Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (5): 766-773.doi: 10.3969/j.issn.2095-4344.0449
Previous Articles Next Articles
Stem cell models for commercialization
Ke Min-xia1, Ji Meng2, Wang Hao2, Hong Dan-ping1, Wu Yue-hong1, Qi Nian-min2
- 1Zhejiang Sci-Tech University, Hangzhou 310018, Zhejiang Province, China; 2Hangzhou Stem Cell Models Biological Technology Co., Ltd., Hangzhou 310018, Zhejiang Province, China
-
Revised:2017-09-15Online:2018-02-18Published:2018-02-18 -
Contact:Qi Nian-min, Ph.D., Professor, Hangzhou Stem Cell Models Biological Technology Co., Ltd., Hangzhou 310018, Zhejiang Province, China; Wu Yue-hong, Ph.D., Associate professor, Zhejiang Sci-Tech University, Hangzhou 310018, Zhejiang Province, China -
About author:Ke Min-xia, Studying for master’s degree, Zhejiang Sci-Tech University, Hangzhou 310018, Zhejiang Province, China -
Supported by:the National Natural Science Foundation of China, No. 31201867; the Zhejiang Provincial Natural Science Foundation of China, No. LY17C120001; the Enterprise Commissioned R & D Project, No. 16040135-J
CLC Number:
Cite this article
Ke Min-xia, Ji Meng, Wang Hao, Hong Dan-ping, Wu Yue-hong, Qi Nian-min. Stem cell models for commercialization[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(5): 766-773.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

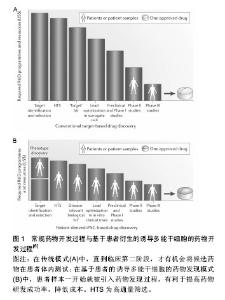
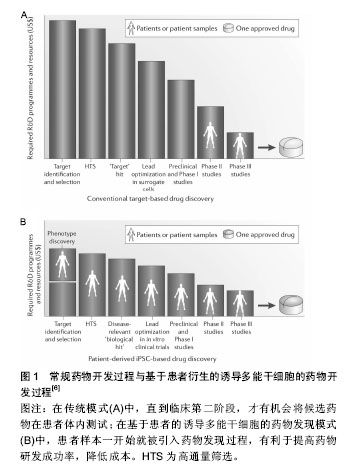
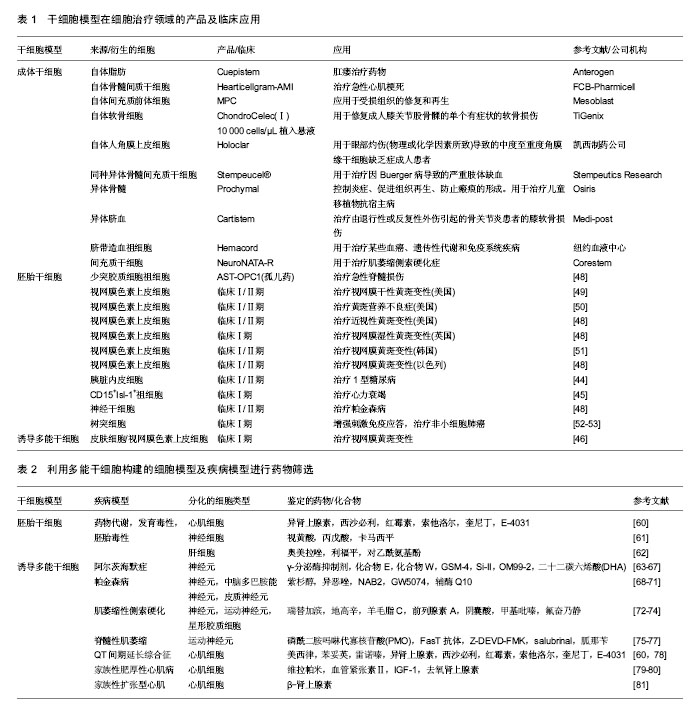
2.1 干细胞模型 2.1.1 胚胎干细胞 胚胎干细胞的研究最为广泛和深入。1998年,Thomson等[1]从人胚胎分离培养了人类胚胎干细胞,被当年的科学杂志称为10大重大科学发现之一。人胚胎干细胞源自早期受精卵的内细胞团,可以长期维持自我更新能力,并具有分化成三胚层(内胚层、中胚层、外胚层)所有细胞类型的潜能[2]。在体外特定的培养条件下,可将胚胎干细胞诱导分化为各种所需的细胞类型,建立各种细胞模型。例如衍生于胚胎干细胞所建立的心肌细胞、肝细胞或神经元细胞模型,对于人类发育生物学、药物发现和细胞替代治疗具有重大的意义和应用价值[3-5]。 相对于常用的永生化或原代细胞,胚胎干细胞具有相当大的理论优势,但它们在应用方面存在着巨大的障碍。首先,人类胚胎干细胞存在相当大的道德争论;其次,由于多因素限制,疾病不能通过植入前遗传诊断(PGD)识别,因此仅能建立罕见的单基因突变或单染色体畸变的疾病特异性胚胎干细胞系;最后,胚胎干细胞的定向诱导分化仍存在挑战性,不同的人胚胎干细胞系发育潜力不同[6-8]。 2.1.2 成体干细胞 成体干细胞是一类存在于已分化组织中的未分化细胞,具有自我更新能力并能够分化成一种或多种特定细胞类型的潜能。成体干细胞种类和数目庞大,可根据其来源的胚层分为:中胚层干细胞、外胚层干细胞和内胚层干细胞。根据来源的组织和功能命名,成体干细胞有骨髓间充质干细胞、造血干细胞、脐带间充质干细胞、神经干细胞、表皮干细胞、心脏干细胞、肝脏干细胞、胰腺干细胞、肠干细胞等。其中,造血干细胞和骨髓间充质干细胞是研究最早、最广和最深的成体干细胞,其临床应用也最为广泛和成熟。 成体干细胞存在于组织的特定区域,大多处于休眠状态,分裂很慢或很少分裂,直到组织受到损伤或发生疾病时被激活,开始分裂分化。成体干细胞不仅具有向原有组织来源的特定细胞分化的潜能,还具有向其他谱系甚至跨胚层分化的潜能,这一特性称为干细胞的可塑性或是横向分化[9]。例如脐带间充质干细胞,既能向中胚层方向如脂肪细胞、成骨细胞、软骨细胞等分化,也可分化成外胚层细胞如神经元、星形胶质细胞和神经胶质细胞,还可向内胚层方向定向分化形成肝实质细胞、胰岛细胞等[10-15]。 目前成体干细胞研究的新发现和新突破不断涌现,综合而言成体干细胞具有以下诸多优点:①成体干细胞源于自身,用于自身,在应用时不存在组织相容性的问题,避免了移植排斥反应以及长期应用免疫抑制剂对患者的伤害;②由于胚胎干细胞和诱导多能干细胞在体内的分化是“非定位性的”,易形成畸胎瘤。理论上成体干细胞致瘤风险很低,而且不存在伦理道德问题[8];③获取相对容易,成体干细胞还具有多向分化潜能。但成体干细胞在研究和应用领域还存在一些尚未解决的问题,如成体干细胞含量极低,分离和纯化困难;体外长期培养可能会导致细胞衰老,产生遗传变异;分化能力受年龄限制,存在遗传错误等[16]。 2.1.3 诱导多潜能干细胞 2006年,日本Takahashi和Yamanaka等科学家从与干细胞维持多能性相关的24个转录因子中筛选出4个转录因子—Oct4,Sox2,klf4,c-Myc,确定这4个转录因子对重编程是至关重要的。他们以反转录病毒为载体,在小鼠的成纤维细胞中导入这4种转录因子,所获得的细胞形态和分化潜能与胚胎干细胞十分相似。由于这种细胞是由成纤维细胞经过诱导转变而成的干细胞,所以命名为诱导性多能干细胞[17]。 继2006年首次报道诱导性多能干细胞后,2007年,两个研究小组成功将小鼠成纤维细胞诱导的诱导性多能干细胞形成嵌合体,并通过生殖细胞系传递到下一代,证明了诱导性多能干细胞具有完整的发育潜能[18-19]。同年Yu和Takahashi等报道将人成纤维细胞重编程获得人诱导性多能干细胞[20-21]。目前科学家已经可以从多种细胞获得诱导性多能干细胞,包括皮肤细胞、神经细胞、头发角质细胞、精原细胞、胰岛β细胞和淋巴细胞等[22-27]。这些多种多样的细胞分别来源于胚胎发育的3个胚层,这说明来源于不同组织的体细胞都有重编程为诱导性多能干细胞的潜能。诱导性多能干细胞可分化为多种类型细胞,包括心肌细胞、神经脊细胞、胰岛β细胞、巨噬细胞、肝细胞等[28-32]。 诱导性多能干细胞与胚胎干细胞类似,具有形成体内所有类型细胞的多向分化潜能和不断自我更新的特性,并且不受细胞来源、免疫排斥、道德伦理、宗教法律等诸多方面的限制[8]。诱导性多能干细胞有着巨大优势,但也存在不少缺陷,比如需要转入慢病毒或反转录病毒载体,目前陆续发展为仙台病毒载体、质粒瞬转、转座子等非整合的导入方式,降低了临床应用中的安全隐患[33-36]。利用诱导性多能干细胞构建的各种细胞模型为特异性疾病模型建立、细胞替代治疗、组织器官再生和移植、发育生物学、药理及毒理学等领域开辟了崭新的研究途径[37-38]。 2.2 商业化应用现状 2.2.1 细胞治疗 干细胞治疗技术应用时间最早,范围最广。骨髓移植技术是最早的干细胞治疗相关的临床应用,并已成为根治白血病等疾病的主要手段。目前不同类型的干细胞其治疗技术成熟度不同,因此其应用现状也处于不同阶段。成体干细胞治疗技术最为成熟,研究最为集中,现临床或已上市的干细胞产品多为此类。胚胎干细胞拥有分化全能性,却存在着免疫排斥的限制和伦理学方面的困扰,在应用方面仍有争议但也选择性地进入临床试验。诱导性多能干细胞由于其独特的优势在再生领域备受期待,目前主要处于基础研究和动物实验阶段,2014年首次应用于人类疾病临床治疗。 国际上已批准多款成体干细胞产品上市,批准这些产品上市的国家或机构包括美国FDA、欧洲EMA、澳洲TGA、韩国FDA、加拿大等。2008年,Osiris公司的干细胞产品Prochymal在美国上市,是一种异体骨髓来源的成体干细胞产品,被设计来控制炎症、促进组织再生、防止瘢痕的形成。2012年,获加拿大药监局批准上市,被称为是世界首个干细胞治疗药物,用于治疗儿童移植物抗宿主病。韩国FDA批准了3种干细胞药物。2011年7月,韩国食品药品管理局宣布,准许由FCB-Pharmicell公司开发的心脏病治疗药物Hearticellgram-AMI自7月1日起投放市场销售。Hearticellgram-AMI用于治疗急性心肌梗死,主要的治疗方法是从患者自身骨髓中提取间质干细胞移植注入冠状动脉。2012年1月,韩国食品药品管理局表示,批准Medi-post公司的软骨再生治疗药物Cartistem(脐血来源)和Anterogen公司的肛瘘治疗药物Cuepistem(自体脂肪来源)等生产许可。CartiStem是全球首创的异基因干细胞药物,来源于他人新生儿脐血干细胞,主要治疗由退行性或反复性外伤引起的骨关节炎患者的膝软骨损伤。 国际干细胞治疗领域上市的多为干细胞产品而非药品。2009年10月,欧盟药品管理局的人用药品委员会(CHMP)推荐医疗产品ChondroCelect(Ⅰ)10 000 cells/μL植入悬液的上市申请,Chondro Celect(Ⅰ) 10 000 cells/μL植入悬液来源于自体软骨细胞,用于修复成人膝关节股骨髁的单个有症状的软骨损伤,目前该产品已在比利时、荷兰、卢森堡、德国、英国、芬兰和西班牙等国上市销售。2010年7月,澳大利亚治疗用品管理局(TGA)批准Mesoblast公司生产和供应自体间充质前体细胞(MPC)产品在澳大利亚上市,该产品主要应用于受损组织的修复和再生。2011年11月,美国食品药品监管局批准了脐血来源的药物Hemacord,这也是第一个得到批准的脐带造血祖细胞(HPCs)疗法。Hmeacord适用于造血系统紊乱患者的造血干细胞移植过程。例如,脐血移植已经被用来治疗某些血癌、遗传性代谢和免疫系统疾病。2015年2月,意大利凯西制药公司的干细胞治疗产品Holoclar获得欧盟委员会有条件批准,用于眼部灼伤(物理或化学因素所致)导致的中度至重度角膜缘干细胞缺乏症成人患者。该产品在10年实验期间,让81例患者重见光明,成功率超过3/4,成为再生医学的一大突破。2015年2月韩国KFDA批准商品名为NeuroNATA-R产品,其来源间充质干细胞,用于治疗肌萎缩侧索硬化症。2016年5月,Stempeutics Research宣布印度药品管理总局批准“Stempeucel®”细胞治疗产品,Stempeucel®是体外培养的成体同种异体骨髓间充质干细胞,用于治疗因Buerger病导致的严重肢体缺血。 人胚胎干细胞是组织器官工程的理想种子细胞,但由于其免疫原性问题和伦理道德而备受争议。目前共有8项临床试验正在进行。涉及脊椎脊柱损伤、黄斑变性、糖尿病、心脏修复、癌症疫苗等多个方面[7]。利用人胚胎干细胞衍生的产品—少突胶质细胞祖细胞1(OPC1)治疗脊椎损伤的临床试验最早开始于2010年,在亚特兰大的牧者中心,由加利福尼亚州Geron公司赞助,治疗了仅5例患者,但由于缺乏投资和支持迫使Geron公司过早地终止这一试验[39]。2013年,Asterias公司重新启动该临床试验,第1例患者于2015年6月在亚特兰大接受治疗。2014年2月,美国食品和药物管理局(FDA)认证了AST-OPC1作为治疗急性脊髓损伤的孤儿药。利用人胚胎干细胞衍生的视网膜色素上皮细胞治疗视网膜黄斑变性目前共有多个临床试验,该疾病可能是通过人胚胎干细胞治疗成功的第1种疾病[39-42]。此外,加利福尼亚Viacyte公司花费了大量时间研发了基于人胚胎干细胞的胰岛素产品VC-01,第1例患者于2014年10月在加州大学圣地亚哥分校接受治疗[43-44]。人胚胎干细胞衍生的心脏方向的CD15+ ISL-1+祖细胞的纤维蛋白贴片用于移植治疗心力衰竭的临床试验于2014年在法国开始,第1例患者在6个月治疗后心力衰竭程度由Ⅲ级改善为Ⅰ级[45]。 人诱导多潜能干细胞的发现为干细胞的治疗带来了新的希望。2017年日本高桥雅代博士团队使用诱导性多能干细胞分化出了视网膜色素上皮细胞,并移植给了1例患老年性黄斑变性的女性患者(临床备案号UMIN000011929),成功阻止了疾病的进展[46]。研究人员用患者自己的皮肤细胞重编程得到了诱导多潜能干细胞,然后分化出了视网膜色素上皮细胞,移植到患者的右眼。术后3个月,研究人员终于在患者的右眼中发现了大量的功能性视网膜色素上皮细胞,黄斑变性的“脚步”被拦住了!患者的视力恢复到了0.1。尽管没有将患者的视力恢复到正常水平,但是干细胞治疗还是阻止了疾病的进展。根据研究人员的随访,直到2016年12月9日,这名患者的情况一直都很稳定,没有出现过排斥和癌变,也没有再接受过其他治疗。这项研究首次证明诱导多潜能干细胞在人体内使用是安全的,也是首例诱导多潜能干细胞克隆技术用于人类疾病临床治疗。来自哈佛医学院的George Daley博士在其关于干细胞临床应用的评论文章中肯定了高桥博士这一临床成果的重要性,称“这是一个里程碑式的研究”[47]。诱导多潜能干细胞治疗技术为干细胞治疗及再生医学开启了新的篇章。 表1罗列了目前在细胞治疗领域成体干细胞的商业化产品及多能干细胞的临床研究[44-53]。 2.2.2 药物筛选 为了提高药物研发产率,同时为了降低开发后期药物损耗的发生率,必须在药物开发项目初期尽早对该化合物的各种潜在毒性以及可能出现的其他不良反应进行全面评估,降低药物开发项目失败的风险。利用实验动物模型检测化合物不良反应方面在新药研发中发挥了十分重要的作用,但是利用动物模型存在几个问题:①动物模型建立困难,实验成本高昂;②动物模型与人类存在种属差异,所建立的疾病临床相关性尚待商榷;③使用动物模型涉及伦理道德问题,受各种相关政策法规的制约。利用多能干细胞建立的细胞模型或疾病模型进行药物筛选是最具有前景的应用之一。 胚胎干细胞实验(embryonic stem cell test,EST)作为欧洲替代动物实验中心(ECVAM)正式批准为体外药物和化合物毒性筛选的动物替代方法,已经进入实际应用阶段,是目前惟一国际公认的利用细胞系的发育毒性进行体外替代实验方法[54]。胚胎干细胞实验方法采用了两种细胞,分化成熟的3T3成纤维细胞和未分化的D3胚胎干细胞。基本流程是通过MTT试验检测受试物的3T3细胞毒性和D3细胞毒性,同时检测抑制胚胎干细胞分化为心肌细胞的能力。ECVAM共挑选了30种受试物,使用胚胎干细胞实验方法进行胚胎发育毒性检测,结果显示预测符合率达到82%[55]。目前胚胎干细胞实验方法仍存在一些不足,例如小鼠与人存在种属差异性、检测指标单一、步骤繁琐、难以实现自动化等问题。研究人员做了大量努力来改进这些不足,例如使用人胚胎干细胞、引入多个分子生物学终点、使用多种检测手段等方法。Roche公司建立的基于受试物对人胚胎干细胞分化过程中SOX17基因表达建立起来的人胚胎干细胞实验模型。Roche公司通过约75种化合物的评价与验证得出SOX17的表达与胚胎毒性呈负相关,结果与已知发育毒性物质的比对证实该模型预测正确率达90%以上。关于胚胎干细胞的使用涉及多方面的伦理道德和法律法规。1990年英国政府出台了《人类生殖与胚胎学法》,是第一个对胚胎干细胞研究立法的国家[56]。英国成立专家组对克隆及胚胎干细胞技术进行评估,并于2001年通过了《人类生殖及胚胎学(研究目的)规则》,允许进行“治疗性克隆”[57]。2009年奥巴马政府宣布解除对用联邦政府资金支持人胚胎干细胞研究的限制。日本政府制定干细胞研究指南,规定用于研究的胚胎细胞只能从那些本该被抛弃、用于生育治疗目的的胚胎中获取。中国关于胚胎干细胞的研究仍存在着争议,尚未形成一套完整有效的管理体系。人胚胎干细胞技术应用于药物安全性评价,需基于伦理学及相关法律法规的正确引导,并建立一系列标准化的规范操作指南。 人诱导多能干细胞与人胚胎干细胞类似,且不存在伦理道德争议,在药物研发、药物安全性评价应用前景十分广阔。基于诱导多能干细胞建立的疾病模型可用于药物开发过程中几乎所有的临床前阶段,包括药物靶点的确认、一般毒性和发育毒性的检测、候选药物的高通量筛选等[6](图1)。iPierian公司通过多个脊髓性肌萎缩症患者自身构建诱导多能干细胞,诱导形成具有该疾病特征的运动神经元模型,利用该模型对20万种化合物进行候选药物的高通量筛选。Johns Hopkins大学的研究人员提取了Riley-Day综合征患者的皮肤细胞,并将其诱导成为多能干细胞,随后他们诱导这些诱导多能干细胞生成受到疾病影响的神经细胞,成功用这些细胞快速简便地进行了药物筛选。在这项研究中,研究人员用体外形成的Riley-Day神经细胞筛选了约7 000种药物的效力。通过自动化分析,研究人员从中选出了8个化合物进行进一步试验,研究显示这些化合物能提高IKBKAP的表达,IKBKAP是Riley-Day综合征中的致病基因。其中,SKF-86466能够通过调节细胞内PKA依赖的CREB磷酸化和cAMP水平来诱导IKBKAP转录,是在细胞水平上中止或逆转疾病进程的潜在分子[58]。Cellular Dynamics公司将来自健康人的诱导多能干细胞培育成心脏细胞出售给制药公司和其他公司用于毒理筛选。利用诱导多能干细胞进行药物筛选,能够提高药物发现的成功率,建立有效的药效和安全性评估系统,并将药物开发成本从目标鉴定到临床试验阶段进行优化[59]。表2列举了部分目前研究最广泛深入的用于药物筛选的衍生的细胞模型和疾病模型[60-81]。"
| [1] Thomson JA, Itskovitz-Eldor J, Shapiro SS, et al. Embryonic stem cell lines derived from human blastocysts. Science. 1998; 282(5391):1145-1147.[2] Ebert AD, Svendsen CN. Human stem cells and drug screening: opportunities and challenges. Nat Rev Drug Discov. 2010;9(5): 367-372.[3] Yang P, Chen X, Kaushal S, et al. High glucose suppresses embryonic stem cell differentiation into cardiomyocytes : High glucose inhibits ES cell cardiogenesis. Stem Cell Res Ther. 2016;7(1):187.[4] Bae J, Lee N, Choi W, et al. Use of Microfluidic Technology to Monitor the Differentiation and Migration of Human ESC-Derived Neural Cells. Methods Mol Biol. 2016;1502:223-235.[5] Waraky A, Aleem E, Larsson O. Downregulation of IGF-1 receptor occurs after hepatic linage commitment during hepatocyte differentiation from human embryonic stem cells. Biochem Biophys Res Commun. 2016;478(4):1575-1581.[6] Grskovic M, Javaherian A, Strulovici B, et al. Induced pluripotent stem cells--opportunities for disease modelling and drug discovery. Nat Rev Drug Discov. 2011;10(12):915-929.[7] Ilic D, Ogilvie C. Concise Review: Human Embryonic Stem Cells-What Have We Done? What Are We Doing? Where Are We Going. Stem Cells. 2017;35(1):17-25.[8] Takahashi K, Yamanaka S. A decade of transcription factor-mediated reprogramming to pluripotency. Nat Rev Mol Cell Biol. 2016;17(3):183-193.[9] Anderson DJ, Gage FH, Weissman IL. Can stem cells cross lineage boundaries. Nat Med. 2001;7(4):393-395.[10] Ciuffreda MC, Malpasso G, Musarò P, et al. Protocols for in vitro Differentiation of Human Mesenchymal Stem Cells into Osteogenic, Chondrogenic and Adipogenic Lineages. Methods Mol Biol. 2016;1416:149-158.[11] Nan C, Guo L, Zhao Z, et al. Tetramethylpyrazine induces differentiation of human umbilical cord-derived mesenchymal stem cells into neuron-like cells in vitro. Int J Oncol. 2016;48(6): 2287-2294.[12] Xuan J, Feng W, An ZT, et al. Anti-TGFβ-1 receptor inhibitor mediates the efficacy of the human umbilical cord mesenchymal stem cells against liver fibrosis through TGFβ-1/Smad pathway. Mol Cell Biochem. 2017;429(1-2):113-122.[13] Zhou X, Cui L, Zhou X, et al. Induction of hepatocyte-like cells from human umbilical cord-derived mesenchymal stem cells by defined microRNAs. J Cell Mol Med. 2017;21(5):881-893.[14] 郑颂浩,哈承志,杨旭,等. 胰岛素促进脐带间充质干细胞成骨分化的作用[J].中国组织工程研究,2016,20(6):807-813.[15] Nasadyuk CM. Umbilical cord stem cells: biological characteristics, approaches to banking and clinical application. Cell and Organ Transplantology. 2016; 4(2):230-235. [16] Kuniakova M, Oravcova L, Varchulova-Novakova Z, et al. Somatic stem cell aging and malignant transformation--impact on therapeutic application. Cell Mol Biol Lett. 2015;20(5): 743-756.[17] Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126(4):663-676.[18] Wernig M, Meissner A, Foreman R, et al. In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature. 2007; 448(7151):318-324.[19] Okita K, Ichisaka T, Yamanaka S. Generation of germline- competent induced pluripotent stem cells. Nature. 2007;448 (7151):313-317.[20] Yu J, Vodyanik MA, Smuga-Otto K, et al. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007; 318(5858):1917-1920.[21] Takahashi K, Tanabe K, Ohnuki M, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131(5):861-872.[22] Chen F, Zhang G, Yu L, et al. High-efficiency generation of induced pluripotent mesenchymal stem cells from human dermal fibroblasts using recombinant proteins. Stem Cell Res Ther. 2016;7(1):99.[23] Eminli S, Utikal J, Arnold K, et al. Reprogramming of neural progenitor cells into induced pluripotent stem cells in the absence of exogenous Sox2 expression. Stem Cells. 2008; 26(10):2467-2474.[24] Hung SS, Pébay A, Wong RC. Generation of Integration-free Human Induced Pluripotent Stem Cells Using Hair-derived Keratinocytes. J Vis Exp. 2015;(102):e53174.[25] Kossack N, Meneses J, Shefi S, et al. Isolation and characterization of pluripotent human spermatogonial stem cell-derived cells. Stem Cells. 2009;27(1):138-149.[26] Stadtfeld M, Brennand K, Hochedlinger K. Reprogramming of pancreatic beta cells into induced pluripotent stem cells. Curr Biol. 2008;18(12):890-894.[27] Hanna J, Markoulaki S, Schorderet P, et al. Direct reprogramming of terminally differentiated mature B lymphocytes to pluripotency. Cell. 2008;133(2):250-264.[28] Shiba Y, Gomibuchi T, Seto T, et al. Allogeneic transplantation of iPS cell-derived cardiomyocytes regenerates primate hearts. Nature. 2016;538(7625):388-391.[29] Liu JA, Cheung M. Neural crest stem cells and their potential therapeutic applications. Dev Biol. 2016;419(2):199-216.[30] Manzar GS, Kim EM, Zavazava N. Demethylation of Induced Pluripotent Stem Cells from Type 1 Diabetic Patients Enhances Differentiation into Functional Pancreatic β-cells. J Biol Chem. 2017 Mar 30. [Epub ahead of print][31] Muffat J, Li Y, Yuan B, et al. Efficient derivation of microglia-like cells from human pluripotent stem cells. Nat Med. 2016;22(11): 1358-1367.[32] Song Z, Cai J, Liu Y, et al. Efficient generation of hepatocyte-like cells from human induced pluripotent stem cells. Cell Res. 2009;19(11):1233-1242.[33] Stadtfeld M, Nagaya M, Utikal J, et al. Induced pluripotent stem cells generated without viral integration. Science. 2008;322 (5903):945-949.[34] Okita K, Yamakawa T, Matsumura Y, et al. An efficient nonviral method to generate integration-free human-induced pluripotent stem cells from cord blood and peripheral blood cells. Stem Cells. 2013;31(3):458-466. [35] Choi IY, Lim H, Lee G. Efficient generation human induced pluripotent stem cells from human somatic cells with Sendai-virus. J Vis Exp. 2014;(86):e51406.[36] Davis RP, Nemes C, Varga E, et al. Generation of induced pluripotent stem cells from human foetal fibroblasts using the Sleeping Beauty transposon gene delivery system. Differentiation. 2013;86(1-2):30-37.[37] Hosoya M, Czysz K. Translational Prospects and Challenges in Human Induced Pluripotent Stem Cell Research in Drug Discovery. Cells. 2016;5(4): E46.[38] Karagiannis P, Eto K. Ten years of induced pluripotency: from basic mechanisms to therapeutic applications. Development. 2016;143(12):2039-2043.[39] Ilic D, Devito L, Miere C, et al. Human embryonic and induced pluripotent stem cells in clinical trials. Br Med Bull. 2015;116: 19-27.[40] Schwartz SD, Regillo CD, Lam BL, et al. Human embryonic stem cell-derived retinal pigment epithelium in patients with age-related macular degeneration and Stargardt's macular dystrophy: follow-up of two open-label phase 1/2 studies. Lancet. 2015;385(9967):509-516.[41] Schwartz SD, Hubschman JP, Heilwell G, et al. Embryonic stem cell trials for macular degeneration: a preliminary report. Lancet. 2012;379(9817):713-720.[42] Song WK, Park KM, Kim HJ, et al. Treatment of macular degeneration using embryonic stem cell-derived retinal pigment epithelium: preliminary results in Asian patients. Stem Cell Reports. 2015;4(5):860-872.[43] Schulz TC. Concise Review: Manufacturing of Pancreatic Endoderm Cells for Clinical Trials in Type 1 Diabetes. Stem Cells Transl Med. 2015;4(8):927-931.[44] Agulnick AD, Ambruzs DM, Moorman MA, et al. Insulin-Producing Endocrine Cells Differentiated In Vitro From Human Embryonic Stem Cells Function in Macroencapsulation Devices In Vivo. Stem Cells Transl Med. 2015;4(10):1214-1222.[45] Menasché P, Vanneaux V, Hagège A, et al. Human embryonic stem cell-derived cardiac progenitors for severe heart failure treatment: first clinical case report. Eur Heart J. 2015;36(30): 2011-2017.[46] Mandai M, Watanabe A, Kurimoto Y, et al. Autologous Induced Stem-Cell-Derived Retinal Cells for Macular Degeneration. N Engl J Med. 2017;376(11):1038-1046.[47] Daley GQ. Polar Extremes in the Clinical Use of Stem Cells. N Engl J Med. 2017;376(11):1075-1077.[48] Trounson A, McDonald C. Stem Cell Therapies in Clinical Trials: Progress and Challenges. Cell Stem Cell. 2015;17(1):11-22.[49] Schwartz SD, Hubschman JP, Heilwell G, et al. Embryonic stem cell trials for macular degeneration: a preliminary report. Lancet. 2012;379(9817):713-720.[50] Schwartz SD, Regillo CD, Lam BL, et al. Human embryonic stem cell-derived retinal pigment epithelium in patients with age-related macular degeneration and Stargardt's macular dystrophy: follow-up of two open-label phase 1/2 studies. Lancet. 2015;385(9967):509-516.[51] Song WK, Park KM, Kim HJ, et al. Treatment of macular degeneration using embryonic stem cell-derived retinal pigment epithelium: preliminary results in Asian patients. Stem Cell Reports. 2015;4(5):860-872.[52] Tseng SY, Nishimoto KP, Silk KM, et al. Generation of immunogenic dendritic cells from human embryonic stem cells without serum and feeder cells. Regen Med. 2009;4(4):513-526.[53] Nishimoto KP, Tseng SY, Lebkowski JS, et al. Modification of human embryonic stem cell-derived dendritic cells with mRNA for efficient antigen presentation and enhanced potency. Regen Med. 2011;6(3):303-318.[54] Seiler AE, Spielmann H. The validated embryonic stem cell test to predict embryotoxicity in vitro. Nat Protoc. 2011;6(7):961-978.[55] Scholz G, Genschow E, Pohl I, et al. Prevalidation of the Embryonic Stem Cell Test (EST)-A New In Vitro Embryotoxicity Test. Toxicol In Vitro. 1999;13(4-5):675-681.[56] Robertson JA. Ethics and policy in embryonic stem cell research. Kennedy Inst Ethics J. 1999;9(2):109-136.[57] Chief Medical Officer's Expert Group Reviewing the Potential of Developments on Stem Cell Research and Cell Nuclear Replacement to Benefit Human Health, Office of the CMO, UK. Stem Cell Research: Stem Cell Research: Medical Progress with Responsibility: Executive Summary. Cloning. 2000;2(2): 91-96.[58] Lee G, Ramirez CN, Kim H, et al. Large-scale screening using familial dysautonomia induced pluripotent stem cells identifies compounds that rescue IKBKAP expression. Nat Biotechnol. 2012;30(12):1244-1248.[59] De Vos J, Bouckenheimer J, Sansac C, et al. Human induced pluripotent stem cells: A disruptive innovation. Curr Res Transl Med. 2016;64(2):91-96.[60] Kuusela J, Kujala VJ, Kiviaho A, et al. Effects of cardioactive drugs on human induced pluripotent stem cell derived long QT syndrome cardiomyocytes. Springerplus. 2016;5:234.[61] Robinson JF, Gormley MJ, Fisher SJ, et al. A genomics-based framework for identifying biomarkers of human neurodevelopmental toxicity. Reprod Toxicol. 2016;60:1-10.[62] Sengupta S, Johnson BP, Swanson SA, et al. Aggregate culture of human embryonic stem cell-derived hepatocytes in suspension are an improved in vitro model for drug metabolism and toxicity testing. Toxicol Sci. 2014;140(1):236-245.[63] Hossini AM, Megges M, Prigione A, et al. Induced pluripotent stem cell-derived neuronal cells from a sporadic Alzheimer's disease donor as a model for investigating AD-associated gene regulatory networks. BMC Genomics. 2015;16:84.[64] Liu Q, Waltz S, Woodruff G, et al. Effect of potent γ-secretase modulator in human neurons derived from multiple presenilin 1-induced pluripotent stem cell mutant carriers. JAMA Neurol. 2014;71(12):1481-1489.[65] Yagi T, Ito D, Okada Y, et al. Modeling familial Alzheimer's disease with induced pluripotent stem cells. Hum Mol Genet. 2011;20(23):4530-4539.[66] Israel MA, Yuan SH, Bardy C, et al. Probing sporadic and familial Alzheimer's disease using induced pluripotent stem cells. Nature. 2012;482(7384):216-220.[67] Kondo T, Asai M, Tsukita K, et al. Modeling Alzheimer's disease with iPSCs reveals stress phenotypes associated with intracellular Aβ and differential drug responsiveness. Cell Stem Cell. 2013;12(4):487-496.[68] Ren Y, Jiang H, Hu Z, et al. Parkin mutations reduce the complexity of neuronal processes in iPSC-derived human neurons. Stem Cells. 2015;33(1):68-78.[69] Cooper O, Seo H, Andrabi S, et al. Pharmacological rescue of mitochondrial deficits in iPSC-derived neural cells from patients with familial Parkinson's disease. Sci Transl Med. 2012;4(141): 979-980.[70] Ryan SD, Dolatabadi N, Chan SF, et al. Isogenic human iPSC Parkinson's model shows nitrosative stress-induced dysfunction in MEF2-PGC1α transcription. Cell. 2013;155(6):1351-1364.[71] Chung CY, Khurana V, Auluck PK, et al. Identification and rescue of α-synuclein toxicity in Parkinson patient-derived neurons. Science. 2013;342(6161):983-987.[72] Burkhardt MF, Martinez FJ, Wright S, et al. A cellular model for sporadic ALS using patient-derived induced pluripotent stem cells. Mol Cell Neurosci. 2013;56:355-364.[73] Barmada SJ, Serio A, Arjun A, et al. Autophagy induction enhances TDP43 turnover and survival in neuronal ALS models. Nat Chem Biol. 2014;10(8):677-685.[74] Avior Y, Sagi I, Benvenisty N. Pluripotent stem cells in disease modelling and drug discovery. Nat Rev Mol Cell Biol. 2016; 17(3):170-182.[75] Yoshida M, Kitaoka S, Egawa N, et al. Modeling the early phenotype at the neuromuscular junction of spinal muscular atrophy using patient-derived iPSCs. Stem Cell Reports. 2015;4(4):561-568.[76] Sareen D, Ebert AD, Heins BM, et al. Inhibition of apoptosis blocks human motor neuron cell death in a stem cell model of spinal muscular atrophy. PLoS One. 2012;7(6):e39113.[77] Ng SY, Soh BS, Rodriguez-Muela N, et al. Genome-wide RNA-Seq of Human Motor Neurons Implicates Selective ER Stress Activation in Spinal Muscular Atrophy. Cell Stem Cell. 2015;17(5):569-584.[78] Malan D, Zhang M, Stallmeyer B, et al. Human iPS cell model of type 3 long QT syndrome recapitulates drug-based phenotype correction. Basic Res Cardiol. 2016;111(2):14.[79] Lan F, Lee AS, Liang P, et al. Abnormal calcium handling properties underlie familial hypertrophic cardiomyopathy pathology in patient-specific induced pluripotent stem cells. Cell Stem Cell. 2013;12(1):101-113.[80] Tanaka A, Yuasa S, Mearini G, et al. Endothelin-1 induces myofibrillar disarray and contractile vector variability in hypertrophic cardiomyopathy-induced pluripotent stem cell-derived cardiomyocytes. J Am Heart Assoc. 2014;3(6): e001263.[81] Wyles SP, Hrstka SC, Reyes S, et al. Pharmacological Modulation of Calcium Homeostasis in Familial Dilated Cardiomyopathy: An In Vitro Analysis From an RBM20 Patient-Derived iPSC Model. Clin Transl Sci. 2016;9(3):158-167. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||