Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (5): 793-798.doi: 10.12307/2023.111
Previous Articles Next Articles
Effect of hypoxic training on the oxygen sensing pathway
Liu Yuan
- Shandong Jianzhu University, Jinan 250101, Shandong Province, China
-
Received:2021-12-15Accepted:2022-05-11Online:2023-02-18Published:2022-07-25 -
About author:Liu Yuan, Master, Lecturer, Shandong Jianzhu University, Jinan 250101, Shandong Province, China
CLC Number:
Cite this article
Liu Yuan. Effect of hypoxic training on the oxygen sensing pathway[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 793-798.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1 低氧诱导因子概述 低氧诱导因子是氧感知通路中调节转录的关键转录因子,而该通路中一系列低氧诱导因子活性的氧感知信号是由相应酶参与调控的。在缺氧期间,酶活性被抑制,低氧诱导因子得以表达。1992年,SEMENZA等在促红细胞生成素调控区(非编码蛋白序列)鉴定出一段受低氧调控的DNA序列——低氧应答元件[2]。经过进一步验证,SEMENZA发现低氧应答元件可与某种被称为低氧诱导因子1的蛋白质特异性结合,增加基因的表达。低氧诱导因子1由诱导型α亚基(低氧诱导因子1α,是低氧敏感的主要成分)和组成型表达的β亚基(低氧诱导因子1β)组成[2]。低氧诱导因子1α是一种新型蛋白,而低氧诱导因子1β则早先被鉴定为芳基烃核转运蛋白(ARNT),它是芳基烃受体(AHR)的二聚体伴侣。低氧诱导因子1α可介导低氧诱导因子复合物的氧敏感性,并通过共享的Per-ARNT-Sim(PAS)域与低氧诱导因子1β相互作用。低氧诱导因子1α和低氧诱导因子1β都包含一个介导DNA结合的N末端碱性螺旋-环-螺旋结构域和诱导基因表达所必需的C末端反式激活结构域。低氧诱导因子1α蛋白的氧敏感性由内部氧依赖性降解域(包括羟基化与von Hippel-Lindau甲基化肿瘤抑制物相互作用的目标脯氨酸残基)和C末端结构域赋予,其中含有天冬酰胺基残基。在缺氧条件下,低氧诱导因子异二聚体与基因启动子及其包含核心序列(RCGTG)的转录增强子中的低氧应答元件结合调控基因的表达。 2.2 酶在缺氧信号中的潜在作用 控制低氧诱导因子活性的氧感知信号是由一系列调节酶产生的,该机制也被称为酶促氧感应机制。低氧诱导因子1α亚基中特定脯氨酸和天冬氨酸残基的羟基化受双氧合酶调控,将分子中的氧原子结合到其底物中。因此,在缺氧期间低氧诱导因子1α活性被抑制。目前来说,已经定义了3种脯氨酸羟化酶(脯氨酸羟化酶1、脯氨酸羟化酶2、脯氨酸羟化酶3)和天冬氨酸羟化酶[4]。 所有的脯氨酸羟化酶都参与低氧诱导因子的调控[5],在大多数细胞中,脯氨酸羟化酶2是低氧诱导因子活性的主要调节剂。有研究发现,小鼠Egln1(编码脯氨酸羟化酶2)基因失活会严重导致胎盘的发育性心脏缺陷,并在胚胎中致死。O2体积分数是影响脯氨酸羟化酶活性的关键因素。有学者指出,相比于对低氧诱导因子1的调控,脯氨酸羟化酶1和脯氨酸羟化酶3对低氧诱导因子2的调节似乎更活跃。这一发现可能是因为脯氨酸羟化酶更偏向于低氧诱导因子多肽底物内不同脯氨酸残基的羟基化,不过相比于低氧诱导因子1,低氧诱导因子2更依赖于C端脯氨酸羟基化。 大多数脯氨酸羟化酶都对O2具有较低的亲和力,在低氧条件下,其催化活性会大幅降低,这种对O2具有特殊敏感性的机制尚不完全清楚,不过有研究发现,在酸性条件下脯氨酸羟化酶2的催化效率更高,因此,脯氨酸羟化酶2的氧敏感性可能在生理范围内受pH值的调节。天冬氨酸羟化酶抑制低氧诱导因子1表达,参与低氧诱导因子1靶基因的激活,在肾脏中,天冬氨酸羟化酶在肾小管的远端高度表达,但这也限制了低氧诱导因子1α靶基因在肾小管中的表达。在对氧依赖性的生化分析中,天冬氨酸羟化酶耐氧性强于脯氨酸羟化酶。这一发现表明,随着O2含量的降低,脯氨酸羟化酶活性会先于天冬氨酸羟化酶降低,而天冬氨酸羟化酶仍可调控低氧诱导因子1表达。当氧体积分数处于极低水平时才能完全抑制脯氨酸羟化酶和天冬氨酸羟化酶,从而最大程度激活低氧诱导因子1反应。虽然已有研究指出,天冬氨酸羟化酶主要抑制低氧诱导因子1的表达[6],甚至可阻碍相关低氧诱导因子1靶基因转录的激活,但也有研究表示天冬氨酸羟化酶可能具有除低氧诱导因子1调控以外的生理功能。尽管有这些见解,但是目前对天冬氨酸羟化酶在介导低氧诱导因子1转录中的作用还没有完全了解。大多数动物,包括人类,都具有控制低氧诱导因子转录的天冬氨酸羟化酶。综上所述,以上研究进展开辟了针对人类氧感知途径治疗干预的可能性。 2.3 低氧训练介导低氧诱导因子1α表达改善骨骼肌对葡萄糖的代谢 细胞对缺氧的反应很大程度上是因为低氧诱导因子1α激活产生的,该反应可在包括骨骼肌在内的大多数组织中表达。低氧诱导因子的靶基因可增加O2的运输,通过促红细胞生成素介导红细胞生成和血管内皮生长因子诱导血管生成等机制增加氧的转运,或通过上调葡萄糖转运蛋白和糖酵解酶的表达在低O2利用率下改善骨骼肌的功能。不过,低氧诱导因子1α也可能抑制线粒体的氧化代谢和细胞耗氧量,不同训练方式对骨骼肌的刺激反馈产生的效果是不同的,急性运动与耐力运动产生的效果就具有显著差异性。有研究表明,骨骼肌具有显著适应功能需求变化的能力,机体多项指标获得改善,包括毛细血管作用、糖原储存、线粒体密度以及糖酵解通量的改变等[8]。 低氧诱导因子1α作为一种运动干预的潜在治疗靶点,在改善骨骼肌的葡萄糖代谢和胰岛素敏感性中起重要作用。胰岛素抵抗是2型糖尿病发生的标志之一[9]。低氧训练对胰岛素抵抗和2型糖尿病的发生具有改善作用,但目前对其改善作用的潜在机制研究仍然不完整。低氧训练刺激骨骼肌内氧水平降低,导致低氧诱导因子1α诱导基因的表达增强。为响应细胞缺氧刺激,低氧诱导因子1α被激活以调节靶基因的转录,例如糖酵解酶和葡萄糖转运蛋白等[10]。如表1所示,SAKAGAMI等[11]发现,低氧诱导因子1α缺失会影响骨骼肌中葡萄糖的摄取,这表明低氧诱导因子1α在调节骨骼肌葡萄糖代谢中可能发挥作用。虽然在常氧状态下低氧诱导因子1α可在骨骼肌中高度表达,但这也仅证明低氧诱导因子1α在维持肌肉稳态中并不只取决于肌肉细胞中的氧含量。更重要的是,已知低氧条件下的耐力运动在改善2型糖尿病血糖调控方面更有效[11],因此印证了低氧训练对低氧诱导因子1α调控基因表达具有促进作用。当低氧诱导因子1α从骨骼肌中条件性敲除时,葡萄糖转运蛋白4、乳酸脱氢酶A、磷酸甘油酸激酶1等基因的表达会减弱[12]。但是也有研究显示,当骨骼肌中低氧诱导因子1敲除可能对机体氧化能力和耐力表现有利,有研究提出,经过一段时间的运动训练后,低氧诱导因子1及其靶基因在急性运动中的表达可能会减弱[13]。不过,这些低氧诱导因子1α敲除鼠在运动时都表现出不同程度的肌肉损伤,这可能是由于低氧诱导因子1α的缺失降低了丙酮酸脱氢酶复合物抑制剂的水平,从而提高了丙酮酸脱氢酶复合物的活性,该复合物导致了线粒体功能受损,使小鼠在运动时出现肌肉损伤。当机体缺氧时,低氧诱导因子1α通过增加丙酮酸脱氢酶激酶的表达来调节细胞中线粒体O2的消耗,丙酮酸脱氢酶激酶抑制丙酮酸脱氢酶复合物,进而抑制线粒体功能,细胞氧消耗降低。以上研究都为低氧诱导因子1α介导基因的表达进而改善机体内环境提供了理论支撑。另外,研究发现RAB20和TXNIP是骨骼肌中2个受低氧诱导因子1α调控的基因。RAB20缺失调控小鼠骨骼肌对葡萄糖的吸收[13-14],HACKENBECK等[15]报道RAB20是低氧诱导因子1α的直接靶点,运动可显著提高RAB20在骨骼肌中的表达,并且其mRNA的表达与全身胰岛素敏感性呈正相关,RAB20缺失可抑制葡萄糖的摄取和胰岛素的敏感性。另外,因为TXNIP是胰岛素负调节剂,低氧诱导因子1α可影响TXNIP的表达。JOHNSON等[16]也得出结论,肌肉中TXNIP下调可以改善人体胰岛素信号传递和葡萄糖代谢。综上所述,运动过程中低氧诱导因子1α为骨骼肌葡萄糖代谢的调控提供了进一步的理论支撑。未来低氧诱导因子1α信号通路可能作为一种新的治疗靶点在改善人体组织功能中发挥其价值。 "


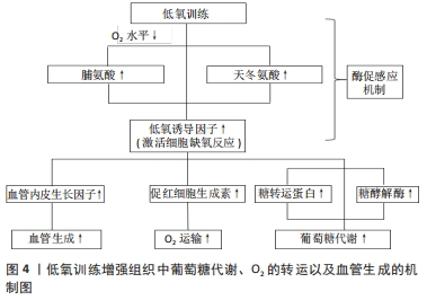
2.4 低氧训练介导低氧诱导因子1α表达增强机体对氧的转运 低氧条件下,当细胞氧张力低时,低氧诱导因子1α稳定诱导红细胞的形成、毛细血管生长和能量代谢基因的转录[17]。与常氧训练比较,低氧训练骨骼肌组织中低氧诱导因子1α mRNA水平更高[3]。低氧诱导因子1α mRNA表达增加也表明了低氧诱导因子1α系统的激活。因此在低氧状态下可更好地促进血管内皮生长因子和肌红蛋白的转录,从而分别增强毛细血管的发育和肌肉纤维内的O2运输[3]。在低氧状态下进行高强度训练可能会刺激氧化酶活性并增加线粒体密度[18-20],而当机体长时间处于低氧环境时会产生肺动脉高压的病理反应并且可能会引起肌纤维萎缩[21-23]。不过也并非所有情况下都会出现该现象,当前有学者提出这可能是低氧剂量引起的。目前常氧训练与低氧训练改善骨骼肌纤维大小、氧化能力和供氧能力的程度尚不清楚。 常氧条件下,低氧诱导因子1α的羟化反应通过脯氨酸羟化酶进行,该酶充当细胞氧张力的传感器,脯氨酸羟化酶调控von Hippel-Lindau(VHL)肿瘤抑制物与低氧诱导因子1α结合,并将其靶向的蛋白酶体降解[21-24]。在缺氧环境中脯氨酸羟化酶羟化反应被抑制,低氧诱导因子1α可稳定表达,与低氧诱导因子1β结合而生成富有活性的复合物。低氧诱导因子1α的激活以类似于运动的方式诱导参与红细胞的生成、血管生成、糖酵解和能量代谢[25-27]。低氧诱导因子1α可通过调控促红细胞生成素的表达增强机体对O2的运输。当然机体对O2的运输不仅与促红细胞生成素的表达有关,还与毛细血管和肌纤维有关。有趣的是,低氧诱导因子1α可通过调控血管内皮生长因子的表达增强毛细血管密度和肌纤维,血管内皮生长因子是已知相应运动最活跃的血管生成因子[28-31],同时血管内皮生长因子也是低氧应答基因,并且在血管内皮生长因子启动子中存在低氧应答元件的共有保守序列[32]。当细胞进入低氧或缺氧的状态,低氧诱导因子1α复合物积累,含有低氧应答元件的靶基因被激活[33],刺激血管内皮生长因子表达上调。当细胞重新暴露于常氧环境后,低氧诱导因子1α降解。因此,低氧诱导因子1α可通过识别低氧应答元件来调控血管内皮生长因子的表达。如表2所示,ZHANG等[34]通过探究miR-140-3p是否参与心肌缺血的分子机制发现,低氧诱导因子1α被确定为miR-140-3p的靶标,缺氧环境下通过低氧诱导因子1α/血管内皮生长因子信号传导促使miR-140-3p的表达上调,导致血管内皮生长因子水平升高。ZHU等[35]也提出miRNA通过靶向调控低氧诱导因子1α影响血管内皮生长因子的表达,进而改变细胞增殖、迁移和血管生成。综上,低氧训练增强机体对O2的吸收与利用是确定的,但其机制还尚未有明确的结论。 "


2.5 低氧训练介导低氧诱导因子1α表达增强机体血管生成 缺氧环境下机体通常需要改变细胞代谢以适应O2可用率降低所造成的代谢反应,这种反应由线粒体氧化磷酸元提供的ATP分解合成[36]。研究发现,低氧诱导因子1α在缺氧条件下的代谢反应中发挥重要作用[37]。低氧诱导因子1α靶基因参与氧转运、糖酵解、葡萄糖转运和血管生成等进程,特别是在血管生成中,低氧诱导因子1α尤为重要[38]。 血管生成是细胞和组织对缺氧的一种极具标志性的适应性反应[39]。低氧诱导因子家族有α和β两个亚基组成,α亚基对氧敏感[40],主要包括低氧诱导因子1α和低氧诱导因子2α。虽然,二者都在血管生成中发挥作用,但根据目前的研究成果显示,低氧诱导因子1α相比于低氧诱导因子 2α更能通过调节血管内皮生长因子来响应缺氧反应[40]。目前的研究表明,机体缺氧条件下的低氧诱导因子1α表达促进血管生成可分为3种方式:第1种为激活血管生成基因及其受体,如血管内皮生长因子[41];第2种是调节促血管生成趋化因子和其受体[42];第3种则是在细胞周期和DNA复制中增强内皮细胞调节基因[43]。以上3种都为低氧诱导因子1α调控血管生成提供了证据。研究证明,低氧诱导因子1α可上调促血管生成素1并下调促血管生成素2,以达到调控血管生成的目的[44]。不止促血管生成因子,促内皮细胞增殖的血管内皮生长因子也是重要的缺氧适应基因,在血管生成中具有重要作用。转录因子低氧诱导因子1α响应血管内皮生长因子的表达。在缺氧条件下,低氧诱导因子1α通过调控血管内皮生长因子的促血管生成活性,控制下游基因表达,最终诱导血管生成[45-47]。而血管内皮生长因子是最为显著的刺激血管生成因子[48]。研究显示,运动诱导的血管生成可以缓解肥胖引起的组织缺氧。如表3所示,AMELN等[49]采用45 min的缺氧单腿伸膝训练,研究发现血管内皮生长因子mRNA显著增强。NAGAHISA等[50]分别采用中等强度和低强度耐力训练6周,两种训练计划都提高了低氧诱导因子1α mRNA的表达和线粒体密度。6周中等训练后,毛血管密度也显著提高;同样的,采用低氧训练干预4周后,研究发现低氧诱导因子1α表达更强,同时血管内皮生长因子mRNA的表达显著提高。 "

| [1] 赵鹏,冯连世.新的低氧训练模式研究及应用进展[J].体育科学, 2005,25(6):71-75,79. [2] WANG GL, SEMENZA GL. Purification and characterization of hypoxia-inducible factor 1. J Biol Chem. 1995;270(3):1230-1237. [3] SEMENZA GL. Regulation of physiological responses to continuous and intermittent hypoxia by hypoxia-inducible factor 1. Exp Physiol. 2006;91(5):803-806. [4] SCHOFIELD CJ, RATCLIFFE PJ. Oxygen sensing by HIF hydroxylases. Nat Rev Mol Cell Biol. 2004;5(5):343-354. [5] BERRA E, BENIZRI E, GINOUVÈS A, et al. HIF prolyl-hydroxylase 2 is the key oxygen sensor setting low steady-state levels of HIF-1alpha in normoxia. EMBO J. 2003;22(16):4082-4090. [6] HAMPTON-SMITH RJ, DAVENPORT BA, NAGARAJAN Y, et al. The conservation and functionality of the oxygen-sensing enzyme Factor Inhibiting HIF (FIH) in non-vertebrates. PLoS One. 2019;14(4):e0216134. [7] DESPLANCHES D, HOPPELER H, LINOSSIER MT, et al. Effects of training in normoxia and normobaric hypoxia on human muscle ultrastructure. Pflugers Arch. 1993;425(3-4):263-267. [8] SUNDBERG CJ. Exercise and training during graded leg ischaemia in healthy man with special reference to effects on skeletal muscle. Acta Physiol Scand Suppl. 1994;615:1-50. [9] GÖRGENS SW, BENNINGHOFF T, ECKARDT K, et al. Hypoxia in Combination With Muscle Contraction Improves Insulin Action and Glucose Metabolism in Human Skeletal Muscle via the HIF-1α Pathway. Diabetes. 2017;66(11):2800-2807. [10] SEMENZA GL. Oxygen sensing, homeostasis, and disease. N Engl J Med. 2011;365(6):537-547. [11] SAKAGAMI H, MAKINO Y, MIZUMOTO K, et al. Loss of HIF-1α impairs GLUT4 translocation and glucose uptake by the skeletal muscle cells. Am J Physiol Endocrinol Metab. 2014;306(9):E1065-1076. [12] MACKENZIE R, MAXWELL N, CASTLE P, et al. Acute hypoxia and exercise improve insulin sensitivity (S(I) (2*)) in individuals with type 2 diabetes. Diabetes Metab Res Rev. 2011;27(1):94-101. [13] MASON SD, HOWLETT RA, KIM MJ, et al. Loss of skeletal muscle HIF-1alpha results in altered exercise endurance. PLoS Biol. 2004;2(10): e288. [14] RICHARDSON RS, WAGNER H, MUDALIAR SR, et al. Exercise adaptation attenuates VEGF gene expression in human skeletal muscle. Am J Physiol Heart Circ Physiol. 2000;279(2):H772-778. [15] HACKENBECK T, HUBER R, SCHIETKE R, et al. The GTPase RAB20 is a HIF target with mitochondrial localization mediating apoptosis in hypoxia. Biochim Biophys Acta. 2011;1813(1):1-13. [16] JOHNSON ML, DISTELMAIER K, LANZA IR, et al. Mechanism by Which Caloric Restriction Improves Insulin Sensitivity in Sedentary Obese Adults. Diabetes. 2016;65(1):74-84. [17] EGAN B, ZIERATH JR. Exercise metabolism and the molecular regulation of skeletal muscle adaptation. Cell Metab. 2013;17(2):162-184. [18] VOGT M, PUNTSCHART A, GEISER J, et al. Molecular adaptations in human skeletal muscle to endurance training under simulated hypoxic conditions. J Appl Physiol (1985). 2001;91(1):173-182. [19] BURGOMASTER KA, HUGHES SC, HEIGENHAUSER GJ, et al. Six sessions of sprint interval training increases muscle oxidative potential and cycle endurance capacity in humans. J Appl Physiol (1985). 2005;98(6):1985-1990. [20] MELISSA L, MACDOUGALL JD, TARNOPOLSKY MA, et al. Skeletal muscle adaptations to training under normobaric hypoxic versus normoxic conditions. Med Sci Sports Exerc. 1997;29(2):238-243. [21] TERRADOS N, JANSSON E, SYLVÉN C, et al. Is hypoxia a stimulus for synthesis of oxidative enzymes and myoglobin? J Appl Physiol (1985). 1990;68(6):2369-2372. [22] GREEN HJ, SUTTON JR, CYMERMAN A, et al. Operation Everest II: adaptations in human skeletal muscle. J Appl Physiol (1985). 1989; 66(5):2454-2461. [23] MACDOUGALL JD, GREEN HJ, SUTTON JR, et al. Operation Everest II: structural adaptations in skeletal muscle in response to extreme simulated altitude. Acta Physiol Scand. 1991;142(3):421-427. [24] MIZUNO M, SAVARD GK, ARESKOG NH, et al. Skeletal muscle adaptations to prolonged exposure to extreme altitude: a role of physical activity? High Alt Med Biol. 2008;9(4):311-317. [25] MAXWELL PH, WIESENER MS, CHANG GW, et al. The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature. 1999;399(6733):271-275. [26] TAYLOR CT. Mitochondria and cellular oxygen sensing in the HIF pathway. Biochem J. 2008;409(1):19-26. [27] MAHONEY DJ, PARISE G, MELOV S, et al. Analysis of global mRNA expression in human skeletal muscle during recovery from endurance exercise. FASEB J. 2005;19(11):1498-1500. [28] SCHMUTZ S, DÄPP C, WITTWER M, et al. Endurance training modulates the muscular transcriptome response to acute exercise. Pflugers Arch. 2006;451(5):678-687. [29] AMARAL SL, LINDERMAN JR, MORSE MM, et al. Angiogenesis induced by electrical stimulation is mediated by angiotensin II and VEGF. Microcirculation. 2001;8(1):57-67. [30] BREEN EC, JOHNSON EC, WAGNER H, et al. Angiogenic growth factor mRNA responses in muscle to a single bout of exercise. J Appl Physiol (1985). 1996;81(1):355-361. [31] GUSTAFSSON T, PUNTSCHART A, KAIJSER L, et al. Exercise-induced expression of angiogenesis-related transcription and growth factors in human skeletal muscle. Am J Physiol. 1999;276(2):H679-685. [32] LLOYD PG, PRIOR BM, YANG HT, et al. Angiogenic growth factor expression in rat skeletal muscle in response to exercise training. Am J Physiol Heart Circ Physiol. 2003;284(5):H1668-1678. [33] FORSYTHE JA, JIANG BH, IYER NV, et al. Activation of vascular endothelial growth factor gene transcription by hypoxia-inducible factor 1. Mol Cell Biol. 1996;16(9):4604-4613. [34] ZHANG L, LUO X, CHEN F, et al. LncRNA SNHG1 regulates cerebrovascular pathologies as a competing endogenous RNA through HIF-1α/VEGF signaling in ischemic stroke. J Cell Biochem. 2018;119(7): 5460-5472. [35] ZHU Y, WANG Y, JIA Y, et al. Roxadustat promotes angiogenesis through HIF-1α/VEGF/VEGFR2 signaling and accelerates cutaneous wound healing in diabetic rats. Wound Repair Regen. 2019;27(4):324-334. [36] HOFER T, WENGER H, GASSMANN M. Oxygen sensing, HIF-1alpha stabilization and potential therapeutic strategies. Pflugers Arch. 2002; 443(4):503-507. [37] THOMAS LW, ASHCROFT M. Exploring the molecular interface between hypoxia-inducible factor signalling and mitochondria. Cell Mol Life Sci. 2019;76(9):1759-1777. [38] SEMENZA GL, JIANG BH, LEUNG SW, et al. Hypoxia response elements in the aldolase A, enolase 1, and lactate dehydrogenase A gene promoters contain essential binding sites for hypoxia-inducible factor 1. J Biol Chem. 1996;271(51):32529-32537. [39] WENGER RH. Cellular adaptation to hypoxia: O2-sensing protein hydroxylases, hypoxia-inducible transcription factors, and O2-regulated gene expression. FASEB J. 2002;16(10):1151-1162. [40] ZIMNA A, KURPISZ M. Hypoxia-Inducible Factor-1 in Physiological and Pathophysiological Angiogenesis: Applications and Therapies. Biomed Res Int. 2015;2015:549412. [41] KIERANS SJ, TAYLOR CT. Regulation of glycolysis by the hypoxia-inducible factor (HIF): implications for cellular physiology. J Physiol. 2021;599(1):23-37. [42] CARROLL VA, ASHCROFT M. Role of hypoxia-inducible factor (HIF)-1alpha versus HIF-2alpha in the regulation of HIF target genes in response to hypoxia, insulin-like growth factor-I, or loss of von Hippel-Lindau function: implications for targeting the HIF pathway. Cancer Res. 2006;66(12):6264-6270. [43] GREIJER AE, VAN DER GROEP P, KEMMING D, et al. Up-regulation of gene expression by hypoxia is mediated predominantly by hypoxia-inducible factor 1 (HIF-1). J Pathol. 2005;206(3):291-304. [44] CERADINI DJ, KULKARNI AR, CALLAGHAN MJ, et al. Progenitor cell trafficking is regulated by hypoxic gradients through HIF-1 induction of SDF-1. Nat Med. 2004;10(8):858-864. [45] MANALO DJ, ROWAN A, LAVOIE T, et al. Transcriptional regulation of vascular endothelial cell responses to hypoxia by HIF-1. Blood. 2005; 105(2):659-669. [46] KELLY BD, HACKETT SF, HIROTA K, et al. Cell type-specific regulation of angiogenic growth factor gene expression and induction of angiogenesis in nonischemic tissue by a constitutively active form of hypoxia-inducible factor 1. Circ Res. 2003;93(11):1074-1081. [47] LEE P, CHANDEL NS, SIMON MC. Cellular adaptation to hypoxia through hypoxia inducible factors and beyond. Nat Rev Mol Cell Biol. 2020; 21(5):268-283. [48] CHEN L, ENDLER A, SHIBASAKI F. Hypoxia and angiogenesis: regulation of hypoxia-inducible factors via novel binding factors. Exp Mol Med. 2009;41(12):849-857. [49] AMELN H, GUSTAFSSON T, SUNDBERG CJ, et al. Physiological activation of hypoxia inducible factor-1 in human skeletal muscle. FASEB J. 2005; 19(8):1009-1011. [50] NAGAHISA H, MUKAI K, OHMURA H, et al. Effect of High-Intensity Training in Normobaric Hypoxia on Thoroughbred Skeletal Muscle. Oxid Med Cell Longev. 2016;2016:1535367. |
| [1] | Fang Xingyan, Tian Zhenli, Zhao Zheyi, Wen Ping, Xie Tingting. Effects of sodium arsenite on human umbilical vein endothelial cell injury and sphingosine kinases 1/sphingosine 1-phosphate signaling axis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [2] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [3] | Cai Zhihao, Xie Zhaoyong. Femoral neck anteversion measurement assessment: how to establish a unified method and standard [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1448-1454. |
| [4] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [5] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [6] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [7] | Xu Cong, Zhao He, Sun Yan. Regeneration of facial nerve injury repaired by biomaterial nerve conduits [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1089-1095. |
| [8] | Chen Shisong, Liu Xiaohong, Xu Zhiyun. Current status and prospects of bioprosthetic heart valves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1096-1102. |
| [9] | Lu Di, Zhang Cheng, Duan Rongquan, Liu Zongxiang. Osteoinductive properties of calcium phosphate ceramic bone repair materials [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1103-1109. |
| [10] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [11] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [12] | Xue Ting, Zhang Xinri, Kong Xiaomei. Mesenchymal stem cell therapy for pneumoconiosis using nanomaterials combined with multi-modal molecular imaging [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1133-1140. |
| [13] | Zhang Qing, Gao Chunlan, Yu Feifei, Zhang Zhenghao, Ma Fang, Gao Yuan, Li Guizhong, Jiang Yideng, Ma Shengchao. Ephrin A receptor 2 DNA methylation increases in pancreatic beta cell apoptosis induced by homocysteine [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 714-719. |
| [14] | Zhou Yinan, Yang Kun, Jiang Jiayi, Zhang Sizhuo, Zhu Yingwen. The relationship between glucose transporter 1/4 gene polymorphisms and athletic ability [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 745-750. |
| [15] | Li Long, Li Guangdi, Shi Hao, Deng Keqi. Circular RNA as a competing endogenous RNA is involved in the regulation of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 751-757. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||