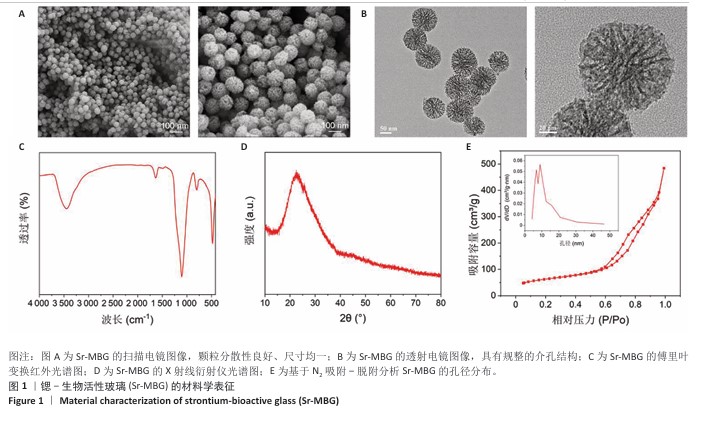
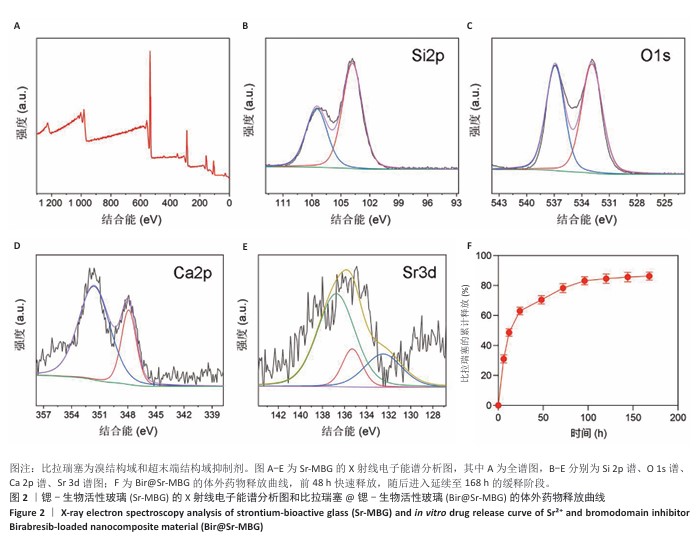
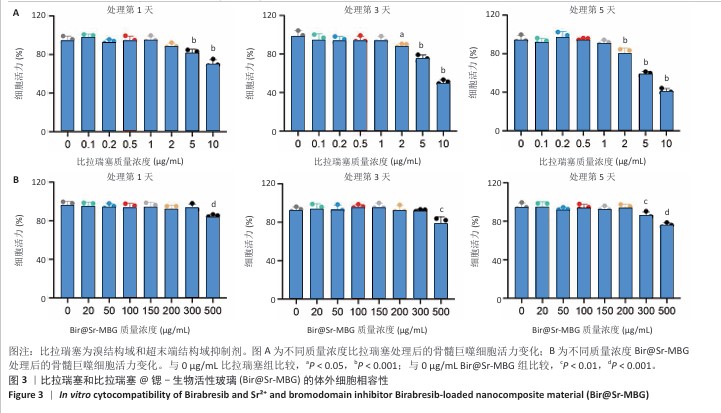
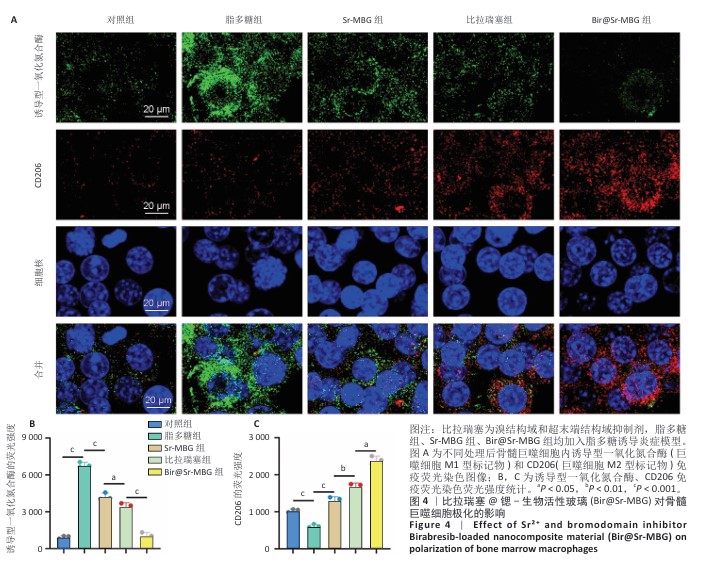
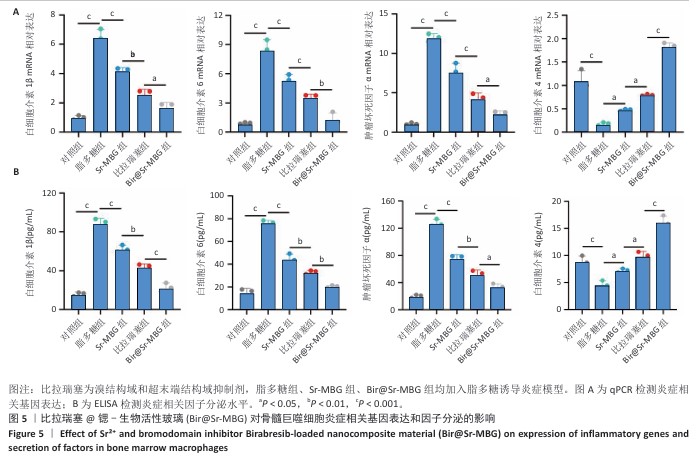
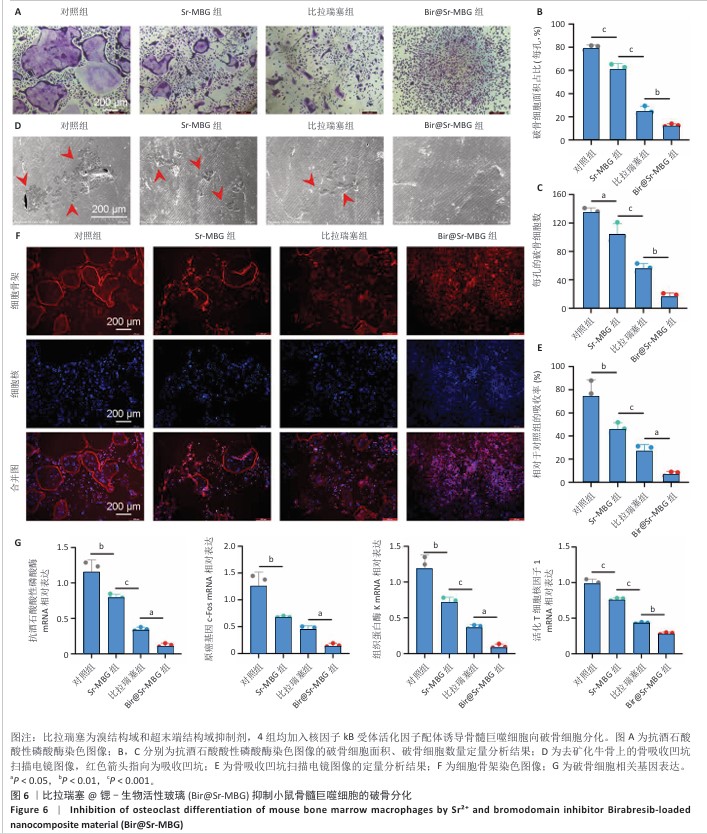
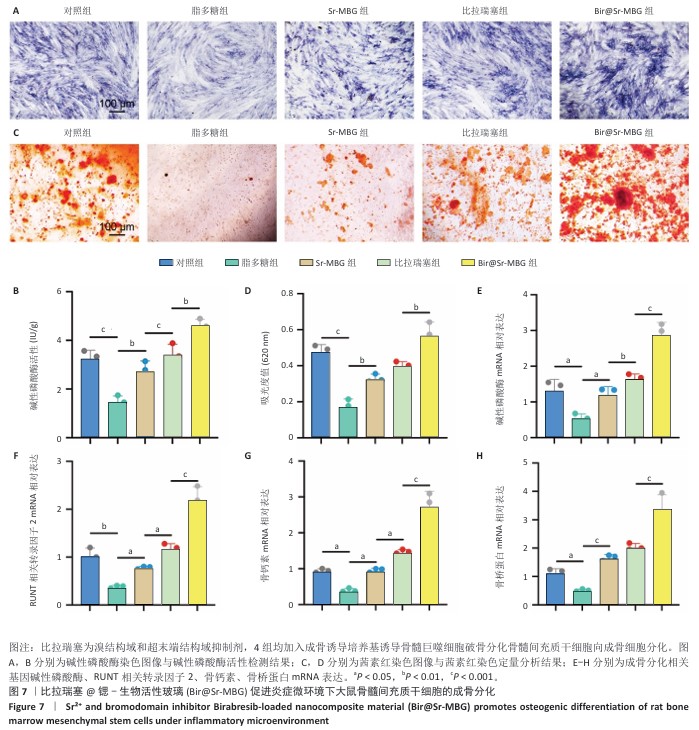
[1] HUANG Y, CHEN T, REN C, et al. High-Strength Gelatin Hydrogel Scaffold with Drug Loading Remodels the Inflammatory Microenvironment to Enhance Osteoporotic Bone Repair. Adv Mater. 2025;37(13):2501051.
[2] HUO S, LIU X, ZHANG S, et al. p300/CBP inhibitor A-485 inhibits the differentiation of osteoclasts and protects against osteoporotic bone loss. Int Immunopharmacol. 2021;94:107458.
[3] XIE Q, DU X, LIANG J, et al. FABP4 inhibition suppresses bone resorption and protects against postmenopausal osteoporosis in ovariectomized mice. Nat Commun. 2025;16(1):4437.
[4] KAHWATI LC, KISTLER CE, BOOTH G, et al. Screening for Osteoporosis to Prevent Fractures: A Systematic Evidence Review for the US Preventive Services Task Force. JAMA. 2025;333(6):509-531.
[5] BOLLAND MJ, NISA Z, MELLAR A, et al. Fracture Prevention with Infrequent Zoledronate in Women 50 to 60 Years of Age. N Engl J Med. 2025;392(3):239-248.
[6] ZHAO W, QIAN J, LI J, et al. From death to birth: how osteocyte death promotes osteoclast formation. Front Immunol. 2025;16:1551542.
[7] MELIS S, TROMPET D, CHAGIN AS, et al. Skeletal stem and progenitor cells in bone physiology, ageing and disease. Nat Rev Endocrinol. 2025;21(3):135-153.
[8] FU W, CHEN M, WANG K, et al. Tau is a receptor with low affinity for glucocorticoids and is required for glucocorticoid-induced bone loss. Cell Res. 2025;35(1):23-44.
[9] SHARMA G, LEE YH, KIM JC, et al. Bone Regeneration Enhanced by Quercetin-Capped Selenium Nanoparticles via miR206/Connexin43, WNT, and BMP signaling pathways. Aging Dis. 2025. doi: 10.14336/AD.2025.0025.
[10] SHAN C, WANG W, LU H, et al. A spatially organized Cd24a+/Pax9+ stem cell core governs postnatal tooth establishment. Sci Adv. 2025; 11(23):eadu5653.
[11] MENG S, LIU Q, DAI R, et al. Development of a novel macroscopic regulation and microscopic intervention mode nanosystem for osteoporosis treatment. Mater Today Bio. 2025;32:101829.
[12] SAPRA L, SAINI C, MISHRA PK, et al. Bacillus coagulans ameliorates inflammatory bone loss in post-menopausal osteoporosis via modulating the “Gut-Immune-Bone” axis. Gut Microbes. 2025;17(1): 2492378.
[13] CUI Y, LV B, LI Z, et al. Bone-Targeted Biomimetic Nanogels Re-Establish Osteoblast/Osteoclast Balance to Treat Postmenopausal Osteoporosis. Small. 2024;20(6):e2303494.
[14] XUE P, WANG J, FU Y, et al. Material-Mediated Immunotherapy to Regulate Bone Aging and Promote Bone Repair. Small. 2025;21(11): e2409886.
[15] KUBI JA, BRAH AS, CHEUNG KMC, et al. Low-molecular-weight estrogenic phytoprotein suppresses osteoporosis development through positive modulation of skeletal estrogen receptors. Bioact Mater. 2024; 42:299-315.
[16] KALYANARAMAN H, PAL CHINA S, CASTEEL DE, et al. Crosstalk between androgen receptor and protein kinase G signaling in bone: implications for osteoporosis therapy. Trends Pharmacol Sci. 2025;46(3):279-294.
[17] LIU X, LI X, HUO S, et al. Magnesium bioactive glass hybrid functionalized polyetheretherketone with immunomodulatory function to guide cell fate and bone regeneration. Colloids Surf B Biointerfaces. 2023;230:113523.
[18] HUO S, LYU Z, SU X, et al. Formation of a novel Cu-containing bioactive glass nano-topography coating with strong bactericidal capability and bone regeneration. Compos B Eng. 2023;253:110521.
[19] HUO S, LIU S, LIU Q, et al. Copper-Zinc-Doped Bilayer Bioactive Glasses Loaded Hydrogel with Spatiotemporal Immunomodulation Supports MRSA-Infected Wound Healing. Adv Sci (Weinh). 2024;11(5):e2302674.
[20] WANG L, JIANG S, ZHOU J, et al. From hard tissues to beyond: Progress and challenges of strontium-containing biomaterials in regenerative medicine applications. Bioact Mater. 2025;49:85-120.
[21] WU D, WU H, YU P, et al. Unraveling the molecular mechanism of aqueous extract of Sargentodoxa cuneata against ulcerative colitis from serum metabolomics and bioinformatics perspectives. J Chromatogr B Analyt Technol Biomed Life Sci. 2024;1249:124372.
[22] SU X, LYU Z, WU Y, et al. Strontium-doped bioactive glass/PDA functionalized polyetheretherketone with immunomodulatory property for enhancing photothermal clearance of Staphylococcus aureus. Mater Design. 2023;225:111552.
[23] BAO X, HUO S, WANG Z, et al. Multifunctional biomimetic hydrogel dressing provides anti-infection treatment and improves immunotherapy by reprogramming the infection-related wound microenvironment. J Nanobiotechnology. 2024;22(1):80.
[24] HUO S, WANG F, LYU Z, et al. Dual-functional polyetheretherketone surface modification for regulating immunity and bone metabolism. Chem Eng J. 2021;426:130806.
[25] MIAO L, ZIHAO X, JUNHAO S, et al. A New Nanoplatform Under NIR Released ROS Enhanced Photodynamic Therapy and Low Temperature Photothermal Therapy for Antibacterial and Wound Repair. Int J Nanomedicine. 2024;19:7509-7527.
[26] ZHANG S, HUO S, LI H, et al. Flufenamic acid inhibits osteoclast formation and bone resorption and act against estrogen-dependent bone loss in mice. Int Immunopharmacol. 2020;78:106014.
[27] HUO S, LIU Y, XU Z, et al. Specific Activation of the STING Pathway by Engineering Piezoelectric Hydrogel Microspheres for Boosting Implant Infection Immunotherapy. ACS Nano. 2025;19(17):16383-16404.
[28] ZHANG X, WANG S, YANG Q, et al. 3D Printed Microsphere-Hydrogel Scaffold Facilitates Restoration of Reinnervation in Bone Regeneration through Programmable Release of NGF/BMP-2 Mimetic Peptides. Adv Healthc Mater. 2025;14(20):e2501594.
[29] SU XJ, LIU S, HUO SC, et al. Polyetheretherketone surface modification by lithium-doped bioglass nanospheres to regulate bone immunity and promote osseointegration. Mater Design. 2024;238:112646.
[30] CHEN Y, WANG T, YAN Z, et al. Bioactive Glass-Induced B Cell Depletion Remodels the Osteoimmunological Microenvironment to Enhance Osteogenesis. Acta Biomater. 2025;201:648-664.
[31] BAI L, LI J, LI G, et al. Skeletal interoception and prospective application in biomaterials for bone regeneration. Bone Res. 2025;13(1):1.
[32] ARTAMONOV MY, SOKOV EL, KORNILOVA LE, et al. The Intraosseous Environment: Physiological Parameters, Regulatory Mechanisms, and Emerging Insights in Bone Biology. Int J Mol Sci. 2025;26(8):3876.
[33] YAO Y, CAI X, CHEN Y, et al. Estrogen deficiency-mediated osteoimmunity in postmenopausal osteoporosis. Med Res Rev. 2025; 45(2):561-575.
[34] MA S, LU Y, SUI S, et al. Unraveling the triad of immunotherapy, tumor microenvironment, and skeletal muscle biomechanics in oncology. Front Immunol. 2025;16:1572821.
[35] SONG S, GUO Y, YANG Y, et al. Advances in pathogenesis and therapeutic strategies for osteoporosis. Pharmacol Ther. 2022;237:108168.
[36] LU J, WU H, JIN H, et al. The influence of modified Qing E Formula on the differential expression of serum exosomal miRNAs in postmenopausal osteoporosis patients. Front Pharmacol. 2024;15: 1467298.
[37] ZENG Q, DU S, YUAN R, et al. Self-Healing Hydrogel Dressing with Solubilized Flavonoids for Whole Layer Regeneration of Diabetic Wound. Adv Healthc Mater. 2025;14(11):e2500734.
[38] WU Y, HUO S, LIU S, et al. Cu-Sr Bilayer Bioactive Glass Nanoparticles/Polydopamine Functionalized Polyetheretherketone Enhances Osteogenic Activity and Prevents Implant-Associated Infections through Spatiotemporal Immunomodulation. Adv Healthc Mater. 2023;12(32):e2301772.
[39] LIU XL, ZHANG CJ, SHI JJ, et al. Nacre-mimetic cerium-doped nano-hydroxyapatite/chitosan layered composite scaffolds regulate bone regeneration via OPG/RANKL signaling pathway. J Nanobiotechnology. 2023;21(1):259.
[40] LIANG QL, XU HG, YU L, et al. Binding-induced fibrillogenesis peptide inhibits RANKL-mediated osteoclast activation against osteoporosis. Biomaterials. 2023;302:122331.
[41] CASEY AM, RYAN DG, PRAG HA, et al. Pro-inflammatory macrophages produce mitochondria-derived superoxide by reverse electron transport at complex I that regulates IL-1β release during NLRP3 inflammasome activation. Nat Metab. 2025;7(3):493-507.
[42] JIN Z, XU H, ZHAO W, et al. Macrophage ATF6 accelerates corticotomy-assisted orthodontic tooth movement through promoting Tnfα transcription. Int J Oral Sci. 2025;17(1):28.
|