Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (18): 2922-2929.doi: 10.12307/2022.704
Previous Articles Next Articles
Characteristics and application on animal models of different species of Perthes disease
Chen Xianxiang, Liao Shijie, Li Boxiang, Li Chong, Huang Qian, Lin Chengsen, Liu Yun, Lu Rongbin, Ding Xiaofei
- First Affiliated Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China
-
Received:2021-10-08Accepted:2021-11-19Online:2022-06-28Published:2022-01-30 -
Contact:Ding Xiaofei, MD, Chief physician, First Affiliated Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
About author:Chen Xianxiang, Master candidate, First Affiliated Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
Supported by:National Natural Science Foundation of China, No. 82060396 (to DXF); National Natural Science Foundation of China, No. 82160809 (to LSJ); Natural Science Foundation of Guangxi Zhuang Autonomous Region, No. 2017GXNSFAA198305 (to DXF); Natural Science Foundation Youth Fund Project of Guangxi Zhuang Autonomous Region, No. 2018GXNSFBA281090 (to LSJ)
CLC Number:
Cite this article
Chen Xianxiang, Liao Shijie, Li Boxiang, Li Chong, Huang Qian, Lin Chengsen, Liu Yun, Lu Rongbin, Ding Xiaofei. Characteristics and application on animal models of different species of Perthes disease[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(18): 2922-2929.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

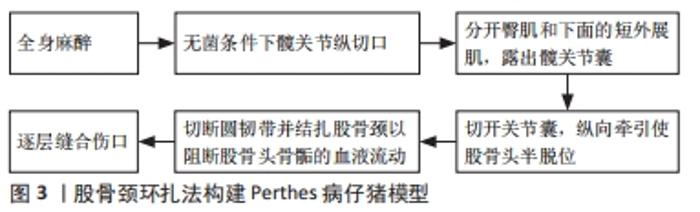
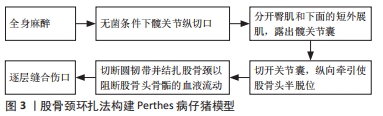
2.1 Perthes病仔猪模型 成年人股骨头主要由来自于股深动脉发出的旋股内、外侧动脉供血,但对于儿童来说,特别是在4-8岁这一年龄阶段,骨外血管从股骨颈基底部的血管环上升至股骨头骺,而股骨头骺板阻断了旋股内侧动脉发出的干骺动脉,从而直接导致血管在骨内干骺端与骨骺端之间无法建立吻合支,血供无法进入骨内,仅靠骨外血管的滋养使得股骨头骺容易因血管损伤导致缺血坏死[4]。 据现有文献报道,仔猪是最常用于Perthes病建模的动物,仔猪的髋关节解剖结构与人类相似[5],是该病较理想的模型动物。作者查阅文献得知绝大多数学者选择的仔猪是雄性、6周龄的[6-8],因为Perthes病在男孩中的发病率要远高于女孩,并且6周大的仔猪股骨头成熟度与4-6岁儿童的股骨头相类似,这是Perthes病发病的典型年龄。学者们若要在仔猪上造模以研究不同的治疗方式,据现有文献报道术后8周取样时机最佳,因为手术诱导缺血性骨坏死后8周是该模型中最大骨吸收和股骨头变形的时间,在术后8周仔猪股骨头和髋臼会变成重度畸形[9-10],此时间点收集样本能更好地看出对比治疗效果。作者还建议动物应该被单独饲养,以最大限度地减少攻击行为并最大限度地减少食物消耗的可变性,否则可能会导致不同的体质量增加[8]。目前Perthes病动物模型最常见的造模方法是股骨颈结扎手术法,由股骨颈结扎并横断圆韧带手术法建立的仔猪股骨头骨骺缺血坏死模型的坏死率较高,早在2001年,KIM等[5]便通过该方法成功建立仔猪股骨头缺血性坏死模型,而后其大体标本、组织学检查和X射线片均证实该模型与Perthes病相似,另有学者也通过相同的方法建模,并经光学和CT成像评估股骨头的血液循环,证实了股骨颈结扎进而阻断血供是导致缺血性骨坏死的主要原因[11]。 股骨颈结扎手术法是横断圆韧带并结扎股骨颈以阻断股骨头骨骺的血液流动,此手术是通过短髋外展肌间隙的外侧手术入路进行的,而非跨肌肉,然后在不损伤或切除唇部的情况下进行外侧囊膜切开术,并通过在圆韧带横断过程中施加牵引力导致股骨头半脱位[12]。在仔猪Perthes病动物模型不仅可观察到股骨头的病理改变,还可观察到髋臼侧畸形[12],为研究髋臼形态学改变提供参考。 SHAPIRO等[13]通过仔猪股骨头缺血坏死模型,发现MRI可准确跟踪股骨头变形;随后有学者通过同样的仔猪股骨头缺血坏死模型研究发现定量MRI在检测股骨头缺血性损伤方面是敏感的[14],说明髋关节的定量MRI可能有助于缺血性损伤的早期检测和评估。 KIM等[15]在仔猪股骨头缺血性坏死模型中观察到的主要修复反应是破骨细胞骨吸收,这种吸收产生了大面积的骨溶解,破坏了股骨头的结构完整性,可以解释临床上股骨头碎裂的现象;纤维血管组织替代吸收骨,缺乏新骨形成,以及纤维血管组织在骨丢失区域的持续存在,是导致股骨头畸形发生的原因。有学者采用股骨颈结扎法对仔猪诱导缺血后,在术后第2,4和8周对造模股骨头以及假手术仔猪的股骨头进行压痕实验[16],结果表明,在出现修复过程和畸形之前,缺血性损伤对生长期股骨头的压痕特性有明显的影响,该结果提示早期Perthes病患者应避免负重。同时,ARUWAJOYE等[7]对所有仔猪股骨头骨骺手术诱导缺血后立即在股骨远端干骺端交界处进行膝上截肢,以模拟临床推荐的患侧免负重,结果发现,仔猪术后免负重可以增加骨骺血运重建和梗死骨骺的吸收率,以减少缺血性股骨头坏死后的畸形。免负重具有机械保护作用,但在生物学上是不理想的,因为免负重不刺激新骨形成,增加了骨吸收和形成的不平衡[17],所以免负重的治疗方式在生物学上仍需进一步探究。REN等[8]对所有仔猪股骨头骨骺手术诱导缺血后并且进行截肢免负重,在此模型上研究托珠单抗(抗白细胞介素6药物)的作用,实验结果显示托珠单抗可减少仔猪髋关节滑膜炎和破骨细胞骨吸收,并增加缺血性骨坏死后的新骨形成,这为免负重和抗白细胞介素6药物的生物学治疗提供临床前证据,值得学者们进一步探讨。 综上所示,仔猪是Perthes病较好的动物模型,造模成功率高,通过对该模型的研究提示免负重、抗白细胞介素6药物或许能成为该病较好的治疗手段,同时可通过该模型探究更深层次的发病机制,以利于临床诊疗。但用仔猪造模也有其缺点:体积相对较大、饲养困难、价格相对昂贵,需要有较大的实验饲养空间并且拥有一定的实验经费。目前国内外学者们研究Perthes病,用仔猪来建模最多见,在仔猪上造模的方法只有一种,即股骨颈环扎法(创伤法)。 作者将主要包含造模方法、造模结论的仔猪Perthes病模型文献按研究时间脉络汇总成表格,见表1。"


2.2 Perthes病大鼠模型 大鼠因骨骺生长板终身存在,干骺端血管与骨骺端血管之间无吻合[18],常被用于制作Perthes病动物模型。NORMAN等[19]于1998年首次在幼龄大鼠上中断股骨头血供、切断圆韧带并且使股骨头脱位诱导股骨头缺血性坏死,手术诱导后第1周骨髓组织学显示坏死,术后第5周股骨头重塑并形成新骨。有学者通过相同方法建立大鼠股骨头缺血性坏死模型来研究缺氧诱导因子1α、血管内皮生长因子和细胞凋亡的可能作用,实验结果提示缺氧可能是股骨头缺血性坏死的主要病因,缺氧诱导因子1α、血管内皮生长因子及凋亡基因在时间和空间上参与了缺血性骨坏死的病理生理过程[20]。也有学者通过相同手术方法建立大鼠股骨头缺血性坏死模型来进行药物治疗研究,研究结果提示药物雷奈酸锶可以有效防止股骨头缺血坏死大鼠股骨头的塌陷,并增加其小梁厚度[21]。 自发性高血压大鼠是从Wistar-Kyoto大鼠中选择性近亲繁殖获得的一种品系,其股骨骨骺容易发生骨坏死,组织学检查与Perthes病非常相似[22]。有文献证明自发性高血压大鼠存在软骨紊乱,导致股骨头骨骺骨化延迟[23],在6-9周龄时,发育不全的股骨头部无法承受正常的负重,导致外侧骺血管闭塞,随后出现缺血和骨坏死,坏死骨在外侧骨骺中再吸收,导致机械性能降低和变形,从而进一步导致血管闭塞,因此骨骺骨化进一步延缓。 Perthes病的特征是未成熟的股骨头骨骺出现缺血性坏死,与成人股骨头缺血性坏死不同,Perthes病在其自然过程中伴随着重塑过程。在重塑过程中,死骨被活骨取代,变形的股骨头部可以重塑[24-26]。KIKKAWA等[27]研究证实,自发性高血压大鼠的骨坏死可立即被髓内肉芽组织吸收,并以类似于人类Perthes病的方式再生。自发性高血压大鼠在骨成熟过程中会出现股骨头缺血性变化和骨骺坏死,并随年龄增长而自行愈合[28],大约50%的自发性高血压大鼠在6-9周发生自发性缺血性损伤,随后股骨头骨骺发生缺血性坏死[29-30],因此,它们也许是一种良好的Perthes病实验动物模型。如果自发性高血压大鼠被限制饮食(导致体质量减轻),Perthes病特征的频率会显著下降[31-32],可能是因为髋关节负荷减少。如果普通的大鼠在喂食时被迫用后腿站立,从而使它们的髋关节超负荷,它们会以与自发性高血压大鼠相似的频率发展出Perthes病的特征[23,33]。因此,虽然存在增加自发性高血压大鼠Perthes病易感性的遗传因素,但髋关节的生物力学超负荷似乎是导致Perthes 病的诱因[34]。有学者用自发性高血压大鼠来研究Perthes病,还发现自发性高血压大鼠的生长板长度、增殖率和血管内皮生长因子的表达减少,而细胞凋亡率增加[28]。选择自发性高血压大鼠模型,比手术诱导的模型似乎更接近Perthes病,但是目前研究者们还不能准确确定自发性高血压大鼠模型的股骨头缺血坏死发生的时间点,以及自发性高血压大鼠股骨头骨骺发生缺血坏死率仅约有50%。 大鼠的基因与人类基因的相似度高达90%,且大鼠饲养时占地面积小、管理成本低,供应充足,易于长周期实验的养殖和管理,切断圆韧带和股骨颈周围的血管(创伤法)造模成功率高;利用自发性高血压大鼠的自身股骨骨骺容易坏死特点来自发诱导骨坏死的造模成功率低且难以判断其股骨头发生缺血坏死的时间点。作者将主要包含造模方法、造模结论的大鼠Perthes病模型文献按研究时间脉络汇总成表格,见表2。"


2.3 Perthes病小鼠模型 大中型动物模型具有较高的临床转化潜力,但高昂的成本、苛刻的饲养条件和缺乏研究分子机制的遗传模型限制了它们的效用,阻碍了它们的使用。相比之下,小鼠模型价格低廉,易于维护,并为机制研究提供了充足的遗传模型[35]。Perthes病是幼年缺血性骨坏死的一种形式[36]。 KAMIYA等[37]用显微外科器械识别并烧灼供应其右股骨远端骨骺的4条血管(即腘动脉分支和膝内侧、中央和外侧血管分支)诱导雄性6周龄C57BL/6小鼠股骨远端骨骺缺血性坏死,研究结果表明促炎细胞因子高迁移率族蛋白B1、肿瘤坏死因子α和白细胞介素1β在Perthes病的发病机制中与白细胞介素6显著相关,提示多个治疗靶点的可用性可能会促进Perthes病治疗策略的发展。 小鼠造模选择股骨远端骨骺作为目标骨而不是股骨近端骨骺,是因为股骨远端是在小鼠中发现的最大的骨节之一,且股骨远端骨骺大约是股骨近端骨骺的4倍大,为研究和分析提供了更多的组织量[38];选择股骨远端骨骺而不是股骨近端骨骺的另一个原因是它们的发育差异,小鼠股骨远端骨骺通过形成类似于人类的次级骨化中心而发育,小鼠的股骨近端骨骺(即股骨头)是通过干骺端血管迁移使钙化软骨直接骨化而形成的[39]。随着对小鼠基因研究的深度了解,有学者用同样造模方法在转基因小鼠上建立股骨远端骨骺缺血坏死模型,KUROYANAGI等[40]对野生型和白细胞介素6敲除小鼠进行手术诱导缺血性骨坏死,研究结果表明白细胞介素6基因敲除刺激了缺血性骨坏死后的血运重建和新骨形成,这提示阻断白细胞介素6的治疗策略可能有利于缺血性骨坏死后的骨愈合和修复[36,40]。在转基因小鼠身上建立缺血性骨坏死模型进行分子生物学操作将为今后开展Perthes病基础研究提供重要基础。 手术诱导小鼠股骨远端骨骺缺血性坏死模型已被证明在产生缺血性骨坏死方面非常可靠[36,40-42]。由于小鼠体型小,容易抓取、饲养管理成本低、造模成功率高加上与人基因相似度高、便于基因修饰研究且现今学者们对小鼠基因研究越来越深入,小鼠逐渐成为研究Perthes病的常用模型。作者将主要包含造模方法、造模结论的小鼠Perthes病模型文献按研究时间脉络汇总成表格,见表3。"


2.4 Perthes病幼兔模型 幼兔体型适中,用其建立Perthes病模型的方法较多,主要造模方法有手术创伤法、TH胶栓塞法、激素注射法和机械运动法。WANG等[43]通过切断幼兔股骨头骨骺外部血管造模,且X射线片、MRI和病理学检查结果均表示该方法可成功建立幼兔股骨头骨骺缺血坏死模型;在幼兔血管损伤模型中,作者研究发现钻穿生长板结合脂肪干细胞和骨形态发生蛋白2治疗可在股骨头骨骺缺血性坏死早期诱导新骨形成并防止股骨头塌陷。 为阐明半脱位对股骨头畸形的影响,ROWE等[44]通过阻断幼兔股骨头骨骺和圆韧带动脉,伸直幼兔膝关节诱导髋关节半脱位,制作半脱位Perthes病实验模型,结果发现:幼兔的股骨头半脱位合并股骨头缺血性坏死可能导致更频繁、更严重的股骨头畸形。 有学者给予幼兔股骨颈注射医用TH胶致骺板缺血建立幼兔Perthes病模型,用骨核素显像检查该幼兔模型发现Perthes病的发生首先与局部静脉回流障碍有关,而不是动脉血流量减少[45]。经股骨颈注射医用TH胶,造成局部静脉内栓塞,静脉回流不畅,造成局部压力升高,并且出现动脉血不能正常进入股骨头内,造成局部骨细胞出现缺血缺氧,髋关节周围组织水肿,软骨面出现充血变性区,关节间隙增宽,重现了Perthes病的基本特征[46]。 刘浩[47]给幼兔臀肌注射7.5 mg/kg醋酸泼尼松龙进行Perthes病造模,每周注射2次共进行8周,结合CT和病理结果:幼兔股骨头坏死发生率为18.75%(6/32)。激素法诱导幼兔股骨头缺血坏死的组织病理学情况:骨小梁排列稀疏,断裂较多,部分缺失;骨细胞数目减少,核固缩;可见较多的脂肪细胞;骺板细胞不规则排列,核缺失较明显[48];影像学情况:股骨头出现骨密度增高,骨纹理消失,骨骺变小,高度下降,髋关节间隙增宽等情况[49]。上述结果表明,单纯臀肌注射糖皮质激素可以导致幼兔股骨头缺血坏死的发生,与临床中Perthes病的早期表现相似,重现Perthes病的早期病程,但股骨头坏死发生率低。 ZHANG等[50]研制了一种二自由度髋外骨骼装置,用于运动性Perthes病动物模型的研究,二自由度髋外骨骼装置驱动幼兔髋关节伸展/弯曲和内收/外展超出其正常运动范围,并伴有中心性撞击来诱导股骨头缺血坏死,4周后,大体观、X射线片、MRI和病理学检查结果证实幼兔股骨头出现早期Perthes病基本特征[50]。该造模方法可以再现体操、杂技等运动项目中对髋部的重复动作和强烈的中心性撞击,是一种建立Perthes病动物模型的机械运动法,而不是传统的医学或生理操作方法,这为新治疗方法的开发和试验提供了条件。 在中型动物幼兔身上造模方法较多,其中手术创伤法造模可在较短时间内完成,操作难度较低,造模成功率高;使用TH胶栓塞法操作性强,稳定,经济,造模成功率也高;激素注射造模法虽然操作难度不高,但此方法造模股骨头坏死发生率低;机械运动法造模与传统的医学或生理操作方法不同,它是一种新的方法,操作难度较大,还有待继续研究。兔作为中型动物,相较于猪、犬的所需费用较低,饲养条件要求也不高,它们的体型适中,易于观察和取材,也是作为研究Perthes病较好的动物模型。作者将主要包含造模方法、造模结论的幼兔Perthes病模型文献按研究时间脉络汇总成表格,见表4。"


2.5 Perthes病幼犬模型 处于生长期的幼犬的股骨头骨骺的主要血液供应是由位于股骨颈的外侧骨骺动脉供应的[51],这与儿童股骨头骨骺血供相似,因此,幼犬也是一种适合建立股骨头缺血坏死模型的动物[52]。经文献得知,学者们在幼犬上造Perthes病模型的方法主要有:TH胶栓塞法、液氮冷冻法、手术创伤法。ALPASLAN等[53]使用明胶海绵栓塞幼犬髂外动脉进行Perthes病造模,通过影像学表现、大体观及显微标本进行了评价,并与临床标本进行了比较,发现介入栓塞股骨头动脉后,幼犬股骨头的变化与Perthes病非常相似。 汪逸等[54]用液氮连续冷冻股骨头15次,持续3 min,再用37 ℃温生理盐水复温3 min,如此循环2次,造成局部冷热交替损伤,成功造成幼犬Perthes病模型。液氮冷冻股骨头可导致股骨头血管发生痉挛,血管内出现凝血和血管内皮细胞遭受伤害,继而会出现股骨头血管通透性增加,股骨头骨髓内发生出血,复温后血管再通会引起缺血再灌注损伤,最终导致股骨头坏死[55]。 股骨头缺血性坏死的特点是微血管损伤、静脉血液淤积及股骨头血供不足导致股骨头缺血。有学者向幼犬股骨颈内注射医用TH胶,使其在局部聚合胶接,封闭穿过骺板的窦状隙, 压闭毛细血管, 从而使静脉回流受阻建立幼犬Perthes病模型[56]。孟海涛等[57]往幼犬股骨颈注射医用TH胶致骺板缺血建立幼犬Perthes病模型,影像及病理变化类似于Perthes病早期表现,为进行同类研究的学者提供了一种制作该病动物模型的可供选择的方法。较少学者用手术创伤法在幼犬上造Perthes病模型,ZAHIR等[58]用手术方法将幼犬的股骨头脱位,电凝灼掉支持带血供,成功造成Perthes病模型。 幼犬股骨头骨骺血供与儿童股骨头骨骺血供相似,且相对较大,方便做局部操作研究,但幼犬价格昂贵,较难获取,饲养管理难度大,这也是较少学者用幼犬来造Perthes病模型的原因。作者将主要包含造模方法、造模结论的幼犬Perthes病模型文献按研究时间脉络汇总成表格,见表5。"

| [1] SINGH K, SHAH H, JOSEPH B, et al. Evolution of Legg-Calvé-Perthes disease following proximal femoral varus osteotomy performed in the avascular necrosis stage:a prospective study. J Child Orthop. 2020; 14(1):58-67. [2] LEROUX J, ABU AMARA S, LECHEVALLIER J. Legg-Calvé-Perthes disease. Orthop Traumatol Surg Res. 2018;104:S107-S112. [3] LAINE J, MARTIN B, NOVOTNY S, et al. Role of Advanced Imaging in the Diagnosis and Management of Active Legg-Calvé-Perthes Disease. J Am Acad Orthop Surg. 2018;26(15):526-536. [4] Chung SM. The arterial supply of the developing proximal end of the human femur. J Bone Joint Surg Am. 1976;58(7):961-970. [5] KIM HK, SU PH, QIU YS. Histopathologic changes in growth-plate cartilage following ischemic necrosis of the capital femoral epiphysis. An experimental investigation in immature pigs. J Bone Joint Surg Am. 2001;83(5):688-697. [6] JOHNSON CP, WANG L, TOTH F, et al. Quantitative susceptibility mapping detects neovascularization of the epiphyseal cartilage after ischemic injury in a piglet model of legg-calve-perthes disease .J Magn Reson Imaging. 2019;50(1):106-113. [7] ARUWAJOYE OO, MONTE F, KIM A, et al. A comparison of transphyseal neck-head tunneling and multiple epiphyseal drilling on femoral head healing following ischemic osteonecrosis: an experimental investigation in immature pigs. J Pediatr Orthop. 2020;40(4):168-175. [8] REN Y, DENG Z, GOKANI V, et al. Anti-interleukin-6 therapy decreases hip synovitis and bone resorption and increases bone formation following ischemic osteonecrosis of the femoral head. J Bone Miner Res. 2021;36(2):357-368. [9] KIM HK, ARUWAJOYE O, DU J, et al. Local administration of bone morphogenetic protein-2 and bisphosphonate during non-weight-bearing treatment of ischemic osteonecrosis of the femoral head: an experimental investigation in immature pigs. J Bone Joint Surg Am. 2014;96(18):1515-1524. [10] SHAPIRO F, CONNOLLY S, ZURAKOWSKI D, et al. Acetabular changes associated with avascularnecrosis of the femoral head in a piglet model. Bone Joint Res. 2014;3(4):130-138. [11] ZHANG P, LIANG Y, KIM H, et al. Evaluation of a pig femoral head osteonecrosis model. J Orthop Surg Res. 2010;5:15. [12] UPASANI VV, JEFFORDS ME, FARNSWORTH CL, et al. Ischemic femoral head osteonecrosis in a piglet model causes three dimensional decrease in acetabular coverage. J Orthop Res. 2018;36(4):1173-1177. [13] SHAPIRO F, CONNOLLY S, ZURAKOWSKI D, et al. Femoral head deformation and repair following induction of ischemic necrosis: a histologic and magnetic resonance imaging study in the piglet. J Bone Joint Surg Am. 2009;91(12):2903-2914. [14] JOHNSON CP, WANG L, TOTH F, et al. Quantitative MRI helps to detect hip ischemia: preclinical model of legg-calve-perthes disease. Radiology. 2018;289(2):386-395. [15] KIM HK, SU PH. Development of flattening and apparent fragmentation following ischemic necrosis of the capital femoral epiphysis in a piglet model. J Bone Joint Surg Am. 2002;84(8):1329-1334. [16] PRINGLE D, KOOB TJ, KIM HK. Indentation properties of growing femoral head following ischemic necrosis. J Orthop Res. 2004;22(1):122-130. [17] KIM HK, ARUWAJOYE O, STETLER J, et al. Effects of non-weight-bearing on the immature femoral head following ischemic osteonecrosis: an experimental investigation in immature pigs. J Bone Joint Surg Am. 2012;94(24):2228-2237. [18] PESKIN B, SHUPAK A, MISSELEVICH I, et al. Transphyseal osseous bridges in experimental osteonecrosis of the femoral head of the rat. Histologic study of the bony bridges connecting the epiphyseal with the metaphyseal bony trabeculae through gaps in the physeal cartilage. J Pediatr Orthop B. 2001;10(3):214-218. [19] NORMAN D, REIS D, ZINMAN C, et al. Vascular deprivation-induced necrosis of the femoral head of the rat. An experimental model of avascular osteonecrosis in the skeletally immature individual or Legg-Perthes disease. Int J Exp Pathol. 1998;79(3):173-181. [20] ZHANG W, YUAN Z, PEI X, et al. In vivo and in vitro characteristic of HIF-1alpha and relative genes in ischemic femoral head necrosis. Int J Clin Exp Pathol. 2015;8(6):7210-7216. [21] CHEN YP, TAN A, HO WP, et al. Effectiveness of strontium ranelate in the treatment of rat model of legg-calve-perthes disease. Indian J Orthop. 2018;52(4):380-386. [22] HIRANO T, IWASAKI K, YAMANE Y. Osteonecrosis of the femoral head of growing, spontaneously hypertensive rats. Acta Orthop Scand. 1988; 59(5):530-535. [23] SUEHIRO M, HIRANO T, MIHARA K, et al. Etiologic factors in femoral head osteonecrosis in growing rats. J Orthop Sci. 2000;5(1):52-56. [24] ZENMYO M, KOMIYA S, KAWABATA R, et al. Morphological and biochemical evidence for apoptosis in the terminal hypertrophic chondrocytes of the growth plate. J Pathol. 1996;180(4):430-433. [25] KIM H, WENGER D. “Functional retroversion” of the femoral head in Legg-Calvé-Perthes disease and epiphyseal dysplasia: analysis of head-neck deformity and its effect on limb position using three-dimensional computed tomography. J Pediatr Orthop. 1997;17(2):240-246. [26] KONG SY, KIM HW, PARK HW, et al. Effects of multiple drilling on the ischemic capital femoral epiphysis of immature piglets. Yonsei Med J. 2011;52(5):809-817. [27] KIKKAWA M, IMAI S, HUKUDA S. Altered postnatal expression of insulin-like growth factor-I (IGF-I) and type X collagen preceding the Perthes’ disease-like lesion of a rat model. J Bone Miner Res. 2000;15(1):111-119. [28] PARK H, KONG SY, KIM HW. Altered cellular kinetics in the growth plate of the femoral head of spontaneously hypertensive rats. Yonsei Med J. 2012;53(3):625-633. [29] HIRANO T, IWASAKI K, ODA J, et al. Osteonecrosis of the femoral head in spontaneously hypertensive rats. Relation to ossific nuclei during growth. Acta Orthop Scand. 1992;63(1):37-40. [30] NAITO S, ITO M, SEKINE I, et al. Femoral head necrosis and osteopenia in stroke-prone spontaneously hypertensive rats (SHRSPs). Bone. 1993; 14(5):745-753. [31] TOMITA M, SHIMOKAWA I, MAEDA H, et al. Dietary restriction reduces the prevalence of osteonecrosis of the caput femoris in spontaneously hypertensive rats. Calcif Tissue Int. 1999;64(3):259-262. [32] KAWAHARA T, SHIMOKAWA I, TOMITA M, et al. Effects of caloric restriction on development of the proximal growth plate and metaphysis of the caput femoris in spontaneously hypertensive rats: microscopic and computer-assisted image analyses. Microsc Res Tech. 2002;59(4):306-312. [33] SUEHIRO M, HIRANO T, SHINDO H. Osteonecrosis induced by standing in growing Wistar Kyoto rats. J Orthop Sci. 2005;10(5):501-507. [34] BERTHAUME MA, PERRY DC, DOBSON CA, et al. Skeletal immaturity, rostral sparing, and disparate hip morphologies as biomechanical causes for Legg-Calve-Perthes’ disease. Clin Anat. 2016;29(6):759-772. [35] YAMAGUCHI R, KAMIYA N, KUROYANAGI G, et al. Development of a murine model of ischemic osteonecrosis to study the effects of aging on bone repair. J Orthop Res. 2021. doi: 10.1002/jor.25006. [36] KAMIYA N, KUROYANAGI G, ARUWAJOYE O, et al. IL6 receptor blockade preserves articular cartilage and increases bone volume following ischemic osteonecrosis in immature mice. Osteoarthritis Cartilage. 2019;27(2):326-335. [37] KAMIYA N, KIM H. Elevation of proinflammatory cytokine hmgb1 in the synovial fluid of patients with legg-calvé-perthes disease and correlation with IL-6. JBMR Plus. 2020;5(2):e10429. [38] KAMIYA N, YAMAGUCHI R, ARUWAJOYE O, et al. Development of a mouse model of ischemic osteonecrosis. Clin Orthop Relat Res. 2015; 473(4):1486-1498. [39] COLE H, YUASA M, HAWLEY G, et al. Differential development of the distal and proximal femoral epiphysis and physis in mice. Bone. 2013; 52(1):337-346. [40] KUROYANAGI G, ADAPALA NS, YAMAGUCHI R, et al. Interleukin-6 deletion stimulates revascularization and new bone formation following ischemic osteonecrosis in a murine model. Bone. 2018;116:221-231. [41] PHIPPS M, HUANG Y, YAMAGUCHI R, et al. In vivo monitoring of activated macrophages and neutrophils in response to ischemic osteonecrosis in a mouse model. J Orthop Res. 2016;34(2):307-313. [42] KAMIYA N, SHAFER S, OXENDINE I, et al. Acute BMP2 upregulation following induction of ischemic osteonecrosis in immature femoral head. Bone. 2013;53(1):239-247. [43] WANG ZL, HE RZ, TU B, et al. Drilling combined with adipose-derived stem cells and bone morphogenetic protein-2 to treat femoral head epiphyseal necrosis in juvenile rabbits. Curr Med Sci. 2018;38(2):277-288. [44] ROWE SM, CHUNG JY, MOON ES, et al. The effects of subluxation of the femoral head with avascular necrosis in growing rabbits. J Pediatr Orthop. 2004;24(6):645-650. [45] 牒军,张开放,王坤正.Perthes病早期骨核素显像的变化[J].中华临床医药杂志(北京),2001,2(11):41-43. [46] 张开放,李政,姚永锋,等.股骨头骨骺缺血性坏死模型兔构建及组织病理学变化和p53与bcl-2的表达[J].中国组织工程研究与临床康复,2011,15(2):219-223. [47] 刘浩.Notch信号通路受体及配体在幼兔激素性股骨头缺血坏死模型中的表达及意义[D].遵义:遵义医科大学,2020. [48] 卓金伟.Wnt/β-catenin信号通路在幼兔激素性股骨头缺血坏死模型中的表达及意义[D].遵义:遵义医学院,2015. [49] 王健.死亡受体通路相关蛋白在激素性股骨头缺血坏死动物模型中的表达[D].遵义:遵义医学院,2014. [50] ZHANG JF, YANG CJ, WU T, et al. A two-degree-of-freedom hip exoskeleton device for an immature animal model of exercise-induced Legg-Calve-Perthes disease. Proc Inst Mech Eng H. 2009;223(8):1059-168. [51] HULTH A, NORBERG I, OLSSON S. Coxa plana in the dog. J Bone Joint Surg Am. 1962;44-A:918-930. [52] AGUADO E, GOYENVALLE E. Legg Calvé Perthes disease in the dog. Morphologie. 2021;105(349):143-147. [53] ALPASLAN AM, AKSOY MC, YAZICI M. Interruption of the blood supply of femoral head:an experimental study on the pathogenesis of Legg-Calve-Perthes Disease. Arch Orthop Trauma Surg. 2007;127(6):485-491. [54] 汪逸,张媛媛,黄绍义,等.骨髓间充质干细胞及其分泌因子对治疗犬股骨头缺血性坏死的效果观察[J].中国兽医杂志,2019,55(8):7. [55] 官建中,周建生,肖玉周,等.建立幼犬股骨头坏死模型的实验研究[J].蚌埠医学院学报,2006,31(5):444-446. [56] 闫宏伟,王坤正,时志斌,等.Legg-Perthes 病动物模型设计与评价[J].中国矫形外科杂志,2002,10(8):795-796. [57] 孟海涛,李林,王翠苹,等.Ⅱ型胶原基因型变化与Legg-perthes病发病的相关性研究[J]实用骨科杂志,2015,21(2):134-135. [58] ZAHIR A, FREEMAN AR. Cartilage changes following a single episode of infarction of the capital femoral epiphysis in the dog. J Bone Joint Surg Am. 1972;54(1):125-136. [59] NEWTON B, CRAWFORD CJ, POWERS DL, et al. The immature goat as an animal model for Legg-Calve-Perthes disease. J Invest Surg. 1994; 7(5):417-430. [60] CRAWFORD CJ, LABERGE M, ALLEN BL JR, et al. Growth profiles and articular cartilage characterization in a goat model of Legg-Calve-Perthes disease. J Invest Surg. 1995;8(6):391-408. [61] MARTINEZ-ALVAREZ S, EPELDEGUI-TORRE T, MANSO-DIAZ G, et al. Experimental induction of Perthes disease in lambs. Rev Esp Cir Ortop Traumatol. 2014;58(2):68-77. |
| [1] | Xu Xinzhong, Wu Zhonghan, Yu Shuisheng, Zhao Yao, Xu Chungui, Zhang Xin, Zheng Meige, Jing Juehua. Biomechanical analysis of different ways of inserting Steinmann Pins into the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1313-1317. |
| [2] | Wei Guoqiang, Li Yunfeng, Wang Yi, Niu Xiaofen, Che Lifang, Wang Haiyan, Li Zhijun, Shi Guopeng, Bai Ling, Mo Kai, Zhang Chenchen, Xu Yangyang, Li Xiaohe. Biomechanical analysis of non-uniform material femur under different loads [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1318-1322. |
| [3] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [4] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [5] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [6] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [7] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [8] | Huang Chenwei, Fei Yankang, Zhu Mengmei, Li Penghao, Yu Bing. Important role of glutathione in stemness and regulation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1119-1124. |
| [9] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [10] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [11] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [12] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| [13] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [14] | Zhou Hongqin, Wu Dandan, Yang Kun, Liu Qi. Exosomes that deliver specific miRNAs can regulate osteogenesis and promote angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1107-1112. |
| [15] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||