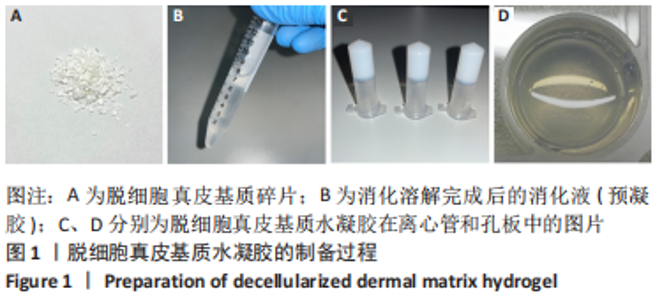
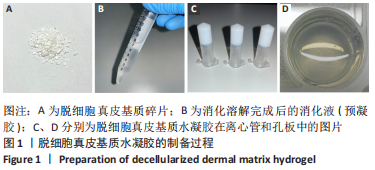
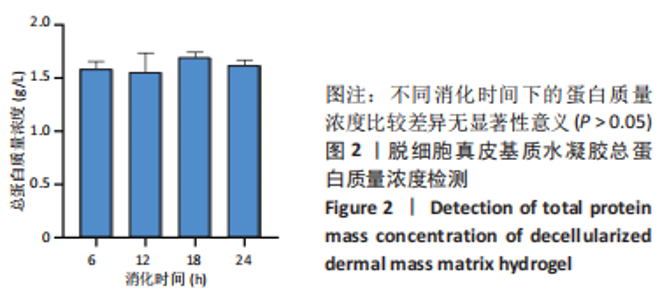
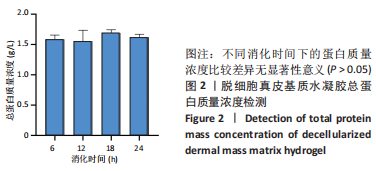
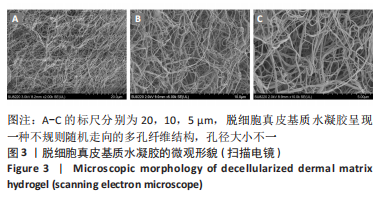
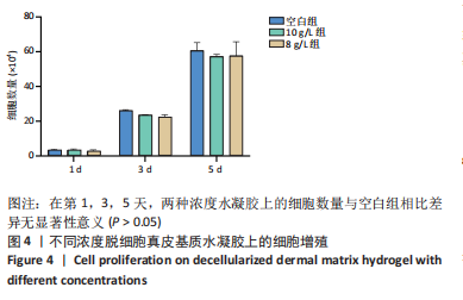
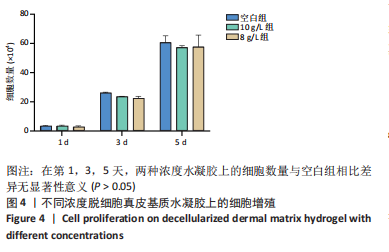
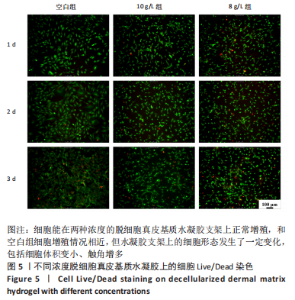
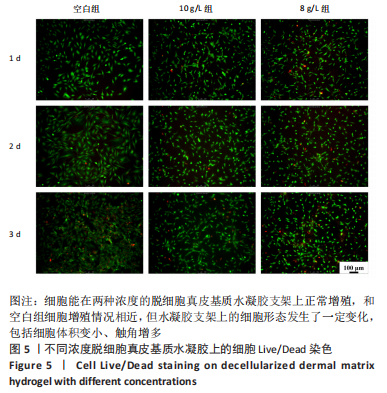
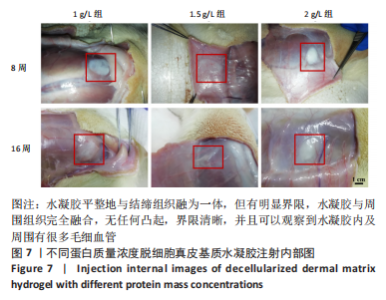
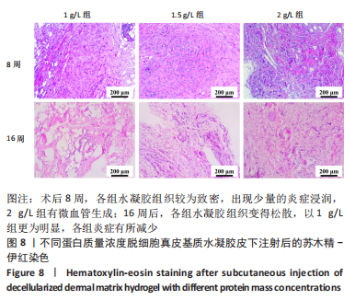
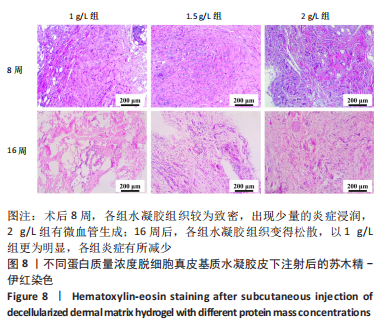
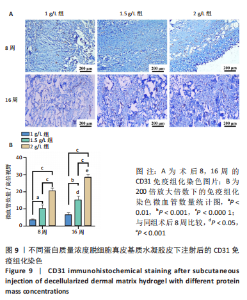
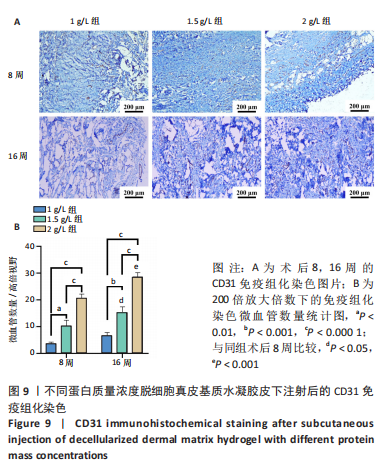
Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (21): 3325-3331.doi: 10.12307/2023.417
Previous Articles Next Articles
Preparation and biological evaluation of decellularized dermal matrix hydrogel
Xu Xin, Liu Yaowei, Mu Yunping, Wang Jianying, Li Fanghong, Zhao Zijian
- School of Biomedical and Pharmaceutical Sciences, Guangdong University of Technology, Guangzhou 510006, Guangdong Province, China
-
Received:2022-03-01Accepted:2022-06-02Online:2023-07-28Published:2022-11-24 -
Contact:Li Fanghong, PhD, Professor, School of Biomedical and Pharmaceutical Sciences, Guangdong University of Technology, Guangzhou 510006, Guangdong Province, China Zhao Zijian, PhD, Professor, School of Biomedical and Pharmaceutical Sciences, Guangdong University of Technology, Guangzhou 510006, Guangdong Province, China -
About author:Xu Xin, Master candidate, School of Biomedical and Pharmaceutical Sciences, Guangdong University of Technology, Guangzhou 510006, Guangdong Province, China -
Supported by:The National Key Research & Development Program of China, No. 2018YFA0800603 (to ZZJ); The Key Research and Development Program of Guangdong Province for “Innovative Drug Creation”, No. 2019B020201015 (to LFH); The Guangdong “Pearl River Talent Plan” Program, No. 2016ZT06Y432 (to ZZJ); The Startup Scientific Research Funding of Guangdong University of Technology, No. 50010102 (to ZZJ and LFH)
CLC Number:
Cite this article
Xu Xin, Liu Yaowei, Mu Yunping, Wang Jianying, Li Fanghong, Zhao Zijian. Preparation and biological evaluation of decellularized dermal matrix hydrogel[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(21): 3325-3331.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
| [1] BAYER IS. Hyaluronic Acid and Controlled Release: A Review. Molecules. 2020;25(11):2649. [2] CHOW KV, URMAN DS, CABRAL ES, et al. Hyaluronic Acid Filler Incidentally Found During Mohs Micrographic Surgery: Observations in 36 Patients Regarding Skin Depth, Degradation Size, and Estimated Persistence Time. Dermatol Surg. 2022;48(4):401-405. [3] 杨彪,郭学平,臧恒昌,等.交联透明质酸凝胶修饰度的测定方法研究进展[J].药物生物技术,2015,22(2):177-180. [4] TANG S, CHI K, XU H, et al. A covalently cross-linked hyaluronic acid/bacterial cellulose composite hydrogel for potential biological applications. Carbohydr Polym. 2021;252:117123. [5] ZERBINATI N, SOMMATIS S, MACCARIO C, et al. Toward Physicochemical and Rheological Characterization of Different Injectable Hyaluronic Acid Dermal Fillers Cross-Linked with Polyethylene Glycol Diglycidyl Ether. Polymers (Basel). 2021;13(6):948. [6] KENNE L, GOHIL S, NILSSON EM, et al. Modification and cross-linking parameters in hyaluronic acid hydrogels--definitions and analytical methods. Carbohydr Polym. 2013;91(1):410-418. [7] RZANY B, CONVERSET-VIETHEL S, HARTMANN M, et al. Efficacy and Safety of 3 New Resilient Hyaluronic Acid Fillers, Crosslinked With Decreased BDDE, for the Treatment of Dynamic Wrinkles: Results of an 18-Month, Randomized Controlled Trial Versus Already Available Comparators. Dermatol Surg. 2019;45(10):1304-1314. [8] CECCHI R, SPOTA A, FRATI P, et al. Migrating granulomatous chronic reaction from hyaluronic acid skin filler (Restylane): review and histopathological study with histochemical stainings. Dermatology. 2014;228(1):14-17. [9] SHOME D, DOSHI K, VADERA S, et al. Delayed hypersensitivity reaction to hyaluronic acid dermal filler post-COVID-19 viral infection. J Cosmet Dermatol. 2021;20(5):1549-1550. [10] SUN W, GREGORY DA, TOMEH MA, et al. Silk Fibroin as a Functional Biomaterial for Tissue Engineering. Int J Mol Sci. 2021;22(3):1499. [11] CHEN D, ZHOU X, CHANG L, et al. Hemostatic Self-Healing Hydrogel with Excellent Biocompatibility Composed of Polyphosphate-Conjugated Functional PNIPAM-Bearing Acylhydrazide. Biomacromolecules. 2021; 22(5):2272-2283. [12] HINDERER S, LAYLAND SL, SCHENKE-LAYLAND K. ECM and ECM-like materials - Biomaterials for applications in regenerative medicine and cancer therapy. Adv Drug Deliv Rev. 2016;97:260-269. [13] KOU L, SUN R, BHUTIA YD, et al. Emerging advances in P-glycoprotein inhibitory nanomaterials for drug delivery. Expert Opin Drug Deliv. 2018;15(9):869-879. [14] HOGANSON DM, OWENS GE, MEPPELINK AM, et al. Decellularized extracellular matrix microparticles as a vehicle for cellular delivery in a model of anastomosis healing. J Biomed Mater Res A. 2016;104(7): 1728-1735. [15] HWANG J, SULLIVAN MO, KIICK KL. Targeted Drug Delivery via the Use of ECM-Mimetic Materials. Front Bioeng Biotechnol. 2020;8:69. [16] PISHAVAR E, KHOSRAVI F, NASERIFAR M, et al. Multifunctional and Self-Healable Intelligent Hydrogels for Cancer Drug Delivery and Promoting Tissue Regeneration In Vivo. Polymers (Basel). 2021;13(16):2680. [17] FREYTES DO, MARTIN J, VELANKAR SS, et al. Preparation and rheological characterization of a gel form of the porcine urinary bladder matrix. Biomaterials. 2008;29(11):1630-1637. [18] WASEEQ UR REHMAN, ASIM M, HUSSAIN S, et al. Hydrogel: A Promising Material in Pharmaceutics. Curr Pharm Des. 2020;26(45):5892-5908. [19] CASCONE S, LAMBERTI G. Hydrogel-based commercial products for biomedical applications: A review. Int J Pharm. 2020;573:118803. [20] ALI F, KHAN I, CHEN J, et al. Emerging Fabrication Strategies of Hydrogels and Its Applications. Gels. 2022;8(4):205. [21] KAMOUN EA, LOUTFY SA, HUSSEIN Y, et al. Recent advances in PVA-polysaccharide based hydrogels and electrospun nanofibers in biomedical applications: A review. Int J Biol Macromol. 2021;187: 755-768. [22] ARKABAN H, BARANI M, AKBARIZADEH MR, et al. Polyacrylic Acid Nanoplatforms: Antimicrobial, Tissue Engineering, and Cancer Theranostic Applications. Polymers (Basel). 2022;14(6):1259. [23] STEPANOVSKA J, SUPOVA M, HANZALEK K, et al. Collagen Bioinks for Bioprinting: A Systematic Review of Hydrogel Properties, Bioprinting Parameters, Protocols, and Bioprinted Structure Characteristics. Biomedicines. 2021;9(9):1137. [24] GOODARZI K, RAO SS. Hyaluronic acid-based hydrogels to study cancer cell behaviors. J Mater Chem B. 2021;9(31):6103-6115. [25] ZOU Z, ZHANG B, NIE X, et al. A sodium alginate-based sustained-release IPN hydrogel and its applications. RSC Adv. 2020;10(65): 39722-39730. [26] RAO F, WANG Y, ZHANG D, et al. Aligned chitosan nanofiber hydrogel grafted with peptides mimicking bioactive brain-derived neurotrophic factor and vascular endothelial growth factor repair long-distance sciatic nerve defects in rats. Theranostics. 2020;10(4):1590-1603. [27] ZHONG G, YAO J, HUANG X, et al. Injectable ECM hydrogel for delivery of BMSCs enabled full-thickness meniscus repair in an orthotopic rat model. Bioact Mater. 2020;5(4):871-879. [28] VOYTIK-HARBIN SL, BRIGHTMAN AO. Small intestinal submucosa: a tissue derived extracellular matrix that promotes tissue-specifific growth and differentiation of cells in vitro. Tissue Eng. 1998;4(2): 157-174. [29] GIOBBE GG, CROWLEY C, LUNI C, et al. Extracellular matrix hydrogel derived from decellularized tissues enables endodermal organoid culture. Nat Commun. 2019;10(1):5658. [30] XU Y, ZHOU J, LIU C, et al. Understanding the role of tissue-specific decellularized spinal cord matrix hydrogel for neural stem/progenitor cell microenvironment reconstruction and spinal cord injury. Biomaterials. 2021;268:120596. [31] CHEN Z, ZHANG B, SHU J, et al. Human decellularized adipose matrix derived hydrogel assists mesenchymal stem cells delivery and accelerates chronic wound healing. J Biomed Mater Res A. 2021; 109(8):1418-1428. [32] BORDBAR S, LOTFI BAKHSHAIESH N, KHANMOHAMMADI M, et al. Production and evaluation of decellularized extracellular matrix hydrogel for cartilage regeneration derived from knee cartilage. J Biomed Mater Res A. 2020;108(4):938-946. [33] YAZDANPANAH G, SHAH R, RAGHURAMA R, et al. In-situ porcine corneal matrix hydrogel as ocular surface bandage. Ocul Surf. 2021;21:27-36. [34] GOLDFRACHT I, EFRAIM Y, SHINNAWI R, et al. Engineered heart tissue models from hiPSC-derived cardiomyocytes and cardiac ECM for disease modeling and drug testing applications. Acta Biomater. 2019;92:145-159. [35] FARNEBO S, WOON CY, SCHMITT T, et al. Design and characterization of an injectable tendon hydrogel: a novel scaffold for guided tissue regeneration in the musculoskeletal system. Tissue Eng Part A. 2014; 20(9-10):1550-1561. [36] FU W, XU P, FENG B, et al. A hydrogel derived from acellular blood vessel extracellular matrix to promote angiogenesis. J Biomater Appl. 2019;33(10):1301-1313. [37] LEE JS, SHIN J, PARK HM, et al. Liver extracellular matrix providing dual functions of two-dimensional substrate coating and three-dimensional injectable hydrogel platform for liver tissue engineering. Biomacromolecules. 2014;15(1):206-218. [38] BANKOTI K, RAMESHBABU AP, DATTA S, et al. Dual Functionalized Injectable Hybrid Extracellular Matrix Hydrogel for Burn Wounds. Biomacromolecules. 2021;22(2):514-533. [39] 梁成宵,古瑞,李婷,等.肝脏基质水凝胶的制备及其细胞相容性研究[J].解放军医学杂志,2018,43(5):409-413. [40] WOLF MT, DALY KA, BRENNAN-PIERCE EP, et al. A hydrogel derived from decellularized dermal extracellular matrix. Biomaterials. 2012; 33(29):7028-7038. [41] KADLER KE, HILL A, CANTY-LAIRD EG. Collagen fibrillogenesis: fibronectin, integrins, and minor collagens as organizers and nucleators. Curr Opin Cell Biol. 2008;20(5):495-501. [42] OZUDOGRU E, ISIK M, EYLEM CC, et al. Decellularized spinal cord meninges extracellular matrix hydrogel that supports neurogenic differentiation and vascular structure formation. J Tissue Eng Regen Med. 2021;15(11):948-963. |
| [1] | Sun Kexin, Zeng Jinshi, Li Jia, Jiang Haiyue, Liu Xia. Mechanical stimulation enhances matrix formation of three-dimensional bioprinted cartilage constructs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [2] | Lian Shilin, Zhang Yan, Jiang Qiang, Zhang Hanshuo, Li Tusheng, Ding Yu. Interventional effects of whole blood and platelet-rich plasma with different preparation methods on nucleus pulposus cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1199-1204. |
| [3] | Liu Xiaolin, Mu Xinyue, Ma Ziyu, Liu Shutai, Wang Wenlong, Han Xiaoqian, Dong Zhiheng. Effect of hydrogel-loaded simvastatin microspheres on osteoblast proliferation and differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 998-1003. |
| [4] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [5] | Yang Yitian, Wang Lu, Yao Wei, Zhao Bin. Application of the interaction between biological scaffolds and macrophages in bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1071-1079. |
| [6] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [7] | Chen Shisong, Liu Xiaohong, Xu Zhiyun. Current status and prospects of bioprosthetic heart valves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1096-1102. |
| [8] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [9] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [10] | Xu Yan, Li Ping, Lai Chunhua, Zhu Peijun, Yang Shuo, Xu Shulan. Piezoelectric materials for vascularized bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1126-1132. |
| [11] | Li Xinyue, Li Xiheng, Mao Tianjiao, Tang Liang, Li Jiang. Three-dimensional culture affects morphology, activity and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 846-852. |
| [12] | Li Xiaoyin, Yang Xiaoqing, Chen Shulian, Li Zhengchao, Wang Ziqi, Song Zhen, Zhu Daren, Chen Xuyi. Collagen/silk fibroin scaffold combined with neural stem cells in the treatment of traumatic spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 890-896. |
| [13] | Yuan Bo, Xie Lide, Fu Xiumei. Schwann cell-derived exosomes promote the repair and regeneration of injured peripheral nerves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 935-940. |
| [14] | Qin Yuxing, Ren Qiangui, Li Zilong, Quan Jiaxing, Shen Peifeng, Sun Tao, Wang Haoyu. Action mechanism and prospect of bone microvascular endothelial cells for treating femoral head necrosis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 955-961. |
| [15] | Li Long, Li Guangdi, Shi Hao, Deng Keqi. Circular RNA as a competing endogenous RNA is involved in the regulation of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 751-757. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||