Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (10): 1610-1617.doi: 10.12307/2022.978
Previous Articles Next Articles
Application and effect of extracellular vesicles in cardiac repair after myocardial infarction
Yue Feifei1, Song Yu1, Wang Xiaobei2, Wang Lin1, 2
- 1Research Center of Tissue Engineering and Regenerative Medicine, 2Department of Laboratory Medicine, Union Hospital Affiliated to Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430022, Hubei Province, China
-
Received:2021-12-31Accepted:2022-02-16Online:2023-04-08Published:2022-09-09 -
Contact:Wang Xiaobei, MD, Associate chief physician, Department of Laboratory Medicine, Union Hospital Affiliated to Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430022, Hubei Province, China Wang Lin, MD, Professor, Research Center of Tissue Engineering and Regenerative Medicine, and Department of Laboratory Medicine, Union Hospital Affiliated to Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430022, Hubei Province, China -
About author:Yue Feifei, Master candidate, Research Center of Tissue Engineering and Regenerative Medicine, Union Hospital Affiliated to Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430022, Hubei Province, China
CLC Number:
Cite this article
Yue Feifei, Song Yu, Wang Xiaobei, Wang Lin. Application and effect of extracellular vesicles in cardiac repair after myocardial infarction[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(10): 1610-1617.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
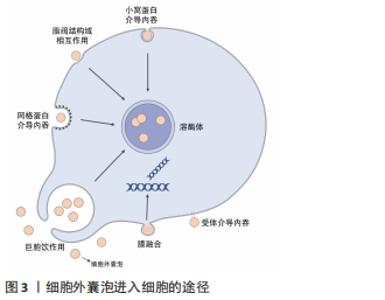
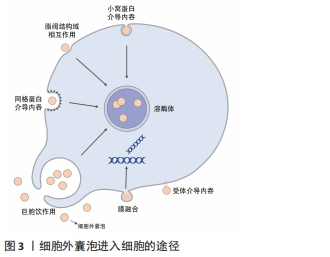
2.1 细胞外囊泡的主要功能 细胞外囊泡是一种由各种类型细胞自然释放到细胞外环境的有脂质双分子层包裹的纳米级结构[2]。在传统意义上,根据细胞外囊泡的大小可以进一步将其分为3类:①外泌体,直径50-100 nm,由多泡体胞吐形成;②微囊泡,直径100-1 000 nm,由质膜出芽形成;③凋亡小体,直径1-5 μm,细胞死亡时释放[3]。目前尚未对特定细胞外囊泡亚型的标记达成共识,因此国际细胞外囊泡学会(International Society for Extracellular Vesicles,ISEV)鼓励作者在文章中更具体地定义细胞外囊泡,例如大小尺寸、细胞来源、产生途径或生化成分(CD63+/CD81+等),避免直接使用外泌体和微囊泡这些术语[2]。 细胞外囊泡中可储存亲代细胞的脂质、蛋白质、核酸,这些内容物的含量和种类在不同的细胞和环境中有很大的区别,特殊的成分可以直接影响细胞外囊泡的行为和功能。细胞外囊泡介导不同细胞之间的通讯,它们可以通过受体介导的内吞作用、网格蛋白的相互作用、脂阀相互作用、吞噬作用以及巨胞饮内化进入细胞,并最终被溶酶体降解,释放内容物,除此之外细胞外囊泡也可以直接与靶细胞的细胞膜融合释放内容物[4],见图3。细胞外囊泡表面富集的蛋白种类可以帮助其靶向不同类型的细胞,例如癌细胞释放的细胞外囊泡表面富含整合素α6β4和αvβ5,能够分别靶向肺成纤维细胞和库普弗细胞[5];细胞外囊泡的脂质成分也会影响其对受体细胞的靶向,例如肿瘤细胞的细胞外囊泡通常富含磷脂酰丝氨酸,可以被巨噬细胞识别并清除[5]。 "

人体各种类型的细胞都能产生细胞外囊泡,心脏组织的细胞也不例外,这些细胞外囊泡中所含有的一些关键蛋白质和RNA,在心肌梗死的发生和发展中发挥重要作用。例如,缺氧可以触发心肌细胞来源细胞外囊泡中肿瘤坏死因子α的包装,刺激炎症反应[6]。除了蛋白质外,细胞外囊泡中的多种miRNA与心肌梗死的发展密切相关,例如心肌损伤引起巨噬细胞激活,导致巨噬细胞来源细胞外囊泡中miR-155的富集,其靶向心脏成纤维细胞导致纤维化和炎症的发生[7]。 这些基于细胞外囊泡的胞间通讯都是治疗心肌梗死的潜在靶点——限制不利于心肌修复的细胞外囊泡产生作用,发挥细胞外囊泡在促进血管生成、抑制炎症反应和纤维化中的益处。目前已有不少利用细胞外囊泡促进心肌修复的研究,治疗思路主要有2种:①直接使用从细胞获得的细胞外囊泡,例如间充质干细胞、胚胎干细胞、诱导多能干细胞等干细胞来源细胞外囊泡或分化成熟的细胞来源细胞外囊泡;②使用改造的细胞外囊泡,人工对细胞外囊泡进行货物装载或修饰以达到靶向心脏组织和促进心肌修复的目的。 2.2 细胞外囊泡自身用于心肌修复的潜力 "


2.2.1 干细胞来源细胞外囊泡在心肌梗死治疗中的作用 (1)间充质干细胞来源的细胞外囊泡:间充质干细胞一直以来都是心肌梗死后细胞治疗的常用选择,但有研究发现间充质干细胞移植进入心肌梗死区域后存活率很低,这说明间充质干细胞的治疗作用可能不是因为分化成心肌细胞[8]。2010年就有研究证实间充质干细胞分泌的细胞外囊泡(半径55-65 nm,表达CD81,CD9和ALIX细胞外囊泡相关蛋白)是减少小鼠心肌缺血再灌注损伤后梗死面积的重要因素[9]。随后,研究者探究了间充质干细胞来源细胞外囊泡修复心肌组织的机制,发现间充质干细胞来源细胞外囊泡可以有效减少心肌细胞凋亡,提高腺嘌呤核苷三磷酸和还原型辅酶Ⅰ水平,减少氧化应激,从而促进梗死后心功能的恢复[10]。间充质干细胞来源细胞外囊泡也可以激活磷脂酰肌醇3激酶/蛋白激酶B(phosphatidylin-ositol-3-kinase/protein kinase B,PI3K/Akt)信号通路,以及抑制c-Jun氨基末端激酶(c-Jun N-terminal kinase,c-JNK)来减少受损心肌细胞的凋亡[11]。除此之外,更多进一步的研究发现间充质干细胞来源细胞外囊泡中装载的一些miRNA可以抑制心肌细胞凋亡,WEN等[12]发现间充质干细胞来源细胞外囊泡中的miR-144可以抑制促凋亡蛋白磷酸酶与张力蛋白同源物的表达,从而激活PI3K/AKT信号通路,抑制缺血心肌细胞的凋亡。间充质干细胞来源细胞外囊泡中还含有miR-21和miR-486-5p,它们也通过调节PI3K/AKT信号通路来减少缺氧心肌细胞的凋亡[10]。另一项研究发现间充质干细胞来源细胞外囊泡中的miR-25-3p可以靶向促凋亡基因FASL和PTEN,降低它们的蛋白水平,使心肌细胞得以存活[13]。 间充质干细胞来源细胞外囊泡不仅可以抑制缺氧心肌细胞的凋亡,还能够通过诱导心肌梗死后的血管生成和发挥抗炎作用从而促进心功能的恢复。TENG等[14]发现间充质干细胞来源细胞外囊泡治疗后,梗死区域新生毛细血管明显增多;XU等[15]发现使用间充质干细胞来源细胞外囊泡处理心肌细胞可以提高乳鼠心肌细胞中血管内皮生长因子、碱性成纤维细胞生长因子和肝细胞生长因子的表达水平,这可能与促心肌梗死区域血管生成相关。此外,心肌梗死引起的慢性炎症反应不利于心肌组织的愈合或修复,反而会扩大心肌损伤。在心肌梗死后的炎症期,中性粒细胞被损伤相关分子模式、趋化因子等募集到梗死区域,并释放蛋白水解酶和活性氧损伤心肌细胞,在受损心肌细胞被清除后,单核细胞、调节性T细胞和M2型巨噬细胞参与进来抑制炎症反应,并通过促进血管生成和减少胶原沉积来帮助心脏修复[16-17]。已有研究证实,使用间充质干细胞来源细胞外囊泡治疗心肌梗死后,梗死区域炎性细胞浸润明显减少[14,18],且能促进巨噬细胞由M1型向M2型转化,使抗炎细胞因子白细胞介素10水平明显上升,这种巨噬细胞的极化与间充质干细胞来源细胞外囊泡中含有的miR-182有关,miR-182通过靶向Toll样受体4(toll like receptor 4,TLR4)/核因子κB(nuclear factor kappa-B,NF-κB)/PI3K/Akt信号级联介导巨噬细胞转化为M2型[18]。M2型巨噬细胞与组织纤维化有关,心肌梗死后的心脏纤维化反应在减少心室壁破裂中发挥重要作用,也是预防随后心力衰竭的关键,M2型巨噬细胞分泌的白细胞介素10减少会导致组织重塑不利和更严重的心脏纤维化[19]。另一项研究发现,间充质干细胞来源细胞外囊泡中富集的miR-4732-3p可以抑制成纤维细胞分化为肌成纤维细胞,防止肌成纤维细胞产生过多含胶原蛋白的细胞外基质,从而减少心肌梗死区域的纤维化。除此之外,miR-4732-3p还可以通过促进缺氧损伤后新生大鼠心肌细胞表达间隙连接蛋白43(connexin 43)来维持心肌细胞的收缩能力[20]。 上述研究都使用了正常培养的间充质干细胞来源细胞外囊泡,但还有一些研究通过对间充质干细胞进行缺氧处理,获得含有促进心肌修复的蛋白或核酸的细胞外囊泡。SUN等[21]发现缺氧条件下的间充质干细胞表达长链非编码RNA(long noncoding RNA,LncRNA)UCA1,其分泌含有lncRNA UCA1的细胞外囊泡能够靶向miR-873-5p,减少miR-873-5p对X-连锁凋亡抑制蛋白(X-linked inhibitor of apoptosis protein,XIAP)的抑制,从而减少缺氧心肌细胞的凋亡。在另一项研究中,JUNG等[22]在低氧环境下培养人诱导多能干细胞分化而来的心肌细胞(human-induced pluripotent stem cell-derived cardiomyocytes,iCMs),获得了高表达miR-106a-363的细胞外囊泡,其可以抑制Notch3的表达,刺激心肌细胞进入细胞周期,诱导心肌细胞增殖,有助于改善心肌梗死后的心功能。 综上,间充质干细胞来源细胞外囊泡不仅可以减少心肌细胞的凋亡,还能帮助促进血管生成,调节免疫反应,促进随后的心肌修复。 (2)胚胎干细胞来源的细胞外囊泡:胚胎干细胞也在治疗心肌梗死相关研究中有广泛的应用,但与间充质干细胞一样,胚胎干细胞在缺血心肌中仍然难以存活和驻留,并且胚胎干细胞的植入还存在形成畸胎瘤的风险。已有研究使用胚胎干细胞来源细胞外囊泡对心肌梗死小鼠模型进行治疗,有助于心肌细胞存活与增殖,促进新生血管的生成,减少梗死后纤维化,从而帮助心功能的改善[23]。胚胎干细胞来源细胞外囊泡中70%的miRNA属于miR-290家族,这些胚胎干细胞特异性高表达的miR-290有助于细胞进入细胞周期,这一发现为后续的研究提供了思路。但由于胚胎干细胞涉及的伦理问题,目前仅有少数研究在心肌梗死的治疗中使用了胚胎干细胞来源细胞外囊泡,大部分研究选择将胚胎干细胞分化为其他细胞后再提取细胞外囊泡[24-25]。 (3)诱导多能干细胞来源的细胞外囊泡:自从发现体细胞可以重编程为诱导多能干细胞以来,为了避免与胚胎干细胞有关的免疫排斥和伦理问题,越来越多的心肌修复相关研究开始把目光放到诱导多能干细胞上[26]。与间充质干细胞及胚胎干细胞类似,诱导多能干细胞来源细胞外囊泡在治疗心肌梗死方面也有与其母细胞相似的作用,ADAMIAK等[27]发现诱导多能干细胞来源细胞外囊泡比诱导多能干细胞更安全,甚至能更有效地帮助心脏修复。他们的研究显示诱导多能干细胞来源细胞外囊泡能有效缓解心肌梗死引起的左心室功能障碍,并能在梗死一段时间后改善左心室的整体功能;相比于心肌内直接注射诱导多能干细胞,注射诱导多能干细胞来源细胞外囊泡能更好地维持左心室的功能。与有效改善心肌梗死后心功能这一治疗效果相符合的是,诱导多能干细胞来源细胞外囊泡还能阻止左心室重构、间质纤维化和左心室肥大。同时,诱导多能干细胞来源细胞外囊泡也有与间充质干细胞来源细胞外囊泡相同的功能——促进梗死边缘区及非缺血区毛细血管生成、减少缺血心肌细胞的凋亡[27]。诱导多能干细胞来源细胞外囊泡所具有的这些治疗作用,也与其所携带的一些分子相关,例如诱导多能干细胞来源细胞外囊泡中miR-21及miR-210水平显著升高,这两种miRNA能有效保护缺氧心肌细胞,防止其凋亡[28-30]。而且诱导多能干细胞来源细胞外囊泡中相关miRNAs可能在心脏组织修复以及血管生成中起到正向作用[27,31-32]。因此,诱导多能干细胞来源细胞外囊泡应用于心肌梗死治疗的潜力是不可忽视的,见表1。 "

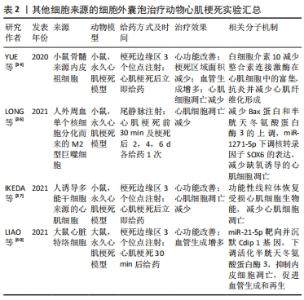
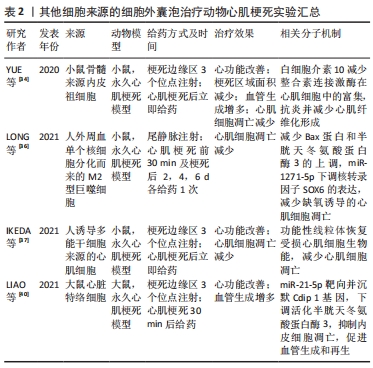
2.2.2 其他类型细胞来源的细胞外囊泡 除了以上提到的干性细胞外,内皮细胞、免疫细胞以及诱导多能干细胞分化形成的心肌细胞等都能产生细胞外囊泡。内皮祖细胞治疗心肌梗死有良好的结果[33],YUE等[34]发现内皮祖细胞来源细胞外囊泡在心肌梗死小鼠模型中也具有改善心功能、抑制细胞凋亡、减小梗死区域瘢痕面积、促进血管生成等功能,而这些治疗效果与内皮祖细胞来源细胞外囊泡中的白细胞介素10相关,由于整合素连接激酶在心脏中有促心肌细胞肥厚、促纤维化以及促炎的不利作用,该研究发现白细胞介素10可以减少整合素连接激酶在心肌细胞中的富集。除此之外,免疫细胞如骨髓树突状细胞来源细胞外囊泡,可以通过调节免疫反应促进梗死后的心肌修复[35]。与此同时,M2型巨噬细胞来源细胞外囊泡在保护心肌细胞免受缺氧诱导的凋亡中起到重要作用[36],M2型巨噬细胞来源细胞外囊泡不仅可以逆转缺氧导致的Bax蛋白和半胱天冬氨酸蛋白酶3的上调,减少心肌细胞凋亡,还能通过其携带的miR-1271-5p下调核转录因子SOX6的表达,减少缺氧诱导的心肌细胞凋亡,因此巨噬细胞来源细胞外囊泡在心肌梗死治疗中发挥的作用不可小觑。由于缺血性心脏病常有线粒体结构异常和ATP减少的表现,从而导致能量生成不足、心肌细胞收缩异常、心肌功能障碍,最终引起代偿性心力衰竭,因此IKEDA等[37]决定从恢复心肌细胞生物能这一方向入手,他们发现人诱导多能干细胞分化而来的心肌细胞分泌的细胞外囊泡中含有大量功能性线粒体,并且该细胞外囊泡可以成功地将功能性线粒体转移到缺氧损伤的心肌细胞中,恢复心肌细胞的生物能,从而减少了心肌细胞的凋亡,达到改善心功能的目的。除了心肌细胞,人诱导多能干细胞也可以分化为内皮细胞、平滑肌细胞,GAO等[38-39]以2∶1∶1的比例混合培养人诱导多能干细胞分化而来的心肌细胞、内皮细胞和平滑肌细胞,并从细胞培养基中获得细胞外囊泡,使用这种人诱导多能干细胞来源细胞联合培养产生的细胞外囊泡对猪心肌梗死模型进行治疗,有效地减少了心肌细胞的凋亡,促进了ATP的产生,同时促进了梗死边缘区的血管生成,使心功能得到了恢复。该细胞外囊泡的疗效与直接以相同比例移植以上3种细胞的疗效相同,但比起基于细胞的产品,细胞外囊泡更便于运输和保存。LIAO等[40]选择了一种新发现的心脏间质细胞群——心脏特络细胞,在此之前已有研究证实在心肌梗死的大鼠心脏内移植治疗性心脏特络细胞可以通过促进血管生成来降低心脏纤维化,改善心功能。而在缺血再灌注损伤中,内皮细胞比心肌细胞更容易受到损伤[41],缺血缺氧诱导的心脏微血管内皮细胞及其他内皮细胞的凋亡先于心肌细胞,在这种环境中内皮细胞释放可溶性促凋亡递质将进一步促进心肌细胞的凋亡[42],因此LIAO等[40]认为心肌梗死时保护内皮细胞凋亡,将是促进梗死区域血管再生和实现更有效的心肌再生的一种方法。该研究发现心脏特络细胞来源细胞外囊泡含有的miR-21-5p可以靶向并沉默 Cdip 1(cell death Inducing P53 target 1)基因,从而下调活化半胱天冬氨酸蛋白酶3,抑制缺血和缺氧条件下内皮细胞的凋亡,促进心肌梗死后血管生成和再生,从而在心肌梗死中发挥治疗作用,见表2。"
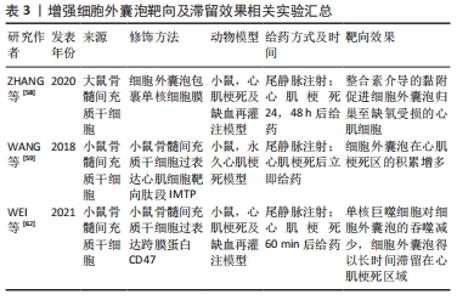
2.3.1 在细胞外囊泡中搭载治疗性药物以增强细胞外囊泡的疗效 目前,已经有一些研究使用细胞外囊泡搭载蛋白用于心肌修复并取得了可观的疗效。由于Notch1基因表达抑制会加重缺血状态下的心肌损伤[45],而Notch1蛋白可以启动心肌损伤后的心肌修复,并且在血管生成中发挥重要作用。XUAN等[46]通过在间充质干细胞中过表达Notch1胞内结构域(N1ICD)来获得含有N1ICD的细胞外囊泡,并将细胞外囊泡注射于心肌梗死小鼠梗死边缘区来进行治疗,结果显示心脏梗死面积减小、纤维化减弱、心功能改善,且效果优于单独使用间充质干细胞来源细胞外囊泡的治疗,进一步发现N1ICD的过表达可以使心肌组织中赖氨酸酰氧化酶同源物2(lysyl oxidase homolog-2,LOXL2)和二聚糖(Biglycan)的表达增加,从而促进缺血损伤后梗死边缘区的血管生成,减少心肌细胞的凋亡。MA等[47]在间充质干细胞中过表达Akt,使间充质干细胞来源细胞外囊泡中富集血小板来源生长因子D,从而促进内皮细胞的增殖与迁移,同样能使心肌梗死边缘区血管生成增多,以达到减少心肌细胞凋亡、促进心肌再生、改善心功能的目的。另一项研究在间充质干细胞中过表达基质细胞衍生因子1以获得含有基质细胞衍生因子1的细胞外囊泡,其对心肌梗死小鼠的治疗效果优于未搭载基质细胞衍生因子1的细胞外囊泡,心肌细胞中抗凋亡因子Bcl-2蛋白表达增多,而促凋亡相关因子Bax蛋白表降低,从而抑制受损心肌细胞的凋亡。基质细胞衍生因子1有促进心肌细胞表达血管内皮生长因子的作用,从而可以促进梗死边缘区微血管再生[48]。还有一项研究以减少心肌梗死后的病理性重构为切入点,在间充质干细胞中过表达基质金属蛋白酶抑制因子2,抑制基质金属蛋白酶2和基质金属蛋白酶9的激活,从而减少心肌细胞外基质中胶原的沉积和纤维化,防止心肌病理性重构[49]。 另一些研究选择在细胞外囊泡中搭载有助于心肌修复的核酸来治疗心肌梗死。MAO等[50]在间充质干细胞来源细胞外囊泡中搭载能抑制软骨细胞凋亡并诱导其增殖的KLF3-AS1,研究结果显示KLF3-AS1 能竞争性结合miR-138-5p,从而减少miR-138-5p对去乙酰化酶Sirtuin 1(Sirt1)表达的抑制,使缺氧心肌细胞的焦亡减少,能获得比单纯使用间充质干细胞来源细胞外囊泡治疗心肌梗死更好的疗效。LIU等[51]在脂肪间充质干细胞中过表达miR-93-5p,然后分离出含有miR-93-5p的细胞外囊泡,miR-93-5p可以特异性结合自噬相关蛋白7 mRNA和Toll样受体4 mRNA的3’端非翻译区来抑制这两种蛋白的表达,从而抑制缺氧诱导的自噬并减轻炎症反应。该研究发现使用负载miR-93-5p的脂肪间充质干细胞来源细胞外囊泡对心肌梗死模型小鼠进行治疗,可以比仅使用脂肪间充质干细胞来源细胞外囊泡能更有效地抑制心肌细胞的自噬,减少心肌梗死面积。WEI等[52]研究发现miR-181a可以通过减少转录因子激活蛋白1的组成成分之一——c-Fos蛋白的表达来促进调节性T细胞的发育,该研究使用搭载了miR-181a的间充质干细胞来源细胞外囊泡对心肌梗死小鼠进行治疗,结果显示心肌梗死面积减小,心功能得到改善且梗死区域的炎性细胞浸润减少,促炎细胞因子白细胞介素6、肿瘤坏死因子α减少,抗炎细胞因子白细胞介素10增多。另一项研究在间充质干细胞中过表达miR-101a后获得细胞外囊泡,miR-101a可以靶向转化生长因子β和Wnt信号通路,减少内皮细胞中转化生长因子β和Ⅰ型胶原α1的表达,从而抑制纤维化[53],结果显示,搭载有miR-101a的细胞外囊泡能更显著地减少心脏梗死区域胶原蛋白的沉积和减小梗死面积[54]。除此之外,SONG等[55]通过人胚胎肾细胞系HEK293T获得细胞外囊泡,在细胞外囊泡中搭载miR-21来靶向缺氧心肌细胞的PDCD4/AP-1,抑制缺氧心肌细胞的凋亡,并激活PTEN/Akt信号通路,刺激内皮细胞表达血管内皮生长因子从而达到促进心功能恢复的目的。间充质干细胞来源细胞外囊泡中含有的miR-486-5p可以保护缺氧心肌细胞防止其凋亡,LI等[56]发现对间充质干细胞进行缺氧处理后,间充质干细胞分泌的细胞外囊泡中miR-486-5p表达上升,由于miR-486-5p可以靶向成纤维细胞中的基质金属蛋白酶19来抑制基质金属蛋白酶19对血管内皮生长因子A的切割,所以含有miR-486-5p的细胞外囊泡能有效诱导血管生成,进一步对心肌梗死模型小鼠和非人类灵长类动物进行治疗,结果显示过表达miR-486-5p的间充质干细胞来源细胞外囊泡可以促进血管生成和心功能恢复,而不会增加心律不齐这一并发症的发生概率。 2.3.2 修饰细胞外囊泡的脂质双分子层以增强细胞外囊泡的靶向性和滞留能力 在动物模型中,静脉注射全身给予细胞外囊泡后,细胞外囊泡会很快到达并被滞留在肝脏、脾和肺,随后被清除[57]。因此,除了在细胞外囊泡中搭载治疗性药物外,还有一些研究对细胞外囊泡的脂质双分子层进行修饰,以增强其对心脏或心肌细胞的靶向性和增加细胞外囊泡在心脏的滞留时间。心肌梗死发生后的30 min内,会有大量的炎性单核细胞被招募至心脏,在梗死边缘区和梗死区分化为巨噬细胞,ZHANG等[58]使用融合挤压法将单核细胞膜包覆在间充质干细胞来源细胞外囊泡上,再通过尾静脉注射的方式对心肌梗死模型小鼠进行治疗,研究发现缺血的心肌细胞膜上细胞间黏附分子1表达增多,引起整合素介导的黏附,单核细胞膜修饰的细胞外囊泡表面富集的整合素Mac-1和LFA-1与细胞间黏附分子1相互作用,使细胞外囊泡归巢至缺氧后受损的心肌细胞,从而达到更好的治疗效果。WANG等[59]在间充质干细胞内同时过表达靶向缺血心肌细胞的肽段IMTP(CSTSMLKAC)和细胞外囊泡富集膜蛋白(Lamp2b),使IMTP能表达在间充质干细胞分泌的细胞外囊泡膜上,并通过尾静脉注射细胞外囊泡的方式对心肌梗死小鼠进行治疗,研究结果显示表达了IMTP的细胞外囊泡在心肌梗死区的积累高于未经修饰的细胞外囊泡,使间充质干细胞来源细胞外囊泡本身的治疗作用得到更好的发挥。除了靶向能力,细胞外囊泡在心脏的滞留时间也会在很大程度上影响细胞外囊泡的治疗效果[60-61]。WEI等[62]在间充质干细胞中过表达跨膜蛋白CD47,使间充质干细胞分泌的细胞外囊泡膜表面修饰CD47,通过CD47和信号调节蛋白α的结合,可以抑制单核巨噬细胞对细胞外囊泡的吞噬作用,研究发现未经修饰的细胞外囊泡由尾静脉注射进入小鼠体内后,只能在血浆中维持3 h,而修饰了CD47的细胞外囊泡在注射后4 h仍能在血浆中检测到,并且在注射8 h后仍大量富集于梗死心肌处,这使心肌梗死小鼠的心功能得到更好地恢复。以上提到的在细胞外囊泡表面修饰靶向蛋白的研究多使用基因操作的方式,但基因操作可能改变细胞外囊泡的生物活性,难以控制其表面靶向表位的密度。目前已有一些物理或化学方法可以控制细胞外囊泡表面修饰的内容[63-64],例如物理方式将修饰有所需蛋白和肽段的脂质掺入细胞外囊泡的膜成分中,或使用无铜催化的点击化学反应将肽段连接到细胞外囊泡表面的官能团上,这些方式可以在细胞外囊泡的纯化步骤中进行,因此更适用于临床转化,见表3。 "

| [1] REED GW, ROSSI JE, CANNON CP. Acute myocardial infarction. Lancet. 2017;389(10065):197-210. [2] THERY C, WITWER KW, AIKAWA E, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7(1):1535750. [3] PEZZANA C, AGNELY F, BOCHOT A, et al. Extracellular Vesicles and Biomaterial Design: New Therapies for Cardiac Repair. Trends Mol Med. 2021;27(3):231-247. [4] VAN NIEL G, D’ANGELO G, RAPOSO G. Shedding light on the cell biology of extracellular vesicles. Nat Rev Mol Cell Biol. 2018;19(4):213-228. [5] HOSHINO A, COSTA-SILVA B, SHEN TL, et al. Tumour exosome integrins determine organotropic metastasis. Nature. 2015;527(7578):329-335. [6] YU X, DENG L, WANG D, et al. Mechanism of TNF-alpha autocrine effects in hypoxic cardiomyocytes: initiated by hypoxia inducible factor 1alpha, presented by exosomes. J Mol Cell Cardiol. 2012;53(6):848-857. [7] WANG C, ZHANG C, LIU L, et al. Macrophage-Derived mir-155-Containing Exosomes Suppress Fibroblast Proliferation and Promote Fibroblast Inflammation during Cardiac Injury. Mol Ther. 2017;25(1): 192-204. [8] MCGINLEY LM, MCMAHON J, STOCCA A, et al. Mesenchymal stem cell survival in the infarcted heart is enhanced by lentivirus vector-mediated heat shock protein 27 expression. Hum Gene Ther. 2013; 24(10):840-851. [9] LAI RC, ARSLAN F, LEE MM, et al. Exosome secreted by MSC reduces myocardial ischemia/reperfusion injury. Stem Cell Res. 2010;4(3):214-222. [10] TAN SJO, FLORIANO JF, NICASTRO L, et al. Novel Applications of Mesenchymal Stem Cell-derived Exosomes for Myocardial Infarction Therapeutics. Biomolecules. 2020;10(5):707. [11] ARSLAN F, LAI RC, SMEETS MB, et al. Mesenchymal stem cell-derived exosomes increase ATP levels, decrease oxidative stress and activate PI3K/Akt pathway to enhance myocardial viability and prevent adverse remodeling after myocardial ischemia/reperfusion injury. Stem Cell Res. 2013;10(3):301-312. [12] WEN Z, MAI Z, ZHU X, et al. Mesenchymal stem cell-derived exosomes ameliorate cardiomyocyte apoptosis in hypoxic conditions through microRNA144 by targeting the PTEN/AKT pathway. Stem Cell Res Ther. 2020;11(1):36. [13] PENG Y, ZHAO JL, PENG ZY, et al. Exosomal miR-25-3p from mesenchymal stem cells alleviates myocardial infarction by targeting pro-apoptotic proteins and EZH2. Cell Death Dis. 2020;11(5):317. [14] TENG X, CHEN L, CHEN W, et al. Mesenchymal Stem Cell-Derived Exosomes Improve the Microenvironment of Infarcted Myocardium Contributing to Angiogenesis and Anti-Inflammation. Cell Physiol Biochem. 2015;37(6):2415-2424. [15] XU H, WANG Z, LIU L, et al. Exosomes derived from adipose tissue, bone marrow, and umbilical cord blood for cardioprotection after myocardial infarction. J Cell Biochem. 2020;121(3):2089-2102. [16] FRANGOGIANNIS NG, SMITH CW, ENTMAN ML. The inflammatory response in myocardial infarction. Cardiovasc Res. 2002;53(1):31-47. [17] NAHRENDORF M, PITTET MJ, SWIRSKI FK. Monocytes: protagonists of infarct inflammation and repair after myocardial infarction. Circulation. 2010;121(22):2437-2445. [18] ZHAO J, LI X, HU J, et al. Mesenchymal stromal cell-derived exosomes attenuate myocardial ischaemia-reperfusion injury through miR-182-regulated macrophage polarization. Cardiovasc Res. 2019;115(7): 1205-1216. [19] TALMAN V, RUSKOAHO H. Cardiac fibrosis in myocardial infarction-from repair and remodeling to regeneration. Cell Tissue Res. 2016;365(3): 563-581. [20] SÁNCHEZ-SÁNCHEZ R, GÓMEZ-FERRER M, REINAL I, et al. miR-4732-3p in Extracellular Vesicles From Mesenchymal Stromal Cells Is Cardioprotective During Myocardial Ischemia. Front Cell Dev Biol. 2021;9:734143. [21] SUN L, ZHU W, ZHAO P, et al. Long noncoding RNA UCA1 from hypoxia-conditioned hMSC-derived exosomes: a novel molecular target for cardioprotection through miR-873-5p/XIAP axis. Cell Death Dis. 2020; 11(8):696. [22] JUNG JH, IKEDA G, TADA Y, et al. miR-106a-363 cluster in extracellular vesicles promotes endogenous myocardial repair via Notch3 pathway in ischemic heart injury. Basic Res Cardiol. 2021;116(1):19. [23] KHAN M, NICKOLOFF E, ABRAMOVA T, et al. Embryonic stem cell-derived exosomes promote endogenous repair mechanisms and enhance cardiac function following myocardial infarction. Circ Res. 2015;117(1):52-64. [24] WU Q, WANG J, TAN WLW, et al. Extracellular vesicles from human embryonic stem cell-derived cardiovascular progenitor cells promote cardiac infarct healing through reducing cardiomyocyte death and promoting angiogenesis. Cell Death Dis. 2020;11(5):354. [25] KERVADEC A, BELLAMY V, EL HARANE N, et al. Cardiovascular progenitor-derived extracellular vesicles recapitulate the beneficial effects of their parent cells in the treatment of chronic heart failure. J Heart Lung Transplant. 2016;35(6):795-807. [26] YOSHIDA Y, YAMANAKA S. Induced Pluripotent Stem Cells 10 Years Later: For Cardiac Applications. Circ Res. 2017;120(12):1958-1968. [27] ADAMIAK M, CHENG G, BOBIS-WOZOWICZ S, et al. Induced Pluripotent Stem Cell (iPSC)-Derived Extracellular Vesicles Are Safer and More Effective for Cardiac Repair Than iPSCs. Circ Res. 2018;122(2):296-309. [28] WANG Y, ZHANG L, LI Y, et al. Exosomes/microvesicles from induced pluripotent stem cells deliver cardioprotective miRNAs and prevent cardiomyocyte apoptosis in the ischemic myocardium. Int J Cardiol. 2015;192:61-69. [29] SAYED D, HE M, HONG C, et al. MicroRNA-21 is a downstream effector of AKT that mediates its antiapoptotic effects via suppression of Fas ligand. J Biol Chem. 2010;285(26):20281-20290. [30] KIM HW, HAIDER HK, JIANG S, et al. Ischemic preconditioning augments survival of stem cells via miR-210 expression by targeting caspase-8-associated protein 2. J Biol Chem. 2009;284(48):33161-33168. [31] GRUBER AJ, GRANDY WA, BALWIERZ PJ, et al. Embryonic stem cell-specific microRNAs contribute to pluripotency by inhibiting regulators of multiple differentiation pathways. Nucleic Acids Research. 2014; 42(14):9313-9326. [32] MENDELL JT. miRiad roles for the miR-17-92 cluster in development and disease. Cell. 2008;133(2):217-222. [33] MÜLLER P, LEMCKE H, DAVID R. Stem Cell Therapy in Heart Diseases - Cell Types, Mechanisms and Improvement Strategies. Cell Physiol Biochem. 2018;48(6):2607-2655. [34] YUE Y, WANG C, BENEDICT C, et al. Interleukin-10 Deficiency Alters Endothelial Progenitor Cell–Derived Exosome Reparative Effect on Myocardial Repair via Integrin-Linked Kinase Enrichment. Circ Res. 2020;126(3):315-329. [35] WEN C, SEEGER RC, FABBRI M, et al. Biological roles and potential applications of immune cell-derived extracellular vesicles. J Extracell Vesicles. 2017;6(1):1400370. [36] LONG R, GAO L, LI Y, et al. M2 macrophage-derived exosomes carry miR-1271-5p to alleviate cardiac injury in acute myocardial infarction through down-regulating SOX6. Mol Immunol. 2021;136:26-35. [37] IKEDA G, SANTOSO MR, TADA Y, et al. Mitochondria-Rich Extracellular Vesicles From Autologous Stem Cell-Derived Cardiomyocytes Restore Energetics of Ischemic Myocardium. J Am Coll Cardiol. 2021;77(8): 1073-1088. [38] GAO L, WANG L, WEI Y, et al. Exosomes secreted by hiPSC-derived cardiac cells improve recovery from myocardial infarction in swine. Sci Transl Med. 2020;12(561):eaay1318. [39] GAO L, GREGORICH ZR, ZHU W, et al. Large Cardiac Muscle Patches Engineered From Human Induced-Pluripotent Stem Cell-Derived Cardiac Cells Improve Recovery From Myocardial Infarction in Swine. Circulation. 2018;137(16):1712-1730. [40] LIAO Z, CHEN Y, DUAN C, et al. Cardiac telocytes inhibit cardiac microvascular endothelial cell apoptosis through exosomal miRNA-21-5p-targeted cdip1 silencing to improve angiogenesis following myocardial infarction. Theranostics. 2021;11(1):268-291. [41] DAVIDSON SM, DUCHEN MR. Endothelial mitochondria: contributing to vascular function and disease. Circ Res. 2007;100(8):1128-1141. [42] SCARABELLI T, STEPHANOU A, RAYMENT N, et al. Apoptosis of endothelial cells precedes myocyte cell apoptosis in ischemia/reperfusion injury. Circulation. 2001;104(3):253-256. [43] SELA, MAGER I, BREAKEFIELD XO, et al. Extracellular vesicles: biology and emerging therapeutic opportunities. Nat Rev Drug Discov. 2013; 12(5):347-357. [44] MURPHY DE, DE JONG OG, BROUWER M, et al. Extracellular vesicle-based therapeutics: natural versus engineered targeting and trafficking. Exp Mol Med. 2019;51(3):1-12. [45] YANG H, SUN W, QUAN N, et al. Cardioprotective actions of Notch1 against myocardial infarction via LKB1-dependent AMPK signaling pathway. Biochem Pharmacol. 2016;108:47-57. [46] XUAN W, KHAN M, ASHRAF M. Extracellular Vesicles From Notch Activated Cardiac Mesenchymal Stem Cells Promote Myocyte Proliferation and Neovasculogenesis. Front Cell Dev Biol. 2020;8:11. [47] MA J, ZHAO Y, SUN L, et al. Exosomes Derived from Akt-Modified Human Umbilical Cord Mesenchymal Stem Cells Improve Cardiac Regeneration and Promote Angiogenesis via Activating Platelet-Derived Growth Factor D. Stem Cells Transl Med. 2017;6(1):51-59. [48] GONG XH, LIU H, WANG SJ, et al. Exosomes derived from SDF1-overexpressing mesenchymal stem cells inhibit ischemic myocardial cell apoptosis and promote cardiac endothelial microvascular regeneration in mice with myocardial infarction. J Cell Physiol. 2019;234(8):13878-13893. [49] NI J, LIU X, YIN Y, et al. Exosomes Derived from TIMP2-Modified Human Umbilical Cord Mesenchymal Stem Cells Enhance the Repair Effect in Rat Model with Myocardial Infarction Possibly by the Akt/Sfrp2 Pathway. Oxid Med Cell Longev. 2019;2019:1958941. [50] MAO Q, LIANG XL, ZHANG CL, et al. LncRNA KLF3-AS1 in human mesenchymal stem cell-derived exosomes ameliorates pyroptosis of cardiomyocytes and myocardial infarction through miR-138-5p/Sirt1 axis. Stem Cell Res Ther. 2019;10(1):393. [51] LIU J, JIANG M, DENG S, et al. miR-93-5p-Containing Exosomes Treatment Attenuates Acute Myocardial Infarction-Induced Myocardial Damage. Mol Ther Nucleic Acids. 2018;11:103-115. [52] WEI Z, QIAO S, ZHAO J, et al. miRNA-181a over-expression in mesenchymal stem cell-derived exosomes influenced inflammatory response after myocardial ischemia-reperfusion injury. Life Sci. 2019; 232:116632. [53] PAN Z, SUN X, SHAN H, et al. MicroRNA-101 inhibited postinfarct cardiac fibrosis and improved left ventricular compliance via the FBJ osteosarcoma oncogene/transforming growth factor-β1 pathway. Circulation. 2012;126(7):840-850. [54] WANG J, LEE CJ, DECI MB, et al. MiR-101a loaded extracellular nanovesicles as bioactive carriers for cardiac repair. Nanomedicine. 2020;27:102201. [55] SONG Y, ZHANG C, ZHANG J, et al. Localized injection of miRNA-21-enriched extracellular vesicles effectively restores cardiac function after myocardial infarction. Theranostics. 2019;9(8):2346-2360. [56] LI Q, XU Y, LV K, et al. Small extracellular vesicles containing miR-486-5p promote angiogenesis after myocardial infarction in mice and nonhuman primates. Sci Transl Med. 2021;13(584):eabb0202. [57] DE ABREU RC, FERNANDES H, DA COSTA MARTINS PA, et al. Native and bioengineered extracellular vesicles for cardiovascular therapeutics. Nat Rev Cardiol. 2020;17(11):685-697. [58] ZHANG N, SONG Y, HUANG Z, et al. Monocyte mimics improve mesenchymal stem cell-derived extracellular vesicle homing in a mouse MI/RI model. Biomaterials. 2020;255:120168. [59] WANG X, CHEN Y, ZHAO Z, et al. Engineered Exosomes With Ischemic Myocardium-Targeting Peptide for Targeted Therapy in Myocardial Infarction. J Am Heart Assoc. 2018;7(15):e008737. [60] LAI CP, MARDINI O, ERICSSON M, et al. Dynamic biodistribution of extracellular vesicles in vivo using a multimodal imaging reporter. ACS Nano. 2014;8(1):483-494. [61] LIU B, LEE BW, NAKANISHI K, et al. Cardiac recovery via extended cell-free delivery of extracellular vesicles secreted by cardiomyocytes derived from induced pluripotent stem cells. Nat Biomed Eng. 2018; 2(5):293-303. [62] WEI Z, CHEN Z, ZHAO Y, et al. Mononuclear phagocyte system blockade using extracellular vesicles modified with CD47 on membrane surface for myocardial infarction reperfusion injury treatment. Biomaterials. 2021;275:121000. [63] ZHU LP, TIAN T, WANG JY, et al. Hypoxia-elicited mesenchymal stem cell-derived exosomes facilitates cardiac repair through miR-125b-mediated prevention of cell death in myocardial infarction. Theranostics. 2018; 8(22):6163-6177. [64] TIAN T, ZHANG HX, HE CP, et al. Surface functionalized exosomes as targeted drug delivery vehicles for cerebral ischemia therapy. Biomaterials. 2018;150:137-149. [65] NAKASE I, NOGUCHI K, FUJII I, et al. Vectorization of biomacromolecules into cells using extracellular vesicles with enhanced internalization induced by macropinocytosis. Sci Rep. 2016;6:34937. [66] NAKASE I, NOGUCHI K, AOKI A, et al. Arginine-rich cell-penetrating peptide-modified extracellular vesicles for active macropinocytosis induction and efficient intracellular delivery. Sci Rep. 2017;7(1):1991. [67] LIU C, BAYADO N, HE D, et al. Therapeutic Applications of Extracellular Vesicles for Myocardial Repair. Front Cardiovasc Med. 2021;8:758050. [68] WATERS R, ALAM P, PACELLI S, et al. Stem cell-inspired secretome-rich injectable hydrogel to repair injured cardiac tissue. Acta Biomater. 2018;69:95-106. [69] LV K, LI Q, ZHANG L, et al. Incorporation of small extracellular vesicles in sodium alginate hydrogel as a novel therapeutic strategy for myocardial infarction. Theranostics. 2019;9(24):7403-7416. [70] HAN C, ZHOU J, LIANG C, et al. Human umbilical cord mesenchymal stem cell derived exosomes encapsulated in functional peptide hydrogels promote cardiac repair. Biomater Sci. 2019;7(7):2920-2933. |
| [1] | Nong Fuxiang, Jiang Zhixiong, Li Yinghao, Xu Wencong, Shi Zhilan, Luo Hui, Zhang Qinglang, Zhong Shuang, Tang Meiwen. Bone cement augmented proximal femoral nail antirotation for type A3.3 intertrochanteric femoral fracturalysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-10. |
| [2] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [3] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [4] | Cai Zhihao, Xie Zhaoyong. Femoral neck anteversion measurement assessment: how to establish a unified method and standard [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1448-1454. |
| [5] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [6] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [7] | Gao Yu, Han Jiahui, Ge Xin. Immunoinflammatory microenvironment after spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1300-1305. |
| [8] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [9] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [10] | Chen Shisong, Liu Xiaohong, Xu Zhiyun. Current status and prospects of bioprosthetic heart valves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1096-1102. |
| [11] | Lu Di, Zhang Cheng, Duan Rongquan, Liu Zongxiang. Osteoinductive properties of calcium phosphate ceramic bone repair materials [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1103-1109. |
| [12] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [13] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [14] | Bai Siqi, Xiao Zhen, Liu Jing. Application potential of adipose-derived stem cells in female pelvic floor dysfunction diseases [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 921-927. |
| [15] | Zhang Qijian, Xu Ximing. Acquisition and application of ectodermal mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 928-934. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||