Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (8): 1270-1276.doi: 10.3969/j.issn.2095-4344.2014.08.020
Previous Articles Next Articles
Application situation of degradable coronary stents
Li Lu-feng, Liu Huan-yun, Zhao Xiao-hui
- Department of Cardiology, Xinqiao Hospital, Third Military Medical University, PLA Institute of Cardiovascular Disease, Chongqing 400037, China
-
Received:2013-12-12Online:2014-02-19Published:2014-02-19 -
Contact:Zhao Xiao-hui, M.D., Master’s supervisor, Department of Cardiology, Xinqiao Hospital, Third Military Medical University, PLA Institute of Cardiovascular Disease, Chongqing 400037, China -
About author:Li Lu-feng, Studying for master’s degree, Department of Cardiology, Xinqiao Hospital, Third Military Medical University, PLA Institute of Cardiovascular Disease, Chongqing 400037, China
CLC Number:
Cite this article
Li Lu-feng, Liu Huan-yun, Zhao Xiao-hui. Application situation of degradable coronary stents[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(8): 1270-1276.
share this article
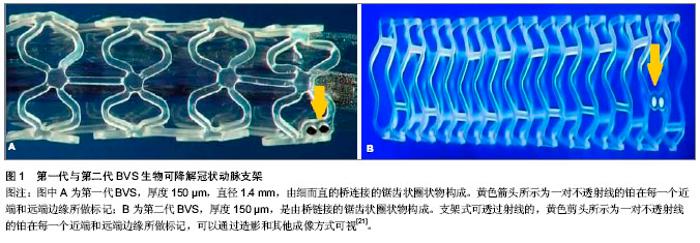
聚羟基乙酸、聚乳酸、聚对二氧环己酮、聚己内酯及聚三亚甲基碳酸酯。其中的聚乳酸有右旋和左旋之分。因只有左旋聚乳酸能被人体降解吸收,故一般选用左旋聚乳酸。在这些可降解材料中,综合考虑机械性能、生物相容性及可降解性,左旋聚乳酸,聚羟基乙酸尤其是它们的共聚物PLGA较适合作为支架材料。聚乳酸结晶度较高,硬度较大,生物降解需时较长,聚羟基乙酸则结晶度较低,硬度较小,生物降解需时较短,将这两种材料按照不同比例共聚得到的不同的PLGA将会具有不同降解速度,从而实现对完全降解时间的调控。Lincoff 等[8]通过实验证实高分子左旋聚乳酸支架(相对分子质量321 000)相比于低分子左旋聚乳酸支架与血管壁的生物相容性更好。Yamawaki等[9]第一次研制出一种由左旋聚乳酸和酪氨酸及酶抑制剂构成的完全的生物可降解支架。通过在猪模型上做实验说明了这个支架的有效性和安全性并支持应用于人。 2.1.2 Igaki-Tamai(IT)支架 IT 支架是第一种在人体内进行临床评估的完全可降解生物支架,由左旋聚乳酸组成,完全降解需要18-24个月。第一代IT支架采用螺旋曲折设计,该支架被安置在一个标准的血管成形的球囊上,随着球囊的扩张进行自我热膨胀。自1998至2012年,在大量动物实验证明左旋聚乳酸支架具有良好的生物相容性及支撑性能后[3],人们对IT支架开展了一系列临床研究[10],FIM(First-in-man)研究表明:IT支架(15例患者,19处病变,25个支架)在30 d内没有发生主要心血管不良事件或支架血栓形成事件,并且与裸金属支架相比,损耗指数为0.48(后期亏损/急性增益),这是生物可降解支架第一次没有引起内膜增生过多。目前Nishio等[11-12]报道了一个前瞻性观察研究对的长期随访结果(>10年),有50例患者(63处病变)共植入84枚IT支架,10年全因死亡、心源性死亡、主要心血管不良事件的累积存活率分别是87%、98%和90%;靶病变血运重建率分别为第1年16%,第5年18%,第10年28%。尽管短期和长期随访结果很不错,但IT支架没能继续取得研究进展,主要是由于植入需要的引导管太大(8F)及需要使用热诱导促使自我膨胀,这可能会引起动脉壁坏死进而导致内膜过度增生或者增加血小板黏附,故难以将其用于临床应用。新一代IT支架整逐渐克服这些缺点,可以使用6F的引导管且不需要再膨胀过程中加热。目前此支架正在进行预临床评估。 2.1.3 BVS支架 BVS支架由美国的雅培公司开发,是一种以左旋聚乳酸为骨架,右旋聚乳酸为涂层,依维莫司为抗增殖药物的生物可降解支架。 在猪模型上进行组织学为基础的研究表明:支架植入后3年被完全吸收[13]。第一代BVS支架(图1A)直径 1.4 mm,厚度150 µm,由细而直的桥连接的锯齿状圈状物构成,进行了ABSORB cohort A试验[14]。该试验的目的是为了评估在单支冠状动脉病变患者置入这种支架的可行性与安全性,选择了30例有稳定或不稳定的冠状动脉缺血患者,主要心血管不良事件的终点是心源性死亡、心肌梗死、缺血性靶病变血运重建。4年跟踪随访显示主要不良心血管事件发生率为3.3%,仅有1例患者发生没有病理性Q波的心肌梗死[15]。造影显示术后6个月和12个月的支架内管腔丢失率没有显著差异。血管内超声显像说明管腔面积的变化是由于支架早期回缩导致内膜增生所致[16]。因为以上的缺陷,BVS支架经过重新设计,新一代BVS拥有更多的支柱能对血管壁提供"

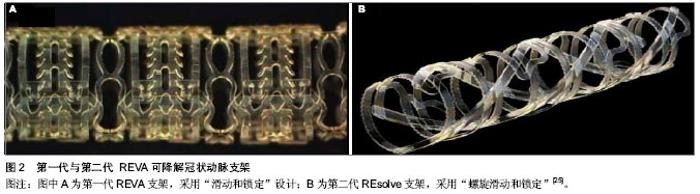
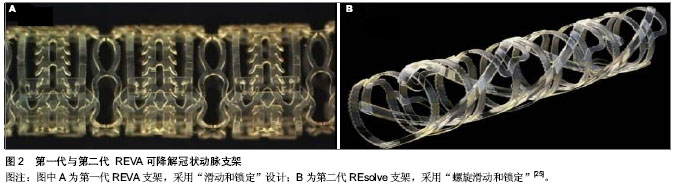
更有力的支撑[17]。第二代BVS支架(图1B)通过ABSORB cohort B试验进行测试。 ABSORB cohort B试验选择101例患者分别植入3.0 mm×18 mm 可吸收BVS支架,分为B1组(n=45),B2组(n=56)[18-19]。B1组在半年和第2年,B2组在第1年和第3年行血管内造影及血管内超声等侵入性检查,两组在随访第18个月是均性断层冠脉造影。发现与第一代BVS支架相比,晚期支架回缩基本消失。6个月时的晚期管腔丢失为(0.19±0.18) mm,3年后随访管腔丢失为(0.27±0.32) mm,与Xience V依维莫司洗脱支架晚期丢失(0.23±0.29) mm相似。除了ABSORB cohort B试验,还有ABSORB physiology、ABSORB 2、ABSORB EXTEND三个试验正在进行。Muramatsu等[20]进行了有关 BVS 支架植入后分支血管阻塞的ABSORB-EXTEND 试验。结果显示BVS比依维莫司洗脱支架能提供更持久的血管支持及将药物输送到血管内皮,没有晚期支架内血栓和未来很难进行外科手术恢复血运重建等问题。该试验还表明:与依维莫司洗脱金属支架相比,BVS有更高的小分支闭塞发生率。随着支架厚度的增加,小分支闭塞的发生率还会进一步增加,此问题有待通过改进支架工艺解决。 2.1.4 REVA支架 是由美国 REVA医药公司研发的多聚(碘化酪氨酸烷基)碳酸酯支架,降解产物有水、二氧化碳、乙醇及碘化酪氨酸烷基,最后均可被人体吸收或排出体外,完全降解需要36个月。第一代REVA支架(图2A)未负载抗增殖药物,采用独特的“滑动和锁定”设计,能在支架置入后提供不错的径向支撑力[22]。第一代REVA支架的长期试验(55个月)在健康猪模型上进行,试验指出:一系列血管超声表明随访期间管腔面积逐渐增大[23]。REVA支架的临床表现通过RESORB研究评价。血管造影显示25例有单支冠脉病变的患者植入此支架后最小管腔直径由(0.88±0.39) mm增加到(2.76± 0.36) mm,与目前金属支架的急性增益相似。然而在随访的4-6个月之间靶病变血运重建率增大,为了解决这一问题,科学家对支架结构进行了重新设计[24]。改进后的第二代ReZolve支架(图2B,抗增殖药物为西罗莫司),药物完全洗脱需要超过30 d,支架完全降解需要1年,通过提高聚合物强度及改进的“螺旋的滑动和锁定”设计进一步提高径向支撑力,以减少支架弹性回缩。REsolve 西罗莫司洗脱可降解冠状动脉支架正在进行RESORB试验,计划选择50例患者调查它的有效性和安全性。目前第二代ReZolve支架已经有了更高剂量的西罗莫司及更慢的药物释放系统,并且支架制作工艺得到了改进,在保证机械性能的前提下减小支架横截面积及厚度,大规模的临床试验即将展开。 2.1.5 IDEAL支架 IDEAL支架是由美国生物吸收治疗公司研发的可完全生物吸收的球囊扩张支架,骨干是聚酐酯和水杨酸,雷帕霉素是涂层药物。预临床试验表明水杨酸有潜在的抗炎和抗血小板的特性,在聚合物洗脱时可以减少狭窄,促进血管恢复正常的生理功能[26-27]。有研究对第一代BTI支架进行了“WHISPER”研究,11例患者参与了这一研究[28]。血管内超声提示由于药物携带量不足和药物释放过快导致了内膜形成增加。故第二代IDEAL支架优化了支架结构,增加携带药物剂量,减慢药物释放,目前新一代的IDEAL支架正在进行预临床试验,不久的将来即将开展临床试验。 2.1.6 DESolve支架 DESolve支架以左旋聚乳酸为骨架,聚乳酸为聚合物涂层,Myolimus和Novolimus 为抗增殖药物。这种支架的径向支撑力可以与Elixir的裸金属支架相比,完全降解吸收需要二三年[29]。为了证明其安全性和有效性,Verheye[30]在16例单支病变长度小于1 cm的冠状动脉病变患者血管内植入直径3 mm的DEsolve支架,1例冠脉旁路移植患者在30 d随访时发生了靶血管血运重建,1例患者在30-180 d的随访时间内发生了靶病变血运重建,而在6个月随访期间没有心源性死亡、心肌梗死、支架内血栓的发生。在总结以上数据后,DEsolve NX试验展开。这个试验计划选取120例患者,随访6个月,终点事件为晚期支架内病变消失。DEsolve NX 2试验计划纳入更多的患者来证实该支架"
| [1] Schomig A,Kastrati A,Dirschinger J,et al.Coronary stenting plus platelet glycoprotein IIb/IIIa blockade compared with tissue plasminogen activator in acute myocardial infarction. Stent versus Thrombolysis for Occluded Coronary Arteries in Patients with Acute Myocardial Infarction Study Investigators. N Engl J Med. 2000;343(6):385-391.[2] Stone GW,Grines CL,Cox DA,et al.Comparison of angioplasty with stenting, with or without abciximab, in acute myocardial infarction.N Engl J Med.2002;346(13):957-966.[3] Tamai H,Igaki K,Tsuji T,et al.A biodegradable poly-l-lacticacid coronary stent in the porcine coronary artery.J Interv Cardiol. 1999;12(6):443-449.[4] Daemen J,Wenaweser P,Tsuchida K,et al.Early and late coronary stent thrombosis of sirolimus-eluting and paclitaxel-eluting stents in routine clinical practice: data from a large two-institutional cohort study.Lancet.2007;369(9562): 667-678.[5] Kuchulakanti PK,Chu WW,Torguson R,et al.Correlates and long-term outcomes of angiographically proven stent thrombosis with sirolimus- and paclitaxel-eluting stents. Circulation.2006;113(8):1108-1113.[6] Cook S,Meier B.Have we been misled by the ESC DES firestorm? EuroIntervention. 2008;3(5):535-537.[7] Waksman R.Lessons learnt from preclinical studies of magnesium scaffolds (Biotronik’s DREAMS program). EuroPCR; Rotterdam, The Netherlands.2012.[8] Lincoff AM,Furst JG,Ellis SG,et al.Sustained local delivery of dexamethasone by a novel intravascular eluting stent to prevent restenosis in the porcine coronary injury model.J Am Coll Cardiol.1997;29(4):808-816.[9] Yamawaki T,Shimokawa H,Kozai T,et al.Intramural delivery of a specific tyrosine kinase inhibitor with biodegradable stent suppresses the restenotic changes of the coronary artery in pigs in vivo.J Am Coll Cardiol.1998;32(3):780-786.[10] Tamai H,Igaki K,Kyo E,et al.Initial and 6-month results of biodegradable poly-l-lactic acid coronary stents in humans.Circulation.2000;102(4):399-404.[11] Nishio S,Kosuga K,Igaki K,et al.Long-Term ( > 10 Years) Clinical outcomes of first-in-human biodegradable poly-l-lactic acid coronary stents igaki-tamai stents.Circulation. 2012; 125(19):2343-2352.[12] Onuma Y,Garg S,Okamura T,et al.Ten-year follow-up of the IGAKI-TAMAI stent. A posthumous tribute to the scientific work of Dr Hideo Tamai. Euro Intervention. 2009;5 Suppl F:F109-F111.[13] Onuma Y,Serruys PW,Perkins LE,et al.Intracoronary optical coherence tomography and histology at 1 month and 2, 3, and 4 years after implantation of everolimus-eluting bioresorbable vascular scaffolds in a porcine coronary artery model: an attempt to decipher the human optical coherence tomography images in the ABSORB trial. Circulation. 2010; 122(22):2288-2300.[14] Ormiston JA,Serruys PW,Regar E,et al.A bioabsorbable everolimus-eluting coronary stent system for patients with single de-novo coronary artery lesions (ABSORB): a prospective open-label trial.Lancet.2008;371:899-907.[15] Serruys PW,Ormiston JA,Onuma Y,et al.A bioabsorbable everolimus-eluting coronary stent system (ABSORB): 2-year outcomes and results from multiple imaging methods. Lancet. 2009;373:897-910.[16] Tanimoto S,Bruining N,van Domburg RT,et al.Late stent recoil of the bioabsorbable everolimus-eluting coronary stent and its relationship with plaque morphology.J Am Coll Cardiol. 2008; 52(20):1616-1620.[17] Tanimoto S,Serruys PW,Thuesen L,et al.Comparison of in vivo acute stent recoil between the bioabsorbable everolimus-eluting coronary stent and the everolimus-eluting cobalt chromium coronary stent: insights from the ABSORB and SPIRIT trials.Catheter Cardiovasc Interv.2007;70(4): 515-523.[18] Diletti R,Onuma Y,Farooq V,et al.6-month clinical outcomes following implantation of the bioresorbable everolimus-eluting vascular scaffold in vessels smaller or larger than 2.5 mm.J Am Coll Cardiol.2011;58(3):258-264.[19] Serruys PW,Onuma Y,Dudek D,et al.Evaluation of the second generation of a bioresorbable everolimus-eluting vascular scaffold for the treatment of de novo coronary artery stenosis: 12-month clinical and imaging outcomes. J Am Coll Cardiol. 2011;58(15):1578-1588.[20] Muramatsu T,Hector M,Christos V,et al.Incidence and Short-Term Clinical Outcomes of Small Side Branch Occlusion After Implantation of an Everolimus-Eluting Bioresorbable Vascular Scaffold.J Am Coll Cardiol.2013; 6(3):247-257.[21] Gogas BD,Farooq V,Onuma Y,et al.The ABSORB Bioresorbable Vascular Scaffold:An Evolution or Revolution in Interventional Cardiology?Hellenic J Cardiol.2012;53(4): 301-309.[22] Pollman MJ.Engineering a bioresorbable stent: REVA programme update. EuroIntervention.2009;5 Suppl F: F54-57.[23] Strandberg E,Zeltinger J,Schulz DG,et al.Late positive remodeling and late lumen gain contribute to vascular restoration by a non-drug eluting bioresorbable scaffold: a four-year intravascular ultrasound study in normal porcine coronary arteries. Circ Cardiovasc Interv.2012; 5(1):39-46.[24] Grube E. The Boston scientific and REVA technology. Euro PCR;Barcelona, Spain.2009.[25] Garg S,Serruys PW.Coronary stents: looking forward.J Am Coll Cardiol. 2010;56(10 Suppl):S43-78.[26] Jabara R,Chronos N,Robinson K.Novel bioabsorbable salicylate-based polymer as a drug-eluting stent coating. Catheter Cardiovasc Interv.2008;72(2):186-194.[27] Jabara R,Pendyala L,Geva S,et al.Novel fully bioabsorbable salicylate-based sirolimus-eluting stent.EuroIntervention. 2009; 5 Suppl F:F58-F64.[28] Jabara R.Poly-anhydride based on salicylic acid and adipic acid anhydride. EuroPCR;Barcelona, Spain.2009. [29] Yan J, Bhat VD. Elixir Medical’s bioresorbable drug eluting stent (BDES) programme: an overview.EuroIntervention.2009;5 Suppl F:F80-F82.[30] Verheye S. DESolve first in man study-preliminary results. EuroPCR; Rotterdam, The Netherlands.2012.[31] Verheye S. DESolve myolimus eluting bioresorbable coronary scaffold first in man trial- imaging and clinical results at 6 months.EuroPCR;Rotterdam,The Netherlands. 2012.[32] 沈雳,齐兵,吴轶喆,等.完全可降解聚乳酸西罗莫司洗脱支架在小型猪冠状动脉模型中的实验研究[J].中国介入心脏病学杂志, 2011,19(6):301-305. [33] Ghimire G,Spiro J,Kharbanda R,et al.Initial evidence for the return of coronary vasoreactivity following the absorption of bioabsorbable magnesium alloy coronary stents. EuroIntervention. 2009;4(4):481-484.[34] Waksman R. PROGRESS was made toward DREAM: magnesium Stent. EuroPCR; Rotterdam, The Netherlands. 2012.[35] Peuster M,Hesse C,Schloo T,et al.Long-term biocompatibility of a corrodible peripheral iron stent in the porcine descending aorta.Biomaterials.2006;27:4955-4962.[36] Waksman R,Pakala R,Baffour R,et al. Short-term effects of biocorrodible iron stents in porcine coronary arteries.J Interv Cardiol.2008;21:15-20.[37] 赵晓辉,尹扬光,方玉强,等.雌激素动员小鼠外周血内皮祖细胞修复损伤血管内皮[J]. 第三军医大学学报,2010,33(1): 94-97. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [3] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [4] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [5] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [6] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [7] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [8] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [9] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [10] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| [11] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [12] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [13] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [14] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [15] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||