[1] DE JONG OG, KOOIJMANS SAA, MURPHY DE, et al. Drug delivery with extracellular vesicles: from imagination to innovation. Acc Chem Res. 2019;52(7):1761-1770.
[2] De Jong OG, Van Balkom BW, Schiffelers RM, et al. Extracellular vesicles: potential roles in regenerative medicine. Front Immunol. 2014;5:608.
[3] 张阳.慢病毒载体的研究进展[J].福建医科大学学报,2014,48(6):407-410.
[4] 吴清胜,李媛媛.腺病毒载体的研究及应用进展[J].国际生物制品学杂志, 2021,44(6):353-360.
[5] 翟贯星,傅卫辉,徐建青,等.腺相关病毒载体优化的研究进展[J].复旦学报(医学版),2021,48(6):827-833.
[6] RAYAMAJHI S, NGUYEN TDT, MARASINI R, et al. Macrophage-derived exosome-mimetic hybrid vesicles for tumortargeted drug delivery. Acta Biomater. 2019;94: 482-494.
[7] 董丽娜,蔡璐璐,赵明明,等.生物大分子载体在肿瘤纳米靶向药物研发中的应用研究进展[J].山东医药,2019,59(35):83-87.
[8] 翟宏强,丁维明,李桂玲.聚合物胶束注射给药系统的研究进展[J].中国医药生物技术,2015,10(5):441-447.
[9] 侯天宇,赖芳芳,李青山.载药纳米聚合物研究进展[J].江西化工,2011(2): 12-15.
[10] SILVA AK, LUCIANI N, GAZEAU F, et al. Combining magnetic nanoparticles with cell derived microvesicles for drug loading and targeting. Nanomedicine. 2015;11(3):645-55.
[11] HERRMANN IK, WOOD MJA, FUHRMANN G. Extracellular vesicles as a next-generation drug delivery platform. Nat Nanotechnol. 2021;16:748-759.
[12] HARDING C, HEUSER J, STAHL P. Receptor-mediated endocytosis of transferrin and recycling of the transferrin receptor in rat reticulocytes. J Cell Biol. 1983;97:329-39.
[13] PAN BT, JOHNSTONE RM. Fate of the transferrin receptor during maturation of sheep reticulocytes in vitro: selective externalization of the receptor. Cell. 1983; 33:967-978.
[14] JOHNSTONE RM, ADAM M, HAMMOND JR, et al. Turbide, Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes). J Biol Chem. 1987;262(19):9412-9420.
[15] RAPOSO G, NIJMAN HW, STOORVOGRL W, et al. B lymphocytes secrete antigen-presenting vesicles. J Exp Med 1996;183:1161-1172.
[16] CAO HQ, WANG H, HE XY, et al. Bioengineered macrophages can responsively transform into nanovesicles to target lung metastasis. Nano Lett. 2018;18:4762-4770.
[17] WEI DG, ZHAN WX, GAO Y, et al. RAB31 marks and controls an ESCRT-independent exosome pathway. Cell Res. 2021;31:157-177.
[18] SCHIAPPARELLI LM, SHARMA P, HE HY, et al. Proteomic screen reveals diverse protein transport between connected neurons in the visual system. Cell Rep. 2022;38:110287.
[19] SUN D, ZHUANG X, XIANG X, et al. A novel nanoparticle drug delivery system: theanti-inflammatory activity of curcumin is enhanced when encapsulated inexosomes. Mol Ther. 2010;18(9):1606-1614.
[20] WALKER S, BUSATTO S, PHAM A, et al. Extracellular vesicle-based drug delivery systems for cancer treatment. Theranostics. 2019;9(26):8001-8017.
[21] O’LOUGHLIN AJ, MAGER I, DE JONG OG, et al. Functional delivery of lipid-conjugated siRNA by extracellular Vesicles. Mol Ther. 2017;25:1580-1587.
[22] WANG J, LEE CJ, DECI MB, et al. MiR-101a loaded extracellular nanovesicles as bioactive carriers for cardiac repair. Nanomedicine. 2020;27:102201.
[23] SATO YT, UMEZAKI K, SAWADA S, et al. Engineering hybrid exosomes by membrane fusion with liposomes. Sci Rep. 2016;6:21933.
[24] OSKOUIE MN, AGHILI MOGHADDAM NS, BUTLER AE, et al. Therapeutic use of curcumin-encapsulated and curcumin-primed exosomes. J Cell Physiol. 2019; 234(6):8182-8191.
[25] KANG JY, KIM H, MUN D, et al. Co-delivery of curcumin and miRNA-144-3p using heart-targeted extracellular vesicles enhances the therapeutic efficacy for myocardial infarction. J Control Release. 2021;331:62-73.
[26] HANEY MJ, KlYACHKO NL, ZhAO Y, et al. Exosomes as drug delivery vehicles for Parkinson’s disease therapy. J Control Release. 2015;207:18-30.
[27] JAMUR MC, OLIVER C. Permeabilization of cell membranes. Methods Mol Biol. 2010;588:63-66.
[28] FUHRMANN G, SERIO A, MAZO M, et al. Active loading into extracellular vesicles significantly improves the cellular uptake and photodynamic effect of porphyrins. J Control Release. 2015;205:35-44.
[29] YI G, SON J, YOO J, et al. Application of click chemistry in nanoparticle modification and its targeted delivery. Biomater Res. 2018;22:13.
[30] HOOD JL. Post isolation modification of exosomes for nanomedicine applications. Nanomedicine (Lond). 2016;11:1745-1756.
[31] SINGH A, TRIVEDI P, JAIN NK. Advances in siRNA delivery in cancer therapy. Artif Cells Nanomed Biotechnol. 2018;46:274-283.
[32] PIFFOUX M, SILVA AKA, WILHELM C, et al. Modification of extracellular vesicles by fusion with liposomes for the design of personalized biogenic drug delivery systems. ACS Nano. 2018;12(7):6830-6842.
[33] ESCUDE MARTINEZ DE CASTILLA P, TONG L, HUANG C, et al. Extracellular vesicles as a drug delivery system: a systematic review of preclinical studies. Adv Drug Deliv Rev. 2021;175:113801.
[34] KOOIJMANS SAA, FLIERVOET LAL, VAN DER MEEL R, et al. PEGylated and targeted extracellular vesicles display enhanced cell specificity and circulation time. J Control Release. 2016;224:77-85.
[35] WAN Y, WANG L, ZHU C, et al, Aptamer-conjugated extracellular nanovesicles for targeted drug delivery. Cancer Res. 2018;78(3):798-808.
[36] KIM MS, HANEY MJ, ZHAO Y, et al. Engineering macrophage-derived exosomes for targeted paclitaxel delivery to pulmonary metastases: in vitro and in vivo evaluations. Nanomedicine. 2018;14(1):195-204.
[37] DAMS ET, LAVERMAN P, OYEN WJ, et al. Accelerated blood clearance and altered biodistribution of repeated injections of sterically stabilized liposomes. J Pharmacol Exp Ther. 2000;292(3):1071-1079.
[38] ISHIDA T, MAEDA R, ICHIHARA M, et al. Accelerated clearance of PEGylated liposomes in rats after repeated injections. J Control Release. 2003;88(1):35-42.
[39] FAN J, PAN J, ZHANG X, et al. A peptide derived from the N-terminus of charged multivesicular body protein 6 (CHMP6) promotes the secretion of gene editing proteins via small extracellular vesicle production. Bioengineered. 2022;13(3):4702-4716.
[40] KIM H, MUN D, KANG JY, et al. Improved cardiac-specific delivery of RAGE siRNA within small extracellular vesicles engineered to express intense cardiac targeting peptide attenuates myocarditis. Mol Ther Nucleic Acids. 2021;24:1024-1032.
[41] LIU B, LEE BW, NAKANISHI K, et al. Cardiac recovery via extended cell-free delivery of extracellular vesicles secreted by cardiomyocytes derived from induced pluripotent stem cells. Nat Biomed Eng. 2018; 2(5):293-303.
[42] HEALLEN TR, MARTIN JF. Heart repair via cardiomyocyte-secreted vesicles. Nat Biomed Eng, 2018;2:271-272.
[43] LE BRAS A. Exosome-based therapy to repair the injured heart. Nat Rev Cardiol. 2018;15(7):382.
[44] WEI Z, CHEN Z, ZHAO Y, et al. Mononuclear phagocyte system blockade using extracellular vesicles modified with CD47 on membrane surface for myocardial infarction reperfusion injury treatment. Biomaterials. 2021;275:121000.
[45] MURPHY DE, DE TONG OG, BROUWER M, et al. Extracellular vesicle-based therapeutics: natural versus engineered targeting and trafficking. Exp Mol Med. 2019;51(3):1-12.
[46] 韩超珊. MiR--675和水凝胶对间充质干细胞外泌体的改造和应用研究[D].苏州:苏州大学,2019.
[47] SONG Y, ZHANG C, ZHANG J, et al. Localized injection of miRNA-21-enriched extracellular vesicles effectively restores cardiac function after myocardial infarction. Theranostics. 2019;9;2346-2360.
[48] FU DL, JIANG H, LI C, et al. MicroRNA-338 in MSCs-derived exosomes inhibits cardiomyocyte apoptosis in myocardial infarction. Eur Rev Med Pharmacol Sci. 2020;24;10107-10117.
[49] LI Y, YANG R, GUO B, et al. Exosomal miR-301 derived from mesenchymal stem cells protects myocardial infarction by inhibiting myocardial autophagy. Biochem Biophys Res Commun. 2019;514:323-328.
[50] WEI Z, QIAO S, ZHAO J, et al. miRNA-181a over-expression in mesenchymal stem cell-derived exosomes inflfluenced inflflammatory response after myocardial ischemia-reperfusion injury. Life Sci. 2019; 232:116632.
[51] DENG S, ZHOU X, Ge Z, et al. Exosomes from adipose-derived mesenchymal stem cells ameliorate cardiac damage after myocardial infarction by activating S1P/SK1/S1PR1 signaling and promoting macrophage M2 polarization. Int J Biochem Cell Biol. 2019;114:105564.
[52] YU DW, GE PP, LIU AL, et al. HSP20-mediated cardiomyocyte exosomes improve cardiac function in mice with myocardial infarction by activating Akt signaling pathway. Eur Rev Med Pharmacol Sci. 2019; 23(11):4873-4881.
[53] ZHANG N, SONG Y, HUANG ZC, et al. Monocyte mimics improve mesenchymal stem cell-derived extracellular vesicle homing in a mouse MI/RI model. Biomaterials. 2020;255:120168.
[54] LI Q, HUANG Z, WANG Q, et al. Targeted immunomodulation therapy for cardiac repair by platelet membrane engineering extracellular vesicles via hitching peripheral monocytes. Biomaterials. 202;284:121529.
[55] HUANG K, OZPINAR EW, SU T, et al. An off-the-shelf artifificial cardiac patch improves cardiac repair after myocardial infarction in rats and pigs. Sci Transl Med. 2020;12(538):eaat9683.
[56] GAO L, KUPFER ME, JUNG JP, et al. Myocardial tissue engineering with cells derived from humaninduced pluripotent stem cells and a native-like, high-resolution, 3- dimensionally printed scaffold. Circ Res. 2017;120(8):1318-1325.
[57] TANG J, VANDERGRIFF A, WANG Z, et al. A regenerative cardiac patch formed by spray painting of biomaterials onto the heart. Tissue Eng Part C. 2017;23(3):146-155.
[58] ZHU D, LI Z, HUANG K, et al. Minimally invasive delivery of therapeutic agents by hydrogel injection into the pericardial cavity for cardiac repair. Nat Commun. 2021;12(1):1412.
[59] ZHANG Y, CAI Z, SHEN Y, et al. Hydrogel-load exosomes derived from dendritic cells improve cardiac function via Treg cells and the polarization of macrophages following myocardial infarction. J Nanobiotechnol. 2021;19(1):271.
[60] MENTKOWSKI KI, LANG JK. Exosomes engineered to express a cardiomyocyte binding peptide demonstrate improved cardiac retention in vivo. Sci Rep. 2019; 9(1):10041.
[61] VANDERGRIFF A, HUANG K, SHEN D, et al. Targeting regenerative exosomes to myocardial infarction using cardiac homing peptide. Theranostics. 018;8(7):1869-1878.
[62] CIULLO A, BIEMMI V, MILANO G, et al. Exosomal expression of CXCR4 targets cardioprotective vesicles to myocardial infarction and improves outcome after systemic administration. Int J Mol Sci. 2019;20(3):468.
[63] MAO S, ZHAO J, ZHANG ZJ, et al. MiR-183-5p overexpression in bone mesenchymal stem cell-derived exosomes protects against myocardial ischemia/reperfusion injury by targeting FOXO1. Immunobiology. 2022;227(3):152204.
[64] WANG Y, DING N, GUAN G, et al. Rapid delivery of Hsa-miR-590-3p using targeted exosomes to treat acute myocardial infarction through regulation of the cell cycle. J Biomed Nanotechnol. 2018;14:968-977
[65] CHEN W, LI L, WANG J, et al. Extracellular vesicle YsRNA in atherosclerosis. Clin Chim Acta. 2021;517:15-22.
[66] PERDOMO L, VIDAL-GOMEZ X, SOLETI R, et al. Large Extracellular vesicle-associated rap1 accumulates in atherosclerotic plaques, correlates with vascular risks and is involved in atherosclerosis. Circ Res. 2020;127(6):747-760.
[67] WU GH, ZHANG JF, ZHAO QR, et al. molecularly engineered macrophage-derived exosomes with inflammation tropism and intrinsic heme biosynthesis for atherosclerosis treatment. Angew Chem Int Ed Engl. 2020;59:4068-4074.
[68] PAPE AC, BAKKER MH, TSENG CC, et al. An injectable and drug-loaded supramolecular hydrogel for local catheter injection into the pig heart. J Vis Exp. 2015;(100):e52450.
[69] CHENG G, ZHU D, HUANG K, et al. Minimally invasive delivery of a hydrogel-based exosome patch to prevent heart failure. J Mol Cell Cardiol. 2022;169:113-121.
[70] SINDI HA, RUSSOMANNO G, SATTA S, et al. Therapeutic potential of KLF2-induced exosomal microRNAs in pulmonary hypertension. Nat Commun. 2020;11:1185.
[71] LIU LN, ZHANG HR, MAO HY, et al. Exosomal miR-320d derived from adipose tissue-derived MSCs inhibits apoptosis in cardiomyocytes with atrial fibrillation (AF). Artif Cells Nanomed Biotechnol. 2019;47:3976-3984.
[72] KIM H, YUN N, MUN D, et al. Cardiac-specific delivery by cardiac tissue-targeting peptide-expressing exosomes. Biochem Biophys Res Commun. 2018;499(4):803-808.
[73] TIAN Y, Li S, SONG J, et al. A doxorubicin delivery platform using engineered natural membrane vesicle exosomes for targeted tumor therapy. Biomaterials. 2014;35(7):2383-2390.
[74] SCHINDLER C, COLLISON A, MATTHEWS C, et al. Exosomal delivery of doxorubicin enables rapid cell entry and enhanced in vitro potency. PLoS One. 2019;14(3):e0214545.
[75] SUN W, ZHAO P, ZHOU Y, et al. Ultrasound targeted microbubble destruction assisted exosomal delivery of miR-21 protects the heart from chemotherapy associated cardiotoxicity. Biochem Biophys Res Commun. 2020;532(1):60-67.
[76] WAN Z, ZHAO L, LU F, et al. Mononuclear phagocyte system blockade improves therapeutic exosome delivery to the myocardium. Theranostics. 2020;10(1):218-230.
[77] LASSER C, ELDH M, LOTVALL J. Isolation and characterization of RNA-containing exosomes. J Vis Exp. 2012;(59):e3037.
[78] 贺继刚,韩金秀,撒亚莲,等.过表达IDO骨髓间充质干细胞分泌外泌体改善移植心脏存活[J].安徽医科大学学报,2018,53(9):1383-1387.
[79] GHARPURE KM, WU SY, LI C, et al. Nanotechnology: future of oncotherapy. Clin Cancer Res. 2015;21(14):3121-3130.
[80] BORRELLI DA, YANKSON K, SHUKLA N, et al. Extracellular vesicle therapeutics for liver disease. J Control Release. 2018;273:86-98.
[81] WIKLANDER OP, NORDIN JZ, O’LOUGHLIN A, et al. Extracellular vesicle in vivo biodistribution is determined by cell source, route of administration and targeting. Journal of extracellular vesicles. 2015;4:26316.
[82] SUH A, PHAM A, CRESS MJ, et al. Adipose-derived cellular and cell-derived regenerative therapies in dermatology and aesthetic rejuvenation. Ageing Res Rev. 2019;54:100933.
[83] AMIRI A, BAGHERIFAR R, ANSARI DEZFOULI E, et al. Exosomes as bio-inspired nanocarriers for RNA delivery:preparation and applications. J Transl Med. 2022;20(1):125. |
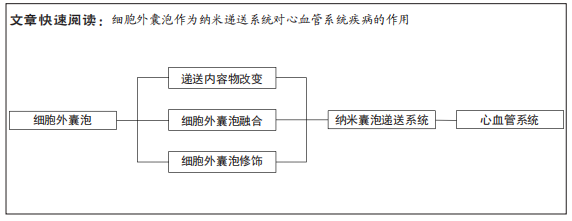
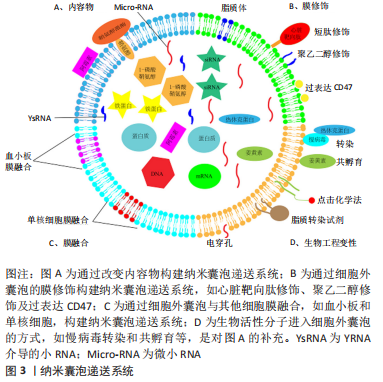
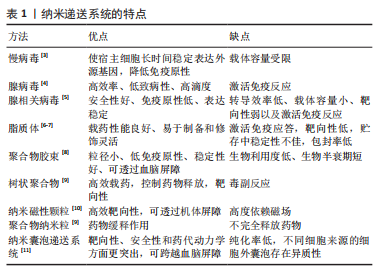


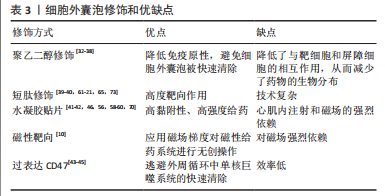
 文章总结了细胞外囊泡修饰和优缺点,见表3。
文章总结了细胞外囊泡修饰和优缺点,见表3。
 3.3 综述的局限性 目前针对纳米囊泡递送系统科学领域仍需解决的问题是细胞外囊泡的大规模生产,或许在不久的将来,有可能会产生与生物系统纯化功能相似的合成外泌体,但产生与自然细胞外囊泡成分相似的细胞外囊泡仍然是一个技术挑战,而且存在免疫原性和安全性等问题。因此,提高细胞外囊泡生产的技术改进值得进一步的临床发展。如果能够开发出产生高产量、低毒性的细胞外囊泡的方法,那么这些药物将在临床上具有巨大的应用前景。并且将大分子物质封装到细胞外囊泡中仍然是一个挑战。因此,建立用于临床应用的大规模的药物负载细胞外囊泡有待进一步关注[83]。
3.3 综述的局限性 目前针对纳米囊泡递送系统科学领域仍需解决的问题是细胞外囊泡的大规模生产,或许在不久的将来,有可能会产生与生物系统纯化功能相似的合成外泌体,但产生与自然细胞外囊泡成分相似的细胞外囊泡仍然是一个技术挑战,而且存在免疫原性和安全性等问题。因此,提高细胞外囊泡生产的技术改进值得进一步的临床发展。如果能够开发出产生高产量、低毒性的细胞外囊泡的方法,那么这些药物将在临床上具有巨大的应用前景。并且将大分子物质封装到细胞外囊泡中仍然是一个挑战。因此,建立用于临床应用的大规模的药物负载细胞外囊泡有待进一步关注[83]。