Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (16): 2606-2613.doi: 10.3969/j.issn.2095-4344.2264
Previous Articles Next Articles
Research and application of antibacterial modification of hydroxyapatite
Luo Jin1, 2, Yan Yajing2, Tan Zhe1, 2, Zhang Han1, 2, Lan Hai2
- 1Zunyi Medical University, Zunyi 563000, Guizhou Province, China; 2Affiliated Hospital of Chengdu University, Chengdu 610000, Sichuan Province, China
-
Received:2019-09-06Revised:2019-09-07Accepted:2019-10-15Online:2020-06-08Published:2020-03-28 -
Contact:Lan Hai, Chief physician, MD, Master’s supervisor, Affiliated Hospital of Chengdu University, Chengdu 610000, Sichuan Province, China -
About author:Luo Jin, Master candidate, Physician, Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
Supported by:the Project of Science & Technology Department of Sichuan Province, No. 2018JY0354; the Project of Science & Technology Bureau of Chengdu, No. 2015-HM01-00511-SF
CLC Number:
Cite this article
Luo Jin, Yan Yajing, Tan Zhe, Zhang Han, Lan Hai. Research and application of antibacterial modification of hydroxyapatite [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(16): 2606-2613.
share this article
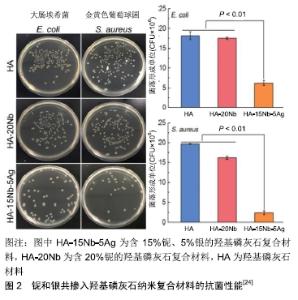
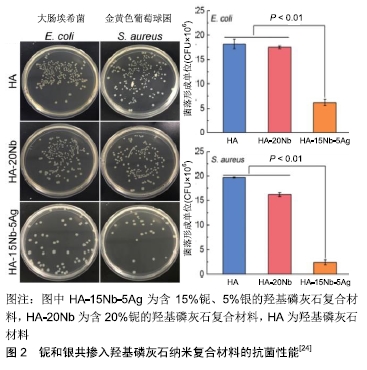
2.1 银掺入羟基磷灰石 金属银很早就被发现具有优良的抗菌性能,中国古代有人发现用银制的器皿盛装牛奶可放得更久而不变质。早在公元1000年前银被用来制造饮用水[17],但银在金属状态下的抗菌性能并不活跃,银系抗菌物质中以银纳米粒子的应用最为广泛和突出。银纳米粒子具有单质银及银离子所不具有的很多优良抗菌特性及安全性,被广泛应用于纺织品、创面敷料和生物医学材料[18]。合成银纳米粒子的方法很多,包括化学、电化学、辐射、光化学方法和生物法等[18]。在银纳米粒子的几种合成方法中,生物方法是简单、快速、无毒、可靠和绿色的方法,可以在优化制备条件下产生明确的尺寸和形态。银及其衍生物对细菌、真菌和病毒具有较强的抑制和杀灭作用,并具有广谱的抗菌活性。银的抗菌性能与银离子的释放量有关,虽然银离子浓度越大抗菌效果越好,但随着离子浓度的增加其毒副作用增加,过量的银离子在人体内可能造成肝、肾、肺、心脏、肠道的损害[19],因此寻找最适宜的抗菌银离子浓度是值得探究的问题。研究表明较低的银离子浓度就能具有抗菌效果,并且不会对机体产生严重的毒副作用[20],但银离子浓度低于0.15%便丧失了抗菌活性,只有在银离子浓度大于0.2%时才能具有抗菌性能。 银系的抗菌机制主要有5种[21-22]:银离子可以进入细菌细胞内与多种重要酶的硫醇基团相互作用,使其失活;银纳米粒子与细菌相互作用时会产生自由基,这些自由基具有改变细胞渗透压并破坏细胞组织的能力,最终导致细菌细胞死亡;银离子还可以通过产生的活性氧对呼吸酶起到抑制作用,从而破坏细菌细胞本身;银离子还可以在进入细菌之后与细菌内的DNA接触,抑制细菌DNA的繁殖,从而导致细菌死亡;银纳米粒子可以改变细菌细胞的底物磷酸化,影响其信号转导,从而抑制细菌繁殖。 纳米银粒子或者银盐可通过多种方法掺入到羟基磷灰石中制备复合物,最常见的有等离子喷涂法、电化学方法、磁控溅射法、溶胶凝胶法等,不同方法制备的复合涂层物理、化学、生物特性都有所差异。银离子还可以与其他具有生物活性或者物理特性的金属与非金属物质共同掺杂进入羟基磷灰石,从而制备出具有抗菌、促进骨细胞生长、促进血管生成、力学性质优异的综合性复合材料。研究表明银和氟用溶胶凝胶法共同掺入羟基磷灰石可制备复合涂层[23],氟掺杂后的复合材料增加了对大肠杆菌的抗菌活性。有研究用高能球磨和火花等离子烧结制备了新型铌和银共掺入羟基磷灰石的纳米复合材料,添加铌、银可显著提高羟基磷灰石的抗压强度和断裂韧性;体外和体内评价进一步表明,铌的加入可以促进成骨细胞增殖、增加成骨分化,增强骨整合能力,并且复合材料具有良好的革兰阳性菌金黄色葡萄球菌和革兰阴性菌大肠杆菌的抗菌活性,见图2[24]。因此,所开发出的新型纳米复合材料可以满足负重骨种植体的机械性能及生物功能需求。还有研究表明银、锶共同掺入羟基磷灰石制备的复合材料,对大肠杆菌(E.coli)和金黄色葡萄球菌(S.aureus)具有良好的抗菌性能,并且由于锶的掺入降低了银的细胞毒性[25]。 "

2.2 铜掺入羟基磷灰石 铜是人体健康不可缺少的微量元素,对促进血管生成具有重要作用,还有助于骨形成和提高抗菌活性[26]。无论是单独的铜离子还是铜配合物中的铜离子,几百年前就被用来对液体、固体和人体组织进行消毒。铜和铜化合物对细菌、真菌和病毒都具有抵抗作用,铜离子的使用减少了医源性病原体、食源性疾病和尘螨的传播及真菌足部感染[27]。目前已经有很多研究报道铜被用于新型医用金属合金材料的制备,以防止感染的发生[28-30]。铜的抗菌性能与其形态结构有一定的关系,在纳米级材料中体积越小抗菌效果越好[31]。纳米级的铜比微米级的铜具有更好的抗菌性能[32],原因可能是纳米粒子表面积与体积的比值更大,铜原子表面与细菌的接触面积更大,同时小颗粒的铜具有更强的穿透细胞能力。铜纳米粒子的制备方法主要分为3大类,即物理、化学、生物法,其中激光烧蚀、微波辅助法、溶胶-凝胶法、共沉淀法、脉冲放电法、真空气相沉积法、高能辐照法、光刻法、机械铣削法、光化学还原法、电化学、电喷雾合成法、水热反应法、微乳液法、化学还原法等多种物理化学方法被广泛应用于纳米粒子的合成[33]。 铜具有广谱的抗菌性能,特别是对抗生素耐药菌依然具有良好的抗菌能力,但铜的抗菌机制还不是很明确。目前铜的可能抗菌机制主要有4个方面[31-32,34]:铜通过直接与细菌细胞膜接触阻断生化代谢,破坏细胞膜,杀死细菌,被称为接触杀灭;铜离子进入细菌体内与DNA相互作用,抑制细菌NDA的增殖;产生活性氧,使得细胞结构出现氧化损伤,从而抑制杀灭细菌;铜离子本身带正电荷,在进入细菌体内后与细菌体内负电荷相互作用,从而杀灭细菌。 铜纳米粒子具有高的表面体积比、延展性、硬度、柔韧性、刚性等优异的性能,因此被广泛用于生物医用材料的合成。铜纳米粒子及其氧化物可以通过多种制备方法掺入羟基磷灰石中。有研究采用连续两次电化学反应合成了抗菌铜-羟基磷灰石复合材料[35],第一阶段采用阴极电解合成法在钛上生长羟基磷灰石纳米晶;在第二反应阶段以羟基磷灰石包覆的钛为阴极,电化学还原金属铜纳米粒子溶液中的Cu2+。通过培养大肠杆菌(革兰阴性菌)和金黄色葡萄球菌(革兰阳性菌)在不同铜含量的涂层存在下测定抗菌活性,发现涂层对两种细菌均表现出铜浓度依赖性的抗菌活性。有研究采用离子交换法和气压印刷法制备了不同铜含量的羟基磷灰石微球,结果证明该复合材料具有良好的降解性能、抗菌性能和细胞存活率,可作为一种良好的骨缺损修复材料[26]。 2.3 金掺入羟基磷灰石 金是一种贵金属。在整个文明史上,黄金以各种形式被用于医学。金及其化合物已被用于治疗风湿性疾病和盘状红斑狼疮及各种炎症性皮肤疾病,如天疱疮、荨麻疹和牛皮癣[36]。金纳米材料具有不同于块状材料的性能,因此金系抗菌材料以纳米粒子的研究最为多见。近年来通过各种物理、化学和生物方法,人们利用金纳米粒子开发了一系列广泛应用于生物技术、工业、电气、制药、医学和农业领域的新材料。 金的抗菌机制依然不是很明确,可能主要有3种情况[37-39]:①金纳米粒子带正电荷,细菌细胞膜总体带负电荷,纳米粒子吸附在细菌表面破坏细胞膜的脂质体并触发其内容物的泄漏。由于纳米粒子与细胞膜之间的相互作用,破坏了细胞膜。②金离子进入细菌细胞内降低ATP水平,细菌代谢过程失调,从而抑制细菌生长繁殖。③金纳米粒子进入细菌体内后致使核糖体亚基与tRNA的结合受到抑制,导致转录机制被破坏。金纳米粒子抵抗革兰阴性细菌主要通过2种方式发挥抗菌作用[40]:破坏膜电位,抑制ATP酶活性,降低ATP水平;抑制核糖体亚基与tRNA结合。此外,巯基嘧啶偶联金纳米团簇通过细胞膜破坏、DNA损伤和活性氧生成等联合机制对耐甲氧西林金黄色葡萄球菌表现出良好的抗菌效果[41]。此外,在细菌细胞中诱导细胞内活性氧的产生主要归因于金纳米粒子的内在氧化酶和过氧化物类催化活性。 由于金良好的抗菌性能,金纳米粒子及金盐被研究者掺入羟基磷灰石以制备具有优异性能的抗菌材料。有研究采用仿生法合成了金掺入羟基磷灰石复合材料,并证明了它的良好抗菌性能,指出金纳米颗粒可能是修复骨感染(骨髓炎)的优良材料,并可作为预防术后感染的潜在支架材料[42]。还有研究采用共沉淀法合成了多孔纳米羟基磷灰石球团,采用真空过滤吸附法将金等纳米粒子有效浸渍到羟基磷灰石颗粒中,所有载纳米粒子羟基磷灰石对大肠杆菌和金黄色葡萄球菌均表现出较强的抗菌活性[43]。 2.4 镁掺入羟基磷灰石 镁是人体内的必需元素之一,也是人体含量十分丰富的元素,约65%的人体镁元素存在于骨骼和牙齿中[44]。体外实验表明镁在维持血管功能中起着重要作用[45-46]。镁诱导内皮细胞产生一氧化氮,这与血管内皮生长因子诱导血管生成的机制基本相同[45]。研究人员还发现通过向钙磷复合材料中添加镁,可以改善成骨细胞的附着、增殖和碱性磷酸酶生成,并且使材料变得更致密[47]。有研究表明,在股骨缺损中植入掺入镁的羟基磷灰石显示出更强的成骨支撑作用及组织相容性[48]。镁已被临床用于制备磷酸镁骨水泥和几种不同的生物玻璃成分。同时,镁还具有抵抗细菌的性能。有研究利用微波辐射法制备了纯羟基磷灰石和镁取代羟基磷灰石[49]。镁的掺入影响了羟基磷灰石纳米粒子的结晶度和形貌,由于镁的合并晶体变得更小、更不规则、更密集。此外,还展示了掺镁羟基磷灰石在医学领域的可能应用,比如它们对人类致病菌具有显著的抗菌活性。还有研究制备了氧化镁颗粒掺杂羟基磷灰石复合物,这种复合物减少了细菌的生长和生物膜的形成,当氧化镁含量在≥3%时可以完全消灭金黄色葡萄球菌,具有较好的抗菌活性和抑制生物膜形成的作用[50]。 镁及其氧化物的抗菌机制依然不是很明确,主要有以下几个特点:①氧化镁纳米颗粒可破坏细菌包膜。可能是由于氧化镁与细菌包膜表面磷酸基结合从而附着在细菌表面,导致细菌包膜被破坏,导致细胞膜渗漏,诱导氧化应激,最终导致细菌死亡。但也可能存在其他的附着机制促进抗菌活性[51-52]。②镁离子的抗菌效果与pH值有很大关系,镁离子的抗菌效果随着pH值的增加而增加[53]。③金属镁可以抑制生物膜的形成,原因可能与镁的降解引起周围碱度的增加有关[54]。 2.5 锶掺入羟基磷灰石 锶是一种非必需元素,占骨骼系统中钙含量的0.035%。锶已被证明在合成骨移植物中具有促进骨再生的作用[55]。本质上由于锶的大小和电荷与钙离子非常相似,它在成骨细胞介导的过程中取代了钙离子。在羟基磷灰石中引入锶离子可以提高复合物的生物相容性和生物活性,简化骨整合过程。此外,锶离子还能改善羟基磷灰石的力学性能。 最近的报道表明锶可以用作抗菌剂。有研究用微波加速的方法将锶掺入羟基磷灰石,使得复合物具有抗金黄色葡萄球菌性能[56]。还有研究认为锶的抗菌性能很差,但与氟化物结合后可提高治疗龋齿方面的抗菌潜力[57]。有文献采用溶胶-凝胶超临界流体干燥法合成锶-羟基磷灰石,证明了该物质含有锶离子,对大肠杆菌、金黄色葡萄球菌和乳酸菌均有一定的抗菌作用[58]。 2.6 锌掺入羟基磷灰石 锌是人类发育过程中重要的必需微量元素。人体内有几种重要的金属酶利用锌进行催化或反应。锌可以创造一个碱性环境,有利于无机磷酸盐沉淀并随后矿化至成骨细胞产生的细胞外基质上。饮食中锌的缺乏与多种骨骼发育缺陷有关,锌可能在预防骨质疏松症中发挥作用。在自然骨重塑过程中锌从骨骼中释放出来[59],微环境中由于这种释放而产生的过量锌,被认为可以阻止破骨细胞的吸收过程并刺激成骨细胞的骨形成过程[60],掺杂锌的磷酸钙材料在体外表现出成骨反应增强,同时在体内也可以促进新的骨形成[61]。磷酸锌骨水泥是牙科工业中最古老、应用最广泛的骨水泥之一。 锌除了具有促进成骨性能外还可以用作抗菌剂。有研究采用化学共沉淀法合成了锌取代羟基磷灰石,随着复合材料中锌离子浓度的增加,复合材料对金黄色葡萄球菌和大肠杆菌具有良好的生物活性和抗菌活性[62]。有研究采用溶胶-凝胶法制备了锌掺杂羟基磷灰石复合涂层,抗菌实验表明复合物对金黄色葡萄球菌的抗菌性能高于大肠埃希菌,并且指出抗菌性能与锌的浓度有很大关系[63]。 2.7 钛掺入羟基磷灰石 钛是一种医学中应用非常广泛的金属,具有良好的物理、化学性质及组织相容性,被广泛用于骨科及牙科材料的制备。不仅如此,钛还具有抗菌活性。有研究将钛掺入羟基磷灰石制备了复合物,并证明了复合物的抗菌性能[64]。钛的氧化物二氧化钛也被广泛用于材料的抗菌应用研究。LOPES等[65]的研究认为二氧化钛掺入羟基磷灰石后能提高其抗菌活性,并且羟基磷灰石与二氧化钛协同作用对条件致病菌有抗菌作用。还有研究通过在阳极氧化钛上电沉积仿生钙磷灰石涂层提高钛的抗菌性能。 钛及其氧化物的抗菌机制目前尚不明确。有研究认为二氧化钛的抗菌机制可能与活性氧产生有关[66]。二氧化钛纳米颗粒可以产生3种类型的活性氧(超氧自由基、羟基自由基和单氧氧基),对不同菌种间的杀菌效率存在较大差异[67],按敏感程度排序为铜绿假单胞菌(革兰阴性菌)、金黄色葡萄球菌(革兰阳性菌)、白色念珠菌(酵母菌)。 2.8 钴掺入羟基磷灰石 钴是人体正常运行的必需元素,是维生素B12的一个组成部分。维生素B12对红细胞的产生、细胞内DNA的合成、髓鞘的形成、神经细胞的保护和神经递质的调节是必要的[68]。钴还被用于成骨材料的制备。有证据表明组织工程支架中钴离子的存在刺激血管生成。有报道称,与纯含孔间隙生物玻璃(MBG)支架相比,掺杂2%和5%钴离子的含孔间隙生物玻璃支架显著增强血管内皮生长因子蛋白的分泌、缺氧诱导因子1a的表达与骨相关基因表达[69]。同样的,有研究将钴掺入羟基磷灰石制备复合材料,并证明了钴的促进血管生长作用[70]。 有研究证实了钴取代羟基磷灰石纳米颗粒的抗菌活性[71]。采用圆盘扩散法和液体培养基定量抗菌实验检测钴取代羟基磷灰石纳米颗粒对大肠杆菌(ATCC 25922)和金黄色葡萄球菌(ATCC 25923)两种病原菌的抗菌效果,结果显示所有被测生物材料均具有一定的抗菌性能,这两种实验的效果在金黄色葡萄球菌中比在大肠杆菌中更明显。钴取代羟基磷灰石纳米粒子晶体结构中钴的含量越高,其抗菌活性越强;同时还证明了材料的无毒性(根据溶血实验),这使得它们有可能作为具有抗菌性能的生物材料运用于临床。 2.9 壳聚糖掺入羟基磷灰石 壳聚糖是Rouget于1859年发现的一种多糖生物聚合物[72],其由几丁质部分去乙酰化反应产生的,几丁质是从虾和蟹壳中提取的。壳聚糖具有独特的生物学特性及广泛的应用前景,包括生物降解性、生物相容性和无毒性,这使得壳聚糖具有很大的商业价值。近年来壳聚糖及其衍生物因其抗真菌、细菌、病毒的性能备受关注。 壳聚糖的抗菌活性受多种因素的影响,包括壳聚糖的类型(如纯壳聚糖或衍生物)、分子质量、去乙酰化程度等。壳聚糖对金黄色葡萄球菌SG511具有剂量依赖性的生长抑制作用。细菌在接触壳聚糖后6 h内大部分被迅速杀灭[72]。然而这个明显的死亡阶段之后是细菌的再生阶段,这可能是由于少部分细菌变异引起的。壳聚糖具有浓度依耐性,能有效地渗透葡萄球菌质膜,并且可引起细菌细胞膜去极化,影响细菌的屏障功能。壳聚糖还可以通过影响细菌能量代谢而影响细菌生长。壳聚糖对耐甲氧西林金黄色葡萄球菌也有很好的抗菌效果[73]。有研究制备了羟基磷灰石-壳聚糖-唑来膦酸复合支架,证明了其对临床致病性金黄色葡萄球菌和大肠杆菌都表现出良好的抗菌活性[74]。还有研究采用电化学沉积法制备了壳聚糖、银和羟基磷灰石复合涂层,发现银和壳聚糖具有协同抗革兰阳性和革兰阴性菌株作用[75]。 2.10 镓掺入羟基磷灰石 镓是一种半金属元素,已被用于自身免疫性疾病和同种异体排斥反应、癌症和传染病等多种疾病的影像学检测和治疗[76]。镓对致病菌生存、定植需要的多种细菌酶酶解过程都有影响,例如影响参与DNA合成、电子传递和氧化应激防御的酶解过程。镓可以简单的盐形式或与有机化合物的复合物形式被细菌的铁捕获系统内化。镓具有某些分子特性如亲脂性,使细菌的细胞壁更容易穿透。硝酸镓的另一个独特特性是能够阻止生物膜的形成并破坏已经建立的生物膜。镓对抑制生物膜中心的细菌也有特别的疗效。这些生物膜中心的细菌通常处于稳定的生长阶段,对抗生素更有抵抗力[77]。 镓可以抑制骨吸收,从而降低血浆中的钙浓度。此外,它还能诱导成骨细胞并帮助骨骼形成。有文献制备了富含镓离子的羟基磷灰石,镓离子含量约占复合材料质量的11%,但他们没有检测这种材料的抗菌活性[78]。 2.11 硒掺入羟基磷灰石 富硒益生菌已被证明在体内和体外对致病性大肠杆菌的生长具有很强的抑制作用。在人体中,硒对保护细胞和组织免受氧化损伤至关重要,其补充用途已被包括组织工程、肿瘤学在内的各学科广泛探索。硒不仅能抑制细菌,而且具有较低的脱靶毒性[79]。硒纳米颗粒可以通过影响细菌内毒素、脂多糖诱导的巨噬细胞信号转导通路来对抗慢性炎症[79]。另外硒纳米粒子表面产生单线态氧引起的光动力学活性可能与其抗菌机制有关[80]。 硒也可以通过多种方法掺入羟基磷灰石。不同方法制备出的复合材料物理化学性质有所差异。有研究采用溶胶-凝胶方法制备了含有硒羟基磷灰石复合材料,并证明了复合材料对金黄色葡萄球菌、大肠杆菌、铜绿假单胞菌、枯草杆菌具有优异的抗菌效果[81]。 3 结论与展望 Conclusions and prospects 羟基磷灰石是良好的生物材料,具有优异的组织相容性、骨传导性,已被广泛应用于骨科及牙科的骨组织修复。但羟基磷灰石不具备抗菌性能,通过向羟基磷灰石中添加具有抗菌性能的材料可有效改善其不具备抗菌性的缺点,向羟基磷灰石中添加金属离子是主要的改性方法。虽然抗菌金属离子众多,但银离子依然是最主要的一个。通过不同的方法将银离子掺杂进羟基磷灰石,制备出了具备优良广谱抗菌性能的复合材料。金属离子掺杂进入羟基磷灰石具有很多优势,避免了有机抗菌物质在制备复合材料不耐高温的缺点。但金属离子加入羟基磷灰石抗菌,因金属离子的毒性研究价值受很大影响。目前抗菌材料的研究还存在几点不足:抗菌离子在羟基磷灰石中的释放曲线还没有办法很好的调控;目前抗菌材料种类较少,能用于人体植入物的就更少,还需要发现更多无毒的具有良好抗菌性能的物质以便今后使用;由于抗菌离子具有的毒性问题,每种抗菌离子的最适抗菌离子浓度还没有一个统一标准。希望今后的研究能解决这几个方面的问题。 "

| [1] ALESSANDRO P, CONSUELO C, ELPIDA P, et al.Engineering of Chitosan-Hydroxyapatite-Magnetite Hierarchical Scaffolds for Guided Bone Growth.Materials(Basel).2019;12:1-13. [2] LI G, QIN S, ZHANG D, et al.Preparation of antibacterial degummed silk fiber/nano-hydroxyapatite/polylactic acid composite scaffold by degummed silk fiber loaded silver nanoparticles.Nanotechnology.2019;30(29):295101. [3] 王天云,张青,朱良均,等.利用电化学法结合丝素膜调控羟基磷灰石沉积及其形貌[J].浙江大学学报(农业与生命科学版),2018, 44(2):209-214. [4] KULPETCHDARA K, LIMPICHAIPANIT A, RANDORN C, et al. Influence of the Nano Hydroxyapatite Powder on Thermally Sprayed HA Coatings onto Stainless Steel.Surf Coat Technol. 2016;306:181-186. [5] VILARDELL AM, CINCA N, GARCIA-GIRALT N, et al. Functionalized coatings by cold spray: An in vitro study of micro- and nanocrystalline hydroxyapatite compared to porous titanium.Mater Sci Eng C Mater Biol Appl.2018;87:41-49. [6] KEYNOOSH B, ABBASI KB, ABDOLLAH A.Characterization of sol-gel derived silver/fluor-hydroxyapatite composite coatings on titanium substrate.Surf Coat Technol.2018;352:522-528. [7] QADIR M, LI Y, WEN C.Ion-substituted calcium phosphate coatings by physical vapor deposition magnetron sputtering for biomedical applications: A review.Acta Biomater.2019;89:14-32. [8] AMANDA B, KATARZYNA S, ELŻBIETA M, et al.Biological effect of hydrothermally synthesized silica nanoparticles within crystalline hydroxyapatite coatings for titanium implants.Mater Sci Eng C.2018;92:88-95. [9] TÜRK S, ALTıNSOY I, EFE GÇ, et al.Biomimetic synthesis of Ag, Zn or Co doped HA and coating of Ag, Zn or Co doped HA/fMWCNT composite on functionalized Ti.Mate Sci Eng C. 2019;99:986-998. [10] WANG J, GONG X, HAI J, et al.Synthesis of silver-hydroxyapatite composite with improved antibacterial properties.Vacuum.2018;152(61):132-137. [11] VILLERET B, DIEU A, STRAUBE M, et al. Silver Nanoparticles Impair Retinoic Acid-Inducible Gene I-Mediated Mitochondrial Antiviral Immunity by Blocking the Autophagic Flux in Lung Epithelial Cells.ACS Nano.2018;12:1188-1202. [12] JALAL M, ANSARI M, ALZOHAIRY MA, et al. Anticandidal activity of biosynthesized silver nanoparticles: effect on growth, cell morphology, and key virulence attributes of Candida species.Int J Nanomedicine.2019;14:4667-4679. [13] DASTJERDI R, MONTAZER M. A review on the application of inorganic nano-structured materials in the modification of textiles: Focus on anti-microbial properties.Colloids Surf.B. 2010;79:5-18. [14] SIMCHI A, TAMJID E, PISHBIN F, et al.Recent progress in inorganic and composite coatings with bactericidal capability for orthopaedic applications. Nanomed.Nanotechnol.Biol Med. 2011;7(1):22-39. [15] HAJIPOUR MJ, FROMM KM, ASHKARRAN AA, et al. Antibacterial properties of nanoparticles.Trends Biotechnol. 2012;30(10):499-511. [16] JOANNA K, EWA G, KWIATKOWSKA-RÓżYCKA D. Substituted Hydroxyapatites with Antibacterial Properties. BioMed Res Int.2014;2014:1-15. [17] ZIELEWICZ J. A Review on Potential Role of Silver Nanoparticles and Possible Mechanisms of their Actions on Bacteria.Drug Res.2017;11(2):70-76. [18] ZHANG XF, LIU ZG, SHEN W, et al.Silver Nanoparticles: Synthesis, Characterization, Properties, Applications, and Therapeutic Approaches.Int J Mol Sci.2016;17(9):1534. [19] EHSAN R, ARAN R, CORMAC MG, et al.Adverse effects of nanosilver on human health and the environment.Acta Biomater.2019;94:145-159. [20] REN L, PAN Y, LIANG Q, et al. Synthesis of Dental Resin Matrix Containing Silver Nanoparticles.J Nanosci Nanotechnol.2019;19:5774-5782. [21] BAHAR K, NAHIDEH A, MORTEZA M, et al.A Review on Potential Role of Silver Nanoparticles and Possible Mechanisms of their Actions on Bacteria.Drug Res (Stuttg). 2017;67:70-76. [22] 叶伟杰,陈楷航,蔡少龄,等.纳米银的合成及其抗菌应用研究进展[J].材料工程,2017,45(9):22-30. [23] KEYHOOSH B, ABBASI KB, ABDOLLAH A.Characterization of sol-gel derived silver/fluor-hydroxyapatite composite coatings on titanium substrate.Surf Coat Technol.2018;352:522-528. [24] WEI P, FANG J, FANG L, et al. Novel niobium and silver toughened hydroxyapatite nanocomposites with enhanced mechanical and biological properties for load-bearing bone implants.Appl Mater Today.2019;15:531-542. [25] GENG Z, CUI Z, LI Z, et al.Strontium incorporation to optimize the antibacterial and biological characteristics of silver-substituted hydroxyapatite coating.Mater Sci Eng C. 2016;58:467-77. [26] CHI W, ZOU J, AI F, et al. Research of Cu-Doped Hydroxyapatite Microbeads Fabricated by Pneumatic Extrusion Printing.Materials(Basel).2019;12:1-11. [27] GABBAY J, BORKOW G. Copper, an Ancient Remedy Returning to Fight Microbial, Fungal and Viral Infections.Curr Chem Biol.2009;3:272-278. [28] RENL, MA Z, LI M, et al.Antibacterial properties of Ti-6Al-4V-xCu alloys.Mater Sci Technol.2014;30:699-705. [29] LIU J, LI F, LIU C, et al.Effect of Cu content on the antibacterial activity of titanium–copper sintered alloys.Mater Sci Eng C. 2014;35:392-400. [30] SCHMIDT MG, TUURI RE, DHARSEE A, et al.Antimicrobial copper alloys decreased bacteria on stethoscope surfaces. Am.J.Infect.Control.2017;45:642-647. [31] VINCENT M, DUVAL RE, HARTEMANN P, et al.Contact killing and antimicrobial properties of copper.J Appl Microbiol. 2017;124(5):1032-1046. [32] 何立伟,高珊.铜的毒性、抗菌性及促进创伤愈合功效研究进展[J].毒理学杂志,2017,31(6):425-429. [33] DIN MI, ARSHAD F, HUSSAIN Z, et al.Green Adeptness in the Synthesis and Stabilization of Copper Nanoparticles: Catalytic, Antibacterial, Cytotoxicity, and Antioxidant Activities. Nanoscale Res Lett.2017;12:638. [34] 王琦,卢珊,胡长鹰.纳米铜食品抗菌包装材料的研究进展[J].包装工程,2019,40(5):64-71. [35] GHOSH R, SWART O, WESTGATE S, et al.Antibacterial copper-hydroxyapatite composite coatings via electrochemical synthesis.Langmuir.2019;35:1-28. [36] SHEDBALKAR U, SINGH R, WADHWANI S, et al.Microbial synthesis of gold nanoparticles: Current status and future prospects.Adv.Colloid Interface Sci.2014;209:40-48. [37] ORTIZ-BENÍTEZ EA, VELÁZQUEZ-GUADARRAMA N, DURÁN FIGUEROA NV, et al.Antibacterial mechanism of gold nanoparticles on Streptococcus pneumoniae. Metallomics. 2019;11:1265-1276. [38] XING X, MA W, ZHAO X, et al. Interaction between Surface Charge-Modified Gold Nanoparticles and Phospholipid Membranes.Langmuir, 2018;34:12583-12589. [39] XIE Y, LIU Y, YAN J, et al.Gold Nanoclusters for Targeting Methicillin-Resistant Staphylococcus aureus In Vivo.Angew Chem Int Ed Engl.2018;57:3958-3962. [40] CUI Y, ZHAO Y, TIAN Y, et al.The molecular mechanism of action of bactericidal gold nanoparticles on Escherichia coli. Biomaterials.2012;33:2327-33. [41] ZHENG Y, LIU W, QIN Z, et al. Mercaptopyrimidine- Conjugated Gold Nanoclusters as Nanoantibiotics for Combating Multidrug-Resistant Superbugs.Bioconjug Chem. 2018;29:3094-3103. [42] KIM H, MONDAL S, JANG B, et al.Biomimetic synthesis of metal–hydroxyapatite (Au-HAp, Ag-HAp, Au-Ag-HAp): Structural analysis, spectroscopic characterization and biomedical application.Ceram Int.2018;44:20490-20500. [43] BAHERJEE S, BAGCHI S, BHANDARY S, et al.A facile vacuum assisted synthesis of nanoparticle impregnated hydroxyapatite composites having excellent antimicrobial properties and biocompatibility.Ceram Int. 2018;44: 1066-1077. [44] RUDE RK, GRUBER HE, NORTON HJ, et al.Dietary magnesium reduction to 25% of nutrient requirement disrupts bone and mineral metabolism in the rat.Bone. 2005;37: 211-219. [45] MAIER JAM, BERNARDINI D, RAYSSIGUIER Y, et al.High concentrations of magnesium modulate vascular endothelial cell behaviour in vitro.Biochim Biophys Acta.2004;1689:6-12. [46] ZICHE M, MORBIDELLI L. Nitric Oxide and Angiogenesis. Circulation.2002;105:2133-2135. [47] XUE W, DAHLQUIST K, BANERIEE A, et al.Synthesis and characterization of tricalcium phosphate with Zn and Mg based dopants.J Mater Sci Mater Med.2008;19:2669-2677. [48] LANDI E, LOGROSCINO G, PROETTI L, et al.Biomimetic Mg-substituted hydroxyapatite: from synthesis to in vivo behaviour.J Mater Sci Mater Med.2008;19:239-247. [49] GAYATHRI B, MUTHUKUMARASAMY N, DHAYALAN V, et al. Magnesium incorporated hydroxyapatite nanoparticles: Preparation,characterization, antibacterial and larvicidal activity. Arabian J Chem.2018;11:645-654. [50] COELHO CC, ARAUJO R, QUADROS PA, et al.Antibacterial bone substitute of hydroxyapatite and magnesium oxide to prevent dental and orthopaedic infections.Mater Sci Eng C Mater Biol Appl.2019;97:529-538. [51] LEUNG Y, NG A, XU X, et al.Mechanisms of antibacterial activity of mgo: non-ros mediated toxicity of mgo nanoparticles towards escherichia coli.Small. 2014;10:1171-1183. [52] HE Y, INGUDAM S, REED S, et al. Study on the mechanism of antibacterial action of magnesium oxide nanoparticles against foodborne pathogens.J Nanobiotechnol.2016;14:1-9. [53] FENG H, WANG G, JIN W, et al.Systematic study of inherent anti-bacterial properties of magnesium-based biomaterials. ACS Appl Mater.Interfaces.2016;8:1-35. [54] LI Y, LIU G, ZHAI Z, et al. Antibacterial properties of magnesium in vitro and in an in vivo model of implant-associated methicillin-resistant staphylococcus aureus infection. Antimicrob. Agents Chemother. 2014;58:7586-7591. [55] SAIDAK Z, MARIE PJ. Strontium signaling: Molecular mechanisms and therapeutic implications in osteoporosis. Pharmacol Ther.2012;136:216-226. [56] RAVI ND, BALU R, KUMAR TSS.Strontium-Substituted Calcium Deficient Hydroxyapatite Nanoparticles: Synthesis, Characterization, and Antibacterial Properties.J Am Ceram Soc.2012;95:2700-2708. [57] BRAUER DS, KARPUKHINA N, KEDIA G, et al.Bactericidal strontium-releasing injectable bone cements based on bioactive glasses.J R Soc Interface.2013;10(78):20120647. [58] LIN Y, YANG Z, CHENG J, et al.Synthesis,Charactcrization and Antibacterial Property of Strontium Half and Totally Substituted Hydroxyapatite Nanoparticles. J Wuhan Univ Technol.2008;23(4):475-479. [59] YAMAGUCHI M. Role of nutritional zinc in the prevention of osteoporosis.Mol Cell Biochem.2010;338(1-2):241-254. [60] XIONG K, ZHANG J, ZHU Y. Zinc doping induced differences in the surface composition, surfacemorphology and osteogenesis performance of the calcium phosphate cementhydration products.Mater.Sci.Eng.C.2019;105:1-9. [61] REBECCA C, JOSé CM, Suelen S, et al.Does the incorporation of zinc into calcium phosphate improve bone repair?A systematic review.Ceram Int.2018;44:1240-1249. [62] OFUDJE EA, ADEOGUN AI, IDOWU MA, et al.Synthesis and characterization of Zn-Doped hydroxyapatite: scaffold application, antibacterial and bioactivity studies.Heliyon. 2019;5:e01716. [63] PREDOI D, ICONARU SL, PREDOI MV, et al.Evaluation of Antibacterial Activity of Zinc-Doped Hydroxyapatite Colloids and Dispersion Stability Using Ultrasounds. Nanomaterials (Basel).2019;9:1-22. [64] WAKAMURA M. Photocatalysis by calcium hydroxyapatite modifiedby Ti(IV).Fujitsu Sci.Tech.J.2005;41(2):181-190. [65] LOPES FS, OLIVEIRA JR, MILANI J, et al.Biomineralized diamond-like carbon films incorporating titanium dioxide nanoparticles improved bioactivity properties and reduced biofilm formation.Mater Sci Eng C.2017;81:373-379. [66] LI Y, ZHANG W, NIU J, et al.Mechanism of Photogenerated Reactive Oxygen Species and Correlation with the Antibacterial Properties of Engineered Metal-Oxide Nanoparticles.ACS Nano.2012;6(6):5164-5173. [67] MACFARLANE J W, JENKINSON H F, SCOTT T B. Sterilization of microorganisms on jet spray formed titanium dioxide surfaces.Appl.Catal.B.2011;(106):181-185. [68] DAPU S, EL CA, CHRISTOFILOPOULOS P, et al.The potential role of cobalt ions released from metal prosthesis on the inhibition of Hv1 proton channels and the decrease in Staphyloccocus epidermidis killing by human neutrophils. Biomaterials.2011;32(7):1769-1777. [69] WU C, ZHOU Y, FAN W, et al.Hypoxia-mimicking mesoporous bioactive glass scaffolds with controllable cobalt ion release for bone tissue engineering.Biomaterials. 2012;33(7):2076-2085. [70] KULANTHAIVEL S, ROY B, AGARWAL T, et al.Cobalt doped Proangiogenic Hydroxyapatite for Bone Tissue Engineering Application. Mater Sci Eng C.2015;58:648. [71] AJDUKOVIĆ ZR, MIHAJILOV-KRSTEV TM, IGNJATOVIĆ NL, et al.In Vitro Evaluation of Nanoscale Hydroxyapatite-Based Bone Reconstructive Materials with Antimicrobial Properties.J Nanosci Nanotechnol. 2016;16(2):1420-1428. [72] DINA R, KRISTINE V B, ALBERT H, et al.Insights into the mode of action of chitosan as an antibacterial compound.Appl Environ Microbiol.2008;74(12):3764-3773. [73] COSTA EM, SILVA S, TAVARIA FK, et al.Insights into chitosan antibiofilm activity against methicillin-resistant Staphylococcus aureus.J Appl Microbiol.2017;122:1547-1557. [74] LU Y, LI M, LI L, et al.High-activity chitosan/ nanohydroxyapatite/zoledronic acid scaffolds for simultaneous tumor inhibition, bone repair and infection eradication.Mater Sci Eng C.2018;82:225-233. [75] YAN Y, ZHANG X, LI C, et al.Preparation and characterization of chitosan-silver/hydroxyapatite composite coatings onTiO2 nanotube for biomedical applications.Appl.Surf.Sci. 2015;332: 62-69. [76] RODRIGUEZ GM, GARDNER R, KAUR N, et al.Utilization of Fe3+ -acinetoferrin analogs as an iron source by Mycobacterium tuberculosis.Biometals 2008;21:93-103. [77] KANEKO Y, THOENDEL M, OLAKANMI O, et al.The transition metal gallium disrupts Pseudomonas aeruginosa iron metabolism and has antimicrobial and antibiofilm activity. J Clin Invest.2007;117(4):877-888. [78] MELNIKOV P, TEIXEIRA AR, MALZAC A, et al. Gallium-containing hydroxyapatite for potential use in orthopedics.Mater Chem Phys.2009;117(1):86-90. [79] LIU W, GOLSHAN N H, DENG X, et al.Selenium nanoparticles incorporated into titania nanotubes inhibit bacterial growth and macrophage proliferation. Nanoscale. 2016;8(34):15783-15794. [80] NASTULYAVICHUS A, KUDRYASHOV S, SMIRNOV N, et al. Antibacterial coatings of Se and Si nanoparticles.Appl Surf Sci.2018;469:220-225. [81] PRIYADARSHINI B, ANJANEYULU U, VIJAYALAKSHMI U. Preparation and characterization of sol-gel derived Ce4+ doped hydroxyapatite and its in vitro biological evaluations for orthopedic applications.Mater Des.2017;119:446-455. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Liu Jiangfeng. Nano-hydroxyapatite/polyamide 66 composite filling combined with locking plate in the treatment of fibrous dysplasia of femoral bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 542-547. |
| [5] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [6] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [7] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [8] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [9] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [10] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [11] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||