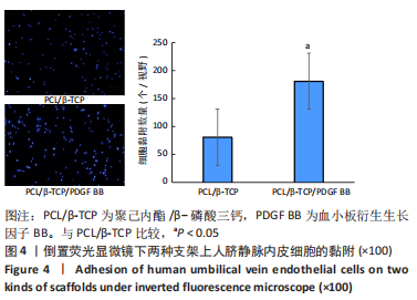
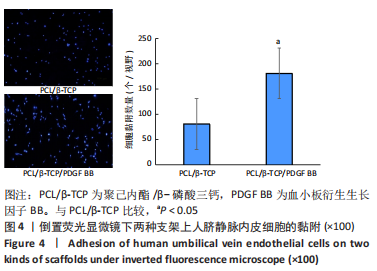
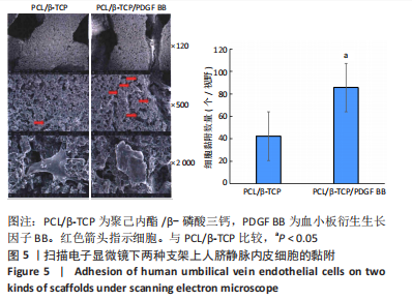
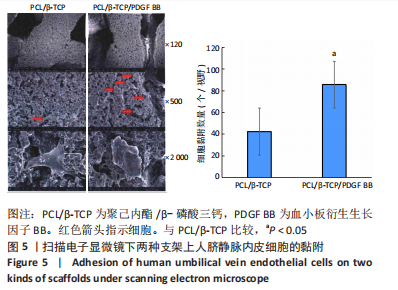
Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (21): 3300-3306.doi: 10.12307/2023.415
Previous Articles Next Articles
Promoting angiogenesis of platelet-derived growth factor BB modified polycaprolactone and beta-tricalcium phosphate composite scaffolds
Zhao Jinlong, Liu Jichao, Yu Yang
- Department of Orthopedics, The 3201 Hospital Affiliated to Xi’an Jiaotong University School of Medicine, Hanzhong 723000, Shaanxi Province, China
-
Received:2022-04-18Accepted:2022-06-02Online:2023-07-28Published:2022-11-24 -
About author:Zhao Jinlong, Master, Associate chief physician, Department of Orthopedics, The 3201 Hospital Affiliated to Xi’an Jiaotong University School of Medicine, Hanzhong 723000, Shaanxi Province, China
CLC Number:
Cite this article
Zhao Jinlong, Liu Jichao, Yu Yang. Promoting angiogenesis of platelet-derived growth factor BB modified polycaprolactone and beta-tricalcium phosphate composite scaffolds[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(21): 3300-3306.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
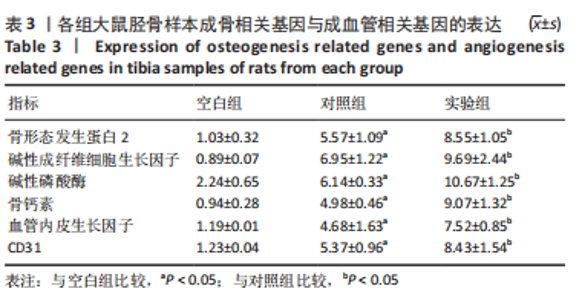
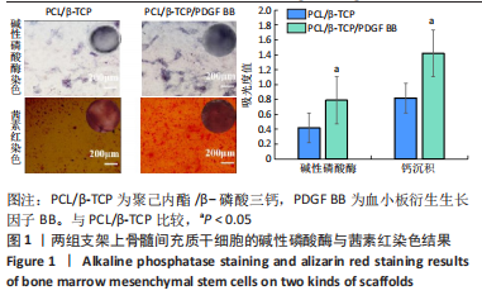
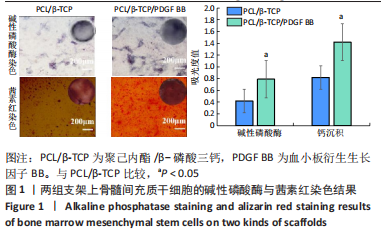
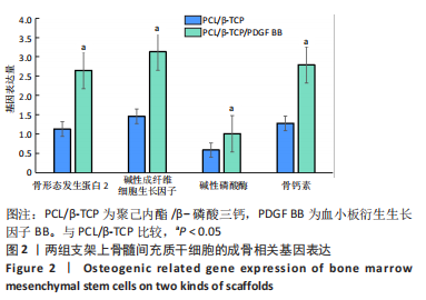
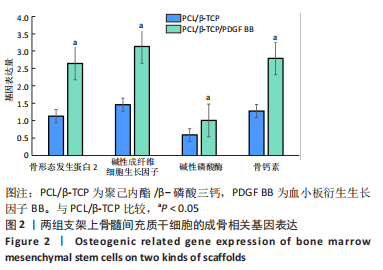
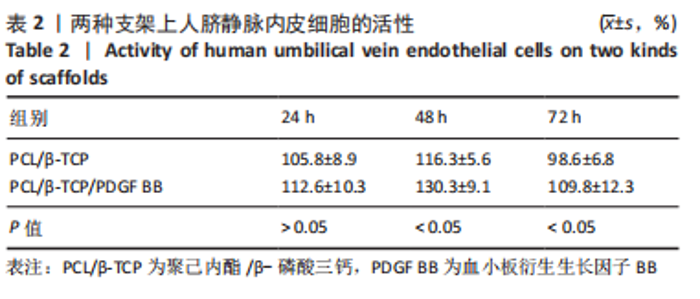
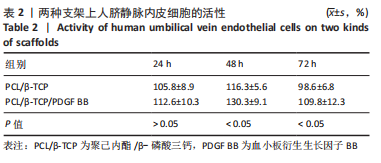
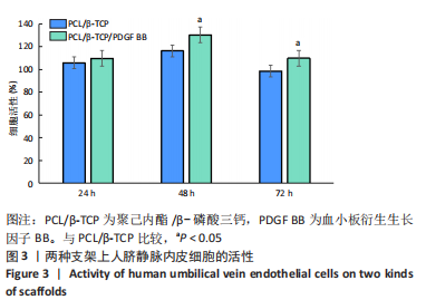
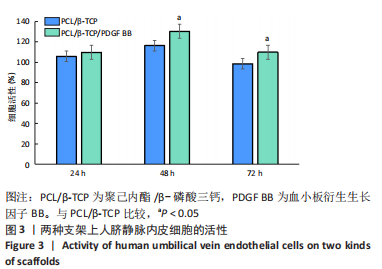
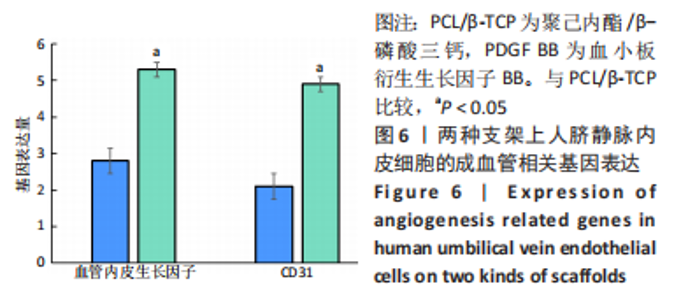
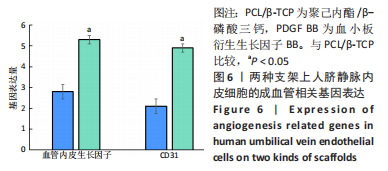
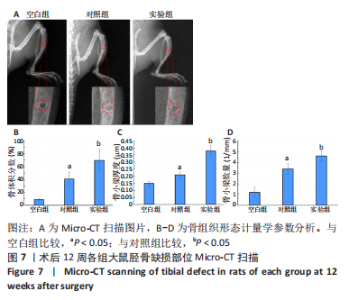
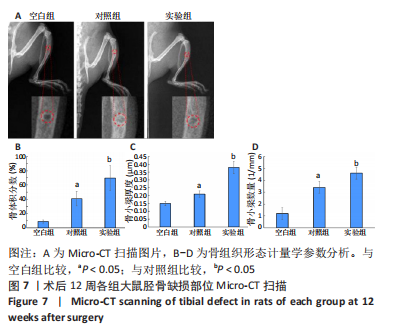
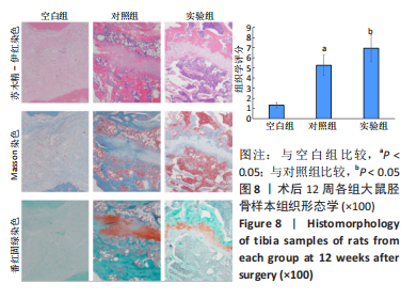
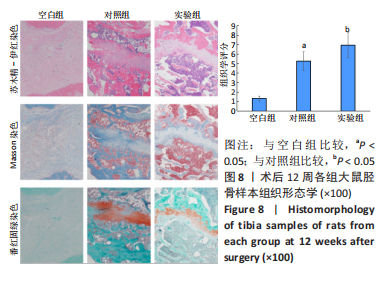
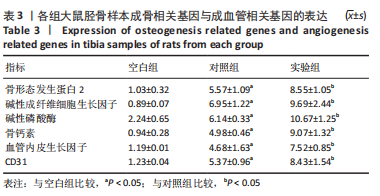
苏木精-伊红染色、Masson和番红固绿染色显示,空白组没有明显骨组织形成,缺损部位主要由少量的血管与大量结缔组织填充;对照组缺损部位可见大量成熟度较高的骨基质及相对较少的不成熟软骨组织;实验组骨缺损部位可见大量的骨样组织及成熟度较高的软骨组织,大部分髓腔再通。 Huddleston组织学评分结果显示,空白组评分低于对照组(P < 0.05),对照组评分低于实验组(P < 0.05)。 2.2.4 RT-PCR检测结果 对照组缺损部位的成骨相关基因骨形态发生蛋白2、碱性成纤维细胞生长因子、碱性磷酸酶、骨钙素的mRNA表达量均高于空白组(P < 0.05),实验组缺损部位骨形态发生蛋白2、碱性成纤维细胞生长因子、碱性磷酸酶、骨钙素的mRNA表达量均高于对照组(P < 0.05),见表3。对照组缺损部位成血管相关基因血管内皮生长因子、CD31的mRNA表达量均高于空白组(P < 0.05),实验组缺损部位血管内皮生长因子、CD31的mRNA表达量均高于对照组(P < 0.05),见表3。"
| [1] KHARE D, BASU B, DUBEY AK. Electrical stimulation and piezoelectric biomaterials for bone tissue engineering applications. Biomaterials. 2020;258:120280. [2] WAN Z, ZHANG P, LIU Y, et al. Four-dimensional bioprinting: Current developments and applications in bone tissue engineering. Acta Biomater. 2020;101:26-42. [3] SHEN C, WITEK L, FLORES RL, et al.Three-Dimensional Printing for Craniofacial Bone Tissue Engineering.Tissue Eng Part A. 2020;26(23-24): 1303-1311. [4] RAJ PREETH D, SARAVANAN S, SHAIRAM M, et al. Bioactive Zinc(II) complex incorporated PCL/gelatin electrospun nanofiber enhanced bone tissue regeneration. Eur J Pharm Sci. 2021;160:105768. [5] MAHALINGAM S, BAYRAM C, GULTEKINOGLU M, et al. Co-Axial Gyro-Spinning of PCL/PVA/HA Core-Sheath Fibrous Scaffolds for Bone Tissue Engineering.Macromol Biosci. 2021;21(10):e2100177. [6] MURUGAN S, PARCHA SR. Fabrication techniques involved in developing the composite scaffolds PCL/HA nanoparticles for bone tissue engineering applications. J Mater Sci Mater Med. 2021;32(8):93. [7] BIGHAM A, SALEHI AOM, RAFIENIA M, et al. Zn-substituted Mg2SiO4 nanoparticles-incorporated PCL-silk fibroin composite scaffold: A multifunctional platform towards bone tissue regeneration. Mater Sci Eng C Mater Biol Appl. 2021;127:112242. [8] PAZARÇEVIREN AE, TEZCANER A, KESKIN D, et al. Boron-doped Biphasic Hydroxyapatite/β-Tricalcium Phosphate for Bone Tissue Engineering. Biol Trace Elem Res. 2021;199(3):968-980. [9] DING X, LI A, YANG F, et al. β-tricalcium phosphate and octacalcium phosphate composite bioceramic material for bone tissue engineering. J Biomater Appl. 2020;34(9):1294-1299. [10] LEE DH, TRIPATHY N, SHIN JH, et al. Enhanced osteogenesis of β-tricalcium phosphate reinforced silk fibroin scaffold for bone tissue biofabrication. Int J Biol Macromol. 2017;95:14-23. [11] NAKHAEE FM, RAJABI M, BAKHSHESHI-RAD HR. In-vitroassessment of β-tricalcium phosphate/bredigite-ciprofloxacin (CPFX) scaffolds for bone treatment applications. Biomed Mater. 2021;16(4). doi: 10.1088/1748-605X/ac0590. [12] LEE S, CHOI D, SHIM JH, et al. Efficacy of three-dimensionally printed polycaprolactone/beta tricalcium phosphate scaffold on mandibular reconstruction. Sci Rep. 2020;10(1):4979. [13] LU L, ZHANG Q, WOOTTON D, et al. Biocompatibility and biodegradation studies of PCL/β-TCP bone tissue scaffold fabricated by structural porogen method. J Mater Sci Mater Med. 2012;23(9):2217-2226. [14] BRUYAS A, LOU F, STAHL AM, et al. Systematic characterization of 3D-printed PCL/β-TCP scaffolds for biomedical devices and bone tissue engineering: influence of composition and porosity. J Mater Res. 2018; 33(14):1948-1959. [15] WANG Q, YE W, MA Z, et al. 3D printed PCL/β-TCP cross-scale scaffold with high-precision fiber for providing cell growth and forming bones in the pores. Mater Sci Eng C Mater Biol Appl. 2021;127:112197. [16] XIE H, CUI Z, WANG L, et al. PDGF-BB secreted by preosteoclasts induces angiogenesis during coupling with osteogenesis. Nat Med. 2014;20(11):1270-1278. [17] HUDDLESTON PM, STECKELBERG JM, HANSSEN AD, et al. Ciprofloxacin inhibition of experimental fracture healing. J Bone Joint Surg Am. 2000;82(2):161-173. [18] YUAN J, MATURAVONGSADIT P, METAVARAYUTH K, et al. Enhanced Bone Defect Repair by Polymeric Substitute Fillers of MultiArm Polyethylene Glycol-Crosslinked Hyaluronic Acid Hydrogels. Macromol Biosci. 2019;19(6):e1900021. [19] WANG D, LIU Y, LIU Y, et al. A dual functional bone-defect-filling material with sequential antibacterial and osteoinductive properties for infected bone defect repair. J Biomed Mater Res A. 2019;107(10):2360-2370. [20] HAN D, LI J. Repair of bone defect by using vascular bundle implantation combined with Runx II gene-transfected adipose-derived stem cells and a biodegradable matrix. Cell Tissue Res. 2013;352(3):561-571. [21] YIN Y, TANG Q, XIE M, et al. Insights into the mechanism of vascular endothelial cells on bone biology.Biosci Rep. 2021;41(1):BSR20203258. [22] ROMEO SG, ALAWI KM, RODRIGUES J, et al. Endothelial proteolytic activity and interaction with non-resorbing osteoclasts mediate bone elongation. Nat Cell Biol. 2019;21(4):430-441. [23] SU W, LIU G, LIU X, et al. Angiogenesis stimulated by elevated PDGF-BB in subchondral bone contributes to osteoarthritis development. JCI Insight. 2020;5(8):e135446. [24] 刘慧,陈慧鸿,廖红兵.破骨细胞衍生的偶联因子鞘氨醇-1-磷酸及血小板衍生生长因子BB对成骨细胞的调节作用[J].中国组织工程研究,2019,23(23):3739-3745. [25] BRUN J, ANDREASEN CM, EJERSTED C, et al. PDGF Receptor Signaling in Osteoblast Lineage Cells Controls Bone Resorption Through Upregulation of Csf1 Expression. J Bone Miner Res. 2020;35(12):2458-2469. [26] 魏琴,张雪,马磊,等.血小板衍生生长因子BB诱导大鼠骨髓间充质干细胞向成骨细胞分化[J].中国组织工程研究,2021,25(19): 2953-2957. [27] 吴硕,魏琴,买买艾力·玉山.rrPDGF-BB基因修饰的自体BMSCs促进大鼠股骨牵张成骨[J].中华显微外科杂志,2021,44(5):526-534. [28] CHENG J, YANG HL, GU CJ, et al. Melatonin restricts the viability and angiogenesis of vascular endothelial cells by suppressing HIF-1α/ROS/VEGF. Int J Mol Med. 2019;43(2):945-955. [29] CHAN CM, HSIAO CY, LI HJ, et al. The Inhibitory Effects of Gold Nanoparticles on VEGF-A-Induced Cell Migration in Choroid-Retina Endothelial Cells.Int J Mol Sci. 2019;21(1):109. [30] POH CK, SHI Z, LIM TY, et al. The effect of VEGF functionalization of titanium on endothelial cells in vitro. Biomaterials. 2010;31(7):1578-1585. [31] HUANG J, ZHANG S. Overexpressed Neuropilin-1 in Endothelial Cells Promotes Endothelial Permeability through Interaction with ANGPTL4 and VEGF in Kawasaki Disease. Mediators Inflamm. 2021; 2021:9914071. |
| [1] | Sun Kexin, Zeng Jinshi, Li Jia, Jiang Haiyue, Liu Xia. Mechanical stimulation enhances matrix formation of three-dimensional bioprinted cartilage constructs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [2] | Wen Xinghua, Ding Huanwen, Cheng Kai, Yan Xiaonan, Peng Yuanhao, Wang Yuning, Liu Kang, Zhang Huiwu. Three-dimensional finite element model analysis of intramedullary nailing fixation design for large femoral defects in Beagle dogs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1371-1376. |
| [3] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [4] | Yang Yitian, Wang Lu, Yao Wei, Zhao Bin. Application of the interaction between biological scaffolds and macrophages in bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1071-1079. |
| [5] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [6] | Lu Di, Zhang Cheng, Duan Rongquan, Liu Zongxiang. Osteoinductive properties of calcium phosphate ceramic bone repair materials [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1103-1109. |
| [7] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [8] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [9] | Xu Yan, Li Ping, Lai Chunhua, Zhu Peijun, Yang Shuo, Xu Shulan. Piezoelectric materials for vascularized bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1126-1132. |
| [10] | Li Xinyue, Li Xiheng, Mao Tianjiao, Tang Liang, Li Jiang. Three-dimensional culture affects morphology, activity and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 846-852. |
| [11] | Li Xiaoyin, Yang Xiaoqing, Chen Shulian, Li Zhengchao, Wang Ziqi, Song Zhen, Zhu Daren, Chen Xuyi. Collagen/silk fibroin scaffold combined with neural stem cells in the treatment of traumatic spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 890-896. |
| [12] | Yuan Bo, Xie Lide, Fu Xiumei. Schwann cell-derived exosomes promote the repair and regeneration of injured peripheral nerves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 935-940. |
| [13] | Qin Yuxing, Ren Qiangui, Li Zilong, Quan Jiaxing, Shen Peifeng, Sun Tao, Wang Haoyu. Action mechanism and prospect of bone microvascular endothelial cells for treating femoral head necrosis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 955-961. |
| [14] | Xiong Bohan, Yu Yang, Lu Xiaojun, Wang Xu, Yang Tengyun, Zhang Yaozhang, Liao Xinyu, Zhou Xiaoxiang, He Lu, Li Yanlin. Research progress in promoting tendon to bone healing during anterior cruciate ligament reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 779-786. |
| [15] | Zhang Min, Zhang Xiaoming, Liu Tongbin. Application potential of naringin in bone tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 787-792. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||