中国组织工程研究 ›› 2017, Vol. 21 ›› Issue (8): 1262-1267.doi: 10.3969/j.issn.2095-4344.2017.08.020
• 组织构建细胞学实验 cytology experiments in tissue construction • 上一篇 下一篇
小胶质细胞对中脑多巴胺能神经元铁代谢的影响及其机制:体外细胞学实验方案
石成奎,陈志强
- 青岛大学医学院,山东省青岛市 266071
-
出版日期:2017-03-18发布日期:2017-04-14 -
作者简介:石成奎,男,1990年生,汉族,硕士研究生,主要从事神经退行性疾病的发病机制及治疗策略研究。 -
基金资助:国家自然科学基金重点项目(99020288)
Effect of microglia on iron metabolism in midbrain dopaminergic neurons and the underlying mechanism: study protocol for an in vitro cellular experiment
Shi Cheng-kui, Chen Zhi-qiang
- Medical College of Qingdao University, Qingdao 266071, Shandong Province, China
-
Online:2017-03-18Published:2017-04-14 -
Supported by:the National Natural Science Foundation of China, No. 99020288
摘要:
中图分类号:
引用本文
石成奎,陈志强. 小胶质细胞对中脑多巴胺能神经元铁代谢的影响及其机制:体外细胞学实验方案[J]. 中国组织工程研究, 2017, 21(8): 1262-1267.
Shi Cheng-kui, Chen Zhi-qiang. Effect of microglia on iron metabolism in midbrain dopaminergic neurons and the underlying mechanism: study protocol for an in vitro cellular experiment[J]. Chinese Journal of Tissue Engineering Research, 2017, 21(8): 1262-1267.

| [1] Mirza B, Hadberg H, Thomsen P, et al. The absence of reactive astrocytosis is indicative of a unique inflammatory process in Parkinson’s disease. Neuroscience. 2000;95: 425-432.[2] Zhang X, Surguladze N, Slagle-Webb B, et al. Cellular iron status influences the functional relationship between microglia and oligodendrocytes. Glia. 2006;54(8):795-804.[3] Wu DC, Jackson-Lewis V, Vila M, et al. Blockade of microglial activation is neuroprotective in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine mouse model of Parkinson disease. J Neurosci. 2002;22(5):1763-1771. [4] Peng J, Stevenson FF, Oo ML, et al. Iron-enhanced paraquat-mediated dopaminergic cell death due to increased oxidative stress as a consequence of microglial activation. Free Radic Biol Med. 2009;46(2):312-320.[5] O'Brien-Ladner AR, Nelson SR, Murphy WJ, et al. Iron is a regulatory component of human IL-1beta production. Support for regional variability in the lung. Am J Respir Cell Mol Biol. 2000;23(1):112-119.[6] Helyar L, Sherman AR. Iron deficiency and interleukin 1 production by rat leukocytes. Am J Clin Nutr. 1987;46(2): 346-352.[7] Smirnov IM, Bailey K, Flowers CH, et al. Effects of TNF-alpha and IL-1beta on iron metabolism by A549 cells and influence on cytotoxicity. Am J Physiol. 1999;277(2 Pt1):L257-263.[8] Eisenstein RS, Tuazon PT, Schalinske KL, et al. Iron-responsive element-binding protein. Phosphorylation by protein kinase C. J Biol Chem. 1993;268:27363-27370.[9] Schalinske KL, Eisenstein RS. Phosphorylation and activation of both iron regulatory proteins 1 and 2 in HL-60 cells. J Biol Chem. 1996;271:7168-7176.[10] Kiemer AK, Förnges AC, Pantopoulos K, et al. ANP-induced decrease of iron regulatory protein activity is independent of HO-1 induction. Am J Physiol Gastrointest Liver Physiol. 2004;287:G518-6526.[11] An L, Sato H, Konishi Y, et al. Expression and localization of lactotransferrin messenger RNA in the cortex of Alzheimer's disease. Neurosci Lett. 2009;452(3):277-280.[12] Leveugle B, Faucheux BA, Bouras C, et al. Cellular distribution of the iron-binding protein lactotransferrin in the mesencephalon of Parkinson's disease cases. Acta Neuropathol. 1996;91(6):566-572.[13] Wang J, Jiang H, Xie JX. Time dependent effects of 6-OHDA lesions on iron level and neuronal loss in rat nigrostriatal system. Neurochem Res. 2004;29(12): 2239-2243.[14] Zhang S, Wang J, Song N, et al. Up-regulation of divalent metal transporter1 is involved in 1-methyl-4-phenylpyridinium (MPP(+))-induced apoptosis in MES23.5 cells. Neurobiol Aging. 2009;30(9):1466-1476.[15] Wang J, Jiang H, Xie JX. Ferroportin1 and hephaestin are involved in the nigral iron accumulation of 6-OHDA- lesioned rats. Eur J Neurosci. 2007;25(9):2766-2772. [16] Xu HM, Jiang H, Wang J, et al. Over-expressed human divalent metal transporter 1 is involved in iron accumulation in MES23.5 cells. Neurochem Int. 2008;52(6):1044-1051.[17] Song N, Wang J, Jiang H, et al. Ferroportin 1 but not hephaestin contributes to iron accumulation in a cell model of Parkinson's disease. Free Radic Biol Med. 2010;48(2): 332-341.[18] Youdim MB, Fridkin M, Zheng H. Bifunctional drug derivatives of MAO-B inhibitor rasagiline and iron chelator VK-28 as a more effective approach to treatment of brain ageing and ageing neurodegenerative diseases. Mech Ageing Dev. 2005;126(2):317-326.[19] Hirsch EC. Altered regulation of iron transport and storage in Parkinson's disease. J Neural Transm Suppl. 2006;(71): 201-204. |
| [1] | 姚晓玲, 彭建城, 许岳荣, 杨志东, 张顺聪. 可变角度零切迹前路椎间融合内固定系统治疗脊髓型颈椎病:30个月随访[J]. 中国组织工程研究, 2022, 26(9): 1377-1382. |
| [2] | 李志一, 贺鹏程, 边天月, 肖玉霞, 高 璐, 刘华胜. 铁死亡机制研究的文献计量学与可视化分析[J]. 中国组织工程研究, 2022, 26(8): 1202-1209. |
| [3] | 陈世坚, 李 舸, 张 钰, 关雅伦, 李雪娇, 刘书华, 李永超, 李韵峰, 高金风, 魏小月, 赵宇红. MPTP诱导帕金森病小鼠亚急性与慢性模型的比较及评价[J]. 中国组织工程研究, 2022, 26(8): 1247-1252. |
| [4] | 朱 婵, 韩栩珂, 姚承佼, 张 强, 刘 静, 邵 明. 针刺治疗帕金森病:动物实验显示的作用机制[J]. 中国组织工程研究, 2022, 26(8): 1272-1277. |
| [5] | 安维政, 何 萧, 任 帅, 刘建宇. 肌源干细胞在周围神经再生中的潜力[J]. 中国组织工程研究, 2022, 26(7): 1130-1136. |
| [6] | 温小雨, 孙玉浩, 夏 猛. 五脏温阳化瘀汤含药血清干预阿尔茨海默症细胞模型磷酸化tau蛋白的变化[J]. 中国组织工程研究, 2022, 26(7): 1068-1073. |
| [7] | 张璟琳, 冷 敏, 朱博恒, 汪 虹. 干细胞源外泌体促进糖尿病创面愈合的机制及应用[J]. 中国组织工程研究, 2022, 26(7): 1113-1118. |
| [8] | 周 颖, 张 幻, 廖 松, 胡凡琦, 易 静, 刘玉斌, 靳继德. 去铁胺联合干扰素γ预处理对人牙髓干细胞的免疫调节作用[J]. 中国组织工程研究, 2022, 26(7): 1012-1019. |
| [9] | 董苗苗, 赖 涵, 李曼玲, 许秀洪, 罗 梦, 王文豪, 周国平. 电针干预脑缺血再灌注损伤模型大鼠核苷酸结合寡聚化结构域样受体蛋白3/半胱天冬氨酸蛋白酶1表达的变化[J]. 中国组织工程研究, 2022, 26(5): 749-755. |
| [10] | 何云影, 李玲婕, 张舒淇, 李雨舟, 杨 生, 季 平. 聚丙烯酸/琼脂糖三维培养构建细胞球的方法[J]. 中国组织工程研究, 2022, 26(4): 553-559. |
| [11] | 何冠宇, 徐宝山, 杜立龙, 张同星, 霍振鑫, 申 力. 天然丝素蛋白构建仿生取向微通道纤维环支架[J]. 中国组织工程研究, 2022, 26(4): 560-566. |
| [12] | 陈晓旭, 罗雅馨, 毕浩然, 杨 琨. 脱细胞支架制备及其在组织工程和再生医学中的应用[J]. 中国组织工程研究, 2022, 26(4): 591-596. |
| [13] | 康坤龙, 王新涛. 生物支架材料促进骨髓间充质干细胞成骨分化的研究热点[J]. 中国组织工程研究, 2022, 26(4): 597-603. |
| [14] | 沈佳华, 付 勇. 基于石墨烯的纳米材料可否在干细胞领域应用[J]. 中国组织工程研究, 2022, 26(4): 604-609. |
| [15] | 张 通, 蔡金池, 袁志发, 赵海燕, 韩兴文, 王文己. 基于透明质酸的复合水凝胶修复骨关节炎软骨损伤:应用与机制[J]. 中国组织工程研究, 2022, 26(4): 617-625. |
中国组织工程研究杂志出版内容重点:组织构建;骨细胞;软骨细胞;细胞培养;成纤维细胞;血管内皮细胞;骨质疏松;组织工程
The measures are shown in Table 1.
|
|
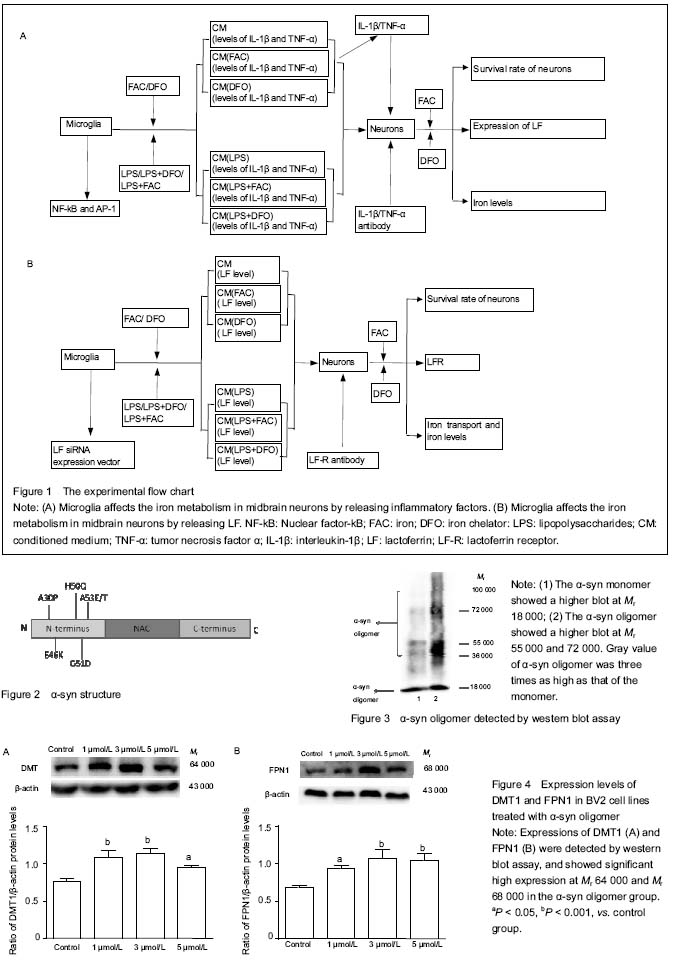
Note: CM: Conditioned medium; FAC: iron; DFO: iron chelator: LPS: lipopolysaccharides.
|
中国组织工程研究杂志出版内容重点:组织构建;骨细胞;软骨细胞;细胞培养;成纤维细胞;血管内皮细胞;骨质疏松;组织工程
中国组织工程研究杂志出版内容重点:组织构建;骨细胞;软骨细胞;细胞培养;成纤维细胞;血管内皮细胞;骨质疏松;组织工程
文题释义: 二价金属离子转运蛋白1(DMT1):是哺乳类跨膜铁转运蛋白,是主要的铁转入蛋白,广泛分布于人体各组织。主要功能是介导小肠上皮细胞的铁吸收以及参与铁从内吞小体移位到胞浆的过程,也参与其他二价金属如Zn2+、Mn2+、Co2+、Cd2+、Cn2+、Ni2+和Pb2+的转运。在帕金森病患者的黑质发现DMT1表达异常增加,因而DMT1可能也与某些神经退行性疾病的形成有关。 中国组织工程研究杂志出版内容重点:组织构建;骨细胞;软骨细胞;细胞培养;成纤维细胞;血管内皮细胞;骨质疏松;组织工程
铁是一切生命体都必不可少的微量金属元素之一,也是人体中最丰富的过渡金属。新的研究显示,铁的功能远比人们原先想象的重要。在生理条件的pH下,二价铁和三价铁可以互相转换,不仅参与了氧的转运和利用,还可以作为多种酶的辅助因子,参与蛋白质的合成、DNA的复制和膜蛋白的构筑等。对于脑而言,铁因为是参与髓鞘和神经递质合成的必要成分而尤为重要。目前大量的研究证实,黑质铁聚集参与了帕金森病的发病。在帕金森病患者及动物模型的黑质存在铁选择性的沉积;铁通过Fenton反应促进过氧化物的降解产生大量的羟自由基造成细胞的死亡;在帕金森病动物模型中应用铁离子螯合剂可起到明显的神经保护作用;应用MRI可以观察到帕金森病患者早期黑质铁含量就增加,而且铁的水平与帕金森病的进程相关,因此黑质铁含量检测可能成为帕金森病诊疗的生物标记物。 中国组织工程研究杂志出版内容重点:组织构建;骨细胞;软骨细胞;细胞培养;成纤维细胞;血管内皮细胞;骨质疏松;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||