| [1] Doerks T, Copley RR, Schultz J, et al. Systematic identification of novel protein domain families associated with nuclear functions. Genome Res. 2002;12(1):47-56.[2] Cannonier SA, Sterling JA. The Role of Hedgehog Signaling in Tumor Induced Bone Disease. Cancers (Basel). 2015;7(3): 1658-1683.[3] McMahon AP, Ingham PW, Tabin CJ, et al. Developmental roles and clinical significance of Hedgehog signaling. Curr Top Dev Biol. 2003;53:1-114.[4] Chiang C, Litingtung Y, Lee E, et al. Cyclopia and defective axial patterning in mice lacking Sonic hedgehog gene function. Nature. 1996;383(6599):407-413.[5] Bitgood MJ, Shen L, McMahon AP, et al. Sertoli cell signaling by Desert hedgehog regulates the male germline. Curr Biol. 1996;6(3):298-304.[6] Mirsky R, Parmantier E, McMahon AP, et al. Schwann Cell‐Derived Desert Hedgehog Signals Nerve Sheath Formation. Ann New York Acad Sci. 1999;883(1):196-202.[7] Marigo V, Davey RA, Zuo Y, et al. Biochemical evidence that patched is the Hedgehog receptor. Nature. 1996; 384(6605): 176-179.[8] Murone M, Rosenthal A, de Sauvage FJ, et al. Sonic hedgehog signaling by the patched–smoothened receptor complex. Curr Biol. 1999;9(2):76-84.[9] Jemtland R, Divieti P, Lee K, et al. Hedgehog promotes primary osteoblast differentiation and increases PTHrP mRNA expression and iPTHrP secretion. Bone. 2003; 32(6):611-620.[10] Zhang XM, Ramalho-Santos M, McMahon AP, et al. Smoothened mutants reveal redundant roles for Shh and Ihh signaling including regulation of L/R symmetry by the mouse node. Cell. 2001;106(2):781-792.[11] McMahon AP. More surprises in the Hedgehog signaling pathway. Cell. 2000;100(2):185-188.[12] Amano K, Densmore MJ, Lanske B, et al. Conditional Deletion of Indian Hedgehog in Limb Mesenchyme Results in Complete Loss of Growth Plate Formation but Allows Mature Osteoblast Differentiation. J Bone Miner Res. 2015;30(12): 2262-2272.[13] Tian Y, Xu Y, Fu Q, et al. Osterix is required for Sonic hedgehog-induced osteoblastic MC3T3-E1 cell differentiation. Cell Biochem Biophys. 2012;64(3):169-176.[14] Nakamura T, Naruse M, Chiba Y, et al. Novel hedgehog agonists promote osteoblast differentiation in mesenchymal stem cells. J Cell Physiol. 2015. 230(4):922-929.[15] Shimoyama A, Wada M, Ikeda F, et al. Ihh/Gli2 signaling promotes osteoblast differentiation by regulating Runx2 expression and function. Mol Biol Cell. 2007;18(7): 2411-2418.[16] Tu X, Joeng KS, Long F, et al. Indian hedgehog requires additional effectors besides Runx2 to induce osteoblast differentiation. Dev Biol. 2012;362(1):76-82.[17] Ohba S, Kawaguchi H, Kugimiya F, et al. Patched1 haploinsufficiency increases adult bone mass and modulates Gli3 repressor activity. Dev Cell. 2008;14(5): 689-699.[18] Ben-awadh AN, Delgado-Calle J, Tu X, et al. Parathyroid hormone receptor signaling induces bone resorption in the adult skeleton by directly regulating the RANKL gene in osteocytes. Endocrinology. 2014;155(8):2797-2809.[19] Mak KK, Bi Y, Wan C, et al. Hedgehog signaling in mature osteoblasts regulates bone formation and resorption by controlling PTHrP and RANKL expression. Dev Cell. 2008; 14(5):674-688.[20] Huang JC, Sakata T, Pfleger LL, et al. PTH differentially regulates expression of RANKL and OPG. J Bone Miner Res. 2004;19(2):235-244.[21] Boabaid F, Berry JE, Koh AJ, et al. The role of parathyroid hormone-related protein in the regulation of osteoclastogenesis by cementoblasts. J Periodontol. 2004; 75(9):1247-1254.[22] Kim S, Yamazaki M, Shevde NK, et al. Transcriptional control of receptor activator of nuclear factor-kappaB ligand by the protein kinase A activator forskolin and the transmembrane glycoprotein 130-activating cytokine, oncostatin M, is exerted through multiple distal enhancers. Mol Endocrinol. 2007; 21(1):197-214.[23] Fu Q, Manolagas SC, and O'Brien CA, et al. Parathyroid hormone controls receptor activator of NF-kappaB ligand gene expression via a distant transcriptional enhancer. Mol Cell Biol. 2006;26(17):6453-6468.[24] Fu Q, Jilka RL, Manolagas SC, et al. Parathyroid hormone stimulates receptor activator of NFkappa B ligand and inhibits osteoprotegerin expression via protein kinase A activation of cAMP-response element-binding protein. J Biol Chem. 2002; 277(50):48868-48875.[25] Zhuang LL, Jin R, Zhu LH, et al. Promoter characterization and role of cAMP/PKA/CREB in the basal transcription of the mouse ORMDL3 gene. PLoS One. 2013;8(4):e60630.[26] Sands WA, Palmer TM. Regulating gene transcription in response to cyclic AMP elevation. Cell Signal. 2008;20(3): 460-466.[27] Bidwell P, Joh K, Leaver HA, et al. Prostaglandin E2 activates cAMP response element-binding protein in glioma cells via a signaling pathway involving PKA-dependent inhibition of ERK. Prostaglandins Other Lipid Mediat. 2010;91(1-2):18-29.[28] Park HJ, Baek K, Baek JH, et al. The cooperation of CREB and NFAT is required for PTHrP-induced RANKL expression in mouse osteoblastic cells. J Cell Physiol. 2015;230(3): 667-679.[29] Lee HL, Bae OY, Baek KH, et al. High extracellular calcium-induced NFATc3 regulates the expression of receptor activator of NF-kappaB ligand in osteoblasts. Bone. 2011;49(2): 242-249.[30] Takami M, Takahashi N, Udagawa N, et al. Intracellular calcium and protein kinase C mediate expression of receptor activator of nuclear factor-kappaB ligand and osteoprotegerin in osteoblasts. Endocrinology. 2000;141(12):4711-4719.[31] Datta NS, Abou-Samra AB. PTH and PTHrP signaling in osteoblasts. Cell Signal. 2009;21(8):1245-1254.[32] Akhtari M, Mansuri J, Newman KA, et al. Biology of breast cancer bone metastasis. Cancer Biol Ther. 2008;7(1):3-9.[33] Hirai T, Kobayashi T, Nishimori S, et al. Bone Is a Major Target of PTH/PTHrP Receptor Signaling in Regulation of Fetal Blood Calcium Homeostasis. Endocrinology. 2015; 156(8):2774-2780.[34] Zayzafoon M. Calcium/calmodulin signaling controls osteoblast growth and differentiation. J Cell Biochem. 2006;97(1):56-70.[35] Shin J, Jang H, Lin J, et al. PKCbeta positively regulates RANKL-induced osteoclastogenesis by inactivating GSK-3beta. Mol Cells. 2014;37(10):747-752.[36] Yao J, Li J, Zhou L, et al. Protein kinase C inhibitor, GF109203X attenuates osteoclastogenesis, bone resorption and RANKL-induced NF-kappaB and NFAT activity. J Cell Physiol. 2015;230(6):1235-1242.[37] Malaval L, Prideaux M, Dallas SL, et al. Parathyroid Hormone Induces Bone Cell Motility and Loss of Mature Osteocyte Phenotype through L-Calcium Channel Dependent and Independent Mechanisms. Plos One. 2015;10(5):e0125731. |
.jpg) 文题释义:
OPG-RANK-RANKL系统:在骨组织代谢平衡中,成骨细胞与破骨细胞的平衡主要通过OPG-RANK-RANKL系统调控轴进行;RANKL即核因子κB受体活化因子配体,是一种促破骨细胞分化因子,可与破骨前体细胞上的RANK结合,刺激破骨前体细胞向破骨细胞分化、成熟;OPG即骨保护素,主要表达于成骨前体细胞,是RANKL的竞争性配体。近几年的研究发现,成骨细胞中Hedgehog信号通路活性增强后,可通过调节其下游信号分子而增强RANKL的表达,RANKL进一步促进破骨前体细胞向破骨细胞分化成熟。
甲状旁腺相关蛋白:甲状旁腺相关蛋白是一种旁分泌因子,是Hedgehog信号通路调节成骨细胞RANKL表达中的重要中间产物,Hedgehog信号增强后显示上调胞内甲状旁腺相关蛋白,再由甲状旁腺相关蛋白激活下游因子环磷酸腺苷应答元件结合蛋白和活化T细胞核因子,并与RANKL基因结合,促进其表达。
文题释义:
OPG-RANK-RANKL系统:在骨组织代谢平衡中,成骨细胞与破骨细胞的平衡主要通过OPG-RANK-RANKL系统调控轴进行;RANKL即核因子κB受体活化因子配体,是一种促破骨细胞分化因子,可与破骨前体细胞上的RANK结合,刺激破骨前体细胞向破骨细胞分化、成熟;OPG即骨保护素,主要表达于成骨前体细胞,是RANKL的竞争性配体。近几年的研究发现,成骨细胞中Hedgehog信号通路活性增强后,可通过调节其下游信号分子而增强RANKL的表达,RANKL进一步促进破骨前体细胞向破骨细胞分化成熟。
甲状旁腺相关蛋白:甲状旁腺相关蛋白是一种旁分泌因子,是Hedgehog信号通路调节成骨细胞RANKL表达中的重要中间产物,Hedgehog信号增强后显示上调胞内甲状旁腺相关蛋白,再由甲状旁腺相关蛋白激活下游因子环磷酸腺苷应答元件结合蛋白和活化T细胞核因子,并与RANKL基因结合,促进其表达。.jpg) 文题释义:
OPG-RANK-RANKL系统:在骨组织代谢平衡中,成骨细胞与破骨细胞的平衡主要通过OPG-RANK-RANKL系统调控轴进行;RANKL即核因子κB受体活化因子配体,是一种促破骨细胞分化因子,可与破骨前体细胞上的RANK结合,刺激破骨前体细胞向破骨细胞分化、成熟;OPG即骨保护素,主要表达于成骨前体细胞,是RANKL的竞争性配体。近几年的研究发现,成骨细胞中Hedgehog信号通路活性增强后,可通过调节其下游信号分子而增强RANKL的表达,RANKL进一步促进破骨前体细胞向破骨细胞分化成熟。
甲状旁腺相关蛋白:甲状旁腺相关蛋白是一种旁分泌因子,是Hedgehog信号通路调节成骨细胞RANKL表达中的重要中间产物,Hedgehog信号增强后显示上调胞内甲状旁腺相关蛋白,再由甲状旁腺相关蛋白激活下游因子环磷酸腺苷应答元件结合蛋白和活化T细胞核因子,并与RANKL基因结合,促进其表达。
文题释义:
OPG-RANK-RANKL系统:在骨组织代谢平衡中,成骨细胞与破骨细胞的平衡主要通过OPG-RANK-RANKL系统调控轴进行;RANKL即核因子κB受体活化因子配体,是一种促破骨细胞分化因子,可与破骨前体细胞上的RANK结合,刺激破骨前体细胞向破骨细胞分化、成熟;OPG即骨保护素,主要表达于成骨前体细胞,是RANKL的竞争性配体。近几年的研究发现,成骨细胞中Hedgehog信号通路活性增强后,可通过调节其下游信号分子而增强RANKL的表达,RANKL进一步促进破骨前体细胞向破骨细胞分化成熟。
甲状旁腺相关蛋白:甲状旁腺相关蛋白是一种旁分泌因子,是Hedgehog信号通路调节成骨细胞RANKL表达中的重要中间产物,Hedgehog信号增强后显示上调胞内甲状旁腺相关蛋白,再由甲状旁腺相关蛋白激活下游因子环磷酸腺苷应答元件结合蛋白和活化T细胞核因子,并与RANKL基因结合,促进其表达。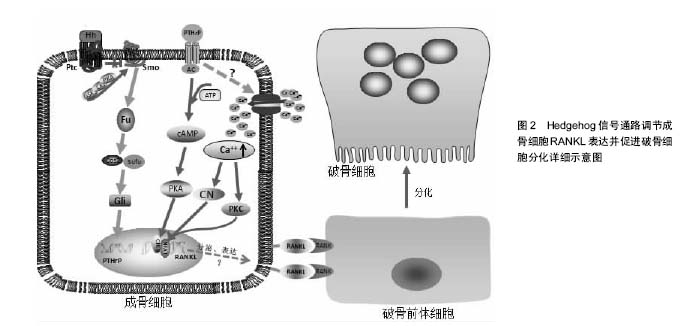
.jpg)
.jpg) 文题释义:
OPG-RANK-RANKL系统:在骨组织代谢平衡中,成骨细胞与破骨细胞的平衡主要通过OPG-RANK-RANKL系统调控轴进行;RANKL即核因子κB受体活化因子配体,是一种促破骨细胞分化因子,可与破骨前体细胞上的RANK结合,刺激破骨前体细胞向破骨细胞分化、成熟;OPG即骨保护素,主要表达于成骨前体细胞,是RANKL的竞争性配体。近几年的研究发现,成骨细胞中Hedgehog信号通路活性增强后,可通过调节其下游信号分子而增强RANKL的表达,RANKL进一步促进破骨前体细胞向破骨细胞分化成熟。
甲状旁腺相关蛋白:甲状旁腺相关蛋白是一种旁分泌因子,是Hedgehog信号通路调节成骨细胞RANKL表达中的重要中间产物,Hedgehog信号增强后显示上调胞内甲状旁腺相关蛋白,再由甲状旁腺相关蛋白激活下游因子环磷酸腺苷应答元件结合蛋白和活化T细胞核因子,并与RANKL基因结合,促进其表达。
文题释义:
OPG-RANK-RANKL系统:在骨组织代谢平衡中,成骨细胞与破骨细胞的平衡主要通过OPG-RANK-RANKL系统调控轴进行;RANKL即核因子κB受体活化因子配体,是一种促破骨细胞分化因子,可与破骨前体细胞上的RANK结合,刺激破骨前体细胞向破骨细胞分化、成熟;OPG即骨保护素,主要表达于成骨前体细胞,是RANKL的竞争性配体。近几年的研究发现,成骨细胞中Hedgehog信号通路活性增强后,可通过调节其下游信号分子而增强RANKL的表达,RANKL进一步促进破骨前体细胞向破骨细胞分化成熟。
甲状旁腺相关蛋白:甲状旁腺相关蛋白是一种旁分泌因子,是Hedgehog信号通路调节成骨细胞RANKL表达中的重要中间产物,Hedgehog信号增强后显示上调胞内甲状旁腺相关蛋白,再由甲状旁腺相关蛋白激活下游因子环磷酸腺苷应答元件结合蛋白和活化T细胞核因子,并与RANKL基因结合,促进其表达。