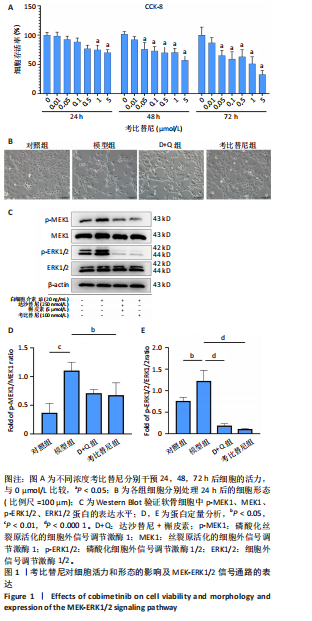
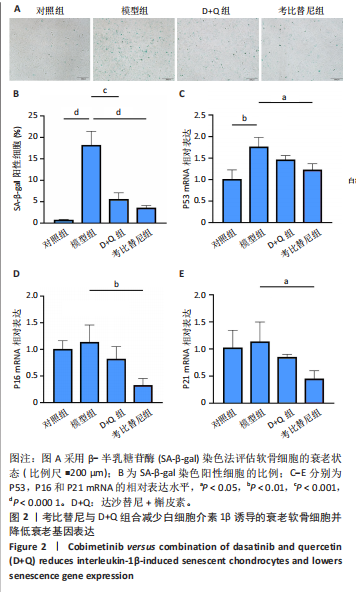
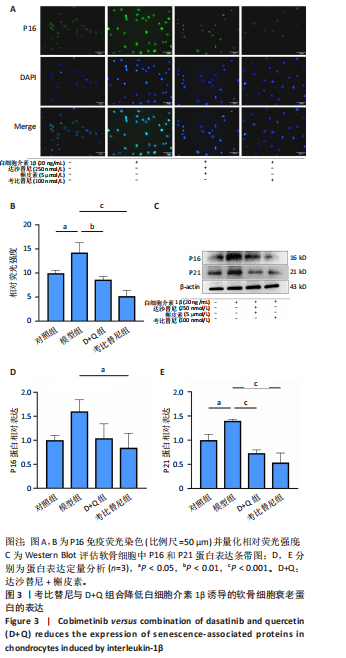
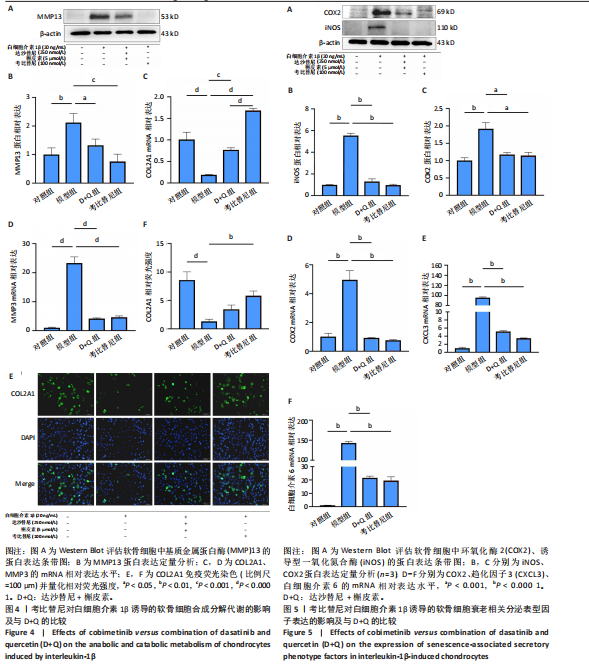
[1] JIANG Y. Osteoarthritis year in review 2021: biology. Osteoarthritis Cartilage. 2022;30(2):207-215.
[2] DOOLITTLE ML, SAUL D, KAUR J, et al. Multiparametric senescent cell phenotyping reveals targets of senolytic therapy in the aged murine skeleton. Nat Commun. 2023;14(1):4587.
[3] VINATIER C, DOMÍNGUEZ E, GUICHEUX J, et al. Role of the Inflammation-Autophagy-Senescence Integrative Network in Osteoarthritis. Front Physiol. 2018;9:706.
[4] DIEKMAN BO, LOESER RF. Aging and the emerging role of cellular senescence in osteoarthritis. Osteoarthritis Cartilage. 2024;32(4):365-371.
[5] GUO Q, CHEN X, CHEN J, et al. STING promotes senescence, apoptosis, and extracellular matrix degradation in osteoarthritis via the NF-κB signaling pathway. Cell Death Dis. 2021;12(1):13.
[6] YANG H, CHEN C, CHEN H, et al. Navitoclax (ABT263) reduces inflammation and promotes chondrogenic phenotype by clearing senescent osteoarthritic chondrocytes in osteoarthritis. Aging (Albany NY). 2020;12(13):12750-12770.
[7] XU M, PIRTSKHALAVA T, FARR JN, et al. Senolytics improve physical function and increase lifespan in old age. Nat Med. 2018;24(8):1246-1256.
[8] KIRKLAND JL, TCHKONIA T. Senolytic drugs: from discovery to translation. J Intern Med. 2020;288(5):518-536.
[9] OGRODNIK M, MIWA S, TCHKONIA T, et al. Cellular senescence drives age-dependent hepatic steatosis. Nat Commun. 2017;8:15691.
[10] KARNEWAR S, KARNEWAR V, SHANKMAN LS, et al. Treatment of advanced atherosclerotic mice with ABT-263 reduced indices of plaque stability and increased mortality. JCI Insight. 2024;9(2):e173863.
[11] YANG C, QIAO W, XUE Q, et al. The senolytic agent ABT263 ameliorates osteoporosis caused by active vitamin D insufficiency through selective clearance of senescent skeletal cells. J Orthop Translat. 2024;49:107-118.
[12] ZHAO J, ZHENG L, DAI G, et al. Senolytics cocktail dasatinib and quercetin alleviate chondrocyte senescence and facet joint osteoarthritis in mice. Spine J. 2025;25(1):184-198.
[13] WU W, WU X, QIU L, et al. Quercetin influences intestinal dysbacteriosis and delays alveolar epithelial cell senescence by regulating PTEN/PI3K/AKT signaling in pulmonary fibrosis. Naunyn Schmiedebergs Arch Pharmacol. 2024;397(7): 4809-4822.
[14] KRZYSTYNIAK A, WESIERSKA M, PETRAZZO G, et al. Combination of dasatinib and quercetin improves cognitive abilities in aged male Wistar rats, alleviates inflammation and changes hippocampal synaptic plasticity and histone H3 methylation profile. Aging (Albany NY). 2022;14(2):572-595.
[15] KANG C. Senolytics and Senostatics: A Two-Pronged Approach to Target Cellular Senescence for Delaying Aging and Age-Related Diseases. Mol Cells. 2019;42(12): 821-827.
[16] HERNANDEZ-SEGURA A, DE JONG TV, MELOV S, et al. Unmasking Transcriptional Heterogeneity in Senescent Cells. Curr Biol. 2017;27(17):2652-2660.
[17] SHARMA A K, ROBERTS R L, BENSON R D J, et al. The Senolytic Drug Navitoclax (ABT-263) Causes Trabecular Bone Loss and Impaired Osteoprogenitor Function in Aged Mice. Front Cell Dev Biol. 2020;8:354.
[18] LARKIN J, ASCIERTO P A, DRÉNO B, et al. Combined vemurafenib and cobimetinib in BRAF-mutated melanoma. N Engl J Med. 2014;371(20):1867-1876.
[19] ZOU J, LEI T, GUO P, et al. Mechanisms shaping the role of ERK1/2 in cellular senescence (Review). Mol Med Rep. 2019;19(2):759-770.
[20] CHAPPELL WH, STEELMAN LS, LONG JM, et al. Ras/Raf/MEK/ERK and PI3K/PTEN/Akt/mTOR inhibitors: rationale and importance to inhibiting these pathways in human health. Oncotarget. 2011;2(3):135-164.
[21] CHEN H, CHEN H, LIANG J, et al. TGF-β1/IL-11/MEK/ERK signaling mediates senescence-associated pulmonary fibrosis in a stress-induced premature senescence model of Bmi-1 deficiency. Exp Mol Med. 2020;52(1):130-151.
[22] BLAGOSKLONNY MV. Cellular senescence: when growth stimulation meets cell cycle arrest. Aging (Albany NY). 2023;15(4):905-913.
[23] WANG Z, MA L, SU M, et al. Baicalin induces cellular senescence in human colon cancer cells via upregulation of DEPP and the activation of Ras/Raf/MEK/ERK signaling. Cell Death Dis. 2018;9(2):217.
[24] CHAPPELL WH, CANDIDO S, ABRAMS SL, et al. Influences of TP53 and the anti-aging DDR1 receptor in controlling Raf/MEK/ERK and PI3K/Akt expression and chemotherapeutic drug sensitivity in prostate cancer cell lines. Aging (Albany NY). 2020;12(11):10194-10210.
[25] JIANG E, DINESH A, JADHAV S, et al. Canagliflozin shares common mTOR and MAPK signaling mechanisms with other lifespan extension treatments. Life Sci. 2023;328:121904.
[26] GKIONI L, NESPITAL T, BAGHDADI M, et al. The geroprotectors trametinib and rapamycin combine additively to extend mouse healthspan and lifespan. Nat Aging. 2025;5(7):1249-1265.
[27] CHEN Y, GUO H, LI L, et al. Long Non-Coding RNA (lncRNA) Small Nucleolar RNA Host Gene 15 (SNHG15) Alleviates Osteoarthritis Progression by Regulation of Extracellular Matrix Homeostasis. Med Sci Monit. 2020;26: e923868.
[28] MAURER S, KIRSCH V, RUTHS L, et al. Senolytic therapy combining Dasatinib and Quercetin restores the chondrogenic phenotype of human osteoarthritic chondrocytes by the release of pro-anabolic mediators. Aging Cell. 2025;24(1):e14361.
[29] COPP ME, FLANDERS MC, GAGLIARDI R, et al. The combination of mitogenic stimulation and DNA damage induces chondrocyte senescence. Osteoarthritis Cartilage. 2021;29(3):402-412.
[30] HAO W, CHANG M, SHI D, et al. Therapeutic targets in aging-related osteoarthritis: A focus on the extracellular matrix homeostasis. Life Sci. 2025;368:123487.
[31] MYAKISHEVA SN, LINKOVA NS, KOZHEVNIKOVA EO, et al. Chondrocytes secretory phenotype associated with aging: role in the pathogenesis of osteoarthritis and prospects for peptide bioregulation. Adv Gerontol. 2023;36(3):313-323.
[32] RIM YA, NAM Y, JU JH. The Role of Chondrocyte Hypertrophy and Senescence in Osteoarthritis Initiation and Progression. Int J Mol Sci. 2020;21(7):2358.
[33] OGRODNIK M, CARLOS ACOSTA J, ADAMS PD, et al. Guidelines for minimal information on cellular senescence experimentation in vivo. Cell. 2024;187(16): 4150-4175.
[34] YAN J, CHEN S, YI Z, et al. The role of p21 in cellular senescence and aging-related diseases. Mol Cells. 2024;47(11):100113.
[35] LI X, LI C, ZHANG W, et al. Inflammation and aging: signaling pathways and intervention therapies. Signal Transduct Target Ther. 2023;8(1):239.
[36] TONUTTI A, GRANATA V, MARRELLA V, et al. The role of WNT and IL-1 signaling in osteoarthritis: therapeutic implications for platelet-rich plasma therapy. Front Aging. 2023;4:1201019.
[37] HUANG X, YOU Y, XI Y, et al. p-Coumaric Acid Attenuates IL-1β-Induced Inflammatory Responses and Cellular Senescence in Rat Chondrocytes. Inflammation. 2020;43(2):619-628.
[38] SHENG J, LIU K, SUN D, et al. Association of RDM1 with osteosarcoma progression via cell cycle and MEK/ERK signalling pathway regulation. J Cell Mol Med. 2021; 25(16):8039-8046.
[39] YU Y, ZHANG X, LIU F, et al. A stress-induced miR-31-CLOCK-ERK pathway is a key driver and therapeutic target for skin aging. Nat Aging. 2021;1(9):795-809.
[40] GROGAN L, SHAPIRO P. Progress in the development of ERK1/2 inhibitors for treating cancer and other diseases. Adv Pharmacol. 2024;100:181-207.
[41] GUÉGAN J, EZAN F, GAILHOUSTE L, et al. MEK1/2 overactivation can promote growth arrest by mediating ERK1/2-dependent phosphorylation of p70S6K. J Cell Physiol. 2014;229(7):903-915.
[42] EL-HOSS J, KOLIND M, JACKSON MT, et al. Modulation of endochondral ossification by MEK inhibitors PD0325901 and AZD6244 (Selumetinib). Bone. 2014;59:151-161.
[43] WANG X, XUE Y, YE W, et al. The MEK-ERK1/2 signaling pathway regulates hyaline cartilage formation and the redifferentiation of dedifferentiated chondrocytes in vitro. Am J Transl Res. 2018,10(10):3068-3085.
[44] PRASADAM I, MAO X, SHI W, et al. Combination of MEK-ERK inhibitor and hyaluronic acid has a synergistic effect on anti-hypertrophic and pro-chondrogenic activities in osteoarthritis treatment. J Mol Med (Berl). 2013;91(3):369-380.
[45] FAN S, CHANG Y, XIONG X, et al. Reversible SAHH inhibitor ameliorates MIA-induced osteoarthritis of rats through suppressing MEK/ERK pathway. Biomed Pharmacother. 2024;170:115975.
[46] WANG M, YUANG-CHI CHANG A. Molecular mechanism of action and potential biomarkers of growth inhibition of synergistic combination of afatinib and dasatinib against gefitinib-resistant non-small cell lung cancer cells. Oncotarget. 2018;9(23):16533-16546.
[47] HASINOFF BB, PATEL D. Mechanisms of the Cardiac Myocyte-Damaging Effects of Dasatinib. Cardiovasc Toxicol. 2020;20(4):380-389.
[48] LI Y, HE Y, HAN X, et al. Dasatinib suppresses invasion and induces apoptosis in nasopharyngeal carcinoma. Int J Clin Exp Pathol. 2015;8(7):7818-7824.
[49] MILANOVIC M, FAN DNY, BELENKI D, et al. Senescence-associated reprogramming promotes cancer stemness. Nature. 2018;553(7686):96-100.
[50] SI H, YANG T, LI L, et al. miR-140 Attenuates the Progression of Early-Stage Osteoarthritis by Retarding Chondrocyte Senescence. Mol Ther Nucleic Acids. 2020;19:15-30.
[51] WU X, ZHOU X, WANG S, et al. DNA damage response (DDR): a link between cellular senescence and human cytomegalovirus. Virol J. 2023;20(1):250.
[52] ZHU Y, TCHKONIA T, PIRTSKHALAVA T, et al. The Achilles’ heel of senescent cells: from transcriptome to senolytic drugs. Aging Cell. 2015;14(4):644-658.
[53] UR RASHID H, XU Y, AHMAD N, et al. Promising anti-inflammatory effects of chalcones via inhibition of cyclooxygenase, prostaglandin E (2), inducible NO synthase and nuclear factor κb activities. Bioorg Chem. 2019;87:335-365.
[54] HAN Z, WANG K, DING S, et al. Cross-talk of inflammation and cellular senescence: a new insight into the occurrence and progression of osteoarthritis. Bone Res. 2024;12(1):69.
[55] FANG C, LIU B, WAN M. “Bone-SASP” in Skeletal Aging. Calcif Tissue Int. 2023; 113(1):68-82.
[56] JEON OH, WILSON DR, CLEMENT CC, et al. Senescence cell-associated extracellular vesicles serve as osteoarthritis disease and therapeutic markers. JCI Insight. 2019;4(7):e125019.
|