Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (28): 4546-4552.doi: 10.12307/2021.071
Previous Articles Next Articles
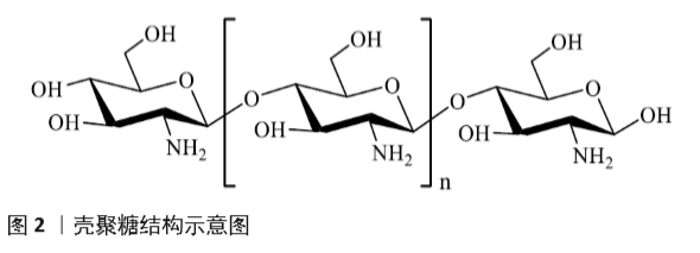
Application of chitosan in nano drug delivery system
Yang Menglu1, 2, Zhang Na1, 2, Wang Fangyuan2, Liu Jianguo1, 2
- 1School of Stomatology, Zunyi Medical University, Zunyi 563099, Guizhou Province, China; 2Special Key Laboratory of Oral Disease Research, Higher Education Institution in Guizhou Province and Zunyi Key Laboratory of Oral Disease Research, Zunyi 563006, Guizhou Province, China
-
Received:2020-11-10Revised:2020-11-13Accepted:2020-12-18Online:2021-10-08Published:2021-05-21 -
Contact:Liu Jianguo, PhD, Professor, School of Stomatology, Zunyi Medical University, Zunyi 563099, Guizhou Province, China; Special Key Laboratory of Oral Disease Research, Higher Education Institution in Guizhou Province and Zunyi Key Laboratory of Oral Disease Research, Zunyi 563006, Guizhou Province, China -
About author:Yang Menglu, Master candidate, Physician, School of Stomatology, Zunyi Medical University, Zunyi 563099, Guizhou Province, China; Special Key Laboratory of Oral Disease Research, Higher Education Institution in Guizhou Province and Zunyi Key Laboratory of Oral Disease Research, Zunyi 563006, Guizhou Province, China -
Supported by:the Guizhou Province and Zunyi City Medical Biomaterials Research & Development Talent Base Construction Project, No. QRLF[2018]3, ZW[2019]69 (to LJG); the Provincial and Municipal Science and Technology Cooperation Special Fund Project, No. SSKH[2014]41 (to LJG); Research Startup Foundation of Zunyi Medical University, No.F-821 (to WFY); the Opening Foundation of Special Key Laboratory of Higher Education Institution in Guizhou province, No. KF201502 (to WFY)
CLC Number:
Cite this article
Yang Menglu, Zhang Na, Wang Fangyuan, Liu Jianguo. Application of chitosan in nano drug delivery system[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(28): 4546-4552.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
| [1] 宋元博,宋策,于丽.纳米药物载体在临床医学中的应用[J].临床医药文献电子杂志,2018,5(70):95-96. [2] SILVA TH, ALVES A, FERREIRA BM, et al. Materials of marine origin: a review onpolymers and ceramics of biomedical interest. Int Mater Rev. 2012;57:276-306. [3] SAYARI N, SILA A, ABDELMALEK BE, et al. Chitin and chitosan from the Norway lobster by products: antimicrobial and anti-proliferative activities, Int J Biol Macromol. 2016;87:163-171. [4] DASH M, CHIELLINI F, OTTENBRITE RM, et al. Chitosan-a versatile semisynthetic polymer in biomedical applications. Prog Polym Sci. 2011;36:981-1014. [5] YOUNES I, RINAUDO M, Chitin and chitosan preparation from marine sources. Structure, properties and applications. Mar Drugs. 2015;13(3):1133-1174. [6] PARK JK, CHUNG MJ, CHOI HN, et al. Effects of the molecular weight and the degree of deacetylation of chitosan oligosaccharides on antitumor activity. Int J Mol Sci. 2011;12:266-277 [7] MARÍA L, DEL PRADO-AUDELO, ISAAC H, et al. Chitosan-decorated nanoparticles for drug delivery. J Drug Deliv Sci Tech. 2020:101896. [8] NAGPAL K, SINGH SK, MISHRA DN. Chitosan nanoparticles: a promising system in novel drug delivery. Chem Pharm Bull (Tokyo). 2010;58(11):1423-1430. [9] DÜNNHAUPT S, BARTHELMES J, RAHMAT D, et al. S-protected thiolated chitosan for oral delivery of hydrophilic macromolecules: evaluation of permeation enhancing and efflux pump inhibitory properties. Mol Pharm. 2012;9:1331-1341. [10] PATRULEA V, OSTAFE V, BORCHARD G, et al. Chitosan as a starting material for wound healing applications. Eur J Pharm Biopharm. 2015; 97;417-426. [11] RIBEIRO CV, VIEIRA RS, MELO IM, et al. Versatility of chitosan-based biomaterials and their use as scaffolds for tissue regeneration. ScientificWorldJournal. 2017;2017:8639898. [12] NATARAJ D, SAKKARA S, MEGHWAL M, et al. Crosslinked chitosan films with controllable properties for commercial applications. Int J Biol Macromol. 2018;120:1256-1264. [13] GUERRERO P, MUXIKA A, ZARANDONA I, et al. Crosslinking of chitosan films processed by compression molding. Carbohydr Polym. 2019;206:820-826. [14] HUANG Y, LAPITSKY Y. On the kinetics of chitosan/tripolyphosphate micro- and nanogel aggregation and their effects on particle polydispersity. J Colloid Interface Sci. 2017;486:27-37. [15] SACCO P, BORGOGNA M, TRAVAN A, et al. Polysaccharide-based networks from homogeneous chitosan-tripolyphosphate hydrogels: synthesis and characterization. Biomacromolecules. 2014;15(9): 3396-3405. [16] XIE H, CHEN X, SHEN X, et al. Preparation of chitosan-collagen-alginate composite dressing and its promoting effects on wound healing. Int J Biol Macromol. 2018;107:93-104. [17] CHEN X, FAN M, TAN H, et al. Magnetic and self-healing chitosan-alginate hydrogel encapsulated gelatin microspheres via covalent cross-linking for drug delivery. Mater Sci Eng C Mater Biol Appl. 2019; 101:619-629. [18] YU X, WEN T, CAO P, et al. Alginate-chitosan coated layered double hydroxide nanocomposites for enhanced oral vaccine delivery. J Colloid Interface Sci. 2019;556:258-265. [19] 赵宇,曹琬晴,刘阳.聚合物纳米药物载体的研究进展[J].高等学校化学学报,2020,41(5):909-923. [20] WANG Y, KHAN A, LIU Y, et al. Chitosan oligosaccharide-based dual pH responsive nano-micelles for targeted delivery of hydrophobic drugs. Carbohydr Polym. 2019;223:115061. [21] LIU X, HE Z, CHEN Y, et al. Dual drug delivery system of photothermal-sensitive carboxymethyl chitosan nanosphere for photothermal-chemotherapy. Int J Biol Macromol. 2020;163:156-166. [22] HORO H, DAS S, MANDAL B, KUNDU LM, et al. Development of a photoresponsive chitosan conjugated prodrug nano-carrier for controlled delivery of antitumor drug 5-fluorouracil. Int J Biol Macromol. 2019;121:1070-1076. [23] XIAO Y, GONG T, JIANG Y, et al. Fabrication and characterization of a glucose-sensitive antibacterial chitosan-polyethylene oxide hydrogel. Polymer. 2016;82:1-10. [24] DESAI KG. Chitosan nanoparticles prepared by ionotropic gelation: an overview of recent advances. Crit Rev Ther Drug Carrier Syst. 2016; 33(2):107-158. [25] CALVO P, REMUNAN-LOPEZ C, VILA-JATO JL, et al. Novel hydro-philic chitosan/polyethylene oxide nanoparticles as proteins carriers. J App Polym Sci. 1997;1(63):125-132. [26] 魏谭军,董德刚,裘梁,等.离子交联法制备壳聚糖纳米颗粒[J].安徽农业科学,2012,40(5):337-338,341. [27] SYDOW S, DE CASSAN D, HÄNSCH R, et al. Layer-by-layer deposition of chitosan nanoparticles as drug-release coatings for PCL nanofibers. Biomater Sci. 2018;7(1):233-246. [28] OHYA Y, SHIRATANI M, KOBAYASHI H, et al. Release behavior of 5-fluorouracil from chitosan-gel nanospheres immobilizing 5-flu-orouracil coated with polysaccharides and their cell specific cyto-toxicity. Pure Appl Chem. 1994;31(1):629-642. [29] XU J, MA L, LIU Y, et al. Design and characterization of antitumor drug paclitaxel-loaded chitosan nanoparticles by W/O emulsions. Int J Biol Macromol. 2012;50(2):438-443. [30] TAREK A, BADER A. Preparation, characterization, and potential application of chitosan, chitosan derivatives, and chitosan metal nanoparticles in pharmaceutical drug delivery. Drug Des Devel Ther. 2016;10:483-507. [31] ESTEVINHO BN, ROCHA F, SANTOS L, et al. Microencapsulation with chitosan by spray drying for industry applications – A review. Trends Food Sci Tech. 2013;31(2):138-155. [32] VISHALI DA, MONISHA J, SUNDARI SSK, et al. Spray freeze drying: Emerging applications in drug delivery. J Control Release. 2019;300: 93-101. [33] SHARMA S, MUKKUR TK, BENSON HA, et al. Enhanced immune response against pertussis toxoid by IgA-loaded chitosan–dextran sulfate nanoparticles. J Pharm Sci. 2012;101:233-244. [34] AKASH MSH, REHMAN K, CHEN S. Polymeric-based particulate systems for delivery of therapeutic proteins. Pharm Dev Technol. 2016;21: 367-378. [35] IMPERIALE JC, SCHLACHET I, LEWICKI M, et al. Oral pharmacokinetics of a chitosan-based nano-drug delivery system of interferon alpha. Polymers (Basel). 2019;11(11):1862. [36] ZHANG Y, ZHU W, ZHANG H, et al. Carboxymethyl chitosan/phospholipid bilayer-capped mesoporous carbon nanoparticles with pH-responsive and prolonged release properties for oral delivery of the antitumor drug. Docetaxel Int J Pharm. 2017;532(1):384-392. [37] MAITY S, MUKHOPADHYAY P, KUNDU PP, et al. Alginate coated chitosan core-shell nanoparticles for efficient oral delivery of naringenin in diabetic animals-an in vitro and in vivo approach. Carbohydr Polym. 2017;170:124-132. [38] MU Y, FU Y, LI J, et al. Multifunctional quercetin conjugated chitosan nano-micelles with P-gp inhibition and permeation enhancement of anticancer drug. Carbohydr Polym. 2019;203:10-18. [39] ZHOU X, HAO Y, YUAN L, et al. Nano-formulations for transdermal drug delivery: A review. Chinese Chem Lett. 2018;12:1713-1724. [40] RADWAN-PRAGOWSKA J, JANUS U, PITKOWSKI M, et al. ZnO nanorods functionalized with chitosan hydrogels crosslinked with azelaic acid for transdermal drug delivery. Colloids Surf B Biointerfaces. 2020;194: 111170. [41] ABNOOS M, MOHSENI M, MOUSAVI SAJ, et al. Chitosan-alginate nano-carrier for transdermal delivery of pirfenidone in idiopathic pulmonary fibrosis. Int J Bio Macromol. 2018:S0141813018311619. [42] ISLAM MA, PARK TE, REESOR E, et al. Mucoadhesive chitosan derivatives as novel drug carriers. Curr Pharm Des. 2015;21(29): 4285-4309. [43] KUMAR A, VIMAL A, KUMAR A. Why chitosan? From properties to perspective of mucosal drug delivery. Int J Biol Macromol. 2016;91: 615-622. [44] ANTER HM, HASHIM IA, AWADIN W, et al. Novel chitosan oligosaccharide -based nanoparticles for gastric mucosal administration of the phytochemical “apocynin”. Int J Nanomed. 2019;14:4911-4929. [45] MARTÍNEZ-PÉREZ B, QUINTANAR-GUERRERO D, TAPIA-TAPIA M, et al. Controlled-release biodegradable nanoparticles: From preparation to vaginal applications. Eur J Pharm Sci. 2018;115:185-195. [46] SUN M, YU X, WANG T, et al. Nasal adaptive chitosan-based nano-vehicles for anti-allergic drug delivery. Int J Bio Macromol. 2019; 135: 1182-1192. [47] LEI X, YA-TONG F, TIAN-JIAO Z, et al. Chemical modification of chitosan for efficient vaccine delivery. Molecules. 2018;23(2):229. [48] SINANI G, SESSEVMEZ M, KORAY G, et al. Modified chitosan-based nano adjuvants enhance immunogenicity of protein antigens after mucosal vaccination. Int J Pharmaceut. 2019;569:118592. [49] LAI JY, LUO LJ. Chitosan-g-poly(N-isopropylacrylamide) copolymers as delivery carriers for intracameral pilocarpine administration. Eur J Pharm Biopharm. 2017;113:140-148. [50] BOLES LR, BUMGARDNER JD, FUJIWARA T, et al. Characterization of trimethyl chitosan/polyethylene glycol derivatized chitosan blend as an injectable and degradable antimicrobial delivery system. Int J Biol Macromol. 2019;133:372-381. [51] CAPRIFICO AE, FOOT PJS, POLYCARPOU E, et al. Overcoming the blood-brain barrier: functionalised chitosan nanocarriers. Pharmaceutics. 2020;12(11):1013. [52] CORTÉS H, ALCALÁ-ALCALÁ S, CABALLERO-FLORÁN IH, et al. A reevaluation of chitosan-decorated nanoparticles to cross the blood-brain barrier. Membranes(Basel). 2020;10(9):212. [53] YAN T, ZHU S, HUI W, et al. Chitosan based pH-responsive polymeric prodrug vector for enhanced tumor targeted co-delivery of doxorubicin and siRNA. Carbohydr Polym. 2020;250:116781. [54] WU D, ZHU L, LI Y, et al. Superparamagnetic chitosan nanocomplexes for colorectal tumor-targeted delivery of irinotecan. Int J Pharm. 2020; 584:119394. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [3] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [4] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [5] | Jiang Hongying, Zhu Liang, Yu Xi, Huang Jing, Xiang Xiaona, Lan Zhengyan, He Hongchen. Effect of platelet-rich plasma on pressure ulcers after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1149-1153. |
| [6] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [7] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [8] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [9] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [10] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [11] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| [12] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [13] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [14] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [15] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||