中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (25): 6554-6565.doi: 10.12307/2026.416
• 组织构建实验造模 experimental modeling in tissue construction • 上一篇 下一篇
黄芪-桃仁缓解慢性肾脏病大鼠肾脏纤维化的作用机制
刘佳勇1,姚静静2,刘诗雨1,唐 艺1,董佳凝1,张 鑫1,侯岚炜1,康建英3,赵怡蕊3
- 1山西中医药大学第三临床学院,山西省晋中市 030619;2山西中医药大学太行本草研究院,山西省晋中市 030619;3山西省中西医结合医院,山西省太原市 030013
Mechanism by which astragalus-peach kernel alleviates renal fibrosis in chronic kidney disease rats
Liu Jiayong1, Yao Jingjing2, Liu Shiyu1, Tang Yi1, Dong Jianing1, Zhang Xin1, Hou Lanwei1, Kang Jianying3, Zhao Yirui3
- 1The Third Clinical College, Shanxi University of Traditional Chinese Medicine, Jinzhong 030619, Shanxi Province, China; 2Taihang Materia Medica Research Institute, Shanxi University of Traditional Chinese Medicine, Jinzhong 030619, Shanxi Province, China; 3Shanxi Provincial Hospital of Integrated Traditional Chinese and Western Medicine, Taiyuan 030013, Shanxi Province, China
摘要:
文题释义:
c-Myc:为重要的原癌基因,与人类癌症的发生高度相关,可参与调控细胞生长、增殖、代谢、凋亡等诸多过程。近年来有研究证实,c-Myc异常表达会引发肾小管损伤、促进炎症因子释放、加速慢性肾脏病的进展。
转化生长因子β/Smad3信号通路:是肾纤维化进展的核心信号传导通路,转化生长因子β通过促进其下游因子Smad3的磷酸化调控肾脏损伤的分子表达。转化生长因子β/Smad3信号通路可促进上皮-间充质转化、加速肌成纤维细胞形成,促进α-平滑肌肌动蛋白表达与Ⅰ型胶原合成,最终导致细胞外基质大量沉积在肾小球、肾小管间质等区域,造成肾脏结构损伤和功能丧失。
背景:课题组前期研究发现黄芪-桃仁可以缓解慢性肾脏病的进展,但作用机制有待进一步验证。
目的:探究黄芪-桃仁防治慢性肾脏病大鼠肾纤维化的作用及机制。
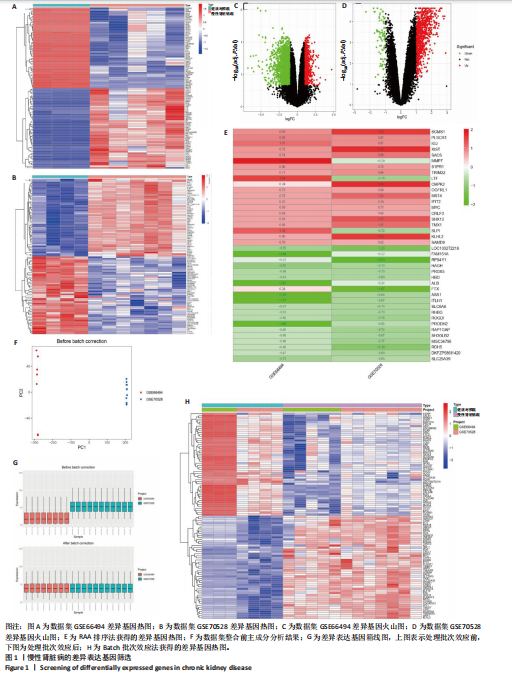
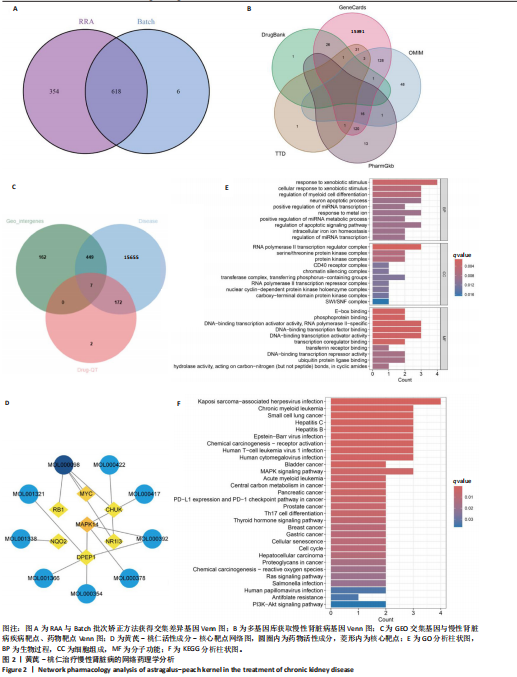
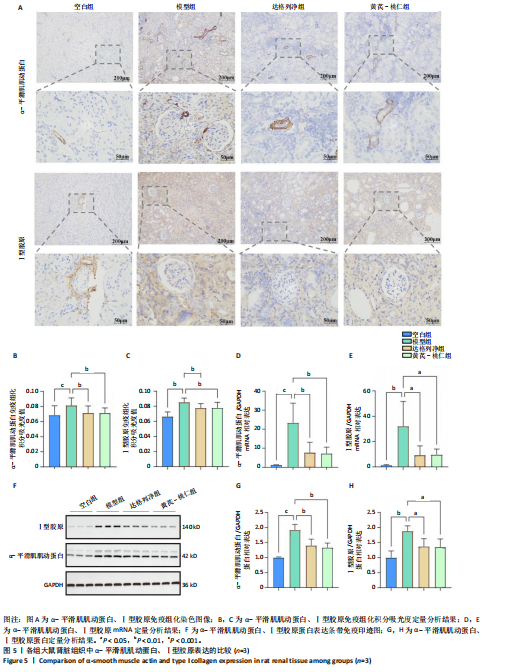
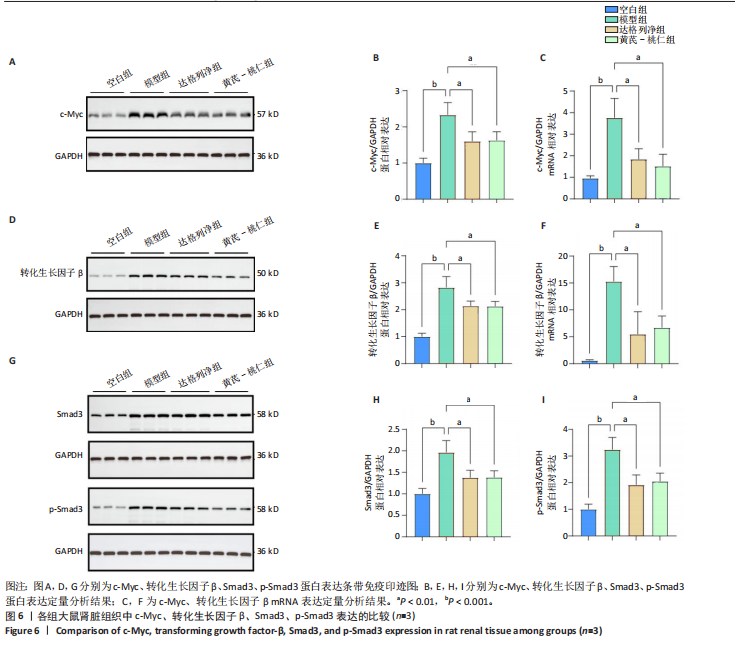
方法:①在GEO数据库(是由美国国立生物技术信息中心创建并维护的公共基因表达数据库,主要用于存储和共享高通量基因组数据,提供免费下载和分析工具,为开放数据库,研究已获得相关机构审查委员会的批准)中检索并筛选慢性肾病相关表达谱芯片数据集,结合网络药理学筛选慢性肾脏病核心靶点,将获得的关键基因进行分子对接验证。②将40只SD大鼠随机分为4组,空白组(n=10)不造模,模型组(n=10)、达格列净组(n=10)、黄芪-桃仁组(n=10)通过灌胃2%腺嘌呤溶液建立慢性肾脏病模型。造模成功后次日,空白组、模型组给予生理盐水灌胃,达格列净组给予达格列净灌胃,黄芪-桃仁组给予黄芪-桃仁(质量比为1∶1)灌胃,1次/d,连续给药8周。末次给药结束后,检测大鼠动脉血血清中肌酐、尿素氮水平,观察肾脏组织苏木精-伊红染色、Masson染色与α-平滑肌肌动蛋白、Ⅰ型胶原免疫组化染色,RT-qRCR检测肾脏组织中α-平滑肌肌动蛋白、Ⅰ型胶原、转化生长因子β、c-Myc mRNA表达,Western blot检测肾脏组织中α-平滑肌肌动蛋白、Ⅰ型胶原、转化生长因子β、c-Myc、Smad3、p-Smad3蛋白表达。
结果与结论:①GEO数据库结合网络药理学分析共获得黄芪-桃仁9个药物活性成分和7个核心疾病靶点(c-Myc、RB1、CHUK、MAPK14、DPEP1、NR1I3、NQO2);KEGG富集分析显示Ras-MAPK-c-Myc信号通路与慢性肾脏病相关;分子对接验证提示c-Myc与核心药物成分均有较强结合能力。②与空白组相比,模型组大鼠肾小球结构数量减少、结构异常,肾小管有大量炎性细胞浸润,大量成纤维细胞增殖,肾间质中有胶原纤维异常募集,血清中肌酐、尿素氮水平以及α-平滑肌肌动蛋白、Ⅰ型胶原的mRNA和蛋白表达上调(P < 0.05);与模型组相比,黄芪-桃仁组肾组织形态更趋完整,炎性细胞浸润明显降低,肾组织成纤维细胞增殖减少,血清中肌酐、尿素氮水平以及α-平滑肌肌动蛋白、Ⅰ型胶原的mRNA和蛋白表达降低(P < 0.05)。与空白组相比,模型组c-Myc、转化生长因子β mRNA和蛋白表达以及Smad3、p-Smad3蛋白表达升高(P < 0.05);与模型组相比,c-Myc、转化生长因子β mRNA和蛋白表达以及Smad3、p-Smad3蛋白表达降低(P < 0.05)。③结果表明黄芪-桃仁可延缓SD大鼠慢性肾脏病的进展,治疗机制可能与c-Myc/转化生长因子β/Smad3信号通路有关。
https://orcid.org/0009-0008-3173-7255 (刘佳勇)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号: