Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (18): 2945-2951.doi: 10.12307/2022.707
Previous Articles Next Articles
Interleukin 6 involved in a series of reaction processes of osteogenesis and bone repair
Wang Jing1, Wang Wanyuji2, Zhang Yi3, Ma Yaping1, Wang Xin1
- 1Department of Orthopedics, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China; 2Zunyi Medical University, Zunyi 563000, Guizhou Province, China; 3School of Public Health, Zunyi Medical University, Zunyi 563000, Guizhou Province, China
-
Received:2021-10-18Accepted:2021-11-05Online:2022-06-28Published:2022-01-30 -
Contact:Wang Xin, MD, Chief physician, Doctoral supervisor, Department of Orthopedics, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China Ma Yaping, Master, Experimentalist, Department of Orthopedics, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China -
About author:Wang Jing, Master candidate, Department of Orthopedics, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China Wang Wanyuji, Zunyi Medical University, Zunyi 563000, Guizhou Province, China Wang Jing and Wang Wanyuji contributed equally to this article. -
Supported by:National Natural Science Foundation of China, No. 31960209, 31760266) (to WX); Basic Research Program of Guizhou Science and Technology Department, No. [2020]1Y093 (to WX); Doctor Starting Fund of Affiliated Hospital of Zunyi Medical University, No. 2017-01 (to WX); Zunyi Joint Fund Project, No. Zunyi Kehe HZ Zi (2021) 40 (to MYP)
CLC Number:
Cite this article
Wang Jing, Wang Wanyuji, Zhang Yi, Ma Yaping, Wang Xin. Interleukin 6 involved in a series of reaction processes of osteogenesis and bone repair[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(18): 2945-2951.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
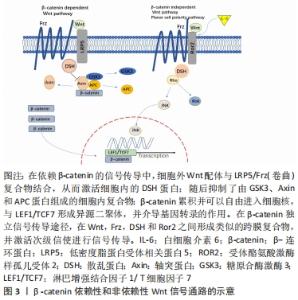
2.2 白细胞介素6对间充质干细胞分化的影响 间充质干细胞能够沿着3条主要的途径分化成不同的细胞类型,分别为成骨细胞、软骨细胞、脂肪细胞等[3]。除了这些基本的潜力,间充质干细胞还可以分化为其他中胚层谱系来源的细胞如造血细胞[4]、肌细胞[5]、肌腱细胞[6]。同时,间充质干细胞也会诱导成骨细胞形成骨[7],这一过程始于成骨祖细胞分化为成骨细胞,终于成骨细胞的发育成熟[8]。间充质干细胞诱导的成骨细胞分化需要关键的Runt相关的转录因子2(recombinant Runt related transcription factor 2,Runx2)的表达[8],但仅仅只有Runx2的表达不足以促进成骨细胞的成熟,其他的转录因子和胞外信号也参与成骨的产生,如:过氧化物酶体增殖物激活受体γ(PPARγ)通常被认为是脂肪生成的主调节器但也有对成骨细胞的作用[9];Wnt信号途径从多种配体信号途径均发挥着成骨的作用[10-14],见图3。"
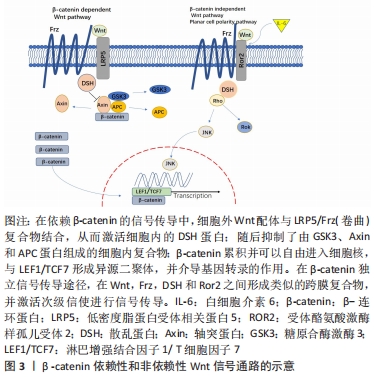
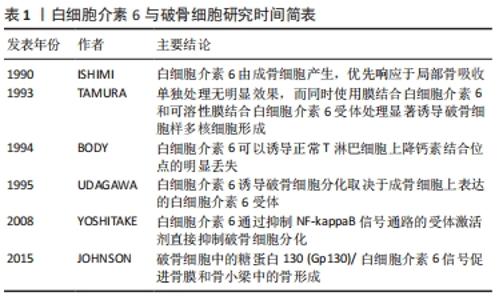
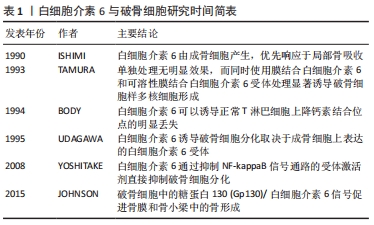
CHEN等[15]发现骨形态发生蛋白可诱导软骨形成和成骨作用;ZHAO等[16]发现刺猬蛋白(Hh)信号传导中通过锌指转录因子Gli2增强骨形态发生蛋白2的表达来促进间充质干细胞的成骨分化;AGHALOO等[17]发现NEL样蛋白1(NELL-1)在颅面成骨分化和骨形成中具有重要作用;胰岛素样生长因子1则可以通过激活哺乳动物雷帕霉素靶标(mammalian target of rapamycin,mTOR)刺激募集的间充质干细胞的成骨细胞分化,从而维持适当的骨微结构和质量[18]。 研究表明IL-6/sIL-6R激活扩增了Runx2的表达及碱性磷酸酶和骨桥蛋白等成骨细胞的分化标记mRNA的表达[19]。并且白细胞介素6可以依赖STAT3途径参与非经典无翼型(WNT)小鼠乳腺肿瘤病毒(MMTV)整合位点通路的受体酪氨酸激酶样孤儿受体2(receptor tyrosine kinase-like orphan receptor 2,ROR2)来加速人脂肪来源间充质干细胞的钙化[20]。MAO等[21]发现在低氧刺激下,间充质干细胞中整合素链接激酶(ILK)的过度表达诱导白细胞介素6的表达和分泌都显著增加,并且缺氧的间充质干细胞的存活和自我更新得到增强;后经检测发现白细胞介素6信号途径中的JAK2/STAT3激活,并显著上调了一个名为lncTCF7的长链非编码RNA,促进Wnt途径的激活从而保持了间充质干细胞的细胞活力和特性。白细胞介素6和胰岛素样生长因子1都是骨骼代谢中很重要的调节剂:胰岛素样生长因子1可通过PI3K/Akt/mTOR途径刺激骨骼的合成代谢[18];白细胞介素6会增加Akt的磷酸化,增强mTOR信号的传导[22]。而胰岛素样生长因子1也可通过激活mTOR刺激募集的间充质干细胞中成骨细胞的分化,从而维持适当的骨微结构和质量[18],所以白细胞介素6和胰岛素样生长因子1通过相同的信号通路mTOR对间充质干细胞的成骨产生协同作用[23]。 2.3 白细胞介素6对成骨细胞的影响 成骨细胞是形成骨组织的主要功能细胞,在骨组织修复中被招募到损伤部位通过合成并逐渐矿化的骨基质参与骨代谢的调节。骨愈合过程中,成骨细胞负责磷酸钙晶体(例如羟基磷灰石)的沉积,并产生骨基质成分(例如Ⅰ型胶原蛋白)。骨基质是由排列有序的胶原纤维与成骨细胞产生的非胶原蛋白复合而成,成骨细胞可以调节基质的钙和磷酸盐局部浓度而影响矿化[24]。随后成骨细胞促进骨基质钙化相关蛋白(唾液蛋白、骨桥蛋白和骨钙蛋白等)的产生使得骨基质进入矿化阶段[25-26]。 白细胞介素6通过2个分子发挥其生物学活性:白细胞介素6受体(IL-6R)和gp130(白细胞介素6家族细胞因子共用的受体和信号转导子)[27]。当白细胞介素6与膜结合白细胞介素6受体(mIL-6R)结合时,会引起gp130的二聚化,并形成白细胞介素6、白细胞介素6受体和gp130的高亲和力功能受体复合物。此外,可溶性白细胞介素6受体也与白细胞介素6结合形成IL-6/sIL-6R复合物在骨代谢中发挥重要作用[28]。KUROZUMI 等[19]发现STAT3的磷酸化会影响IL-6/sIL-6R诱导的人血管平滑肌细胞分化为成骨细胞;并且IL-6/sIL-6R还可以加速Runx2基因的表达,同时上调成骨细胞分化的相关基因。信号转导研究表明,白细胞介素6激活了两个主要的信号传导途径:SHP2/MEK/ERK和JAK/STAT3,还有SHP2/PI3K/Akt2信号通路[29]。KANESHIRO等[30]使用siRNA转染MEK2和Akt2增强了碱性磷酸酶活性和Runx2的基因表达,发现白细胞介素6对成骨细胞分化的负面影响可以通过抑制MEK和PI3K来恢复,并且可以通过抑制STAT3来增强。这些结果表明白细胞介素6通过SHP2/MEK2/ERK和SHP2/PI3K/Akt2途径负调节成骨细胞的分化,同时通过JAK/STAT3对其产生正向影响,故抑制成骨细胞中的MEK2和Akt2信号可能潜在地用于治疗骨质疏松症。JOHNSON等[31]使用STAT3抑制剂可减少成骨、STAT3磷酸化以及成骨标记物如osterix的表达,并且染色质免疫沉淀(ChIP)分析表明,在白细胞介素6刺激的成骨过程中,STAT3与osterix启动子上的STAT3结合位点的结合增加。故白细胞介素6可通过STAT3途径诱导osterix的表达进而促进成骨。 PERUZZI等[32]发现络氨酸激酶(c-Src)和白细胞介素6对成骨细胞的影响是相互串扰的:白细胞介素6可以通过胰岛素样生长因子结合蛋白5(Insulin-like Growth Factor Binding Protein 5,IGFBP5)途径激活络氨酸激酶;络氨酸激酶不仅在白细胞介素6诱导的成骨细胞减少中会发挥作用,而且还可以通过STAT3途径调控白细胞介素6的表达。成骨细胞除了生理性的成骨以外,在病理性条件例如肿瘤发生转移性骨化的过程中也会起到一定的作用,ZHENG等[33]在体外用白细胞介素6处理人乳腺癌细胞(MDA-MB-231)后,检测到RANK mRNA的表达显著提高,这表明肿瘤细胞RANK表达的增加可能是通过白细胞介素6自分泌机制介导的。源自癌细胞的白细胞介素6通过自分泌机制诱导肿瘤细胞表达RANK,从而使癌症对RANKL的作用敏感。不仅如此,白细胞介素6还会促进成骨细胞和成骨细胞谱系其他细胞中的RANKL表达[34]。这些相互依赖的、自我放大的信号通路协同作用,会增加骨骼微环境中细胞因子的输出并刺激骨骼中转移性肿瘤的生长[33]。 骨形态发生蛋白是一组信号分子,属于转化生长因子β超家族,可以诱导骨骼的形成[35]。当体内炎症递质如肿瘤坏死因子α和白细胞介素1β很高时,它们会抑制骨髓基质细胞中骨形态发生蛋白2诱导的成骨分化作用[36]。但是有意思的是在动物模型中发现白细胞介素6可以协同骨形态发生蛋白2来增强骨骼再生[37],并且在骨代谢过程中白细胞介素6会促进成骨细胞前体和脂肪干细胞的成骨分化[38]。鉴于白细胞介素6参与了骨形态发生蛋白2诱导的脂肪成骨过程,HUANG等[37]将重组人骨形态发生蛋白2和其载体ACS一起装在海绵中放入小鼠损伤部位,然后分别用生理盐水和100 μg白细胞介素6处理,获得第4,8周的重组人骨形态发生蛋白2/ACS标本后检测,发现白细胞介素6通过促进骨形态发生蛋白1A型受体(BMPR1A)的细胞表面易位并刺激两个不同的下游途径来增强重组人骨形态发生蛋白2诱导的成骨和脂肪形成。 2.4 白细胞介素6对破骨细胞分化的影响 研究脉络见表1。"
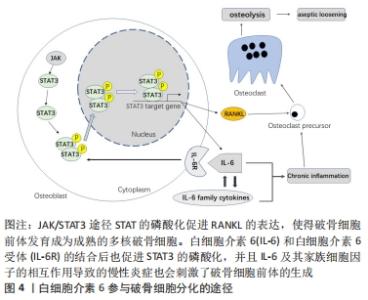
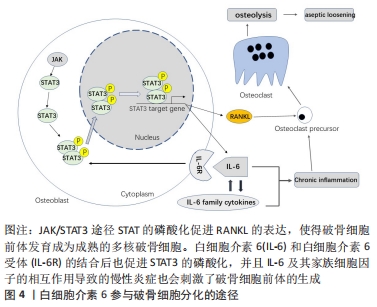
白细胞介素6有两种作用方式:顺式和反式[39],其中经典(顺式)信号仅在表达膜结合白细胞介素6受体的细胞中发生,而反式信号中白细胞介素6在招募gp130的同型二聚体之前结合可溶性白细胞介素6受体。最新的研究发现尽管白细胞介素6顺式信号可以提高STAT3应答基因Socs3和Cebpd的mRNA水平,但顺式信号并没有促进骨的增殖;相反,白细胞介素6反式作用信号转导可以产生效应,促进骨细胞和破骨细胞的形成[40]。gp130是白细胞介素6家族细胞因子共用的受体和信号转导分子[41],JOHNSON等[42]使用组织蛋白酶K Cre(CtskCre)处理破骨细胞中的gp130信号缺失的小鼠,发现处理组小鼠骨髓中产生了更多的破骨细胞并且形成的破骨细胞的核更多,而且处理组小鼠比对照组的股骨更细、骨膜表面骨形成更少,这表明白细胞介素6/gp130信号对于正常的骨吸收不是必须的,但可以刺激破骨细胞衍生的“偶联因子”和“成骨递质”提高成骨细胞活性进而维持骨的形成。UDAGAWA等[43]发现单独的白细胞介素6并不能诱导小鼠骨髓和成骨细胞共培养物中的破骨细胞形成,但是在可溶性白细胞介素6受体的协助下则可以触发这一过程,这表明白细胞介素6诱导的破骨细胞分化需要可溶性白细胞介素6受体的共同作用。 白细胞介素6、肿瘤坏死因子、白细胞介素1、甲状旁腺激素相关蛋白(PTHrP)和1,25-二羟基维生素D3都可以诱导人类破骨细胞样细胞的形成[44]。白细胞介素6可以诱导白细胞介素1的释放来促进破骨细胞前体形成并诱导破骨细胞多核化[45];AL-HUMIDAN等[46]发现白细胞介素6在一定程度上可以影响甲状旁腺激素/甲状旁腺激素相关蛋白对破骨细胞形成和破骨细胞介导的骨吸收。核因子kB受体活化因子配体RANKL对于破骨细胞分化十分重要。FENG等[47]发现IL-6/sIL-6R促进了低水平(10 μg/L)RANKL却抑制高水平(50 μg/L)RANKL诱导的破骨细胞分化,细胞信号转导分析发现IL-6/sIL-6R的组合通过NF-κB、ERK和JNK信号通路差异性调节RANKL诱导的破骨细胞形成的水平,证实了IL-6/sIL-6R对RANKL诱导的破骨细胞形成存在不同的调节。白细胞介素6既可以作为人类破骨细胞的自分泌因子,又可以作为旁分泌因子,并且在破骨细胞的骨吸收能力中发挥重要作用[48]。 白细胞介素6参与成骨细胞对破骨细胞分化的调节,见图4。双膦酸盐主要用于抑制破骨细胞的活性,近期研究发现双膦酸盐通过自分泌白细胞介素6的STAT3信号路径上调RANKL的表达[49],而成骨细胞产生RANKL对破骨细胞分化产生影响,所以双膦酸盐可以通过白细胞介素6/RANKL轴增强成骨细胞介导的破骨细胞分化。此外,也有研究表明,白细胞介素6可能在绝经后骨质疏松症[50]、 Paget病[51]、多发性骨髓瘤相关的骨质破坏中起主要作用[51]。因此,使用白细胞介素6的拮抗剂例如Sant 5可以抑制破骨细胞的形成[52]。此外,一种新型的免疫抑制剂盐酸芬戈莫德(FTY720)也可通过调节白细胞介素6、白细胞介素4和金属蛋白酶2的表达抑制破骨细胞生成[53]。而这些有效的炎症因子抑制剂对患者的骨重塑有重大意义。"
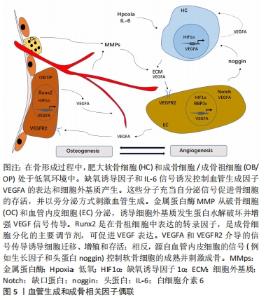
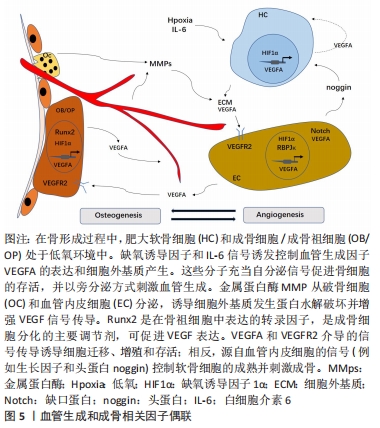
2.5 白细胞介素6对血管化的影响 骨骼是高度血管化的,除少数(如生长板和关节软骨)外在骨骼系统的所有区域都发现了血管[54]。近年来,发现了调控骨骼血管生成相关的细胞因子,见图5。如低氧和血管内皮生长因子(vascular endothelial growth factor,VEGF)家族生长因子的产生是驱动软骨内血管生成的主要信号[55],而这些信号中的大多数都影响血管化和成骨,表明血管化与成骨过程相互关联。细胞因子如VEGF、缺氧诱导因子(HIF)、基质金属蛋白酶(MMP)、成纤维细胞生长因子(FGF)、结缔组织生长因子(CTGF)、激酶c-RAF(RAF1)和Notch信号等都会影响软骨内血管的生成进而影响成骨[54]。成纤维细胞生长因子对血管内皮细胞有较强的趋化作用和促增殖作用,并刺激血管内皮细胞产生胶原酶和纤维蛋白溶解酶原激活物,促进毛细血管的生成,提高血管弹性,从而增强成骨过程中血管化进程[56]。结缔组织生长因子是分泌型母细胞蛋白CCN家族的成员,它与生长因子和整联蛋白相互作用[57],同时结缔组织生长因子也参与VEGF-A表达的调节[58]。在软骨内血管生成过程中,结缔组织生长因子在软骨膜和肥大软骨细胞中强表达,参与了骨血管的生成[59]。Notch信号是细胞生长分化的多功能调节剂,它控制着血管生成、内皮细胞的增殖以及动脉和静脉的分化[60]。并且VEGF信号在内皮细胞中的促血管生成作用也由Notch途径调节[61]。"
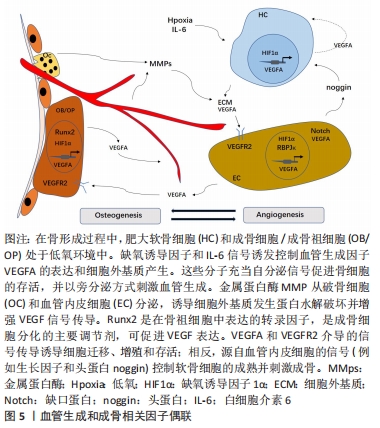
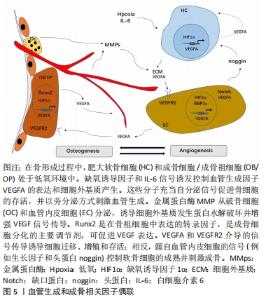
2.5 白细胞介素6对血管化的影响 骨骼是高度血管化的,除少数(如生长板和关节软骨)外在骨骼系统的所有区域都发现了血管[54]。近年来,发现了调控骨骼血管生成相关的细胞因子,见图5。如低氧和血管内皮生长因子(vascular endothelial growth factor,VEGF)家族生长因子的产生是驱动软骨内血管生成的主要信号[55],而这些信号中的大多数都影响血管化和成骨,表明血管化与成骨过程相互关联。细胞因子如VEGF、缺氧诱导因子(HIF)、基质金属蛋白酶(MMP)、成纤维细胞生长因子(FGF)、结缔组织生长因子(CTGF)、激酶c-RAF(RAF1)和Notch信号等都会影响软骨内血管的生成进而影响成骨[54]。成纤维细胞生长因子对血管内皮细胞有较强的趋化作用和促增殖作用,并刺激血管内皮细胞产生胶原酶和纤维蛋白溶解酶原激活物,促进毛细血管的生成,提高血管弹性,从而增强成骨过程中血管化进程[56]。结缔组织生长因子是分泌型母细胞蛋白CCN家族的成员,它与生长因子和整联蛋白相互作用[57],同时结缔组织生长因子也参与VEGF-A表达的调节[58]。在软骨内血管生成过程中,结缔组织生长因子在软骨膜和肥大软骨细胞中强表达,参与了骨血管的生成[59]。Notch信号是细胞生长分化的多功能调节剂,它控制着血管生成、内皮细胞的增殖以及动脉和静脉的分化[60]。并且VEGF信号在内皮细胞中的促血管生成作用也由Notch途径调节[61]。"

VEGF-A作为血管生成中的主要调节因子,对骨的血管生成、软骨生成和骨化都有极大的影响[62]。白细胞介素6的经典信号途径可以诱导VEGF-A的信号传导[63]。GAO等[64]使用siRNA处理的白细胞介素6抑制了禽白血病J亚组(ALV-J)诱导的VEGF-A在血管内皮细胞中的表达,表明白细胞介素6参与ALV-J诱导的VEGF-A表达;同时通过抑制或激活白STAT3信号途径观察对VEGF-A表达的影响,表明白细胞介素6诱导VEGF-A的表达通过STAT3信号途径。研究发现,肥大细胞产生促血管生成的VEGF并表达VEGF受体[65]。肥大细胞不仅可以通过与IgE高亲和受体(FcεRI)交联,还可以通过前列腺素E2[66]、腺苷和白细胞介素9产生VEGF[67-68]。最新研究发现,单独的白细胞介素6就可以诱导人皮肤肥大细胞产生VEGF[69],所以白细胞介素6对骨缺损处肥大细胞的作用还需要继续探索。肥大细胞自发分泌白细胞介素6诱导VEGF的产生[70],并且还将其他肥大细胞募集到炎症部位,在该部位可以产生其他促血管生成因子,促进血管生成[69]。所以白细胞介素6通过诱导VEGF的形成可促进血管化的进程进而影响成骨。成骨过程中涉及到血管的重塑,而金属蛋白酶因其降解细胞外基质的能力在血管重塑中发挥重要作用。缺氧会增加血清白细胞介素6水平,进而增加JAK2和STAT3磷酸化,从而上调基质金属蛋白酶9,而基质金属蛋白酶9的过表达显著促进了大鼠骨髓微血管增殖和基底膜的降解[71]。故白细胞介素6可以通过JAK/STAT3途径影响金属蛋白酶的产生进而影响血管化。Notch信号传导与白细胞介素6的STAT3途径相互作用,高水平的白细胞介素6可以激活STAT3 Tyr705磷酸化诱导Notch信号传导[72]。故白细胞介素6可以通过调节Notch信号、金属蛋白酶和VEGF等的表达影响血管化进程从而影响成骨。 2.6 白细胞介素6对血凝块结构的影响 骨折愈合分为4个不同阶段:①初始/炎症阶段;②软愈伤组织形成;③硬愈伤组织形成;④重塑阶段[73]。骨折后,骨髓中大量细胞(如间充质干细胞、造血干细胞、免疫细胞及其前体细胞)释放与破裂血管处细胞在骨折间隙内混合,形成骨折血肿[74]。而骨折血肿的形成是骨折愈合中非常重要的阶段,它引发炎症反应和细胞进程,使得愈伤组织形成和骨骼连续性恢复[75]。研究表明,移除小鼠早期骨折血肿可能会损害整个骨骼的愈合过程[76]。骨缺损的早期阶段,血凝块不仅仅充当“天然支架”的角色,同时它可以提供纤维蛋白矩阵供细胞的浸润、增殖和分化,并作为短期的生长因子“储备库”而发挥着作用[77]。血凝块中释放的转化生长因子β、血小板衍生生长因子、VEGF、成纤维细胞生长因子等对骨折愈合中细胞增殖分化和骨基质合成产生积极的作用[78]。血凝块的结构特征包括纤维直径、凝块密度、刚度或渗透性,可以确定整体凝块完整性和功能特征对凝血和纤溶是有影响的[79]。血凝块对骨折愈合产生影响,但血凝块结构影响成骨的作用近年来才被证明。WANG等[80]通过研究大鼠骨缺损模型,发现与正常骨愈合相比,延迟愈合的骨折模型中血凝块表现为更细和更密集的凝块结构。而应用S-亚硝基谷胱甘肽(GSNO)处理骨折模型得到了具有较多粗纤维并且比较松散的多孔凝块结构,而疏松粗大的血凝块明显增强了骨愈合。 纤维蛋白凝块的结构特性(如纤维厚度、密度和凝块孔隙率)直接影响凝块的力学性能,稳定性和对纤维蛋白溶解的敏感性[79]。纤维蛋白由可溶性纤维蛋白原形成,纤维蛋白原是肝脏中合成的340 kD的二聚糖蛋白[81]。纤维蛋白原的每个二聚体由3个链组成,分别称为Aα,Bβ和γ链,这6个多肽链的N末端会聚在分子的中心“E”结构域中,Bβ和γ链的C末端向外延伸至远端“D”结构域。纤维蛋白聚合需要凝血酶诱导,研究证明血纤蛋白聚合过程中旋钮孔的结合是由强度高、稳定性强和高度特异性的A:a驱动的,而不是A:b,B:b或B:a的作用[82]。而“旋钮B”和“孔b”的相互作用促进横向纤维聚集体,同时增加纤维蛋白网络的孔隙率和单纤维强度[83],而大孔隙的纤维蛋白形成的血凝块对早期骨愈合有积极的效应。纤维蛋白原作为一种急性期反应物,在受到炎症细胞因子(主要是白细胞介素6)刺激后会上调[84]。凝血因子XIII负责血凝块形成初期与纤维蛋白γ链的交联,而 γ链的交联影响纤维蛋白纤维的出现时间和纤维密度[85]。REIN-SMITH等[86]使用白细胞介素6处理人肝癌细胞,发现总纤维蛋白原的最大产量增加了约2.3倍,γ纤维蛋白原的产量约多增加了3.2倍,且γ链的mRNA水平增加了8.3倍,所以白细胞介素6能够差异性地上调总纤维蛋白原和γ纤维蛋白原的产生。SJ?LAND等[87]发现在高白细胞介素6情况下纤维蛋白凝块渗透性低、可压实性弱且对纤维蛋白降解的敏感性低(P < 0.001)。此外,白介素家族中白细胞介素1β也通过直接影响在纤维蛋白原的γ链的羧基末端位点影响纤维尺寸[88]。白细胞介素1细胞因子家族包含11个成员,包括7种促炎性激动剂(IL-1α,IL-1β,IL-18 ,IL-33,IL-36α,IL-36β和IL-36γ)和4种抗炎活性拮抗剂(IL-1Ra,IL-36Ra,IL-37和IL-38)。白细胞介素6家族包括白细胞介素6、白细胞介素12、白细胞介素23、白细胞介素27、白细胞介素35。目前已经发现白细胞介素1β和白细胞介素6通过对血凝块结构的调控来影响骨的愈合,所以考虑其他同族的白细胞介素成员对血凝块结构也会产生一定的影响,这需要进一步的深入研究。"

| [1] ZURA R, XIONG Z, EINHORN T, et al. Epidemiology of Fracture Nonunion in 18 Human Bones. JAMA Surg. 2016;151(11):e162775. [2] ROCHA-RODRIGUES S, RODRÍGUEZ A, GOUVEIA AM, et al. Effects of physical exercise on myokines expression and brown adipose-like phenotype modulation in rats fed a high-fat diet. Life Sci. 2016;165:100-108. [3] PAJARINEN J, LIN T, GIBON E, et al. Mesenchymal stem cell-macrophage crosstalk and bone healing. Biomaterials. 2019;196:80-89. [4] ZHAO L, CHEN S, YANG P, et al. The role of mesenchymal stem cells in hematopoietic stem cell transplantation: prevention and treatment of graft-versus-host disease. Stem Cell Res Ther. 2019;10(1):182. [5] BROWN C, MCKEE C, BAKSHI S, et al. Mesenchymal stem cells: Cell therapy and regeneration potential. J Tissue Eng Regen Med. 2019;13(9):1738-1755. [6] VERONESI F, SALAMANNA F, TSCHON M, et al. Mesenchymal stem cells for tendon healing: what is on the horizon? J Tissue Eng Regen Med. 2017; 11(11):3202-3219. [7] GRAFE I, ALEXANDER S, PETERSON JR, et al. TGF-β Family Signaling in Mesenchymal Differentiation. Cold Spring Harb Perspect Biol. 2018;10(5): a022202. [8] Melnik S, Gabler J, Dreher SI, et al. MiR-218 affects hypertrophic differentiation of human mesenchymal stromal cells during chondrogenesis via targeting RUNX2, MEF2C, and COL10A1. Stem Cell Res Ther. 2020;11(1): 532. [9] Pierce JL, Begun DL, Westendorf JJ, et al. Defining osteoblast and adipocyte lineages in the bone marrow. Bone. 2019;118:2-7. [10] CASE N, RUBIN J. Beta-catenin--a supporting role in the skeleton. J Cell Biochem. 2010;110(3):545-553. [11] LITTLE RD, CARULLI JP, DEL MASTRO RG, et al. A mutation in the LDL receptor-related protein 5 gene results in the autosomal dominant high-bone-mass trait. Am J Hum Genet. 2002;70(1):11-19. [12] CHEN J, LONG F. β-catenin promotes bone formation and suppresses bone resorption in postnatal growing mice. J Bone Miner Res. 2013;28(5):1160-1169. [13] TAKAHASHI N, MAEDA K, ISHIHARA A, et al. Regulatory mechanism of osteoclastogenesis by RANKL and Wnt signals. Front Biosci (Landmark Ed). 2011;16:21-30. [14] NIE B, ZHOU S, FANG X, et al. Implication of receptor activator of NF-κB ligand in Wnt/β-catenin pathway promoting osteoblast-like cell differentiation. J Huazhong Univ Sci Technolog Med Sci. 2012;32(6):818-822. [15] CHEN D, ZHAO M, MUNDY GR. Bone morphogenetic proteins. Growth Factors. 2004;22(4):233-241. [16] ZHAO M, QIAO M, HARRIS SE, et al. The zinc finger transcription factor Gli2 mediates bone morphogenetic protein 2 expression in osteoblasts in response to hedgehog signaling. Mol Cell Biol. 2006;26(16):6197-6208. [17] AGHALOO T, COWAN CM, CHOU YF, et al. Nell-1-induced bone regeneration in calvarial defects. Am J Pathol. 2006;169(3):903-915. [18] XIAN L, WU X, PANG L, et al. Matrix IGF-1 maintains bone mass by activation of mTOR in mesenchymal stem cells. Nat Med. 2012;18(7):1095-1101. [19] KUROZUMI A, NAKANO K, YAMAGATA K, et al. IL-6 and sIL-6R induces STAT3-dependent differentiation of human VSMCs into osteoblast-like cells through JMJD2B-mediated histone demethylation of RUNX2. Bone. 2019;124:53-61. [20] FUKUYO S, YAMAOKA K, SONOMOTO K, et al. IL-6-accelerated calcification by induction of ROR2 in human adipose tissue-derived mesenchymal stem cells is STAT3 dependent. Rheumatology (Oxford). 2014;53(7):1282-1290. [21] MAO Q, LIANG XL, WU YF, et al. ILK promotes survival and self-renewal of hypoxic MSCs via the activation of lncTCF7-Wnt pathway induced by IL-6/STAT3 signaling. Gene Ther. 2019;26(5):165-176. [22] FAJGENBAUM DC, LANGAN RA, JAPP AS, et al. Identifying and targeting pathogenic PI3K/AKT/mTOR signaling in IL-6-blockade-refractory idiopathic multicentric Castleman disease. J Clin Invest. 2019;129(10):4451-4463. [23] BAKKER AD, JASPERS RT. IL-6 and IGF-1 Signaling Within and Between Muscle and Bone: How Important is the mTOR Pathway for Bone Metabolism? Curr Osteoporos Rep. 2015;13(3):131-139. [24] KAUR M, NAGPAL M, SINGH M. Osteoblast-n-Osteoclast: Making Headway to Osteoporosis Treatment. Curr Drug Targets. 2020;21(16):1640-1651. [25] MARUOTTI N, CORRADO A, NEVE A, et al. Bisphosphonates: effects on osteoblast. Eur J Clin Pharmacol. 2012;68(7):1013-1018. [26] NEVE A, CORRADO A, CANTATORE FP. Osteoblast physiology in normal and pathological conditions. Cell Tissue Res. 2011;343(2):289-302. [27] YOUSIF AS, RONSARD L, SHAH P, et al. The persistence of interleukin-6 is regulated by a blood buffer system derived from dendritic cells. Immunity. 2021;54(2):235-246.e5. [28] BARAN P, HANSEN S, WAETZIG GH, et al. The balance of interleukin (IL)-6, IL-6·soluble IL-6 receptor (sIL-6R), and IL-6·sIL-6R·sgp130 complexes allows simultaneous classic and trans-signaling. J Biol Chem. 2018;293(18): 6762-6775. [29] WANG SW, SUN YM. The IL-6/JAK/STAT3 pathway: potential therapeutic strategies in treating colorectal cancer (Review). Int J Oncol. 2014;44(4): 1032-1040. [30] KANESHIRO S, EBINA K, SHI K, et al. IL-6 negatively regulates osteoblast differentiation through the SHP2/MEK2 and SHP2/Akt2 pathways in vitro. J Bone Miner Metab. 2014;32(4):378-392. [31] JOHNSON DE, O’KEEFE RA, GRANDIS JR. Targeting the IL-6/JAK/STAT3 signalling axis in cancer. Nat Rev Clin Oncol. 2018;15(4):234-248. [32] PERUZZI B, CAPPARIELLO A, DEL FATTORE A, et al. c-Src and IL-6 inhibit osteoblast differentiation and integrate IGFBP5 signalling. Nat Commun. 2012;3:630. [33] ZHENG Y, CHOW SO, BOERNERT K, et al. Direct crosstalk between cancer and osteoblast lineage cells fuels metastatic growth in bone via auto-amplification of IL-6 and RANKL signaling pathways J Bone Miner Res. 2014; 29(9):1938-1949. [34] SUDA T, TAKAHASHI N, UDAGAWA N, et al. Modulation of osteoclast differentiation and function by the new members of the tumor necrosis factor receptor and ligand families. Endocr Rev. 1999;20(3):345-357. [35] ZIMMERMANN A, HERCHER D, REGNER B, et al. Evaluation of BMP-2 Minicircle DNA for Enhanced Bone Engineering and Regeneration. Curr Gene Ther. 2020;20(1):55-63. [36] HUANG RL, YUAN Y, TU J, et al. Exaggerated inflammatory environment decreases BMP-2/ACS-induced ectopic bone mass in a rat model: implications for clinical use of BMP-2. Osteoarthritis Cartilage. 2014;22(8):1186-1196. [37] HUANG RL, SUN Y, HO CK, et al. IL-6 potentiates BMP-2-induced osteogenesis and adipogenesis via two different BMPR1A-mediated pathways. Cell Death Dis. 2018;9(2):144. [38] BASTIDAS-CORAL AP, BAKKER AD, ZANDIEH-DOULABI B, et al. Cytokines TNF-α, IL-6, IL-17F, and IL-4 Differentially Affect Osteogenic Differentiation of Human Adipose Stem Cells. Stem Cells Int. 2016;2016:1318256. [39] SCHELLER J, GARBERS C, ROSE-JOHN S. Interleukin-6: from basic biology to selective blockade of pro-inflammatory activities. Semin Immunol. 2014; 26(1):2-12. [40] MCGREGOR NE, MURAT M, ELANGO J, et al. IL-6 exhibits both cis- and trans-signaling in osteocytes and osteoblasts, but only trans-signali ng promotes bone formation and osteoclastogenesis. J Biol Chem. 2019;294(19):7850-7863. [41] KAUR S, BANSAL Y, KUMAR R, et al. A panoramic review of IL-6: Structure, pathophysiological roles and inhibitors. Bioorg Med Chem. 2020;28(5):115327. [42] JOHNSON RW, MCGREGOR NE, BRENNAN HJ, et al. Glycoprotein130 (Gp130)/interleukin-6 (IL-6) signalling in osteoclasts promotes bone formation in periosteal and trabecular bone. Bone. 2015;81:343-351. [43] UDAGAWA N, TAKAHASHI N, KATAGIRI T, et al. Interleukin (IL)-6 induction of osteoclast differentiation depends on IL-6 receptors expressed on osteoblastic cells but not on osteoclast progenitors. J Exp Med. 1995;182(5): 1461-1468. [44] RAGIPOGLU D, DUDECK A, HAFFNER-LUNTZER M, et al. The Role of Mast Cells in Bone Metabolism and Bone Disorders. Front Immunol. 2020;11:163. [45] AMARASEKARA DS, YUN H, KIM S, et al. Regulation of Osteoclast Differentiation by Cytokine Networks. Immune Netw. 2018;18(1):e8. [46] AL-HUMIDAN A, RALSTON SH, HUGHES DE, et al. Interleukin-6 does not stimulate bone resorption in neonatal mouse calvariae. J Bone Miner Res. 1991;6(1):3-8. [47] FENG W, LIU H, LUO T, et al. Combination of IL-6 and sIL-6R differentially regulate varying levels of RANKL-induced osteoclastogenesis through NF-κB, ERK and JNK signaling pathways. Sci Rep. 2017;7:41411. [48] QING H, DESROULEAUX R, ISRANI-WINGER K, et al. Origin and Function of Stress-Induced IL-6 in Murine Models. Cell. 2020;182(2):372-387.e14. [49] KIM HJ, KIM HJ, CHOI Y, et al. Zoledronate Enhances Osteocyte-Mediated Osteoclast Differentiation by IL-6/RANKL Axis. Int J Mol Sci. 2019;20(6):1467. [50] HADJI P, COLLI E, REGIDOR PA. Bone health in estrogen-free contraception. Osteoporos Int. 2019;30(12):2391-2400. [51] ROODMAN GD, KURIHARA N, OHSAKI Y, et al. Interleukin 6. A potential autocrine/paracrine factor in Paget’s disease of bone]. J Clin Invest. 1992; 89(1):46-52. [52] DEVLIN RD, REDDY SV, SAVINO R, et al. IL-6 mediates the effects of IL-1 or TNF, but not PTHrP or 1,25(OH)2D3, on osteoclast-like cell formation in normal human bone marrow cultures. J Bone Miner Res. 1998;13(3):393-399. [53] ZHANG D, HUANG Y, HUANG Z, et al. FTY-720P Suppresses Osteoclast Formation by Regulating Expression of Interleukin-6 (IL-6), Interleukin-4 (IL-4), and Matrix Metalloproteinase 2 (MMP-2). Med Sci Monit. 2016;22:2187-94. [54] SIVARAJ KK, ADAMS RH. Blood vessel formation and function in bone. Development. 2016;143(15):2706-15. [55] ZHA Y, LI Y, LIN T, et al. Progenitor cell-derived exosomes endowed with VEGF plasmids enhance osteogenic induction and vascular remodeling in large segmental bone defects. Theranostics. 2021;11(1):397-409. [56] MAJIDINIA M, SADEGHPOUR A, YOUSEFI B. The roles of signaling pathways in bone repair and regeneration. J Cell Physiol. 2018;233(4):2937-2948. [57] RAMAZANI Y, KNOPS N, ELMONEM MA, et al. Connective tissue growth factor (CTGF) from basics to clinics. Matrix Biol. 2018;68-69:44-66. [58] WANG Y, DANG Z, CUI W, et al. Mechanical stretch and hypoxia inducible factor-1 alpha affect the vascular endothelial growth factor and the connective tissue growth factor in cultured ACL fibroblasts. Connect Tissue Res. 2017;58(5):407-413. [59] WANG WB, LI HP, YAN J, et al. CTGF regulates cyclic stretch-induced vascular smooth muscle cell proliferation via microRNA-19b-3p. Exp Cell Res. 2019; 376(1):77-85. [60] RAMASAMY SK, KUSUMBE AP, SCHILLER M, et al. Blood flow controls bone vascular function and osteogenesis. Nat Commun. 2016;7:13601. [61] CHEN Y, ZHAO B, ZHU Y, et al. HIF-1-VEGF-Notch mediates angiogenesis in temporomandibular joint osteoarthritis. Am J Transl Res. 2019;11(5):2969-2982. [62] BAKSHI R, HOKUGO A, KHALIL D, et al. A Chemotactic Functional Scaffold with VEGF-Releasing Peptide Amphiphiles Facilitates Bone Regeneration by BMP-2 in a Large-Scale Rodent Cranial Defect Model. Plast Reconstr Surg. 2021;147(2):386-397. [63] COUGHLIN BA, TROMBLEY BT, MOHR S. Interleukin-6 (IL-6) mediates protection against glucose toxicity in human Müller cells via activation of VEGF-A signaling. Biochem Biophys Res Commun. 2019;517(2):227-232. [64] GAO Y, ZHANG Y, YAO Y, et al. Avian leukosis virus subgroup J induces VEGF expression via NF-κB/PI3K-dependent IL-6 production. Oncotarget. 2016;7(49):80275-80287. [65] MARONE G, VARRICCHI G, LOFFREDO S, et al. Mast cells and basophils in inflammatory and tumor angiogenesis and lymphangiogenesis. Eur J Pharmacol. 2016;778:146-151. [66] GRABAUSKAS G, WU X, GAO J, et al. Prostaglandin E(2), Produced by Mast Cells in Colon Tissues From Patients With Irritable Bowel Syndrome, Contributes to Visceral Hypersensitivity in Mice. Gastroenterology. 2020; 158(8):2195-2207.e6. [67] PARDANANI A. Systemic mastocytosis in adults: 2019 update on diagnosis, risk stratification and management. Am J Hematol. 2019;94(3):363-377. [68] KOCH S, SOPEL N, FINOTTO S. Th9 and other IL-9-producing cells in allergic asthma. Semin Immunopathol. 2017;39(1):55-68. [69] MCHALE C, MOHAMMED Z, DEPPEN J, et al. Interleukin-6 potentiates FcεRI-induced PGD(2) biosynthesis and induces VEGF from human in situ-matured skin mast cells. Biochim Biophys Acta Gen Subj. 2018;1862(5):1069-1078. [70] KOMI DEA, REDEGELD FA. Role of Mast Cells in Shaping the Tumor Microenvironment. Clin Rev Allergy Immunol. 2020;58(3):313-325. [71] ZHU M, YANG M, YANG Q, et al. Chronic Hypoxia-Induced Microvessel Proliferation and Basal Membrane Degradation in the Bone Marrow of Rats Regulated through the IL-6/JAK2/STAT3/MMP-9 Pathway. Biomed Res Int. 2020;2020:9204708. [72] XIONG S, WANG R, CHEN Q, et al. Cancer-associated fibroblasts promote stem cell-like properties of hepatocellular carcinoma cells through IL-6/STAT3/Notch signaling. Am J Cancer Res. 2018;8(2):302-316. [73] EINHORN TA, GERSTENFELD LC. Fracture healing: mechanisms and interventions. Nat Rev Rheumatol. 2015;11(1):45-54. [74] MA Y, ZHOU Y, WU F, et al. The Bidirectional Interactions Between Inflammation and Coagulation in Fracture Hematoma. Tissue Eng Part B Rev. 2019;25(1):46-54. [75] WALTERS G, POUNTOS I, GIANNOUDIS PV. The cytokines and micro-environment of fracture haematoma: Current evidence. J Tissue Eng Regen Med. 2018;12(3):e1662-e1677. [76] WANG C, QI B, ZHANG C, et al. Identification of key genes influenced by fixation stability in early fracture hematoma and elucidation of their roles in fracture healing. Exp Ther Med. 2017;14(5):4633-4638. [77] WANG X, LUO Y, MASCI PP, et al. Influence of Interleukin-1 Beta on Platelet-Poor Plasma Clot Formation: A Potential Impact on Early Bone Healing. PLoS One. 2016;11(2):e0149775. [78] LIM ZXH, RAI B, TAN TC, et al. Autologous bone marrow clot as an alternative to autograft for bone defect healing. Bone Joint Res. 2019;8(3):107-117. [79] MIHALKO E, BROWN AC. Clot Structure and Implications for Bleeding and Thrombosis. Semin Thromb Hemost. 2020;6(1):96-104. [80] WANG X, FRIIS TE, MASCI PP, et al. Alteration of blood clot structures by interleukin-1 beta in association with bone defects healing. Sci Rep. 2016;6: 35645. [81] LUYENDYK JP, SCHOENECKER JG, FLICK MJ. The multifaceted role of fibrinogen in tissue injury and inflammation. Blood. 2019;133(6):511-520. [82] GARCÍA X, SEYVE L, TELLIER Z, et al. Aggregates Dramatically Alter Fibrin Ultrastructure. Biophys J. 2020;118(1):172-181. [83] WEISEL JW, LITVINOV RI. Fibrin Formation, Structure and Properties. Subcell Biochem. 2017;82:405-456. [84] CRONJÉ HT, NIENABER-ROUSSEAU C, ZANDBERG L, et al. Fibrinogen and clot-related phenotypes determined by fibrinogen polymorphisms: Independent and IL-6-interactive associations. PLoS One. 2017;12(11):e0187712. [85] NOORI A, ASHRAFI SJ, VAEZ-GHAEMI R, et al. A review of fibrin and fibrin composites for bone tissue engineering. Int J Nanomedicine. 2017;12:4937-4961. [86] REIN-SMITH CM, ANDERSON NW, FARRELL DH. Differential regulation of fibrinogen γ chain splice isoforms by interleukin-6. Thromb Res. 2013; 131(1):89-93. [87] SJØLAND JA, SIDELMANN JJ, BRABRAND M, et al. Fibrin clot structure in patients with end-stage renal disease. Thromb Haemost. 2007;98(2):339-345. [88] Sahni A, Francis CW. Plasmic degradation modulates activity of fibrinogen-bound fibroblast growth factor-2. J Thromb Haemost. 2003;1(6): 1271-1277. |
| [1] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [2] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [3] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [4] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [5] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [6] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [7] | Huang Chenwei, Fei Yankang, Zhu Mengmei, Li Penghao, Yu Bing. Important role of glutathione in stemness and regulation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1119-1124. |
| [8] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [9] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [10] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [11] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| [12] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [13] | Zhou Hongqin, Wu Dandan, Yang Kun, Liu Qi. Exosomes that deliver specific miRNAs can regulate osteogenesis and promote angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1107-1112. |
| [14] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [15] | Xu Lei, Han Xiaoqiang, Zhang Jintao, Sun Haibiao. Hyaluronic acid around articular chondrocytes: production, transformation and function characteristics [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 768-773. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||



