengxiao, Li Xiaofeng. An insight into biomarkers of osteoarthritis synovium based on bioinformatics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 785-790. |
| [14] | Deng Zhenhan, Huang Yong, Xiao Lulu, Chen Yulin, Zhu Weimin, Lu Wei, Wang Daping. Role and application of bone morphogenetic proteins in articular cartilage regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 798-806. |
| [15] | Zheng Li, Li Dadi, Hu Weifan, Tang Jinlong, Zhao Fengchao. Risk assessment of contralateral knee arthroplasty after unilateral total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 374-379. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||