中国组织工程研究 ›› 2014, Vol. 18 ›› Issue (2): 251-258.doi: 10.3969/j.issn.2095-4344.2014.02.015
• 组织构建与生物活性因子 tissue construction and bioactive factors • 上一篇 下一篇
角膜基质细胞基质金属蛋白酶1,2活性与组织因子途径抑制物2的效应
俞建雄1,袁 静2,周炼红2
- 武汉大学人民医院,1外科,2眼科,湖北省武汉市 430060
-
收稿日期:2013-10-23出版日期:2014-01-08发布日期:2014-01-08 -
通讯作者:袁静,博士,副主任医师,武汉大学人民医院眼科,湖北省武汉市430060 -
作者简介:俞建雄,男,汉族,1974年生,江西省婺源县人,2004年华中科技大学附属同济医学院毕业,博士,主治医师,主要从事基因工程与外科治疗研究。 -
基金资助:国家自然科学基金资助项目(81100664);武汉大学自主科研基金资助项目(111091)
Effect of tissue factor pathway inhibitor-2 on the expressions of matrix metalloproteinase 1 and 2 in keratocytes
Yu Jian-xiong1, Yuan Jing2, Zhou Lian-hong2
- 1 Department of Gastrointestinal Surgery, Renmin Hospital of Wuhan University, Wuhan 430060, Hubei Province, China
2 Eye Center, Renmin Hospital of Wuhan University, Wuhan 430060, Hubei Province, China
-
Received:2013-10-23Online:2014-01-08Published:2014-01-08 -
Contact:Yuan Jing, M.D., Associate chief physician, Eye Center, Renmin Hospital of Wuhan University, Wuhan 430060, Hubei Province, China -
About author:Yu Jian-xiong, M.D., Attending physician, Department of Gastrointestinal Surgery, Renmin Hospital of Wuhan University, Wuhan 430060, Hubei Province, China -
Supported by:the National Natural Science Foundation of China, No. 81100664; Scientific Research Project of Wuhan University, No. 111091
摘要:
背景:有研究表明,基质金属蛋白酶所参与的细胞外基质降解在角膜新生血管形成过程中起关键作用,组织因子途径抑制物2是新近发现的一种新型的丝氨酸蛋白酶抑制物,能有效抑制基质金属蛋白酶的活性。 目的:观察组织因子途径抑制物2对体外角膜基质细胞表达基质金属蛋白酶活性的关系。 方法:在体外对兔角膜基质细胞进行原代及传代培养,用脂质体介导的人类组织因子途径抑制物2真核表达载体转染兔角膜基质细胞,G418筛选阳性细胞。 结果与结论:RT-PCR,Western blot及明胶酶谱法分析结果显示,转染后角膜基质细胞组织因子途径抑制物2 mRNA和蛋白质的表达均上调(P < 0.05),而基质金属蛋白酶1,2的活性下降(P < 0.05)。结果提示,组织因子途径抑制物2可明显抑制角膜基质细胞中基质金属蛋白酶1,2的活性。
中图分类号:
引用本文
俞建雄,袁 静,周炼红. 角膜基质细胞基质金属蛋白酶1,2活性与组织因子途径抑制物2的效应[J]. 中国组织工程研究, 2014, 18(2): 251-258.
Yu Jian-xiong, Yuan Jing, Zhou Lian-hong. Effect of tissue factor pathway inhibitor-2 on the expressions of matrix metalloproteinase 1 and 2 in keratocytes[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(2): 251-258.
Restriction enzymes were analyzed
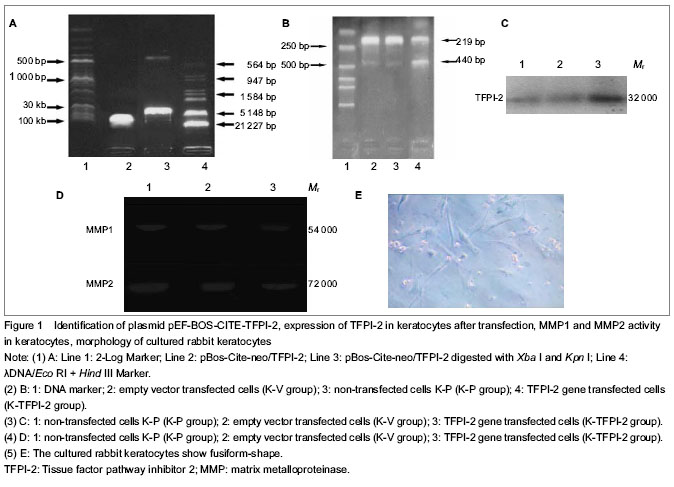
TFPI-2 protein expression in keratocytes as detected by western blot analysis

| [1] Zhou Q, Yang L, Qu M, et al. Role of senescent fibroblasts on alkali-induced corneal neovascularization. J Cell Physiol. 2012;227(3):1148-1156.[2] Shi W, Liu J, Li M, et al. Expression of MMP, HPSE, and FAP in stroma promoted corneal neovascularization induced by different etiological factors. Curr Eye Res. 2010;35(11):967-977.[3] Ebrahem Q, Chaurasia SS, Vasanji A, et al. Cross-talk between vascular endothelial growth factor and matrix metalloproteinases in the induction of neovascularization in vivo. Am J Pathol. 2010;176(1):496-503. [4] Chang JH, Han KY, Azar DT. Wound healing fibroblasts modulate corneal angiogenic privilege: interplay of basic fibroblast growth factor and matrix metalloproteinases in corneal angiogenesis. Jpn J Ophthalmol. 2010;54(3): 199-205. [5] Yu JX, Yuan J. Inhibition of corneal neovascularization by tissue factor pathway inhibitor 2 gene transfection in situ. Zhongguo Zuzhi Gongcheng Yanjiu yu Linchuang Kangfu. 2007;11(37):7498-7502.[6] Lee S, Zheng M, Kim B, et al. Role of matrix metalloproteinase-9 in angiogenesis caused by ocular infection with herpes simplex virus. J Clin Invest. 2002; 110(8):1105-1111.[7] Praidou A, Androudi S, Brazitikos P, et al. Angiogenic growth factors and their inhibitors in diabetic retinopathy. Curr Diabetes Rev. 2010;6(5):304-312.[8] Stellas D, Patsavoudi E. Inhibiting matrix metalloproteinases, an old story with new potentials for cancer treatment. Anticancer Agents Med Chem. 2012;12(7):707-717.[9] Bogaczewicz J, Dudek W, Zubilewicz T, et al. The role of matrix metalloproteinases and their tissue inhibitors in angiogenesis. Pol Merkur Lekarski. 2006;21(121):80-85. [10] Rundhaug JE. Matrix metalloproteinases and angiogenesis. J Cell Mol Med. 2005;9(2):267-285.[11] Kong DS, Guo HS, Cai X, et al. Cloning, expression and biological characterization of hTFPI-2 gene. Zhonghua Xue Ye Xue Za Zhi. 2006;27(9):606-610. [12] Chand HS, Foster DC, Kisiel W. Structure, function and biology of tissue factor pathway inhibitor-2. Thromb Haemost. 2005;94(6):1122-1130.[13] Kong D, Ma D, Bai H, et al. Expression and characterization of the first kunitz domain of human tissue factor pathway inhibitor-2. Biochem Biophys Res Commun. 2004;324(4):1179-1185.[14] Sierko E, Zawadzki RJ, Wojtukiewicz MZ. Tissue factor pathway inhibitors. Pol Merkur Lekarski. 2002;13(73):66-69. [15] Kamei S, Kazama Y, Kuijper JL, et al. Genomic structure and promoter activity of the human tissue factor pathway inhibitor-2 gene. Biochim Biophys Acta. 2001;1517(3):430-435.[16] Chand HS, Schmidt AE, Bajaj SP, et al. Structure-function analysis of the reactive site in the first Kunitz-type domain of human tissue factor pathway inhibitor-2. J Biol Chem. 2004;279(17):17500-17507. [17] Rao CN, Reddy P, Reeder DJ, et al. Prokaryotic expression, purification, and reconstitution of biological activities (Antiprotease, antitumor, and heparin-binding) for tissue factor pathway inhibitor-2. Biochem Biophys Res Commun. 2000;276(3):1286-1294.[18] The Ministry of Science and Technology of the People’s Republic of China. Guidance Suggestions for the Care and Use of Laboratory Animals. 2006-09-30.[19] Dropcova S, Denyer SP, Lloyd AW, et al. A standard strain of human ocular keratocytes. Ophthalmic Res. 1999;31(1):33-41. [20] Neaud V, Hisaka T, Monvoisin A, et al. Paradoxical pro-invasive effect of the serine proteinase inhibitor tissue factor pathway inhibitor-2 on human hepatocellular carcinoma cells. J Biol Chem. 2000;275(45):35565-35569.[21] Jin M, Udagawa K, Miyagi E, et al. Expression of serine proteinase inhibitor PP5/TFPI-2/MSPI decreases the invasive potential of human choriocarcinoma cells in vitro and in vivo. Gynecol Oncol. 2001;83(2):325-333. [22] Chang JH, Gabison EE, Kato T, et al. Corneal neovascularization. Curr Opin Ophthalmol. 2001;12(4):242-249. [23] Perera C, Jhanji V, Vajpayee RB. Factors influencing outcomes of the treatment of allograft corneal rejection. Am J Ophthalmol. 2011;152(3):358-363. [24] Niederkorn JY. High-risk corneal allografts and why they lose their immune privilege. Curr Opin Allergy Clin Immunol. 2010;10(5):493-497. [25] Garcia DD, Shtein RM, Musch DC, et al. Herpes simplex virus keratitis: histopathologic neovascularization and corneal allograft failure. Cornea. 2009;28(9):963-965. [26] Shtein RM, Garcia DD, Musch DC, et al. HSV keratitis: histopathologic predictors of corneal allograft complications. Trans Am Ophthalmol Soc. 2008;106:161-168; discussion 168-170.[27] Levine HA, Pamuk S, Sleeman BD, et al. Mathematical modeling of capillary formation and development in tumor angiogenesis: penetration into the stroma. Bull Math Biol. 2001;63(5):801-863.[28] Bauer AL, Jackson TL, Jiang Y. Topography of extracellular matrix mediates vascular morphogenesis and migration speeds in angiogenesis. PLoS Comput Biol. 2009;5(7):e1000445. [29] Pytliak M, Vargová V, Mechírová V. Matrix metalloproteinases and their role in oncogenesis: a review. Onkologie. 2012;35(1-2):49-53. [30] Benjamin MM, Khalil RA. Matrix metalloproteinase inhibitors as investigative tools in the pathogenesis and management of vascular disease. EXS. 2012;103:209-279. [31] Vargová V, Pytliak M, Mechírová V. Matrix metalloproteinases. EXS. 2012;103:1-33. [32] Martins VL, Caley M, O'Toole EA. Matrix metalloproteinases and epidermal wound repair. Cell Tissue Res. 2013;351(2):255-268.[33] Gomis-Rüth FX, Maskos K, Betz M, et al. Mechanism of inhibition of the human matrix metalloproteinase stromelysin-1 by TIMP-1. Nature. 1997;389(6646):77-81.[34] Olson TM, Hirohata S, Ye J, et al. Cloning of the human tissue inhibitor of metalloproteinase-4 gene (TIMP4) and localization of the TIMP4 and Timp4 genes to human chromosome 3p25 and mouse chromosome 6, respectively. Genomics. 1998;51(1):148-151.[35] Brew K, Dinakarpandian D, Nagase H. Tissue inhibitors of metalloproteinases: evolution, structure and function. Biochim Biophys Acta. 2000;1477(1-2):267-283.[36] Woessner JF Jr Matrix metalloproteinase inhibition. From the Jurassic to the third millennium. Ann NY Acad Sci. 1999;878:388-403.[37] Takahashi C, Sheng Z, Horan TP, et al. Regulation of matrix metalloproteinase-9 and inhibition of tumor invasion by the membrane-anchored glycoprotein RECK. Proc Natl Acad Sci U S A. 1998;95(22):13221-13226. |
| [1] | 张同同, 王中华, 文 杰, 宋玉鑫, 刘 林. 3D打印模型在颈椎肿瘤手术切除与重建中的应用[J]. 中国组织工程研究, 2021, 25(9): 1335-1339. |
| [2] | 曾燕华, 郝延磊. 许旺细胞体外培养及纯化的系统性综述[J]. 中国组织工程研究, 2021, 25(7): 1135-1141. |
| [3] | 徐东紫, 张 婷, 欧阳昭连. 心脏组织工程领域全球专利竞争态势分析[J]. 中国组织工程研究, 2021, 25(5): 807-812. |
| [4] | 马泽涛, 曾 晖, 王德利, 翁 鉴, 冯 松. 微小RNA-138-5p与软骨细胞增殖和自噬的关系[J]. 中国组织工程研究, 2021, 25(5): 674-678. |
| [5] | 吴子健, 胡昭端, 谢有琼, 王 峰, 李 佳, 李柏村, 蔡国伟, 彭 锐. 3D打印技术与骨组织工程研究文献计量及研究热点可视化分析[J]. 中国组织工程研究, 2021, 25(4): 564-569. |
| [6] | 常文辽, 赵 杰, 孙晓亮, 王 锟, 吴国锋, 周 剑, 李树祥, 孙 晗. 人工骨膜的材料选择、理论设计及生物仿生功能[J]. 中国组织工程研究, 2021, 25(4): 600-606. |
| [7] | 刘 旒, 周箐竹, 龚 桌, 刘博言, 杨 斌, 赵 娴. 胶原/无机材料构建组织工程骨的特点及制造技术[J]. 中国组织工程研究, 2021, 25(4): 607-613. |
| [8] | 刘 飞, 崔宇韬, 刘 贺. 局部抗生素递送系统治疗骨髓炎的优势与问题[J]. 中国组织工程研究, 2021, 25(4): 614-620. |
| [9] | 李晓壮, 段 浩, 王伟舟, 唐志宏, 王旸昊, 何 飞. 骨组织工程材料治疗骨缺损疾病在体内实验中的应用[J]. 中国组织工程研究, 2021, 25(4): 626-631. |
| [10] | 张振坤, 李 喆, 李 亚, 王莹莹, 王亚苹, 周馨魁, 马珊珊, 关方霞. 海藻酸盐基水凝胶/敷料在创面愈合中的应用:持续、动态与顺序释放[J]. 中国组织工程研究, 2021, 25(4): 638-643. |
| [11] | 陈佳娜, 邱燕玲, 聂敏海, 刘旭倩. 组织工程支架材料修复口腔颌面部软组织缺损[J]. 中国组织工程研究, 2021, 25(4): 644-650. |
| [12] | 邢 浩, 张永红, 王 栋. 长骨大段骨缺损修复方法的优势与不足[J]. 中国组织工程研究, 2021, 25(3): 426-430. |
| [13] | 陈思奇, 先德彬, 徐荣胜, 覃中杰, 张 磊, 夏德林. 羟基磷灰石-磷酸三钙支架复合骨髓间充质干细胞和人脐静脉内皮细胞对大鼠颅骨缺损修复早期成血管的影响[J]. 中国组织工程研究, 2021, 25(22): 3458-3465. |
| [14] | 刘 鋆, 杨 龙, 王伟宇, 周玉虎, 吴 颖, 卢 涛, 舒莉萍, 马敏先, 叶 川. 聚3-羟基丁酸酯4-羟基丁酸酯/聚乙二醇/氧化石墨烯组织工程支架的制备和性能评价[J]. 中国组织工程研究, 2021, 25(22): 3466-3472. |
| [15] | 王 皓, 陈明学, 李俊康, 罗旭江, 彭礼庆, 李 获, 黄 波, 田广招, 刘舒云, 眭 翔, 黄靖香, 郭全义, 鲁晓波. 脱细胞猪皮基质构建组织工程半月板支架[J]. 中国组织工程研究, 2021, 25(22): 3473-3478. |
|
||||||||||||||||||||||||
|
Results are shown as mean ± SEM. Statistical analyses were performed using one-way analysis of variance test with SPSS 13.0 statistical software. A value of P < 0.05 was considered statistically significant.
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||