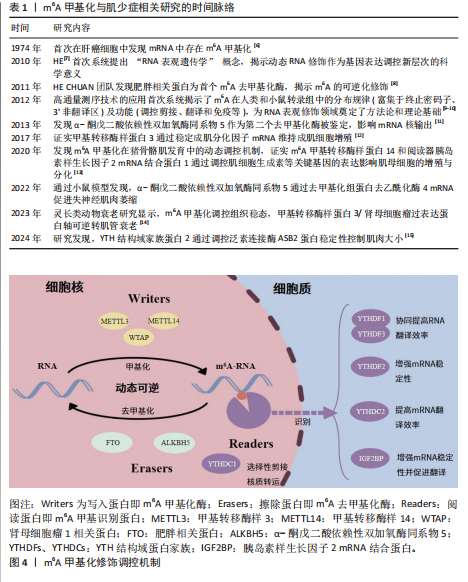
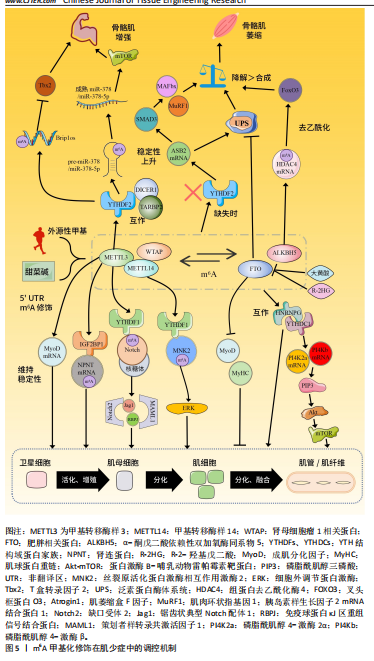
[1] CHEN LK, WOO J, ASSANTACHAI P, et al. Asian working group for sarcopenia: 2019 consensus update on sarcopenia diagnosis and treatment. J Am Med Dir Assoc. 2020;21(3):300-307.e2.
[2] YUAN S, LARSSON SC. Epidemiology of sarcopenia: Prevalence, risk factors, and consequences. Metabolism. 2023;144:155533.
[3] PETERMANN-ROCHA F, BALNTZI V, GRAY SR, et al. Global prevalence of sarcopenia and severe sarcopenia: A systematic review and meta‐analysis. J Cachexia Sarcopenia Muscle. 2022; 13(1):86-99.
[4] LIANG W, XU F, LI L, et al. Epigenetic control of skeletal muscle atrophy. Cell Mol Biol Lett. 2024; 29(1):99.
[5] ZACCARA S, RIES RJ, JAFFREY SR. Reading, writing and erasing mRNA methylation. Nat Rev Mol Cell Biol. 2019;20(10):608-624.
[6] DESROSIERS R, FRIDERICI K, ROTTMAN F. Identification of methylated nucleosides in messenger RNA from novikoff hepatoma cells. Proc Natl Acad Sci U S A. 1974;71(10):3971-3975.
[7] HE C. Grand challenge commentary: RNA epigenetics? Nat Chem Biol. 2010;6(12):863-865.
[8] JIA G, FU Y, ZHAO X, et al. N6-methyladenosine in nuclear RNA is a major substrate of the obesity-associated FTO. Nat Chem Biol. 2011;7(12):885-887.
[9] DOMINISSINI D, MOSHITCH-MOSHKOVITZ S, SCHWARTZ S, et al. Topology of the human and mouse m6A RNA methylomes revealed by m6A-seq. Nature. 2012;485(7397):201-206.
[10] MEYER KD, SALETORE Y, ZUMBO P, et al. Comprehensive analysis of mRNA methylation reveals enrichment in 3′ UTRs and near stop codons. Cell. 2012;149(7):1635-1646.
[11] ZHENG G, DAHL J A, NIU Y, et al. ALKBH5 is a mammalian RNA demethylase that impacts RNA metabolism and mouse fertility. Mol Cell. 2013;49(1):18-29.
[12] KUDOU K, KOMATSU T, NOGAMI J, et al. The requirement of Mettl3-promoted MyoD mRNA maintenance in proliferative myoblasts for skeletal muscle differentiation. Open Biol. 2017;7(9): 170119.
[13] ZHANG X, YAO Y, HAN J, et al. Longitudinal epitranscriptome profiling reveals the crucial role of N6-methyladenosine methylation in porcine prenatal skeletal muscle development. J Genet Genomics. 2020;47(8):466-476.
[14] WU Z, LU M, LIU D, et al. m6A epitranscriptomic regulation of tissue homeostasis during primate aging. Nat Aging. 2023;3(6):705-721.
[15] GILBERT CJ, RABOLLI CP, GOLUBEVA VA, et al. YTHDF2 governs muscle size through a targeted modulation of proteostasis. Nat Commun. 2024; 15(1):2176.
[16] JIANG X, LIU B, NIE Z, et al. The role of m6A modification in the biological functions and diseases. Signal Transduct Target Ther. 2021; 6(1):74.
[17] ZHAO Y, SHI Y, SHEN H, et al. m6A-binding proteins: The emerging crucial performers in epigenetics. J Hematol Oncol. 2020;13(1):35.
[18] WENG H, HUANG F, YU Z, et al. The m6A reader IGF2BP2 regulates glutamine metabolism and represents a therapeutic target in acute myeloid leukemia. Cancer Cell. 2022;40(12):1566-1582.e10.
[19] YIN J, DING F, CHENG Z, et al. METTL3-mediated m6A modification of LINC00839 maintains glioma stem cells and radiation resistance by activating wnt/β-catenin signaling. Cell Death Dis. 2023;14(7):417.
[20] CHEN A, ZHANG VX, ZHANG Q, et al. Targeting the oncogenic m6A demethylase FTO suppresses tumourigenesis and potentiates immune response in hepatocellular carcinoma. Gut. 2025; 74(1):90-102.
[21] LI X, YANG Y, LI Z, et al. Deficiency of WTAP in islet beta cells results in beta cell failure and diabetes in mice. Diabetologia. 2023;66(6):1084-1096.
[22] YIN H, JU Z, ZHENG M, et al. Loss of the m6A methyltransferase METTL3 in monocyte-derived macrophages ameliorates alzheimer’s disease pathology in mice. PLoS Biol. 2023;21(3):e3002017.
[23] QIN Y, LI L, LUO E, et al. Role of m6A RNA methylation in cardiovascular disease (review). Int J Mol Med. 2020;46(6):1958-1972.
[24] YANG Y, HSU PJ, CHEN YS, et al. Dynamic transcriptomic m6A decoration: Writers, erasers, readers and functions in RNA metabolism. Cell Res. 2018;28(6):616-624.
[25] PING XL, SUN BF, WANG L, et al. Mammalian WTAP is a regulatory subunit of the RNA N6-methyladenosine methyltransferase. Cell Res. 2014;24(2):177-189.
[26] FU Y, JIA G, PANG X, et al. FTO-mediated formation of N6-hydroxymethyladenosine and N6-formyladenosine in mammalian RNA. Nat Commun. 2013;4(1):1798.
[27] REICHEL M, KÖSTER T, STAIGER D. Marking RNA: m6A writers, readers, and functions in arabidopsis. J Mol Cell Biol. 2019;11(10):899-910.
[28] SHI H, WANG X, LU Z, et al. YTHDF3 facilitates translation and decay of N6-methyladenosine-modified RNA. Cell Res. 2017;27(3):315-328.
[29] WANG X, LU Z, GOMEZ A, et al. N6-methyladenosine-dependent regulation of messenger RNA stability. Nature. 2014;505(7481): 117-120.
[30] XIAO W, ADHIKARI S, DAHAL U, et al. Nuclear m 6 a reader YTHDC1 regulates mRNA splicing. Mol Cell. 2016;61(4):507-519.
[31] ROUNDTREE IA, LUO GZ, ZHANG Z, et al. YTHDC1 mediates nuclear export of N6-methyladenosine methylated mRNAs. eLife. 2017;6:e31311.
[32] HSU PJ, ZHU Y, MA H, et al. Ythdc2 is an N6-methyladenosine binding protein that regulates mammalian spermatogenesis. Cell Res. 2017; 27(9):1115-1127.
[33] HUANG H, WENG H, SUN W, et al. Recognition of RNA N6-methyladenosine by IGF2BP proteins enhances mRNA stability and translation. Nat Cell Biol. 2018;20(3):285-295.
[34] ALARCÓN CR, GOODARZI H, LEE H, et al. HNRNPA2B1 is a mediator of m6A-dependent nuclear RNA processing events. Cell. 2015;162(6): 1299-1308.
[35] CHEN JJ, LU TZ, WANG T, et al. The m6A reader HNRNPC promotes glioma progression by enhancing the stability of IRAK1 mRNA through the MAPK pathway. Cell Death Dis. 2024;15(6):390.
[36] WANG N, SHI B, DING L, et al. FMRP protects breast cancer cells from ferroptosis by promoting SLC7A11 alternative splicing through interacting with hnRNPM. Redox Biol. 2024;77:103382.
[37] LAI Y, RAMÍREZ-PARDO I, ISERN J, et al. Multimodal cell atlas of the ageing human skeletal muscle. Nature. 2024;629(8010):154-164.
[38] NEDERVEEN JP, FORTINO SA, BAKER JM, et al. Consistent expression pattern of myogenic regulatory factors in whole muscle and isolated human muscle satellite cells after eccentric contractions in humans. J Appl Physiol. 2019; 127(5):1419-1426.
[39] GHELLER BJ, BLUM JE, FONG EHH, et al. A defined N6-methyladenosine (m6A) profile conferred by METTL3 regulates muscle stem cell/myoblast state transitions. Cell Death Discov. 2020;6(1):95.
[40] LIANG Y, HAN H, XIONG Q, et al. METTL3-mediated m6A methylation regulates muscle stem cells and muscle regeneration by notch signaling pathway. Stem Cells Int. 2021;2021:1-13.
[41] XIE SJ, LEI H, YANG B, et al. Dynamic m6A mRNA methylation reveals the role of METTL3/14-m6A-MNK2-ERK signaling axis in skeletal muscle differentiation and regeneration. Front Cell Dev Biol. 2021;9:744171.
[42] LI R, CAO Y, WU W, et al. Inhibitor of FTO, rhein, restrains the differentiation of myoblasts and delays skeletal muscle regeneration. Animals. 2024;14(16):2434.
[43] YANG X, MEI C, MA X, et al. m6A methylases regulate myoblast proliferation, apoptosis and differentiation. Animals. 2022;12(6):773.
[44] QIAO Y, SUN Q, CHEN X, et al. Nuclear m6A reader YTHDC1 promotes muscle stem cell activation/proliferation by regulating mRNA splicing and nuclear export. eLife. 2023;12: e82703.
[45] LIU J, ZUO H, WANG Z, et al. The m6A reader YTHDC1 regulates muscle stem cell proliferation via PI4K–Akt–mTOR signalling. Cell Prolif. 2023; 56(8):e13410.
[46] CHEN Z, LI L, WU W, et al. Exercise protects proliferative muscle satellite cells against exhaustion via the Igfbp7-akt-mTOR axis. Theranostics. 2020;10(14):6448-6466.
[47] HWANG J, KANG S, JUNG H. Effects of American wild ginseng and korean cultivated wild ginseng pharmacopuncture extracts on the regulation of C2C12 myoblasts differentiation through AMPK and PI3K/akt/mTOR signaling pathway. Mol Med Rep. 2022;25(6):192.
[48] LI Y, LIU Z, YAN H, et al. Polygonatum sibiricum polysaccharide ameliorates skeletal muscle aging and mitochondrial dysfunction via PI3K/akt/mTOR signaling pathway. Phytomedicine. 2025;136:156316.
[49] YEH TS, LEI TH, LIU JF, et al. Astragalus membranaceus enhances myotube hypertrophy through PI3K-mediated akt/mTOR signaling phosphorylation. Nutrients. 2022;14(8):1670.
[50] ZHAO T, ZHAO R, YI X, et al. METTL3 promotes proliferation and myogenic differentiation through m6A RNA methylation/YTHDF1/2 signaling axis in myoblasts. Life Sci. 2022;298:120496.
[51] HUANG X, XU C, ZHANG J, et al. Endurance exercise remodels skeletal muscle by suppressing Ythdf1-mediated myostatin expression. Cell Death Dis. 2025;16(1):96.
[52] PETROSINO JM, HINGER SA, GOLUBEVA VA, et al. The m6A methyltransferase METTL3 regulates muscle maintenance and growth in mice. Nat Commun. 2022;13(1):168.
[53] FENG S, ZHOU H, LIN X, et al. Exercise promotes skeletal muscle growth in adolescents via modulating Mettl3-mediated m6A methylation of MyoD in muscle satellite cells. Cell Mol Biol Lett. 2024;29(1):150.
[54] FERNANDO R, DRESCHER C, NOWOTNY K, et al. Impaired proteostasis during skeletal muscle aging. Free Radic Biol Med. 2019;132:58-66.
[55] ROM O, REZNICK AZ. The role of E3 ubiquitin-ligases MuRF-1 and MAFbx in loss of skeletal muscle mass. Free Radic Biol Med. 2016;98:218-230.
[56] HABERECHT-MÜLLER S, KRÜGER E, FIELITZ J. Out of control: The role of the ubiquitin proteasome system in skeletal muscle during inflammation. Biomolecules. 2021;11(9):1327.
[57] LAN XQ, DENG CJ, WANG QQ, et al. The role of TGF-β signaling in muscle atrophy, sarcopenia and cancer cachexia. Gen Comp Endocrinol. 2024; 353:114513.
[58] SUN J, ZHOU H, CHEN Z, et al. Altered m6A RNA methylation governs denervation-induced muscle atrophy by regulating ubiquitin proteasome pathway. J Transl Med. 2023;21(1):845.
[59] YOSHIDA T, DELAFONTAINE P. Mechanisms of IGF-1-mediated regulation of skeletal muscle hypertrophy and atrophy. Cells. 2020;9(9):1970.
[60] LIU Y, ZHOU T, WANG Q, et al. m6 a demethylase ALKBH5 drives denervation‐induced muscle atrophy by targeting HDAC4 to activate FoxO3 signalling. J Cachexia Sarcopenia Muscle. 2022; 13(2):1210-1223.
[61] DIAO LT, XIE SJ, LEI H, et al. METTL3 regulates skeletal muscle specific miRNAs at both transcriptional and post-transcriptional levels. Biochem Biophys Res Commun. 2021;552:52-58.
[62] DENG K, SU Y, LIU Z, et al. Ythdf2 facilitates precursor miR-378/miR-378-5p maturation to support myogenic differentiation. Cell Mol Life Sci. 2024;81(1):445.
[63] XIE SJ, TAO S, DIAO LT, et al. Characterization of long non-coding RNAs modified by m6A RNA methylation in skeletal myogenesis. Front Cell Dev Biol. 2021;9:762669.
[64] LIU J, LI K, CAI J, et al. Landscape and regulation of m6A and m6Am methylome across human and mouse tissues. Mol Cell. 2020;77(2):426-440.e6.
[65] YANG X, MEI C, RAZA SHA, et al. Interactive regulation of DNA demethylase gene TET1 and m6A methyltransferase gene METTL3 in myoblast differentiation. Int J Biol Macromol. 2022;223: 916-930.
[66] KHARRAZ Y, GUERRA J, MANN CJ, et al. Macrophage plasticity and the role of inflammation in skeletal muscle repair. Mediators Inflamm. 2013;2013:1-9.
[67] TENG Y, YI J, CHEN J, et al. N6-methyladenosine (m6A) modification in natural immune cell-mediated inflammatory diseases. J Innate Immun. 2023;15(1):804-821.
|