Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (27): 4401-4407.doi: 10.12307/2023.629
Previous Articles Next Articles
Endoscopic transforaminal lumbar interbody fusion for degenerative diseases of the lumbar spine: decompression fusion and preservation of posterior spine anatomy
Jin Xinjie, Lu Xiangdong, Zhao Yibo, Zhao Xiaofeng, Qi Detai, Zhao Bin
- Second Hospital of Shanxi Medical University, Taiyuan 030000, Shanxi Province, China
-
Received:2022-03-02Accepted:2022-08-15Online:2023-09-28Published:2022-11-08 -
Contact:Zhao Bin, MD, Chief physician, Second Hospital of Shanxi Medical University, Taiyuan 030000, Shanxi Province, China -
About author:Jin Xinjie, Master candidate, Second Hospital of Shanxi Medical University, Taiyuan 030000, Shanxi Province, China
CLC Number:
Cite this article
Jin Xinjie, Lu Xiangdong, Zhao Yibo, Zhao Xiaofeng, Qi Detai, Zhao Bin. Endoscopic transforaminal lumbar interbody fusion for degenerative diseases of the lumbar spine: decompression fusion and preservation of posterior spine anatomy[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(27): 4401-4407.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
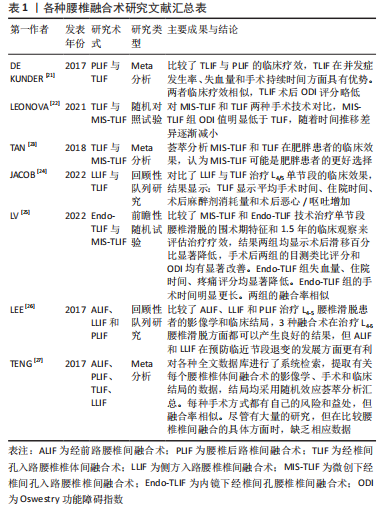
2.1 发展概述 1944年BRIGGS和MILLIGAN [11]首次描述腰椎后路椎间融合术(posterior lumbar interbody fusion,PLIF),随着技术的不断发展,PLIF的疗效不断改善,PLIF作为腰椎最为传统的入路,通过后路对神经的直接减压,最大限度扩大了椎管的有效容积,然而后方肌肉、韧带、棘突等解剖结构的破坏会导致患者长期的腰痛[12]。1982年HARMS和ROLINGER [13]在PLIF的基础上提出经椎间孔入路腰椎椎间融合术(transforaminal lumbar interbody fusion ,TLIF),通过单侧椎间孔途径实现减压和融合器的植入,不侵入中央管,保留了对侧椎板,同时保留了后方棘突和韧带,但仍然对椎旁肌造成损伤。LOWERY和KULKARNIS [14]在2000年使用钛棒改进了经皮椎弓根钉内固定术。2002年,FOLEY等[15]基于TLIF技术提出了微创下经椎间孔入路腰椎椎间融合术(minimally invasive transforaminal lumbar interbody fusion, MIS-TLIF),该技术无需大的正中切口和显著的椎旁肌肉剥离,椎弓根螺钉和预成型棒通过小切口置入,减轻了组织损伤,钉棒系统放置的解剖位置标准化,优化了固定物生物力学。但与传统术式TLIF相比,增加了术者和患者接受X射线的暴露时间,该术式存在陡峭的学习曲线,且初始开展MIS-TLIF需具有熟练的开放手术经验。与此同时,PARVIZ和HARRIS等[16]在1983年率先报道了使用经皮后外侧入路关节镜下腰椎髓核摘除术 (arthroscopic micro discectomy,AMD)治疗腰椎间盘突出症,并在1991年提出“安全三角区”的概念,奠定了经皮脊柱内镜技术的基础。1999年YEUNG[17]研制具有代表性的脊柱内窥镜操作系统(yeung endoscocip spine system,YESS),YESS系统是将工作套管通过安全三角区置入病变的椎间盘内,并不能进入病变部分的椎管内,因此只能实现间接减压。2006年,HOOGLAND等[18]在YESS的基础上研发出TESS系统,该技术包括一套不同直径的椎间孔铰刀,通过逐级铰除部分上关节突前下缘骨质结构,扩大椎间孔,通过椎间孔入路将工作套管直接伸入椎管内,达到直接减压效果。YEUNG和HOOGLAND对经椎间孔入路PELD技术的理论革新、设备器械改进做出了开拓性贡献。CHOI等[19]于2006年提出了经椎板间隙入路椎间盘摘除术。随后,RUNTTEN [20]提出全可视化镜下操作的概念,在镜下完成软组织剥离、椎板关节处理、直视下进入椎管。随着微创理念逐渐为业内人士所认知,脊柱内镜手术逐渐由单一减压模式向减压融合发展,催生了现如今的内镜下经椎间孔腰椎椎间融合术,见表1[21-27]。"

2.2 Endo-TLIF的适应证及禁忌证 2.2.1 Endo-TLIF的适应证 Endo-TLIF是腰椎退行性疾病患者的重要治疗手段。近年来,Endo-TLIF已逐渐应用于腰椎退行性疾病的治疗,患者痛苦小,术后生活质量高,但行Endo-TLIF治疗必须严格明确患者有无手术指征,包括腰椎椎管狭窄、椎间孔狭窄、腰椎峡部裂、Ⅰ、Ⅱ度腰椎滑脱、腰椎不稳、腰椎侧隐窝狭窄、椎间盘突出、术后不稳定或背部手术失败综合征等[28-32]。 2.2.2 Endo-TLIF的禁忌证 Endo-TLIF的禁忌证包括肿瘤、椎体骨折、感染、代谢性疾病、椎管内病变、全身性疾病、Ⅲ、Ⅳ度腰椎滑脱 (滑脱超过50%)、显著骨质疏松症、马尾综合征、先天性脊柱畸形、出血疾病、严重钙化碎片、既往腰椎手术(翻修)、多发不稳。特别需要注意对于经Kambin入路,严重的椎间孔狭窄、严重的中央管狭窄、L5/S1节段伴高髂骨和极度塌陷的椎间盘间隙是相对的禁忌证[30]。 2.3 手术简介 2.3.1 Endo-TLIF的分类 根据使用内镜系统的类型,可分为经皮内窥镜或全内窥镜TLIF、双通道内镜TLIF及显微内镜下TLIF。①经皮内窥镜或全内窥镜TLIF:是临床实践中最常用的内镜下融合技术,该内镜具有的特点是将光学系统和操作器械合并在同一工作管道,经皮穿刺,持续盐水灌洗[30]。手术通路示意图见图3。此种手术可在局麻或硬膜外麻醉下进行,这对不能耐受全麻手术的患者来说有重大意义,但持续的盐水灌洗导致术后患者低体温,且造成的隐性失血量无法计算。②双通道内镜下TLIF:该技术的特点是具有独立的光学系统通道和操作器械通道,类似于关节镜技术,光学系统通道用于持续冲洗和观察手术区域,而器械通道用于手术操作,手术也是在盐水持续冲洗下进行的[30]。③显微内镜下TLIF:显微内窥镜系统的光学设备附在管状牵开器上,最常用的显微内镜系统是METRx系统,内窥镜主要用于减压和融合,整个过程不使用恒定的盐水灌洗,通过肌肉扩张而不是肌肉收缩来减少创伤[30]。然而,由于临床研究证据不足,内镜系统的选择通常由外科医生决定,目前尚无相关指南或规范明确不同适应证下内镜系统的选择,因此仍需设计良好的长期随访和大样本的研究来比较不同内镜手术之间的临床特征。"
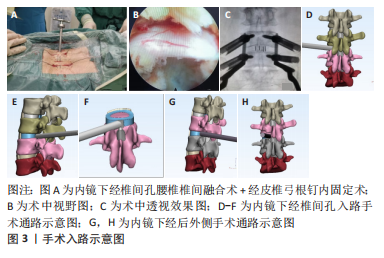
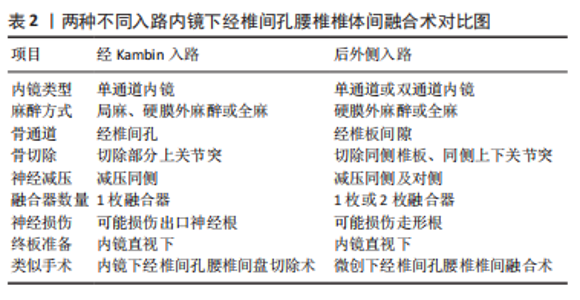
2.3.2 手术入路 内镜下腰椎融合术有两种手术入路:经Kambin入路和后外侧入路[29,33]。经Kambin入路采用Kambin三角,使用单通道内镜,麻醉方式灵活,可在局麻、硬膜外麻醉或全麻下进行,其入路与经椎间孔入路腰椎间盘切除术手术入路相同,切除部分上关节突,对同侧神经减压,然后切除椎间盘,进行终板准备,在内窥镜检查和引导下插入融合器[34-36],这种手术入路通常只能放置1个融合器[7],有损伤术侧出口神经根的可能性[37]。后外侧入路与MIS-TLIF相似[38],可使用单通道内镜或双通道内镜,在硬膜外麻醉或全身麻醉下,经后外侧入路行同侧椎板切开术,同侧下关节突全切除、同侧上关节突全切除或部分切除,对同侧和对侧神经根及椎管进行减压,可放置一到两个融合器,手术操作时可能会损伤走行根[37]。目前尚无研究对两种手术入路的临床结果进行比较。两种手术入路对比见表2,手术入路示意图见图3。"

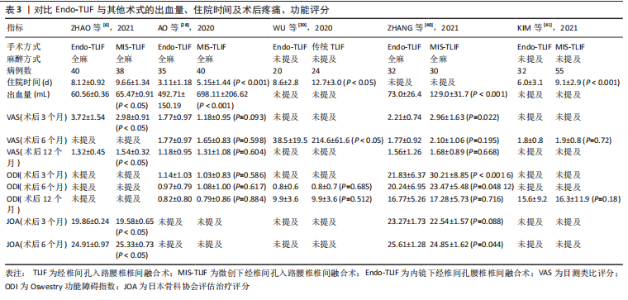
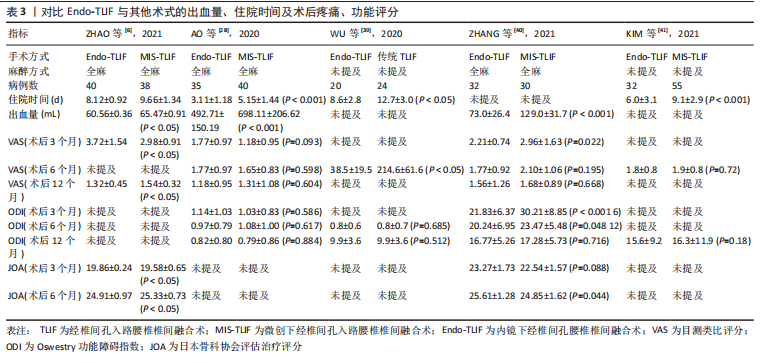
2.4 内镜下腰椎融合术的优势、并发症及局限性 2.4.1 优势 内镜下腰椎融合术在腰椎退行性疾病的临床治疗中取得了良好的临床结果。在腰椎退行性疾病治疗方面,Endo-TLIF具有显著的优势:①保存脊柱后方结构。内窥镜经后外侧或Kambin三角入路,椎管减压,置入融合器,安置经皮椎弓根螺钉,内镜下的直接视觉化,减少了椎旁肌的剥离,保留脊柱后部结构,如棘突、双侧小关节、椎板等,减少了手术创伤及内固定系统的负荷,同时,术中减少了对马尾神经的牵拉[6]。②由于手术破坏小,Endo-TLIF在术中拥有更少的出血量、术后疼痛明显减少、更短的住院时间,见表3[6,28,39-41]。③更好的终板准备。内镜下的直接视觉化,视野更加清晰,软骨终板可在清晰放大的内镜下完全切除而不损伤骨终板[36]。良好终板的准备,能够减少融合器移位及下沉的概率,提高融合率,是促进椎间融合的重要环节[42]。④麻醉方式灵活。Endo-TLIF手术无需全身麻醉即可进行,特别是经皮内镜下TLIF可在局麻或硬膜外麻醉下进行[8],这对不能耐受全麻的患者尤其中老年人及肾损害患者具有重要意义。有文献报道,内镜下TLIF局部麻醉可提供与开放TLIF全身麻醉患者相似的结果[43]。不使用全身麻醉的内镜下TLIF是一种新颖且有前景的短节段腰椎融合术,持续的技术创新可能对短期和长期恢复带来更大的改善。⑤可撑开融合器的应用。与传统融合器相比,撑开式融合器体积小,便于植入,需要切除的骨性结构也较少,可避免过度牵拉神经根[44]。目前临床使用最多的是DePuy Synthes可撑开融合器,是一种最新式的膨胀式钛合金融合器,该融合器增加了终板接触面积、携带更多的骨移植物,支撑强度大且操作简便灵活,最大限度恢复椎间隙正常高度,能够有效促进骨的融合及防止术后融合器沉降[45-46]。⑥氧化应激指标降低。当患者机体处于应激状态时,中枢神经系统发出信号并传导至下丘脑,激活交感肾上腺髓质,促进儿茶酚胺大量分泌,进而促进糖皮质激素的合成和释放,引发外周效应,最终导致血糖、皮质醇水平上升[47]。姚楚亮等[47]使用内镜下腰椎融合治疗腰椎退行性疾病的研究结果显示,术前血糖平均值为(4.15±0.21) mmol/L,术后12,24 h的血糖平均值为(4.98±0.24),(5.26±0.19) mmol/L;术前皮质醇平均值为85.89±11.98,术后12,24 h皮质醇平均值为92.81±12.38,115.68±11.47,两者差异均有显著性意义(P < 0.05)。 2.4.2 并发症 Endo-TLIF虽然有上述诸多优势,但也会出现一些并发症。①经Kambin入路的Endo-TLIF由于椎间盘切除、终板准备、插入融合器是通过Kambin三角操作,因此易导致出口神经根的损伤,切除部分上关节突以扩展Kambin三角、可撑开融合器的使用及术中神经监护有助于减少出口神经根的损伤[48]。②由于经后外侧入路Endo-TLIF的操作与MIS-TLIF中管状牵开系统的操作相似,手术并发症也相似,如硬脊膜撕裂、术后血肿、短暂性神经麻痹、感染、股四头肌肌力下降等,这些并发症比较轻微,不需要手术翻修,可以通过保守治疗自行缓解[34]。HEO等[34]研究比较了Endo-TLIF与MIS-TLIF的并发症,结果显示两者并发症差异无统计学意义。另一项研究对 Endo-TLIF与PLIF的并发症进行比较,结果显示两者并发症发生率无显著差异(P > 0.05)。在王放等[49]的报道中,使用内镜下融合技术治疗31例退行性腰椎疾病患者,1例患者在麻醉苏醒阶段突然出现单侧下肢肌力下降,术后CT检查示椎弓根钉位置良好,稳固无移位,手术切开探查见术区血肿压迫,清理血肿并放置引流,术后给予激素冲击、脱水及营养神经治疗后症状逐渐缓解。另在任伟剑等[50]的研究中,1例腰椎滑脱的患者在接受脊柱内镜下融合手术治疗后出现低体温、麻醉后苏醒困难,给予复温等对症治疗后恢复良好。综上所述,内镜下腰椎间融合术的并发症可概括为与手术入路相关的并发症及其他并发症,大多数并发症轻微且经保守治疗可得到改善,加强脊柱外科医师对脊柱结构尤其椎间孔解剖结构的掌握、发展先进手术器械可有效避免一些并发症的发生。"

2.4.3 局限性 Endo-TLIF具有出血量少、手术破坏小、恢复时间短等优点,但仍然存在一定的局限性,具体如下: (1)辐射量高。Endo-TLIF术中需反复透视以确定工作导管、椎弓根螺钉及融合器的位置,因此患者和医生将暴露于较高的辐射剂量[40]。 (2)学习曲线陡峭。在Endo-TLIF手术过程中,外科医生需深入掌握椎间孔解剖结构,并且有丰富的脊柱内镜减压手术经验[9,40],这对于年轻的脊柱外科医生来说是一个巨大的挑战,可能会延长手术时间,增加术中辐射量,增加术中出血量和神经损伤的风险[51]。CHOI等[52]分析了一位有8年脊柱外科内镜手术经验的术者开展的68例脊柱内镜手术的学习曲线,研究中该术者的手术学习曲线在36例之后手术时间趋于稳定,术中视野的清晰度和对手术出血点的控制可能是影响手术时间的关键原因,术野的清晰度会显著耗费创建操作空间和寻找手术标志的时间。因此,建议术者在熟练掌握经皮椎间孔入路椎间盘切除术或经皮椎板间入路椎间盘切除术基础上并达到平台期后再进行Endo-TLIF手术,避免手术时间过长对患者产生不良影响及不必要的医患纠纷。 (3)适应证局限。尽管Endo-TLIF有诸多优点,但其仍是一项具有挑战性和技术复杂的手术,适应证有限[29],对于椎间盘间隙狭窄或者Kambin三角非常狭窄的患者,可能很难为安全置入融合器做好充足的椎间盘准备,从而导致融合失败或者延迟融合、下沉、移位或出口神经根的损伤[30]。 (4) Endo-TLIF在内镜直视下完成终板的处理,视野清晰,安全可靠,终板处理更加精准彻底,同时可以避免终板的损伤,防止术后融合器的移位及塌陷。由于受到工具和通道角度的限制,该项技术对终板处理的广度不如传统手术,植骨量和植骨面积小,融合率有待进一步随访,随着工具的不断改进和技术的提高,以期这一问题能够得到解决[53]。 2.4.4 技术要点 脊柱内镜下融合技术需术者熟练掌握人体解剖及微创技术,在初始阶段容易出现镜下“迷路”现象,因此,①建议术中在镜下融合手术前先熟练掌握脊柱内镜下减压技术,必要时可借助C臂机透视确认内镜位置[49]。②关于术中止血,镜下操作止血至关重要,可在手术入路过程中采用射频电极严格止血,必要时结合血压及调节冲洗水压力来控制椎管内静脉丛止血,在切除黄韧带及骨性结构时,可借助骨蜡有效控制出血[49]。③经Kambin入路需要充分显露Kambin三角,做好关节突成型术,为融合器的置入提供一个安全可靠的通道,否则极易损伤神经根。如Endo-TLIF术中需先显露下关节突内缘与上位椎体的下椎板交界处,然后向尾端分离并显露上关节突内缘与下位椎体的上椎板交界处,以此骨性结构为标志,逐步完成椎骨减压[53]。④值得注意的是,显露黄韧带后不能盲目进入椎管,否则有造成神经根损伤及椎管内出血的可能性。建议先完成椎体间融合的步骤,再完成减压的操作,这样操作的优点是黄韧带可以保护椎管内神经阻滞以及减少黄韧带切除导致的椎管内出血,阻碍手术进程 [53]。 2.5 电磁导航在镜下融合中的应用 脊柱内镜手术离不开导航系统的辅助,在手术过程中,导航系统可以对患者的解剖结构提供实时精确的定位。光学导航和电磁导航是最常用的导航系统[54],光学导航系统是基于与红外LED相关的光学跟踪技术,通过摄像头捕捉到的LED光可实时对手术操作过程进行反馈。光学导航系统使用的设备体积大且重,视线也经常受到干扰,从而影响手术操作[55]。相比于传统的光学导航系统,新出现的电磁导航系统基于一种四维多阵列传感器技术,其能够在手术期间实现智能、精确地持续反馈手术过程。电磁导航系统的内部相关电极尺寸较小,使用灵活,可以实现更小的手术切口和瘢痕,电磁场对于视线跟踪的限制更小,更适合微创内镜手术。电磁导航系统提供的精准定位和引导,提高了Endo-TLIF经皮椎弓根螺钉置入、内镜下减压的手术效率,减少了螺钉的调整,减少了术中医生和患者暴露的辐射量[9]。Endo-TLIF是一种复杂的经椎间孔入路手术,学习曲线陡峭。在某些特殊情况下,如椎间隙塌陷、严重骨质增生、手术翻修,即使是经验丰富的外科医生也可能会被复杂的解剖结构所困扰,从而限制了该术式的广泛应用。电磁导航系统中使用的小型探头可清晰地显示椎间孔周围的解剖结构,帮助外科医生更快地学习,降低了手术难度,探头还可进一步识别小关节、峡部和部分椎板,明确患者具体病情。通过探头还可评估融合器插入前的处理深度,这些都极大地提高了手术的安全性和准确性。导航技术的进一步发展将有助于Endo-TLIF手术的安全性[9],有利于外科医生更好地学习及手术的广泛开展。"

| [1] KAISER MG, ECK JC, GROFF MW, et al. Guideline update for the performance of fusion procedures for degenerative disease of the lumbar spine. Part 1: introduction and methodology. J Neurosurg Spine. 2014;21(1):2-6. [2] LI L, LIU Y, ZHANG P, et al. Comparison of posterior lumbar interbody fusion with transforaminal lumbar interbody fusion for treatment of recurrent lumbar disc herniation: A retrospective study. J Int Med Res. 2016;44(6):1424-1429. [3] 陈培,戴春宏,彭和兵,等.椎间孔镜治疗退行性腰椎管狭窄的临床疗效及其对腰椎功能评分的影响[J]. 医学综述,2020,26(7):1402-1406. [4] PRICE JP, DAWSON JM, SCHWENDER JD, et al. Clinical and Radiologic Comparison of Minimally Invasive Surgery With Traditional Open Transforaminal Lumbar Interbody Fusion: A Review of 452 Patients From a Single Center. Clin Spine Surg. 2018;31(2):E121-E126. [5] 李佳奇,王林峰,高显达,等.MIS-TLIF治疗腰椎退变性疾病的研究进展[J].中国矫形外科杂志,2018,26(7):631-634. [6] ZHAO XB, MA HJ, GENG B, et al. Early Clinical Evaluation of Percutaneous Full-endoscopic Transforaminal Lumbar Interbody Fusion with Pedicle Screw Insertion for Treating Degenerative Lumbar Spinal Stenosis. Orthop Surg. 2021;13(1):328-337. [7] JIN M, ZHANG J, SHAO H, et al. Percutaneous Transforaminal Endoscopic Lumbar Interbody Fusion for Degenerative Lumbar Diseases: A Consecutive Case Series with Mean 2-Year Follow-Up. Pain Physician. 2020;23(2):165-174. [8] WANG MY, GROSSMAN J. Endoscopic minimally invasive transforaminal interbody fusion without general anesthesia: initial clinical experience with 1-year follow-up. Neurosurg Focus. 2016;40(2):E13. [9] XU D, HAN S, WANG C, et al. The technical feasibility and preliminary results of minimally invasive endoscopic-TLIF based on electromagnetic navigation: a case series. BMC Surg. 2021;21(1):149. [10] ATTIVISSIMO F, LANZOLLA A, CARLONE S, et al. A novel electromagnetic tracking system for surgery navigation. Comput Assist Surg (Abingdon). 2018;23(1):42-52. [11] BRIGGS H, MILLIGAN PR. Chip fusion of the low back following exploration of the spinal canal. J Bone Joint Surg Am. 1944;26:125-130. [12] BAEESA SS, MEDRANO BG, NORIEGA DC. Long-Term Outcomes of Posterior Lumbar Interbody Fusion Using Stand-Alone Ray Threaded Cage for Degenerative Disk Disease: A 20-Year Follow-Up. Asian Spine J. 2016;10(6):1100-1105. [13] HARMS J, ROLINGER H. A one-stager procedure in operative treatment of spondylolistheses: dorsal traction-reposition and anterior fusion (author’s transl). Z Orthop Ihre Grenzgeb. 1982;120(3):343-347. [14] LOWERY GL, KULKARNI SS. Posterior percutaneous spine instrumentation. Eur Spine J. 2000;9 Suppl 1(Suppl 1):S126-S130. [15] FOLEY KT, GUPTA SK. Percutaneous pedicle screw fixation of the lumbar spine: preliminary clinical results. J Neurosurg. 2002;97(1 Suppl):7-12. [16] PARVIZ K, HARRIS G. Percutaneous Lateral Discectomy of the Lumbar Spine. Clin Orthop Relat Res. 1983;174:127-132. [17] YEUNG AT. Minimally Invasive Disc Surgery with the Yeung Endoscopic Spine System (YESS). Surg Technol Int. 1999;8:267-277. [18] HOOGLAND T, SCHUBERT M, MIKLITZ B, et al. Transforaminal posterolateral endoscopic discectomy with or without the combination of a low-dose chymopapain: a prospective randomized study in 280 consecutive cases. Spine (Phila Pa 1976). 2006;31(24):E890-E897. [19] CHOI G, LEE SH, RAITURKER PP, et al. Percutaneous endoscopic interlaminar discectomy for intracanalicular disc herniations at L5-S1 using a rigid working channel endoscope. Neurosurgery. 2006;58(1 Suppl):ONS59-68; discussion ONS59-68. [20] RUETTEN S, KOMP M, GODOLIAS G. A New full-endoscopic technique for the interlaminar operation of lumbar disc herniations using 6-mm endoscopes: prospective 2-year results of 331 patients. Minim Invasive Neurosurg. 2006;49(2):80-87. [21] DE KUNDER SL, VAN KUIJK S, RIJKERS K, et al. Transforaminal lumbar interbody fusion (TLIF) versus posterior lumbar interbody fusion (PLIF) in lumbar spondylolisthesis: a systematic review and meta-analysis. Spine J. 2017;17(11):1712-1721. [22] LEONOVA ON, CHEREPANOV EA, KRUTKO AV. MIS-TLIF versus O-TLIF for single-level degenerative stenosis: study protocol for randomised controlled trial. BMJ Open. 2021;11(3):e041134. [23] TAN JH, LIU G, NG R, et al. Is MIS-TLIF superior to open TLIF in obese patients?: A systematic review and meta-analysis. Eur Spine J. 2018; 27(8):1877-1886. [24] JACOB KC, PATEL MR, RIBOT MA, et al. Single-level TLIF Versus LLIF at L4-5: A Comparison of Patient-reported Outcomes and Recovery Ratios. J Am Acad Orthop Surg. 2022;30(4):e495-e505. [25] LV Y, CHEN M, WANG SL, et al. Endo-TLIF versus MIS-TLIF in 1-segment lumbar spondylolisthesis: A prospective randomized pilot study. Clin Neurol Neurosurg. 2022;212:107082. [26] LEE CW, YOON KJ, HA SS. Which Approach Is Advantageous to Preventing Development of Adjacent Segment Disease? Comparative Analysis of 3 Different Lumbar Interbody Fusion Techniques (ALIF, LLIF, and PLIF) in L4-5 Spondylolisthesis. World Neurosurg. 2017;105:612-622. [27] TENG I, HAN J, PHAN K, et al. A meta-analysis comparing ALIF, PLIF, TLIF and LLIF. J Clin Neurosci. 2017;44:11-17. [28] AO S, ZHENG W, WU J, et al. Comparison of Preliminary clinical outcomes between percutaneous endoscopic and minimally invasive transforaminal lumbar interbody fusion for lumbar degenerative diseases in a tertiary hospital: Is percutaneous endoscopic procedure superior to MIS-TLIF? A prospective cohort study. Int J Surg. 2020;76: 136-143. [29] WU J, LIU H, AO S, et al. Percutaneous Endoscopic Lumbar Interbody Fusion: Technical Note and Preliminary Clinical Experience with 2-Year Follow-Up. Biomed Res Int. 2018;2018:5806037. [30] AHN Y, YOUN MS, HEO DH. Endoscopic transforaminal lumbar interbody fusion: a comprehensive review. Expert Rev Med Devices. 2019;16(5):373-380. [31] SHEN J. Fully Endoscopic Lumbar Laminectomy and Transforaminal Lumbar Interbody Fusion Under Local Anesthesia with Conscious Sedation: A Case Series. World Neurosurg. 2019;127:e745-e750. [32] QUILLO-OLVERA J, QUILLO-RESÉNDIZ J, QUILLO-OLVERA D, et al. Ten-Step Biportal Endoscopic Transforaminal Lumbar Interbody Fusion Under Computed Tomography-Based Intraoperative Navigation: Technical Report and Preliminary Outcomes in Mexico. Oper Neurosurg (Hagerstown). 2020;19(5):608-618. [33] MORGENSTERN C, YUE JJ, MORGENSTERN R. Full Percutaneous Transforaminal Lumbar Interbody Fusion Using the Facet-sparing, Trans-Kambin Approach. Clin Spine Surg. 2020;33(1):40-45. [34] HEO DH, SON SK, EUM JH, et al. Fully endoscopic lumbar interbody fusion using a percutaneous unilateral biportal endoscopic technique: technical note and preliminary clinical results. Neurosurg Focus. 2017; 43(2):E8. [35] HEO DH, HONG YH, LEE DC, et al. Technique of Biportal Endoscopic Transforaminal Lumbar Interbody Fusion. Neurospine. 2020;17(Suppl 1): S129-S137. [36] PARK MK, PARK SA, SON SK, et al. Clinical and radiological outcomes of unilateral biportal endoscopic lumbar interbody fusion (ULIF) compared with conventional posterior lumbar interbody fusion (PLIF): 1-year follow-up. Neurosurg Rev. 2019;42(3):753-761. [37] HEO DH, PARK CK. Clinical results of percutaneous biportal endoscopic lumbar interbody fusion with application of enhanced recovery after surgery. Neurosurg Focus. 2019;46(4):E18. [38] WU PH, KIM HS, LEE YJ, et al. Uniportal Full Endoscopic Posterolateral Transforaminal Lumbar Interbody Fusion with Endoscopic Disc Drilling Preparation Technique for Symptomatic Foraminal Stenosis Secondary to Severe Collapsed Disc Space: A Clinical and Computer Tomographic Study with Technical Note. Brain Sci. 2020;10(6):373. [39] WU W, YANG S, DIAO W, et al. Analysis of clinical efficacy of endo-LIF in the treatment of single-segment lumbar degenerative diseases. J Clin Neurosci. 2020;71:51-57. [40] ZHANG H, ZHOU C, WANG C, et al. Percutaneous Endoscopic Transforaminal Lumbar Interbody Fusion: Technique Note and Comparison of Early Outcomes with Minimally Invasive Transforaminal Lumbar Interbody Fusion for Lumbar Spondylolisthesis. Int J Gen Med. 2021;14:549-558. [41] KIM JE, YOO HS, CHOI DJ, et al. Comparison of Minimal Invasive Versus Biportal Endoscopic Transforaminal Lumbar Interbody Fusion for Single-level Lumbar Disease. Clin Spine Surg. 2021;34(2):E64-E71. [42] POSTACCHINI R, CINOTTI G, POSTACCHINI F. Injury to major abdominal vessels during posterior lumbar interbody fusion. A case report and review of the literature. Spine J. 2013;13(1):e7-e11. [43] STONE CE, MYERS BL, GUPTA S, et al. Surgical Outcomes After Single-Level Endoscopic Transforaminal Lumbar Interbody Fusion: A Systematic Review and Meta-Analysis. Cureus. 2020;12(10):e11052. [44] 赵志明,王玉召,钟浩,等.脊柱微创通道镜系统辅助下椎间孔腰椎椎体间融合术对腰椎退行性疾病患者血清氧化应激指标、疼痛介质及脊髓功能的影响[J].中国临床医生杂志,2019,47(1):72-76. [45] 刘镠,李莹,吴从俊,等. 经皮内镜下腰椎间融合技术研究进展[J].中国中医骨伤科杂志,2019,27(11):85-88. [46] CRIPTON PA, JAIN GM, WITTENBERG RH, et al. Load-sharing characteristics of stabilized lumbar spine segments. Spine (Phila Pa 1976). 2000;25(2):170-179. [47] 姚楚亮,陈杰,杜杰明,等.脊柱微创通道镜下改良经椎间孔入路椎间植骨融合术治疗腰椎退行性疾病106例[J].中国中医骨伤科杂志,2022,30(1):74-77. [48] HEO DH, LEE DC, KIM HS, et al. Clinical Results and Complications of Endoscopic Lumbar Interbody Fusion for Lumbar Degenerative Disease: A Meta-Analysis. World Neurosurg. 2021;145:396-404. [49] 王放,王永虎,乔浩,等.脊柱内镜下融合技术治疗退行性腰椎疾病的早期疗效[J].西安交通大学学报(医学版),2022,43(1):143-148. [50] 任伟剑,朱志勇,吴骏,等.脊柱内镜下融合术治疗腰椎滑脱症的疗效[J].局解手术学杂志,2021,30(12):1038-1042. [51] KIM HS, RAORANE HD, WU PH, et al. Incidental Durotomy During Endoscopic Stenosis Lumbar Decompression: Incidence, Classification, and Proposed Management Strategies. World Neurosurg. 2020;139: e13-e22. [52] CHOI DJ, CHOI CM, JUNG JT, et al. Learning Curve Associated with Complications in Biportal Endoscopic Spinal Surgery: Challenges and Strategies. Asian Spine J. 2016;10(4):624-629. [53] 郭时空,高全有,周程沛,等.全内镜下腰椎椎间融合术治疗腰椎退行性病变伴腰椎失稳的早期临床疗效[J].西安交通大学学报(医学版),2022,43(1):149-153. [54] HAHN P, OEZDEMIR S, KOMP M, et al. A New Electromagnetic Navigation System for Pedicle Screws Placement: A Human Cadaver Study at the Lumbar Spine. PLoS One. 2015;10(7):e0133708. [55] USUDA J. Virtual Bronchoscopic Navigation( VBN) and Electromagnetic Navigation System. Kyobu Geka. 2018;71(10):843-849. [56] The classic. The treatment of ruptured lumbar intervertebral discs by vertebral body fusion. I. Indications, operative technique, after care. By Ralph B. Cloward, 1953. Clin Orthop Relat Res. 1985;(193):5-15. [57] PHAN K, MOBBS RJ. Evolution of Design of Interbody Cages for Anterior Lumbar Interbody Fusion. Orthop Surg. 2016;8(3):270-277. [58] 唐杰,汪洋,应放,等.经侧方入路腰椎融合术(LLIF)的研究进展[J].中国矫形外科杂志,2016,24(15):1399-1402. [59] GAMMAL ID, SPIVAK JM, BENDO JA. Systematic Review of Thigh Symptoms after Lateral Transpsoas Interbody Fusion for Adult Patients with Degenerative Lumbar Spine Disease. Int J Spine Surg. 2015;9:62. [60] 张蒂, 冯世庆. 斜外侧腰椎椎间融合术[J].中华骨科杂志,2017, 37(16):1029-1035. |
| [1] | Nong Fuxiang, Jiang Zhixiong, Li Yinghao, Xu Wencong, Shi Zhilan, Luo Hui, Zhang Qinglang, Zhong Shuang, Tang Meiwen. Bone cement augmented proximal femoral nail antirotation for type A3.3 intertrochanteric femoral fracturalysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-10. |
| [2] | Liu Jinyu, Zhang Hanshuo, Cui Hongpeng, Pan Lingzhi, Zhao Boran, Li Fei, Ding Yu. Finite element biomechanical analysis of minimally invasive treatment of cervical spondylotic myelopathy and accurate exercise rehabilitation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1359-1364. |
| [3] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [4] | Cai Zhihao, Xie Zhaoyong. Femoral neck anteversion measurement assessment: how to establish a unified method and standard [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1448-1454. |
| [5] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [6] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [7] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [8] | Gao Yu, Han Jiahui, Ge Xin. Immunoinflammatory microenvironment after spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1300-1305. |
| [9] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [10] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [11] | Chen Shisong, Liu Xiaohong, Xu Zhiyun. Current status and prospects of bioprosthetic heart valves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1096-1102. |
| [12] | Lu Di, Zhang Cheng, Duan Rongquan, Liu Zongxiang. Osteoinductive properties of calcium phosphate ceramic bone repair materials [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1103-1109. |
| [13] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [14] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [15] | Bai Siqi, Xiao Zhen, Liu Jing. Application potential of adipose-derived stem cells in female pelvic floor dysfunction diseases [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 921-927. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||