Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (12): 2091-2099.doi: 10.3969/j.issn.2095-4344.2013.12.001
Self-made Fe3O4 versus gelatin sponge embolic agents in rabbit pulmonary microcirculation
Yang Qing, Tong Yu-yun, Guo Li, Wang Jia-ping, Li Ying-chun, Jiang Hua
- Department of Interventional Radiology, Second Hospital of Kunming Medical University, Kunming 650101, Yunnan Province, China
-
Received:2012-12-12Revised:2013-02-05Online:2013-03-19Published:2013-03-19 -
Contact:Tong Yu-yun, Master, Attending physician, Department of Interventional Radiology, Second Hospital of Kunming Medical University, Kunming 650101, Yunnan Province, China 1012580@qq.com -
About author:Yang Qing☆, Doctor, Attending physician, Department of Interventional Radiology, Second Hospital of Kunming Medical University, Kunming 650101, Yunnan Province, China yq7119@hotmail.com
CLC Number:
Cite this article
Yang Qing, Tong Yu-yun, Guo Li, Wang Jia-ping, Li Ying-chun, Jiang Hua. Self-made Fe3O4 versus gelatin sponge embolic agents in rabbit pulmonary microcirculation[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(12): 2091-2099.
share this article
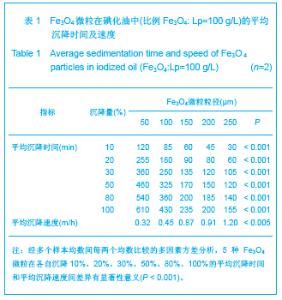
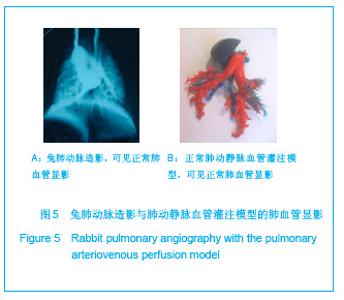
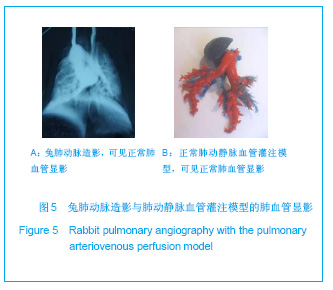
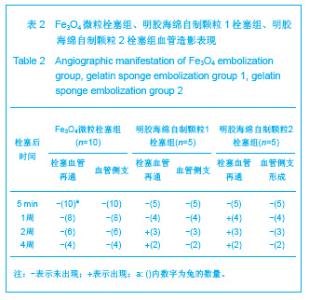
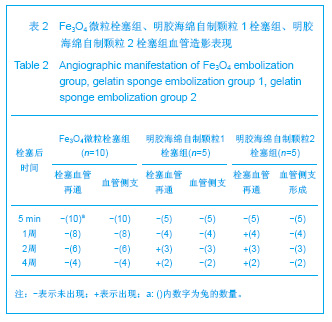
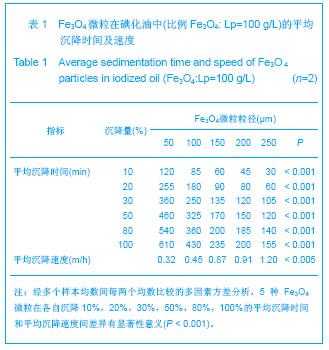
2.1 实验动物数量分析 实验共用兔21只,其中1只行肺血管铸型,实验过程中死亡12只,死亡兔经解剖排除与异位栓塞相关,死因与麻醉过量、感染相关,及时补充实验兔达实验组规定数目,栓塞成功,最终计入结果分析兔共20只。 2.2 Fe3O4微粒体外形态 Fe3O4微粒加入碘化油中,初期微粒聚集呈大小不等团块沉于碘化油底部。充分振荡后,磁微粒团块离散,微粒呈均匀、分散状悬浮于碘化油中,然后缓慢沉降。用注射器推注可顺利通过直径为3-6F的动脉导管。5种规格及不同比例的Fe3O4微粒在碘化油中的悬浮情况见表1。5种Fe3O4微粒在各自沉降10%,20%,30%,50%,80%,100%的平均沉降时间和平均沉降速度间差异有显著性意义(P < 0.001)。"
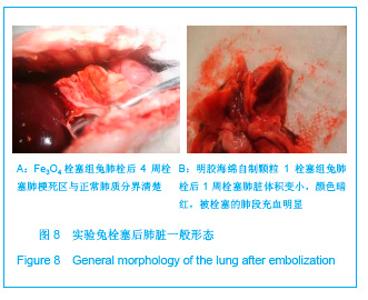
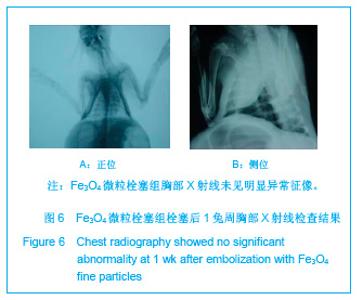
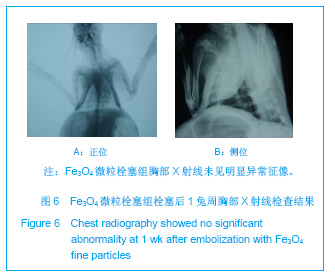
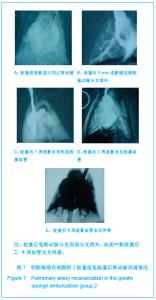
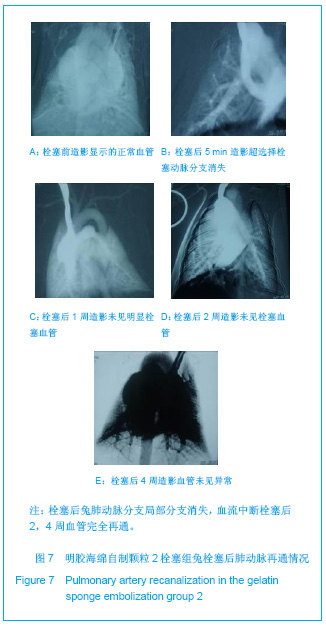
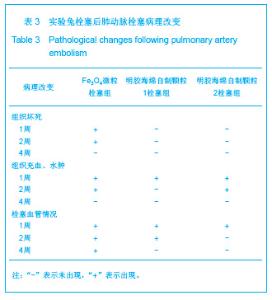
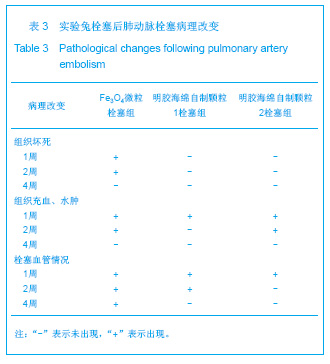
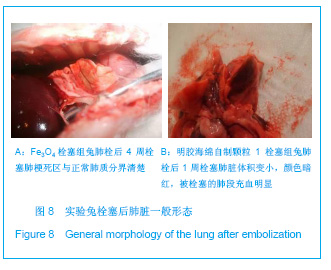
2.5 实验兔栓塞后1,2,4周肺组织病理分析结果 大体观察结果:Fe3O4微粒栓塞组兔肺栓塞后1周(2只),栓塞肺脏体积变小,颜色暗红,被栓塞的肺段充血明显。2周(2只)、4周(6只),栓塞肺脏,表面有散在出血点,质地变韧,实质出现梗死区域,梗死区与正常肺质分界清楚。明胶海绵自制颗粒1栓塞组兔栓塞肺动脉后1周(1只) ,栓塞肺体积稍增大,颜色深红,2周时(1只),栓塞肺体积肺恢复正常,未见有明显梗死区域。4周时(2只),仍未见有明显梗死改变。明胶海绵自制颗粒2栓塞组兔栓塞肺动脉后1周(1只)栓塞肺体积稍增大,颜色正常,2周时(1只),栓塞肺体积肺恢复正常,未见有明显梗死区域。4周时(2只),仍未见有明显梗死改变。见图8。"
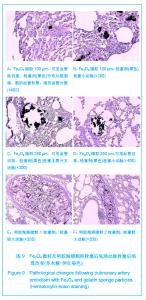
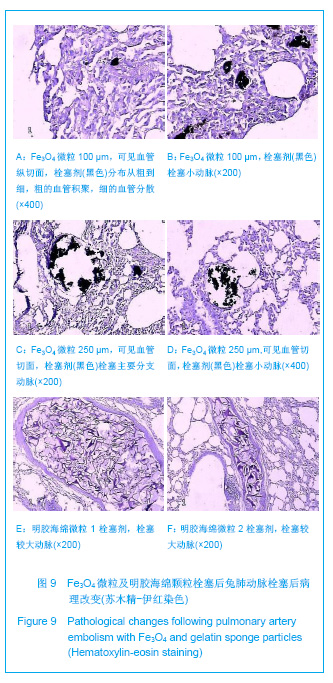
Fe3O4微粒栓塞组兔肺栓塞后1,2,4周,苏木精-伊红染色示50-250 μm的Fe3O4微粒:黑色Fe3O4微粒栓塞肺细小血管内,在较大血管中颗粒互相集聚成团,较小血管中偶可见单颗粒,各组颗粒栓塞血管程度不同,50 μm组栓塞可见少量位于肺泡周毛细血管及毛细血管前小动脉,多数集聚于小动脉内,栓塞彻底。100 μm粒径很少栓塞毛细血管,常五六个视野内未见栓塞毛细血管,毛细血管前小动脉见栓塞,多数集聚于5级分支小动脉内,150-250 μm栓塞范围在小动脉四五级小动脉内。明胶海绵自制颗粒1栓塞组兔栓塞肺动脉后1周(1只),可见3级大动脉及4级小动脉内栓塞剂栓塞,远端小动脉未见栓塞。明胶海绵自制颗粒2栓塞组基本与明胶海绵自制颗粒1栓塞组相同。Fe3O4微粒栓塞组可见局部组织坏死,其他两组未见异常。见图9。"
| [1] Bilbao JI, Martínez-Cuesta A, Urtasun F. Complications of embolization. Semin Intervent Radiol. 2006;23(2):126-142. [2] Walsh KP. Advanced embolization techniques. Pediatr Cardiol. 2005;26(3):275-288.[3] Al-Shahi R, Warlow C. Arteriovenous malformations of the brain: ready to randomisel. J Neurol Neurosurg Psychiatry. 2005;76(10):1327-1329[4] White RI Jr. Pulmonary arteriovenous malformations: how do I embolize? Tech Vasc Interv Radiol. 2007;10(4):283-290. [5] Kjeldsen AD, Andersen PE, Oxhøj H, et al. Percutaneous transluminal embolization of pulmonary arteriovenous malformations. Ugeskr Laeger. 1998;160(10):1465-1469.[6] Vaidya S, Kathleen R. An overview of embolic agents. Semin Intervent Radiol. 2008;25(3):204-215. [7] Becker TA, Preul MC, Bichard WD, et al. Calcium alginate gel as a biocompatible material for endovascular arteriovenous malformation embolization: six-month results in an animal model. Neurosurgery. 2005;56:793-801. [8] Becker TA, Preul MC, Bichard WD, et al. Preliminary investigation of calcium alginate gel as a biocompatible material for endovascular aneurysm embolization in vivo. Neurosurgery. 2007;60:1119-1128. [9] Vanninen R L, Manninen I. Onyx, a new liquid embolic material for peripheral interventions: preliminary experience in aneurysm, pseudoaneurysm, and pulmonary arteriovenous malformation embolization. Cardiovasc Intervent Radiol. 2007; 30(2):196-200. [10] Raffi L, Simonetti L, Cenni P, et al. Use of Glubran 2 acrylic glue in interventional neuroradiology. Neuroradiology. 2007; 49:829-836. [11] Pollak JS, White RI. The use of cyanoacrylate adhesives in peripheral embolization. J Vasc Interv Radiol. 2001;12: 907-913. [12] Osuga K, Mikami K, Higashihara H, et al. Principles and techniques of transcatheter embolotherapy for peripheral vascular lesions. Radiat Med. 2006;24(4):309-314. [13] Spies J B, Cornell C, Worthington-Kirsch R, et al. Long-term outcome from uterine fibroid embolization with tris-acryl gelatin microspheres: results of a multicenter study. J Vasc Interv Radiol. 2007;18(2):203-207. [14] Becker TA, Kipke DR, Brandon T. Calcium alginate gel: a biocompatible and mechanically stable polymer for endovascular embolization. J Biomed Mater Res. 2001; 54(1):76-86. [15] López-Benítez R, Richter GM, Kauczor HU, et al. Analysis of nontarget embolization mechanisms during embolization and chemoembolization procedures. Cardiovasc Intervent Radiol. 2009;32(4):615-622. [16] Hidaka K, Nakamura M, Osuga K, et al. Elastic characteristics of microspherical embolic agents used for vascular interventional radiology. 2010;3(7):497-503.[17] Hidaka K, Moine L, Collin G, et al. Elasticity and viscoelasticity of embolization microspheres. J Mech Behav Biomed Mater. 2011;4(8):2161-2167.[18] Yoon W. Embolic agents used for bronchial artery embolisation in massive haemoptysis. Expert Opin Pharmacother. 2004;5(2):361-367.[19] Loffroy R, Guiu B, Cercueil JP, et al. Endovascular therapeutic embolisation: an overview of occluding agents and their effects on embolised tissues. Curr Vasc Pharmacol. 2009;7(2):250-263.[20] Lewis AL, Adams C, Busby W etal.Comparative in vitro evaluation of microspherical embolisation agents. J Mater Sci Mater Med. 2006;17(12):1193-1204.[21] Stampfl S, Bellemann N, Stampfl U, et al. Arterial distribution characteristics of Embozene particles and comparison with other spherical embolic agents in the porcine acute embolization model. J Vasc Interv Radiol. 2009;20(12): 1597-607. [22] Stampfl S, Bellemann N, Stampfl U, et al. Inflammation and recanalization of four different spherical embolization agents in the porcine kidney model. J Vasc Interv Radiol. 2008;19(4): 577-586. [23] Bilbao JI, de Luis E, García de Jalón JA, et al. Comparative study of four different spherical embolic particles in an animal model: a morphologic and histologic evaluation. J Vasc Interv Radiol. 2008;19(11):1625-1638. [24] Abada HT, Golzarian J. Gelatine sponge particles: handling characteristics for endovascular use. Tech Vasc Interv Radiol. 2007;10(4):257-260. [25] White RI Jr, Pollak JS, Wirth JA. Pulmonary arteriovenous malformations: diagnosis and transcatheter embolotherapy. J Vasc Interv Radiol. 1996;7(6):787-804.[26] Gulati MS, Srinivasan A, Paul SB, et al. Uterine restoration following fibroid expulsion after uterine artery embolisation using gelfoam. J Postgrad Med. 2004;50(1):80.[27] Katsumori T, Kasahara T, Akazawa K. Long-term outcomes of uterine artery embolization using gelatin sponge particles alone for symptomatic fibroids. AJR Am J Roentgenol. 2006; 186(3):848-854.[28] The Ministry of Science and Technology of the People’s Republic of China. Guidance Suggestions for the Care and Use of Laboratory Animals. 2006-09-30. |
| [1] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Intravenous, topical tranexamic acid alone or their combination in total knee arthroplasty: a meta-analysis of randomized controlled trials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 948-956. |
| [2] | Li Li, Ma Li. Immobilization of lactase on magnetic chitosan microspheres and its effect on enzymatic properties [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 576-581. |
| [3] | Zhou Anqi, Tang Yufei, Wu Bingfeng, Xiang Lin. Designing of periosteum tissue engineering: combination of generality and individuality [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3551-3557. |
| [4] | Lang Limin, He Sheng, Jiang Zengyu, Hu Yiyi, Zhang Zhixing, Liang Minqian. Application progress of conductive composite materials in the field of tissue engineering treatment of myocardial infarction [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3584-3590. |
| [5] | Xie Jian, Su Jiansheng. Advantages and characteristics of electrospun aligned nanofibers as scaffolds for tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2575-2581. |
| [6] | Ji Qi, Yu Zhengwen, Zhang Jian. Problems and trends of technique and clinical application of metallic biomaterials prepared by three-dimensional printing technology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2597-2604. |
| [7] | Qian Nannan, Zhang Qian, Yang Rui, Ao Jun, Zhang Tao. Mesenchymal stem cells in the treatment of spinal cord injury: cell therapy and combination of new drugs and biomaterials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2114-2120. |
| [8] | Jia Wei, Zhang Mandong, Chen Weiyi, Wang Chenyan, Guo Yuan. Effects of femoral prosthetic materials on artificial knee arthroplasty performance [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1477-1481. |
| [9] | Wang Qian, Li Lu, Shu Jingyuan, Dong Zhiheng, Jin Youshi, Wang Qingshan. Micro-morphology and phase of zirconia-based nano-hydroxyapatite functional gradient biomaterials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1517-1521. |
| [10] | Li Xiao, Pan Jinbing, Ma Yun, Qian Haoyu, Zhang Quncheng, Wang Zheng. Silicone stent insertion for treating tracheobronchomalacia in adults [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 549-554. |
| [11] | Xie Xiufeng, Zhang Yue, Qu Ze. Clinical outcomes of drug-eluting balloons and drug-eluting stents for the treatment of in-stent restenosis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 555-560. |
| [12] | Li Li, Ma Li, Li He. Preparation and characterization of magnetic chitosan microspheres [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 577-582. |
| [13] | Tang Mengmeng, Chen Hechun, Xie Hongchen, Zhang Yu, Tan Xiaoshuang, Sun Yixuan, Huang Yina. Histocompatibility of poly(L-lactide-co-ε-caprolactone)/cross-linked polyvinylpyrrolidone ureteral stent grafted into the rat bladder [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 583-588. |
| [14] | Chen Zegang, Ding Haili, Li Long, Wang Chun. Changes of bone metabolism after different intensity endurance exercises in growing rats [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(35): 5582-5588. |
| [15] | Guo Enhui, Xu Zitong, Liang Yize, Zhou Liang, Lu Zhaoxiang, You Liang, Xia Yujun. Properties of a novel photocrosslinked fish collagen peptide-hyaluronic acid hydrogel [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(28): 4518-4525. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||