中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (34): 9048-9055.doi: 10.12307/2026.824
• 组织构建综述 tissue construction review • 上一篇 下一篇
机械应力调控骨关节炎的分子机制与治疗靶点
张益瑞1,顾 叶1,钱正韬1,吴泽睿1,谢 恒1,唐奕涵1,顾赢楚1,方 涛1,王秋霏1,彭育沁1,耿德春2,徐耀增2
- 1苏州大学附属常熟医院常熟市第一人民医院骨科,江苏省常熟市 215500;2苏州大学附属第一医院骨科,江苏省苏州市 215006
-
收稿日期:2025-10-15修回日期:2025-12-31出版日期:2026-12-08发布日期:2026-04-15 -
通讯作者:顾叶,博士,主任医师,苏州大学附属常熟医院常熟市第一人民医院骨科,江苏省常熟市 215500 通讯作者:钱正韬,硕士,主管检验师,苏州大学附属常熟医院常熟市第一人民医院骨科,江苏省常熟市 215500 -
作者简介:张益瑞,男,2002年生,河南省安阳市人,汉族,苏州大学在读硕士,主要从事关节外科的基础与临床研究。 -
基金资助:常熟市科技局科技发展项目(CS202217),项目负责人:方涛;常熟市科技局科技发展项目(CY202325),项目负责人:顾赢楚;常熟市科技计划项目(CS202451),项目负责人:顾叶;常熟市卫健委科技发展计划项目(CSWS202305),项目负责人:
顾叶;苏州市姑苏卫生人才科研项目(gsws2024062),项目负责人:顾叶
Molecular mechanisms and therapeutic targets of mechanical stress regulating osteoarthritis
Zhang Yirui1, Gu Ye1, Qian Zhengtao1, Wu Zerui1, Xie Heng1, Tang Yihan1, Gu Yingchu1, Fang Tao1, Wang Qiufei1, Peng Yuqin1, Geng Dechun2, Xu Yaozeng2
- 1Department of Orthopedics, Changshu First People’s Hospital, Changshu Affiliated Hospital of Soochow University, Changshu 215500, Jiangsu Province, China; 2Department of Orthopedics, First Affiliated Hospital of Soochow University, Suzhou 215006, Jiangsu Province, China
-
Received:2025-10-15Revised:2025-12-31Online:2026-12-08Published:2026-04-15 -
Contact:Qian Zhengtao, MS, Chief laboratorian, Department of Orthopedics, Changshu First People’s Hospital, Changshu Affiliated Hospital of Soochow University, Changshu 215500, Jiangsu Province, China -
About author:Zhang Yirui, MS candidate, Department of Orthopedics, Changshu First People’s Hospital, Changshu Affiliated Hospital of Soochow University, Changshu 215500, Jiangsu Province, China Corresponding author: Gu Ye, MD, Chief physician, Department of Orthopedics, Changshu First People’s Hospital, Changshu Affiliated Hospital of Soochow University, Changshu 215500, Jiangsu Province, China -
Supported by:Science and Technology Development Project of Changshu Science and Technology Bureau, No. CS202217 (to FT); Science and Technology Development Project of Changshu Science and Technology Bureau, No. CY202325 (to GYC); Changshu Science and Technology Plan Project, No. CS202451 (to GY); Changshu Health Commission Science and Technology Development Plan, No. CSWS202305 (to GY); Suzhou Gusu Health Talent Research Project, No. gsws2024062 (to GY)
摘要:
文题释义:
压电型机械门控离子通道组件(Piezo):是一种以钙离子内流为主的机械门控阳离子通道蛋白,在骨关节炎的发生发展中起到关键作用。Piezo蛋白能够感知机械力(如压力、拉伸或剪切力)并将其转化为电信号或化学信号,作用于下游相应信号传导通路,最终引起疼痛并加速骨关节炎的发生发展。
骨关节炎:是一种以关节软骨进行性退变、关节边缘骨质增生和软骨下骨硬化或囊性变为主要病理特征的慢性退行性关节病,常与年龄和劳损密切相关,主要导致关节疼痛、僵硬和功能障碍。
背景:压电型机械门控离子通道组件(piezo-type mechanosensitive ion channel component,PIEZO)离子通道通过感知机械刺激调控钙信号,在骨关节炎的软骨退变、炎症及疼痛中发挥关键作用,或许可以成为治疗骨关节炎的重要潜在靶点。
目的:系统综述PIEZO离子通道在骨关节炎病理机制中的作用,评估它作为新型治疗靶点的可能性。
方法:由第一作者应用计算机检索中国知网与PubMed数据库,以“机械应力,Piezo,骨关节炎,软骨细胞,成骨细胞,破骨细胞,滑膜细胞,免疫细胞”等为中文检索词,以“Piezo1,osteoarthritis,chondrocyte,osteoblast,osteoclast,synovial membrane,immune cell,GsMTx4”等为英文检索词,选择2000-2025年发表的文献,按照入选标准,最终纳入102篇文献进行综述。
结果与结论:机械应力在关节软骨和周围组织的退行性变中起核心作用,长期的过度机械应力或不均衡负荷会引起软骨细胞的损伤、凋亡以及炎症反应,进而加速骨关节炎的进展。目前已知的机械应力感受器包括瞬时受体电位通道家族、双孔域钾通道家族、退化因子/上皮钠通道家族以及整合素家族等。PIEZO家族是在哺乳动物细胞中发现的第一组机械敏感阳离子通道孔蛋白,在人体细胞中广泛存在,通过感受周围环境压力变化控制Ca2+内流,进而影响细胞功能。PIEZO离子通道通过感知细胞膜的机械刺激调控Ca2+流入,进而影响软骨细胞、成骨功能、滑膜细胞、免疫细胞及疼痛感知等过程,通过抑制PIEZO离子通道或可成为治疗关节炎的有效方法。
https://orcid.org/0009-0000-6865-6661(张益瑞)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
张益瑞, 顾 叶, 钱正韬, 吴泽睿, 谢 恒, 唐奕涵, 顾赢楚, 方 涛, 王秋霏, 彭育沁, 耿德春, 徐耀增. 机械应力调控骨关节炎的分子机制与治疗靶点[J]. 中国组织工程研究, 2026, 30(34): 9048-9055.
Zhang Yirui, Gu Ye, Qian Zhengtao, Wu Zerui, Xie Heng, Tang Yihan, Gu Yingchu, Fang Tao, Wang Qiufei, Peng Yuqin, Geng Dechun, Xu Yaozeng. Molecular mechanisms and therapeutic targets of mechanical stress regulating osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 9048-9055.
2.2 机械应力与骨关节炎的关系
2.2.1 机械应力 生理条件下,身体关节承受数百万次循环的负荷,导致高达体质量10倍的力通过关节,正常情况下这种负荷对软骨或其他关节组织很少造成不利影响[23],并且这些机械应力作用于关节软骨、骨骼和滑膜等组织,通常会促进软骨细胞的修复和代谢[24];而由多种原因如肥胖、制动、关节不稳定、过度使用或创伤等引起的异常负荷常常与骨关节炎密切相关。由于关节不稳定或损伤而导致的关节负荷改变,是骨关节炎发生和进展的重要危险因素[25]。长期的过度机械应力或不均衡负荷会引起软骨细胞的损伤、凋亡以及炎症反应,进而加速骨关节炎的进展[26]。在骨关节炎中,关节的负荷能力下降,应力传导不均,软骨出现裂隙、破损等退行性变,这些变化与机械应力密切相关,机械应力的变化会触发关节处生物学反应,如细胞增殖、基质合成以及软骨降解等。
2.2.2 机械应力引发的炎症反应 机械应力所致的损伤是关节炎的诱因之一,贯穿于整个炎症的病理过程[27],不仅影响关节软骨的力学特性,还能激活细胞内的炎症反应[28-29]。在受到

机械应力时,软骨细胞和滑膜细胞能够通过多种途径如核因子κB、丝裂原活化蛋白激酶等激活炎症反应通路[30-31]。有研究表明,机械应力可以通过激活软骨细胞中的Hippo-蛋白激酶Cζ-核因子κB通路,导致与炎症相关的核因子κB靶基因表达[32];此外,机械应力还可以通过调节白细胞介素1β、肿瘤坏死因子α、白细胞介素6、白细胞介素8、神经生长因子等细胞因子的产生进一步加重关节的炎症反应,这在骨关节炎的发生和进展中起着重要作用[33]。白细胞介素1β是骨关节炎的关键炎性递质[34],可激活核因子κB和丝裂原活化蛋白激酶信号通路[35],诱导其他炎症因子如肿瘤坏死因子α、白细胞介素6和基质金属蛋白酶(如基质金属蛋白酶1和基质金属蛋白酶13)的表达,导致软骨基质降解和滑膜炎[36]。肿瘤坏死因子α水平与软骨损伤程度呈正相关,在促进炎症反应、诱导软骨细胞凋亡、滑膜炎和产生基质金属降解酶3、含血小板反应蛋白基序的解聚素样金属蛋白酶5的过程中发挥重要作用[37]。白细胞介素6是炎症反应早期的重要递质,可通过Janus激酶/信号转导及转录激活子通路增强炎症信号,间接促进基质金属蛋白酶和趋化因子的表达[38]。白细胞介素8通过与CXC基序趋化因子受体1和2结合诱导嗜中性粒细胞的趋化性,进而有助于骨关节炎的启动与维持[39]。同时,白细胞介素8是一种由成骨细胞产生的细胞因子,以自分泌方式促进受体活化核因子κB配体诱导的破骨细胞生成[40]。此外,女性关节炎患者滑膜液中的白细胞介素8水平通常升高,并且该细胞因子升高水平与骨关节炎严重程度相关[41]。神经生长因子在骨关节炎滑膜成纤维细胞中高度表达,导致关节疼痛[42]。在炎症状态下,通过激活细胞表面受体Toll 样受体激活核因子κB通路可诱导成纤维细胞分泌多种炎症因子,包括白细胞介素6、血管内皮生长因子、基质金属蛋白酶1和基质金属蛋白酶3,进一步加剧滑膜炎症和软骨破坏[43]。不同细胞因子在骨关节炎发生发展过程中扮演的角色,见表2。
2.3 PIEZO离子通道概述
PIEZO离子通道的发现与分类:PIEZO离子通道是一种膜跨膜的结构,含有多个跨膜螺旋[44]。PIEZO 家族是在哺乳动物细胞中发现的第一组机械敏感阳离子通道孔蛋白[14]。PIEZO1离子通道可以在机械应力下非选择性诱导Ca2+、 Mg2+、K+、Na+等内流,但是更偏向Ca2+[45]。PIEZO1离子通道在感应到作用在细胞膜上的机械应力的变化后,开放并调节Ca2+、Na+等的流入,机械脉冲转化为电或化学信号,进而激活细胞中的信号转导通路[44]。PIEZO蛋白家族目前发现PIEZO1和PIEZO2两种,哺乳动物的PIEZO蛋白由2 500-2 800个氨基酸组成[46]。最近的研究发现,PIEZO1主要表达在非兴奋性细胞类型中[47],并且PIEZO1对于血管机械转导[48]、尿渗透压[49]、软骨机械转导[50]、外周三叉神经的伤害性感受等病理生理状况非常重要[51]。PIEZO2主要表达在感觉神经元中,如躯体感觉神经节、外毛细胞、肠道内肠嗜铬细胞等,并在气道伸展和肺膨胀中发挥作用[47,51-52]。与其他机械激活通道相比,PIEZO1对侧向膜张力具有极其敏感的反应,而静息张力可以驱动通道失活,从而调节PIEZO1的整

体机械敏感性[53]。PIEZO2蛋白的机械激活阈值对刺激速度高度敏感,随速度增加而显著降低,这提示了PIEZO2离子通道蛋白面对快速刺激更易被激活[54]。在骨关节炎中,PIEZO离子通道通过调节不同细胞内的钙离子浓度进一步影响关节组织的生理功能,见表3。Yoda1是首个发现的PIEZO1特异性小分子激动剂,可在无机械刺激情况下激活PIEZO1离子通道,被广泛用于研究PIEZO1机械转导机制[55]。
Dooku1(PIEZO1离子通道抑制剂)通过竞争性结合阻断Yoda1的效应,但对PIEZO1的基线机械激活(如膜张力或压力诱导的电流)无显著抑制作用,也不对PIEZO2产生影响[56]。PIEZO2通道蛋白暂时缺乏特异性激活剂或抑制剂的研究成果。此外,LEE等[57]的研究报道,PIEZO1的表达主要由p38丝裂原活化蛋白激酶以及cAMP反应元件结合蛋白1、肝细胞核因子4和激活转录因子2等直接与 PIEZO1启动子结合的转录因子决定。
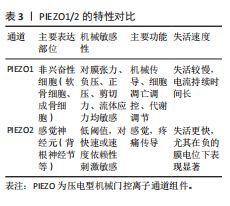
2.4 PIEZO离子通道在关节炎中的作用
2.4.1 PIEZO离子通道在软骨细胞中的作用 见图4。
PIEZO在软骨细胞中表达显著,并在软骨细胞的机械转导中起着至关重要的作用[58]。研究表明,软骨细胞膜上的机械门控离子通道在256 nm的平均凹陷下可诱导Ca2+内流,而这种反应在敲低PIEZO1基因后显著减弱[59]。生理条件下,适度的机械应力通过激活PIEZO1通道,诱导钙信号传导促进软骨细胞合成Ⅱ型胶原和蛋白聚糖,从而增强软骨的抗压能力[50]。LI等[60]的实验证明,在异常机械应力下,PIEZO1通道会激活丝裂原活化蛋白激酶/胞外信号调节激酶1/2将机械信号传递到细胞核,调控核内凋亡相关基因如Bcl-2、Bax和Caspase-3的表达,诱导软骨细胞凋亡。软骨细胞凋亡后刺激关节内环境会产生大量氧自由基和炎症递质,如白细胞介素1β、肿瘤坏死因子α、前列腺素E2等物质,对新生软骨组织和血管造成伤害;软骨细胞丢失和慢性炎症浸润会导致关节液增多,对关节内外结构施加压力,进一步激活机械感受器和损伤感受器,并形成恶性循环[61]。
近年来越来越多的证据表明,PIEZO离子通道不仅通过调节钙离子内流影响软骨细胞的凋亡,还可能与内质网应激密切相关。在机械应力或炎症因子刺激下,PIEZO1激活导致胞内
Ca2?超载,这会干扰内质网的钙稳态,引发内质网应激[62]。内质网应激信号通路中的关键分子,如C/EBP同源蛋白,已被证明在软骨细胞凋亡中发挥重要作用,C/EBP同源蛋白的上调与骨关节炎软骨退变密切相关,其表达水平与软骨退变程度呈正相关[63]。此外,Caspase-12作为内质网应激介导凋亡通路中的重要执行分子,也在骨关节炎软骨细胞死亡中发挥了显著作用[64]。虽然目前对PIEZO1-内质网应激轴的研究局限于软骨细胞凋亡,但有证据表明这一通路的激活与骨关节炎的整体疾病进展紧密相关。软骨细胞凋亡可促进基质金属蛋白酶和炎症因子的分泌,加速软骨基质降解,导致骨关节炎的病情恶化[65]。因此,PIEZO1离子通道通过调节Ca2?内流并启动内质网应激信号通路,可能在骨关节炎的早期软骨退变及晚期结构破坏中发挥桥梁作用。未来,深入探索PIEZO1-内质网应激的交互作用,不仅有助于揭示骨关节炎的发病机制,也有望成为骨关节炎治疗的新靶点。
软骨细胞死亡在骨关节炎的发病机制中起到关键作用,铁死亡是一种最近发现的细胞死亡形式,会导致线粒体功能障碍和细胞氧化损伤。PIEZO离子通道与细胞的铁死亡和氧化应激之间也存在重要联系。谷胱甘肽过氧化物酶4在调节铁死亡中起着主要作用[66],而谷胱甘肽过氧化物酶4的产生与谷胱甘肽合成相关,该合成可以保护细胞免受铁死亡[67]。机械超负荷通过激活PIEZO1离子通道导致细胞内钙超载,抑制谷胱甘肽合成,进而降低谷胱甘肽过氧化物酶4活性,最终触发脂质过氧化和铁死亡,并加剧小鼠内侧半月板失稳化模型中的软骨退变,加重骨关节炎程度[68]。
越来越多的证据表明,衰老细胞通过衰老相关分泌表促进关节组织中的炎症状态和微环境变化。由衰老关节细胞产生的衰老相关分泌表型因子包括促炎细胞因子(如白细胞介素1、白细胞介素6、白细胞介素8、肿瘤坏死因子α)、趋化因子(如C-C 基序趋化因子配体2、C-C 基序趋化因子配体4)、蛋白酶(基质金属蛋白酶1、基质金属蛋白酶3、基质金属蛋白酶12、基质金属蛋白酶13和含血小板反应蛋白基序的解聚素样金属蛋白酶)、生长因子、小分子代谢物以及microRNA[69-71]。衰老相关分泌表型因子通过旁分泌和自分泌机制传播衰老,进一步促进骨关节炎的发展[72]。LIU等[73]的实验分别使用p38抑制剂SB203580降低白细胞介素6表达、核因子κB抑制剂BMS-345541降低白细胞介素1β表达,证明p38丝裂原活化蛋白激酶和核因子κB的激活分别负责PIEZO1激活后白细胞介素6和白细胞介素1β的产生,提示在骨关节炎病理情况下,PIEZO1通过促进衰老相关分泌表型因子产生加速骨关节炎恶化的可能。
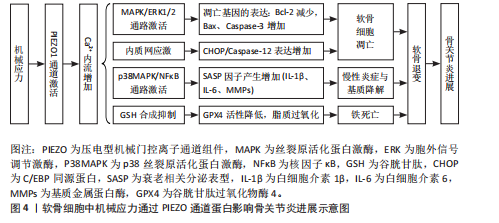
另有研究表明,白细胞介素1α与白细胞介素1受体1型复合物通过丝裂原活化蛋白激酶激酶3/6激活p38丝裂原活化蛋白激酶,磷酸化的p38将信号传递至核内,激活转录因子cAMP反应元件结合蛋白1[57];cAMP反应元件结合蛋白1与转录因子(肝细胞核因子4和活化转录因子2等)共同上调PIEZO1基因的表达,导致胞内静息Ca2+和机械应力诱发的Ca2+增加程度增加。因PIEZO1过表达而增加的胞内静息Ca2+导致F-肌动蛋白稀疏,从而降低软骨细胞的刚度,这导致了机械负荷下细胞的变形程度增加,上调了PIEZO离子通道打开的概率,而PIEZO1过表达进一步放大了这一效应[57];稀疏化的F-肌动蛋白细胞骨架可能通过增加细胞质中p38的可及性促进其在白细胞介素1受体1型激活后的磷酸化。综上所述,这些信号传导和基因重编程会引起一个有害的Ca2+正反馈过程,使得PIEZO1机械传导敏感化并成为骨关节炎的正反馈机制,见图5。
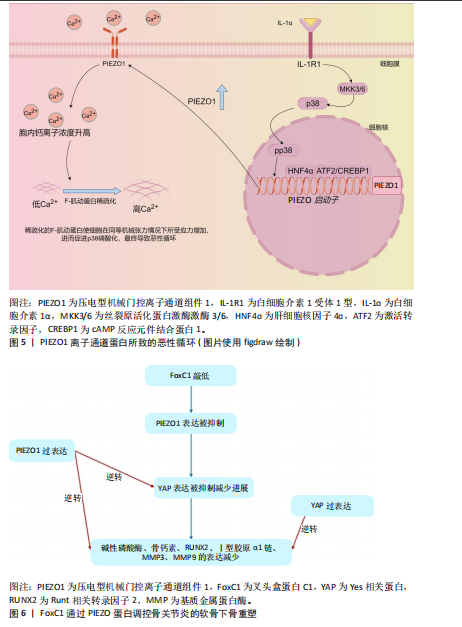
2.4.2 PIEZO离子通道在成骨中的作用 在骨关节炎中,软骨下骨重塑和骨赘形成是导致关节功能障碍和疼痛的核心机制。机械敏感离子通道PIEZO1在成骨细胞中高表达,通过感知机械应力(如关节过载或压缩)介导钙离子内流,调控成骨细胞分化、骨基质沉积及软骨下骨硬化[13]。LI等[74]最近的研究发现,叉头盒蛋白C1敲低降低了PIEZO1和Yes相关蛋白的表达水平,并减少了成骨细胞中碱性磷酸酶、骨钙素、Ⅰ型胶原α1链、Runt相关转录因子2、基质金属蛋白酶3和基质金属蛋白酶9的表达,PIEZO1的过表达逆转了叉头盒蛋白C1敲低对上述蛋白表达水平的抑制作用;此外,Yes相关蛋白过表达增加了成骨细胞中上述蛋白的表达水平,而叉头盒蛋白敲低则降低了这些表达水平;在小鼠中条件性敲除叉头盒蛋白或PIEZO1,小鼠膝关节中软骨下骨的异常增生和硬化程度降低、微血管增生也减少,凸显了PIEZO1在骨关节炎病理中的关键作用,这为理清PIEZO1信号通路提供了新的观点。FoxC1通过PIEZO蛋白调控骨关节炎的软骨下骨重塑的机制,见图6。
碱性磷酸酶是成骨细胞早期分化的标志物,碱性磷酸酶高度表达于骨形成活跃的成骨细胞表面,参与骨基质矿化。在骨关节炎软骨下骨成骨细胞中,碱性磷酸酶活性增强与软骨下骨硬化相关,反映机械应力(如通过PIEZO1感知)诱导的骨形成活跃。骨钙素是成熟成骨细胞分泌的非胶原蛋白,为成骨细胞晚期分化标志物,调控骨基质矿化和钙代谢,促进羟基磷灰石沉积。在骨关节炎中,骨钙素高表达与软骨下骨硬化和骨赘形成相关,反映病理骨重建活跃。Runt相关转录因子2是成骨细胞分化的主调控转录因子,调控间充质干细胞向成骨前体细胞分化,驱动碱性磷酸酶、Ⅰ型胶原α1、骨钙素等基因的表达。Ⅰ型胶原α1链编码Ⅰ型胶原,是骨基质中最丰富的胶原蛋白,构成骨组织的结构框架,为矿化提供基质。成骨细胞在基质成熟阶段高表达Ⅰ型胶原α1,与碱性磷酸酶协同促进基质矿化。在骨关节炎软骨下骨成骨细胞中,Ⅰ型胶原α1过表达与软骨下骨增厚相关,驱动骨关节炎病理骨重塑。基质金属蛋白酶3降解胶原(Ⅱ、Ⅲ型胶原)、蛋白聚糖等基质成分,在骨重塑中调控基质更新。在骨关节炎软骨下骨成骨细胞中,基质金属蛋白酶3表达上调,促进软骨下骨基质降解,增强骨重塑活性,与骨关节炎病理炎症和骨赘形成相关。基质金属蛋白酶9降解Ⅰ型胶原和基质成分,调控骨基质重塑和成骨-破骨细胞交互作用。在骨关节炎软骨下骨成骨细胞中,基质金属蛋白酶9表达增加,促进软骨下骨基质降解,增强骨重塑和炎症反应,与骨赘形成和软骨退变相关。
另外,PIEZO1离子通过促进其下游效应因子Yes相关蛋白的核定位,增强成骨细胞系中Ⅱ型和Ⅸ型胶原蛋白的表达,从而影响破骨细胞生成[75]。ZHOU等[76]的实验发现,在Prrx1启动子驱动的Cre重组酶诱导的PIEZO1条件性敲除和PIEZO1/2双基因敲除突变体的P0骨骼中,Yes相关蛋白和β-连环蛋白含量均降低,而在PIEZO1/2双基因敲除的骨骼样本中这种降低更为明显;此外,Wnt/β-连环蛋白和Yes相关蛋白的转录靶标(如Ctgf/CCN2、Cyr61/CCN1和Tcf7/Lef1)表达在 PIEZO1条件性敲除和PIEZO1/2双基因敲除突变体骨骼中均下调,这些结果表明,PIEZO1/2除通过上调Yes相关蛋白的活性之外也可能通过上调Wnt/β-连环蛋白活性来促进成骨细胞分化,并且使用PIEZO1激动剂Yoda1后,Yoda1也可以促进T 细胞核因子的转录活性。研究结果表明,PIEZO1介导的Ca2?可以协同激活T细胞核因子c1、Yes相关蛋白和β-连环蛋白3个关键转录因子,三者共同调控基因转录变化,进而促进成骨细胞分化与骨形成。
BRYLKA等[77]利用软骨细胞特异性PIEZO1敲除小鼠模型,进一步揭示了PIEZO1在骨关节炎中的双重作用:在生理条件下,PIEZO1缺
失导致出生后早期生长板下方的小梁骨显著消失,并出现肋骨多发性骨折,提示PIEZO1对骨骼发育至关重要;在60周龄未手术小鼠和前交叉韧带切断诱导的骨关节炎模型中,软骨细胞特异性PIEZO1敲除小鼠的软骨退变、骨赘形成及骨赘成熟均显著减轻,同时骨赘组织体积较对照组下降超过70%,髌骨及滑膜钙化程度也明显降低,在来自骨关节炎患者的骨赘样本中亦检测到 PIEZO1在软骨化区的表达明显上调,证实了PIEZO1在病理性骨化中的驱动作用。研究结果表明,软骨中的PIEZO1驱动了生理性及病理性内源性软骨内成骨过程:在生理条件下维持小梁骨形成,而在骨关节炎条件下促进了病理性骨赘的生成,因此,针对 PIEZO1的拮抗策略有望成为抑制骨关节炎骨赘形成的新型分子治疗思路。
2.4.3 PIEZO离子通道在滑膜细胞中的作用 有研究表明,至少50%骨关节炎患者存在不同程度的滑膜炎性改变[78]。滑膜纤维化与骨关节炎病情进展高度相关,是引起患者的关节僵硬、活动受限甚至功能丧失的重要原因之一。滑膜纤维化的组织学特征是滑膜内壁细胞增厚、基质血管化增加、炎症细胞浸润及亚内膜滑膜细胞纤维化,主要原因在于成纤维细胞过度增殖以及胶原合成分解代谢之间的不平衡[79-80]。于利凯等[81]研究发现,使用PIEZO1抑制剂GsMTx4和细胞外信号调节激酶抑制剂PD98059以及两者联合使用均可抑制滑膜纤维化的进展,其中单独使用GsMTx4可以抑制PIEZO1和磷酸化/总细胞外信号调节激酶以及相关mRNA表达,单独使用PD98059仅可以抑制磷酸化/总细胞外信号调节激酶,对PIEZO1无显著影响,这提示细胞外信号调节激酶1/2可能位于PIEZO1的下游,高强度机械应力负荷可通过激活PIEZO1通道启动细胞外信号调节激酶1/2信号通路,从而诱导滑膜组织中Ⅰ型胶原、α-平滑肌肌动蛋白等纤维化标志物表达上调,导致滑膜纤维化的发生与加重;而抑制PIEZO或细胞外信号调节激酶可以缓解纤维化症状并改善滑膜病理状态[81],提示PIEZO1作为机械应力感受器可能在骨关节炎滑膜纤维化的发生发展中发挥关键调控作用。有研究表明,PIEZO1蛋白的过度表达可以促进人类风湿关节炎滑膜细胞的凋亡,PIEZO1蛋白的siRNA沉默表达可以抑制滑膜细胞的凋亡[82],可能为滑膜细胞内PIEZO离子通道在骨关节炎进展中所发挥的作用起到提示作用。
2.4.4 PIEZO离子通道在免疫细胞中的作用 在骨关节炎的炎症微环境中,免疫细胞尤其是T细胞在关节滑膜和软骨组织中发挥着重要的调控作用。近年来,机械敏感性离子通道PIEZO1被证实在T细胞中表达,并且通过介导机械信号的传导参与骨关节炎的慢性炎症反应和关节破坏过程[83]。值得关注的是,ZHOU等[83]在近期研究中提出了一个新的骨关节炎免疫调控机制:机械负荷可通过激活间充质干细胞中的PIEZO1离子通道诱导糖酵解代谢通路的上调(包括己糖激酶2和乳酸脱氢酶A表达增加),进而上调巨噬细胞迁移抑制因子的表达,增强间充质干细胞与T细胞之间的相互作用,最终驱动辅助性T细胞h17细胞的极化,促进白细胞介素17和白细胞介素22等炎症因子的释放,从而加重骨关节炎的炎症进展与组织损伤。该研究不仅拓展了PIEZO1在骨关节炎中的作用范围,也首次揭示了其通过跨细胞代谢信号轴参与间充质干细胞–辅助性T细胞17免疫网络调控的可能机制。
2.4.5 PIEZO离子通道在疼痛感受中的作用 骨关节炎相关疼痛是一种慢性机制性感觉异常,关键机制之一是关节局部炎症与组织重塑所诱导的机械敏化。神经生长因子被广泛认为是骨关节炎疼痛发展的主要递质之一,神经生长因子不仅可通过酪氨酸受体激酶A激活伤害感受神经元,还可诱导中枢神经系统中与疼痛相关的神经肽(如降钙素基因相关肽)表达,从而增强疼痛传导。近期研究表明,机械敏感离子通道PIEZO2在外周伤害性感受器中高度表达,直接参与神经生长因子诱导的机械敏化过程。例如,OBEIDAT等[84]研究显示,伤害感受器特异性PIEZO2条件敲除可以保护雌性小鼠免受与炎性关节疼痛相关的机械致敏、雄性小鼠与骨关节炎相关的关节疼痛以及雄性小鼠与反复关节内注射神经生长因子相关的膝关节肿胀和关节疼痛,证明PIEZO2缺失可显著抑制神经生长因子诱导的降钙素基因相关肽基因表达及疼痛行为反应,PIEZO2对神经生长因子介导的疼痛感受具有关键调控作用。炎症信号缓激肽通过B2受体激活蛋白激酶A和蛋白激酶C信号通路,增强PIEZO2离子通道介导的机械敏感电流,导致电流幅度增大和失活减缓;在原代背根神经节神经元中,这种现象同样存在,PIEZO2离子通道活性被缓激肽显著放大,抑制蛋白激酶A/C或缓激肽信号可缓解炎症相关疼痛(如关节炎)[85]。
PIEZO2在炎症诱导的伤害性感受器敏化中发挥关键作用,PIEZO2下调可显著减弱降钙素基因相关肽的表达和疼痛行为反应[86]。近期有研究提示,PIEZO2离子通道调控过程可能涉及环磷酸腺苷-环磷腺苷直接激活的交换蛋白1-蛋白激酶 A信号轴,或依赖G蛋白偶联受体激酶2对环磷腺苷直接激活的交换蛋白1磷酸化修饰,这一机制已在炎症性痛觉过敏模型中得到验证[87-88]。尽管尚缺乏直接证据证明环磷腺苷直接激活的交换蛋白1与PIEZO2之间存在明确的调控联系,但上述信号路径为进一步探索PIEZO2调控网络提供了可能方向。目前尚未发现PIEZO1直接参与外周伤害性感受的确切机制,但PIEZO1在滑膜细胞和免疫细胞中调节炎症的功能可能通过间接方式放大疼痛信号。整体来看,PIEZO2是机械性疼痛感知的关键感受器,它在骨关节炎疼痛中的作用日益受到关注,可能成为未来镇痛靶点之一。
2.5 PIEZO通道作为骨关节炎治疗的新靶点
PIEZO通道抑制与治疗策略:在前临床模型中,抑制PIEZO1活性已被证明可以减少软骨降解,揭示了抑制PIEZO1逆转骨关节炎的潜能[77,89]。在分子水平上,PIEZO1缺失显著降低了关节软骨细胞中Runt相关转录因子2及其下游含血小板反应蛋白基序的解聚素样金属蛋白酶5的表达水平,这有助于抑制骨关节炎的发生发展[90]。
GsMTx4是一种来源于狼蛛毒液的34个氨基酸肽,可以抑制PIEZO1和PIEZO2离子通道[91-92]。GsMTx4在低张力下通过赖氨酸残基锚定到外膜表面,当膜处于张力状态时,GsMTx4能够更深入地沉入膜中,导致膜的外单层部分松弛[93],这破坏了PIEZO1离子通道附近的张力分布,导致力的传递效率降低,使PIEZO1在机械刺激下更难打开[94]。使用GsMTx4处理软骨外植体,发现GsMTx4能够显著减少机械诱导的软骨细胞死亡[50],这表明抑制PIEZO离子通道活性具有保护软骨的作用,可能为治疗骨关节炎提供潜在策略。然而,骨骼以外的各种细胞类型中也存在PIEZO通道蛋白控制生物体中的关键功能,所以PIEZO离子通道蛋白拮抗剂可能需要关节内注射,以尽可能减轻不良反应[95]。此外,包括GsMTx4在内的阻断剂(如钌红和Gd3+)对PIEZO不具有特异性,因此,通过抑制PIEZO通道蛋白来治疗骨关节炎的药物仍然存在挑战[95]。
GAN等[96]研究发现,青蒿素可以有效抑制软骨细胞中的钙流动、磷脂酰肌醇3激酶/蛋白激酶B的磷酸化以及可能由于PIEZO1离子通道蛋白激活引起的分解代谢相关蛋白标志物的表达,青蒿素既可以防止内侧半月板失稳化诱导的小鼠骨关节炎,又可以改善Yoda1通过激动PIEZO1诱导的骨关节炎病理变化,表明青蒿素至少部分通过靶向作用于PIEZO1蛋白发挥抗骨关节炎功能。
有研究表明,雌激素或其调节剂可有效缓解骨关节炎患者的疼痛,并轻微降低关节置换的患病率和发病率[97]。雌激素的生理浓度可调节软骨细胞活力,并促进软骨细胞增殖和分化,而雌激素缺乏可激活软骨细胞的凋亡机制并抑制软骨细胞增值,导致骨关节炎的发生和发展[98-99]。近期研究发现,雌激素通路也参与PIEZO1介导的软骨细胞机械信号传导,G蛋白偶联雌激素受体的激活在机械应激诱导的软骨细胞凋亡中起关键作用[100]。G-1(G蛋白偶联雌激素受体激动剂)激活G蛋白偶联雌激素受体,可显著促进转录共刺激分子Yes相关蛋白从细胞质转移到细胞核,Yes相关蛋白的核定位上调了ARHGAP29表达,下调了Ras同源基因家族成员A、LIM激酶和肌动蛋白解聚因子的磷酸化,导致肌动蛋白解聚,从而阻断了机械应力对PIEZO1的刺激,最终提升了软骨细胞活力。
尽管目前尚无已上市药物专门针对PIEZO离子通道,但PIEZO1在骨关节炎关键细胞(滑膜细胞、软骨细胞、免疫细胞)中的普遍表达以及其调控钙信号、炎症反应与细胞命运的能力,使其成为未来骨关节炎治疗研究的重要靶点之一,未来或可通过靶向通道结构域、调节膜张力感应机制或设计RNA干预策略实现更精准的干预。

| [1] HUNTER DJ, BIERMA-ZEINSTRA S. Osteoarthritis. Lancet. 2019;393(10182):1745-1759. [2] BIJLSMA JW, BERENBAUM F, LAFEBER FP. Osteoarthritis: an update with relevance for clinical practice. Lancet. 2011;377(9783):2115-2126. [3] TURKIEWICZ A, KIADALIRI AA, ENGLUND M. Cause-specific mortality in osteoarthritis of peripheral joints. Osteoarthritis Cartilage. 2019;27(6):848-854. [4] LIU Q, NIU J, LI H, et al. Knee Symptomatic Osteoarthritis, Walking Disability, NSAIDs Use and All-cause Mortality: Population-based Wuchuan Osteoarthritis Study. Sci Rep. 2017;7(1):3309. [5] SHARMA L. Osteoarthritis of the Knee. N Engl J Med. 2021;384(1):51-59. [6] CAO F, XU Z, LI XX, et al. Trends and cross-country inequalities in the global burden of osteoarthritis, 1990-2019: A population-based study. Ageing Res Rev. 2024;99(102382. [7] LONG H, LIU Q, YIN H, et al. Prevalence Trends of Site-Specific Osteoarthritis From 1990 to 2019: Findings From the Global Burden of Disease Study 2019. Arthritis Rheumatol. 2022;74(7):1172-1183. [8] JIANG W, LIU H, WAN R, et al. Mechanisms linking mitochondrial mechanotransduction and chondrocyte biology in the pathogenesis of osteoarthritis. Ageing Res Rev. 2021;67:101315. [9] XIA B, DI C, ZHANG J, et al. Osteoarthritis pathogenesis: a review of molecular mechanisms. Calcif Tissue Int. 2014;95(6):495-505. [10] MOBASHERI A, RAYMAN MP, GUALILLO O, et al. The role of metabolism in the pathogenesis of osteoarthritis. Nat Rev Rheumatol. 2017;13(5): 302-311. [11] YAO Q, WU X, TAO C, et al. Osteoarthritis: pathogenic signaling pathways and therapeutic targets. Signal Transduct Target Ther. 2023;8(1):56. [12] ZHANG Y, ZHUANG H, REN X, et al. Implications of mechanosensitive ion channels in the pathogenesis of osteoarthritis: a comprehensive review. Front Cell Dev Biol. 2025;13:1549812. [13] LEI L, WEN Z, CAO M, et al. The emerging role of Piezo1 in the musculoskeletal system and disease. Theranostics. 2024;14(10):3963-3983. [14] COSTE B, MATHUR J, SCHMIDT M, et al. Piezo1 and Piezo2 are essential components of distinct mechanically activated cation channels. Science. 2010;330(6000):55-60. [15] BAE C, SACHS F, GOTTLIEB PA. The mechanosensitive ion channel Piezo1 is inhibited by the peptide GsMTx4. Biochemistry. 2011;50(29):6295-6300. [16] RANADE SS, WOO SH, DUBIN AE, et al. Piezo2 is the major transducer of mechanical forces for touch sensation in mice. Nature. 2014; 516(7529):121-125. [17] GE J, LI W, ZHAO Q, et al. Architecture of the mammalian mechanosensitive Piezo1 channel. Nature. 2015;527(7576):64-69. [18] ZHANG T, CHI S, JIANG F, et al. A protein interaction mechanism for suppressing the mechanosensitive Piezo channels. Nat Commun. 2017;8(1):1797. [19] XIAO B. Levering Mechanically Activated Piezo Channels for Potential Pharmacological Intervention. Annu Rev Pharmacol Toxicol. 2020; 60:195-218. [20] ATCHA H, JAIRAMAN A, HOLT JR, et al. Mechanically activated ion channel Piezo1 modulates macrophage polarization and stiffness sensing. Nat Commun. 2021;12(1):3256. [21] JIANG W, WIJERATHNE TD, ZHANG H, et al. Structural and thermodynamic framework for PIEZO1 modulation by small molecules. Proc Natl Acad Sci U S A. 2023;120(50):e2310933120. [22] KOSTER AK, YARISHKIN O, DUBIN AE, et al. Chemical mapping of the surface interactome of PIEZO1 identifies CADM1 as a modulator of channel inactivation. Proc Natl Acad Sci U S A. 2024;121(41):e2415934121. [23] GUILAK F. Biomechanical factors in osteoarthritis. Best Pract Res Clin Rheumatol. 2011;25(6):815-823. [24] ZHANG J, HAO X, CHI R, et al. Moderate mechanical stress suppresses the IL-1β-induced chondrocyte apoptosis by regulating mitochondrial dynamics. J Cell Physiol. 2021;236(11):7504-7515. [25] BLALOCK D, MILLER A, TILLEY M, et al. Joint instability and osteoarthritis. Clin Med Insights Arthritis Musculoskelet Disord. 2015;8:15-23. [26] XU B, XING R, HUANG Z, et al. Excessive mechanical stress induces chondrocyte apoptosis through TRPV4 in an anterior cruciate ligament-transected rat osteoarthritis model. Life Sci. 2019; 228:158-166. [27] BUCKWALTER JA, ANDERSON DD, BROWN TD, et al. The Roles of Mechanical Stresses in the Pathogenesis of Osteoarthritis: Implications for Treatment of Joint Injuries. Cartilage. 2013;4(4): 286-294. [28] VINCENT TL. Mechanoflammation in osteoarthritis pathogenesis. Semin Arthritis Rheum. 2019;49(3s): S36-S38. [29] VAN DEN BOSCH MHJ, BLOM AB, VAN DER KRAAN PM. Inflammation in osteoarthritis: Our view on its presence and involvement in disease development over the years. Osteoarthritis Cartilage. 2024;32(4):355-364. [30] YANG Y, WANG Y, KONG Y, et al. Mechanical stress protects against osteoarthritis via regulation of the AMPK/NF-κB signaling pathway. J Cell Physiol. 2019;234(6):9156-9167. [31] TAKEBE K, NISHIYAMA T, HAYASHI S, et al. Regulation of p38 MAPK phosphorylation inhibits chondrocyte apoptosis in response to heat stress or mechanical stress. Int J Mol Med. 2011;27(3): 329-335. [32] CAI X, WARBURTON C, PEREZ OF, et al. Hippo-PKCζ-NFκB signaling axis: A druggable modulator of chondrocyte responses to mechanical stress. iScience. 2024;27(6):109983. [33] WANG T, HE C. Pro-inflammatory cytokines: The link between obesity and osteoarthritis. Cytokine Growth Factor Rev. 2018;44:38-50. [34] BONNELYE E, REBOUL P, DUVAL N, et al. Estrogen receptor-related receptor α regulation by interleukin-1β in prostaglandin E(2)- and cAMP-dependent pathways in osteoarthritic chondrocytes. Arthritis Rheum. 2011;63(8):2374-2384. [35] OHASHI H, NISHIDA K, YOSHIDA A, et al. Adipose-Derived Extract Suppresses IL-1β-Induced Inflammatory Signaling Pathways in Human Chondrocytes and Ameliorates the Cartilage Destruction of Experimental Osteoarthritis in Rats. Int J Mol Sci. 2021;22(18):9781. [36] LEE YA, CHOI HM, LEE SH, et al. Synergy between adiponectin and interleukin-1β on the expression of interleukin-6, interleukin-8, and cyclooxygenase-2 in fibroblast-like synoviocytes. Exp Mol Med. 2012;44(7):440-447. [37] LAI Y, BAI X, ZHAO Y, et al. ADAMTS-7 forms a positive feedback loop with TNF-α in the pathogenesis of osteoarthritis. Ann Rheum Dis. 2014;73(8):1575-1584. [38] ZHOU Q, REN Q, JIAO L, et al. The potential roles of JAK/STAT signaling in the progression of osteoarthritis. Front Endocrinol (Lausanne). 2022; 13:1069057. [39] TEIJEIRA A, GARASA S, OCHOA MDC, et al. Differential Interleukin-8 thresholds for chemotaxis and netosis in human neutrophils. Eur J Immunol. 2021;51(9):2274-2280. [40] KOPESKY P, TIEDEMANN K, ALKEKHIA D, et al. Autocrine signaling is a key regulatory element during osteoclastogenesis. Biol Open. 2014;3(8): 767-776. [41] GARCíA-MANRIQUE M, CALVET J, ORELLANA C, et al. Synovial fluid but not plasma interleukin-8 is associated with clinical severity and inflammatory markers in knee osteoarthritis women with joint effusion. Sci Rep. 2021;11(1):5258. [42] OHASHI Y, UCHIDA K, FUKUSHIMA K, et al. NGF Expression and Elevation in Hip Osteoarthritis Patients with Pain and Central Sensitization. Biomed Res Int. 2021;2021:9212585. [43] ZHU LJ, YANG TC, WU Q, et al. Tumor necrosis factor receptor-associated factor (TRAF) 6 inhibition mitigates the pro-inflammatory roles and proliferation of rheumatoid arthritis fibroblast-like synoviocytes. Cytokine. 2017;93:26-33. [44] FANG X Z, ZHOU T, XU J Q, et al. Structure, kinetic properties and biological function of mechanosensitive Piezo channels. Cell Biosci. 2021;11(1):13. [45] GNANASAMBANDAM R, BAE C, GOTTLIEB PA, et al. Ionic Selectivity and Permeation Properties of Human PIEZO1 Channels. PLoS One. 2015;10(5):e0125503. [46] BAGRIANTSEV SN, GRACHEVA EO, GALLAGHER PG. Piezo proteins: regulators of mechanosensation and other cellular processes. J Biol Chem. 2014; 289(46):31673-31681. [47] JIANG Y, YANG X, JIANG J, et al. Structural Designs and Mechanogating Mechanisms of the Mechanosensitive Piezo Channels. Trends Biochem Sci. 2021;46(6):472-488. [48] ZENG WZ, MARSHALL KL, MIN S, et al. PIEZOs mediate neuronal sensing of blood pressure and the baroreceptor reflex. Science. 2018; 362(6413):464-467. [49] MARTINS JR, PENTON D, PEYRONNET R, et al. Piezo1-dependent regulation of urinary osmolarity. Pflugers Arch. 2016;468(7):1197-1206. [50] LEE W, LEDDY HA, CHEN Y, et al. Synergy between Piezo1 and Piezo2 channels confers high-strain mechanosensitivity to articular cartilage. Proc Natl Acad Sci U S A. 2014;111(47):E5114-E5122. [51] MIKHAILOV N, LESKINEN J, FAGERLUND I, et al. Mechanosensitive meningeal nociception via Piezo channels: Implications for pulsatile pain in migraine? Neuropharmacology. 2019;149:113-123. [52] WOO SH, RANADE S, WEYER AD, et al. Piezo2 is required for Merkel-cell mechanotransduction. Nature. 2014;509(7502):622-626. [53] LEWIS AH, GRANDL J. Mechanical sensitivity of Piezo1 ion channels can be tuned by cellular membrane tension. Elife. 2015;4:e12088. [54] ZEITZSCHEL N, LECHNER SG. The activation thresholds and inactivation kinetics of poking-evoked PIEZO1 and PIEZO2 currents are sensitive to subtle variations in mechanical stimulation parameters. Channels (Austin). 2024; 18(1):2355123. [55] SYEDA R, XU J, DUBIN AE, et al. Chemical activation of the mechanotransduction channel Piezo1. Elife. 2015;4:e07369. [56] EVANS EL, CUTHBERTSON K, ENDESH N, et al. Yoda1 analogue (Dooku1) which antagonizes Yoda1-evoked activation of Piezo1 and aortic relaxation. Br J Pharmacol. 2018;175(10):1744-1759. [57] LEE W, NIMS RJ, SAVADIPOUR A, et al. Inflammatory signaling sensitizes Piezo1 mechanotransduction in articular chondrocytes as a pathogenic feed-forward mechanism in osteoarthritis. Proc Natl Acad Sci U S A. 2021; 118(13):e2001611118. [58] LEE W, GUILAK F, LIEDTKE W. Role of Piezo Channels in Joint Health and Injury. Curr Top Membr. 2017;79:263-273. [59] SERVIN-VENCES MR, MORONI M, LEWIN GR, et al. Direct measurement of TRPV4 and PIEZO1 activity reveals multiple mechanotransduction pathways in chondrocytes. Elife. 2017;6:e21074. [60] LI XF, ZHANG Z, LI XD, et al. [Mechanism of the Piezo1 protein-induced apoptosis of the chondrocytes through the MAPK/ERK1/2 signal pathway]. Zhonghua Yi Xue Za Zhi. 2016;96(31): 2472-2477. [61] HWANG HS, KIM HA. Chondrocyte Apoptosis in the Pathogenesis of Osteoarthritis. Int J Mol Sci. 2015;16(11): 26035-26054. [62] WANG X, TAO J, ZHOU J, et al. Excessive load promotes temporomandibular joint chondrocyte apoptosis via Piezo1/endoplasmic reticulum stress pathway. J Cell Mol Med. 2024;28(11):e18472. [63] UEHARA Y, HIROSE J, YAMABE S, et al. Endoplasmic reticulum stress-induced apoptosis contributes to articular cartilage degeneration via C/EBP homologous protein. Osteoarthritis Cartilage. 2014;22(7):1007-1017. [64] LIU C, CAO Y, YANG X, et al. Tauroursodeoxycholic acid suppresses endoplasmic reticulum stress in the chondrocytes of patients with osteoarthritis . Int J Mol Med. 2015;36(4):1081-1087. [65] MEHANA EE, KHAFAGA AF, EL-BLEHI SS. The role of matrix metalloproteinases in osteoarthritis pathogenesis: An updated review. Life Sci. 2019; 234:116786. [66] ZHANG Y, SWANDA RV, NIE L, et al. mTORC1 couples cyst(e)ine availability with GPX4 protein synthesis and ferroptosis regulation. Nat Commun. 2021;12(1):1589. [67] LEI G, MAO C, YAN Y, et al. Ferroptosis, radiotherapy, and combination therapeutic strategies. Protein Cell. 2021;12(11):836-857. [68] WANG S, LI W, ZHANG P, et al. Mechanical overloading induces GPX4-regulated chondrocyte ferroptosis in osteoarthritis via Piezo1 channel facilitated calcium influx. J Adv Res. 2022;41:63-75. [69] ASTRIKE-DAVIS EM, CORYELL P, LOESER RF. Targeting cellular senescence as a novel treatment for osteoarthritis. Curr Opin Pharmacol. 2022;64:102213. [70] GASEK NS, KUCHEL GA, KIRKLAND JL, et al. Strategies for Targeting Senescent Cells in Human Disease. Nat Aging. 2021;1(10) 870-879. [71] LIU Y, ZHANG Z, LI T, et al. Senescence in osteoarthritis: from mechanism to potential treatment. Arthritis Res Ther. 2022;24(1):174. [72] FAUST HJ, ZHANG H, HAN J, et al. IL-17 and immunologically induced senescence regulate response to injury in osteoarthritis. J Clin Invest. 2020;130(10):5493-5507. [73] LIU Y, ZHANG Z, LI J, et al. Piezo1 transforms mechanical stress into pro senescence signals and promotes osteoarthritis severity. Mech Ageing Dev. 2023;216:111880. [74] LI Z, HAO L, CHEN S, et al. Forkhead box C1 promotes the pathology of osteoarthritis in subchondral bone osteoblasts via the Piezo1/YAP axis. Cell Signal. 2024;124:111463. [75] WANG L, YOU X, LOTINUN S, et al. Mechanical sensing protein PIEZO1 regulates bone homeostasis via osteoblast-osteoclast crosstalk. Nat Commun. 2020;11(1):282. [76] ZHOU T, GAO B, FAN Y, et al. Piezo1/2 mediate mechanotransduction essential for bone formation through concerted activation of NFAT-YAP1-ß-catenin. Elife. 2020;9:e52779. [77] BRYLKA LJ, ALIMY AR, TSCHAFFON-MÜLLER MEA, et al. Piezo1 expression in chondrocytes controls endochondral ossification and osteoarthritis development. Bone Res. 2024;12(1):12. [78] SAKKAS LI, PLATSOUCAS CD. The role of T cells in the pathogenesis of osteoarthritis. Arthritis Rheum. 2007;56(2):409-424. [79] MATHIESSEN A, CONAGHAN PG. Synovitis in osteoarthritis: current understanding with therapeutic implications. Arthritis Res Ther. 2017; 19(1):18. [80] KUROWSKA-STOLARSKA M, ALIVERNINI S. Synovial tissue macrophages: friend or foe? RMD Open. 2017;3(2):e000527. [81] 于利凯,田地,苏子珊,等.机械应力经Piezo1/ERK1/2轴介导KOA滑膜纤维化的机制研究[J].中国比较医学杂志,2024,34(10):47-56. [82] 曲向阳,宋钦勇.类风湿关节炎成纤维样滑膜细胞凋亡与MAPK/ERK5信号通路的介导[J].中国组织工程研究,2021,25(26):4156-4161. [83] ZHOU Y, LI M, LIN S, et al. Mechanical sensing protein PIEZO1 controls osteoarthritis via glycolysis mediated mesenchymal stem cells-Th17 cells crosstalk. Cell Death Dis. 2025;16(1): 231. [84] OBEIDAT AM, WOOD MJ, ADAMCZYK NS, et al. Piezo2 expressing nociceptors mediate mechanical sensitization in experimental osteoarthritis. Nat Commun. 2023;14(1):2479. [85] DUBIN AE, SCHMIDT M, MATHUR J, et al. Inflammatory signals enhance piezo2-mediated mechanosensitive currents. Cell Rep. 2012;2(3):511-517. [86] NENCINI S, MORGAN M, THAI J, et al. Piezo2 Knockdown Inhibits Noxious Mechanical Stimulation and NGF-Induced Sensitization in A-Delta Bone Afferent Neurons. Front Physiol. 2021; 12:644929. [87] EIJKELKAMP N, LINLEY JE, TORRES JM, et al. A role for Piezo2 in EPAC1-dependent mechanical allodynia. Nat Commun. 2013;4:1682. [88] SINGHMAR P, HUO X, EIJKELKAMP N, et al. Critical role for Epac1 in inflammatory pain controlled by GRK2-mediated phosphorylation of Epac1. Proc Natl Acad Sci U S A. 2016;113(11):3036-3041. [89] SAVADIPOUR A, NIMS RJ, RASHIDI N, et al. Membrane stretch as the mechanism of activation of PIEZO1 ion channels in chondrocytes. Proc Natl Acad Sci U S A. 2023;120(30):e2221958120. [90] CHEN D, KIM DJ, SHEN J, et al. Runx2 plays a central role in Osteoarthritis development. J Orthop Translat. 2020;23:132-139. [91] BOWMAN CL, GOTTLIEB PA, SUCHYNA TM, et al. Mechanosensitive ion channels and the peptide inhibitor GsMTx-4: history, properties, mechanisms and pharmacology. Toxicon. 2007;49(2):249-270. [92] ALCAINO C, KNUTSON K, GOTTLIEB PA, et al. Mechanosensitive ion channel Piezo2 is inhibited by D-GsMTx4. Channels (Austin). 2017;11(3):245-253. [93] GAO W, HASAN H, ANDERSON DE, et al. The Role of Mechanically-Activated Ion Channels Piezo1, Piezo2, and TRPV4 in Chondrocyte Mechanotransduction and Mechano-Therapeutics for Osteoarthritis. Front Cell Dev Biol. 2022;10: 885224. [94] GNANASAMBANDAM R, GHATAK C, YASMANN A, et al. GsMTx4: Mechanism of Inhibiting Mechanosensitive Ion Channels. Biophys J. 2017; 112(1):31-45. [95] SYEDA R. Physiology and Pathophysiology of Mechanically Activated PIEZO Channels. Annu Rev Neurosci. 2021;44:383-402. [96] GAN D, TAO C, JIN X, et al. Piezo1 activation accelerates osteoarthritis progression and the targeted therapy effect of artemisinin. J Adv Res. 2024;62:105-117. [97] XIAO YP, TIAN FM, DAI MW, et al. Are estrogen-related drugs new alternatives for the management of osteoarthritis? Arthritis Res Ther. 2016;18:151. [98] GE Y, ZHOU S, LI Y, et al. Estrogen prevents articular cartilage destruction in a mouse model of AMPK deficiency via ERK-mTOR pathway. Ann Transl Med. 2019;7(14):336. [99] TALWAR RM, WONG BS, SVOBODA K, et al. Effects of estrogen on chondrocyte proliferation and collagen synthesis in skeletally mature articular cartilage. J Oral Maxillofac Surg. 2006;64(4):600-609. [100] SUN Y, LENG P, GUO P, et al. G protein coupled estrogen receptor attenuates mechanical stress-mediated apoptosis of chondrocyte in osteoarthritis via suppression of Piezo1. Mol Med. 2021;27(1):96. [101] STEINECKER-FROHNWIESER B, LOHBERGER B, TOEGEL S, et al. Activation of the Mechanosensitive Ion Channels Piezo1 and TRPV4 in Primary Human Healthy and Osteoarthritic Chondrocytes Exhibits Ion Channel Crosstalk and Modulates Gene Expression. Int J Mol Sci. 2023; 24(9):7868. [102] WAN Y, ZHOU J, LI H. The Role of Mechanosensitive Piezo Channels in Chronic Pain. J Pain Res. 2024; 17:4199-4212. |
| [1] | 于晨锜, 刘洋, 余建锋, 康康, 邓垚歌, 夏小伟, 张一健, 朱雪松. 仿生黑磷纳米系统调控滑膜巨噬细胞极化治疗骨关节炎[J]. 中国组织工程研究, 2026, 30(在线): 1-13. |
| [2] | 张 楠, 孟庆华, 鲍春雨. 踝关节有限元模型的特性及临床应用[J]. 中国组织工程研究, 2026, 30(9): 2343-2349. |
| [3] | 陈秋函, 杨 龙, 袁代柱, 吴展羽, 邹梓豪, 叶 川. 膝关节周围截骨治疗膝骨关节炎:治疗策略的优化[J]. 中国组织工程研究, 2026, 30(9): 2303-2312. |
| [4] | 张子峥, 罗 旺, 刘长路. 膝内侧间室骨关节炎单髁置换中有限元分析的应用价值[J]. 中国组织工程研究, 2026, 30(9): 2313-2322. |
| [5] | 黎清斌, 林建辉, 黄文杰, 王明爽, 杜间开, 劳永锵. 膝关节周围骨巨细胞瘤病灶扩大刮除后填充骨水泥:软骨下植骨与不植骨的比较[J]. 中国组织工程研究, 2026, 30(8): 1896-1902. |
| [6] | 宋浦蓁, 马贺宾, 陈宏广, 章亚东. 骨髓间充质干细胞外泌体联合转化生长因子β1对巨噬细胞的作用[J]. 中国组织工程研究, 2026, 30(7): 1616-1623. |
| [7] | 李林臻, 焦泓焯, 陈伟南, 张铭哲, 王建龙, 张君涛. 淫羊藿苷含药血清对脂多糖诱导人软骨细胞炎症损伤的影响[J]. 中国组织工程研究, 2026, 30(6): 1368-1374. |
| [8] | 李 豪, 陶红成, 曾 平, 刘金富, 丁 强, 牛驰程, 黄 凯, 康宏誉. 丝裂原活化蛋白激酶信号通路调控骨关节炎的发生发展:指导中药靶点治疗[J]. 中国组织工程研究, 2026, 30(6): 1476-1485. |
| [9] | 陈 驹, 郑锦畅, 梁 振, 黄成硕, 林 颢, 曾 莉. β-石竹烯对小鼠膝骨关节炎的作用及机制[J]. 中国组织工程研究, 2026, 30(6): 1341-1347. |
| [10] | 吕国庆, 艾孜麦提江·肉孜, 熊道海. 鸢尾素抑制人关节软骨细胞中铁死亡的作用及其机制[J]. 中国组织工程研究, 2026, 30(6): 1359-1367. |
| [11] | 张 倩, 黄东锋. 加权基因共表达网络分析结合机器学习筛选及验证骨关节炎生物标记物[J]. 中国组织工程研究, 2026, 30(5): 1096-1105. |
| [12] | 部洋洋, 宁新丽, 赵 琛. 关节腔注射治疗颞下颌关节骨关节炎:不同药物与多种联合治疗方案[J]. 中国组织工程研究, 2026, 30(5): 1215-1224. |
| [13] | 刘凤芝, 董玉娜, 田文艺, 王春雷, 梁晓东, 鲍 霖. 基因预测731种免疫细胞表型与类风湿性关节炎的关系[J]. 中国组织工程研究, 2026, 30(5): 1311-1319. |
| [14] | 陈伊娴, 陈 晨, 卢立恒, 汤锦鹏, 于晓巍. 雷公藤甲素治疗骨关节炎的网络药理学分析与实验验证[J]. 中国组织工程研究, 2026, 30(4): 805-815. |
| [15] | 包卓玛, 侯孜明, 江 露, 李玮怡, 张宗星, 刘道忠, 袁 林. 枫杨总黄酮调控类风湿关节炎成纤维样滑膜细胞增殖、迁移及凋亡的作用及机制[J]. 中国组织工程研究, 2026, 30(4): 816-823. |
机械应力在关节软骨和周围组织的退行性变中起着核心作用[11],目前已知的机械应力感受器包括瞬时受体电位通道家族、双孔域钾通道家族、退化因子/上皮钠通道家族以及整合素家族等。压电型机械门控离子通道组件(piezo-type mechanosensitive ion channel component,PIEZO)1和PIEZO2首次被鉴定为机械敏感钙离子通道[12],在骨关节炎中发挥着关键作用,包括调控软骨细胞凋亡和炎症反应等[13]。PIEZO通道通过感知细胞膜的机械刺激调控钙离子流入,进而影响软骨细胞、成骨功能、滑膜细胞、免疫细胞及疼痛感知等过程。该文旨在系统综述PIEZO离子通道在骨关节炎病理机制中的作用,探讨它在上述细胞与疼痛感受中的功能,评估其作为新型治疗靶点的可能性,为未来骨关节炎的精准治疗提供理论基础。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1 文章框架设计 见图1。
1.2 资料来源
1.2.1 检索人及检索时间 由第一作者于2025年4月进行检索。
1.2.2 文献检索时限 2000-2025年。
1.2.3 检索数据库 PubMed数据库、中国知网。
1.2.5 检索文献类型 包括原始研究、荟萃分析、综述和临床试验。
1.2.6 检索策略 以PubMed数据库检索策略为例,见图2。
1.2.7 检索文献量 初步检索到526篇文献。
1.3 入组标准
1.3.1 纳入标准 ①PIEZO对骨关节炎影响及作用机制的相关文献;②PIEZO与软骨细胞、成骨细胞、滑膜细胞、免疫细胞、疼痛相关研究的文献;③PIEZO抑制剂对骨关节炎作用的相关文献。
1.3.2 排除标准 与文章主题无关、研究质量较差、重复性及陈旧性文献。
1.4 文献质量评估及数据提取 通过计算机检索与手工检索,共检索到526篇参考文献,按入选标准进行人工筛选,排除与主题相关性差及重复、陈旧的文献,最终纳入102篇文献,纳入研究的文献包括研究原著、综述等。文献筛选流程见图3。
3.2 该综述区别于他人他篇的特点 该综述的独特之处在于从PIEZO蛋白作为核心切入点,系统阐述了它在多种细胞类型中的作用,包括软骨细胞、成骨细胞、滑膜细胞、免疫细胞以及疼痛感知过程等,这与其他综述不同,后者往往仅聚焦于单一细胞类型或机制。与以往相关综述相比,该文强调了PIEZO离子通道在骨关节炎整体病理网络中的整合作用,例如如何通过钙离子内流连接机械应力与下游信号通路,从而驱动细胞凋亡、炎症和骨重塑。此外,该文介绍了PIEZO2离子通道蛋白在骨关节炎疼痛中的贡献,这一部分内容在很多综述中未被提及,该综述提示PIEZO2离子通道蛋白在控制骨关节炎疼痛方面的重要潜力。文章还提示了PIEZO蛋白作为靶点在预防和治疗骨关节炎的潜力,为今后开发治疗关节炎的新技术、新药物提供了全面的研究思路。
3.3 该综述的局限性 尽管PIEZO离子通道在骨关节炎中的研究取得了进展,但当前证据仍存在显著局限性,仍然需要更加深入的研究:目前许多研究仍然依赖于动物模型,如小鼠膝骨关节炎模型,结论与研究结果在人类骨关节炎中是否适用或是否存在差异表达仍然需要进一步探索;体外研究往往使用机械应力模拟,这与真实关节内的复杂环境存在差异,结论是否适用仍然存疑;当前关于关节炎中PIEZO的研究并不充分,临床数据匮乏,大多停留在基础阶段,缺乏大型队列研究验证PIEZO变异与骨关节炎风险的相关性。下一步研究应该着重于PIEZO离子通道蛋白在骨关节炎中各信号通路之间的交互与联系,只有这样才能更深入了解PIEZO离子通道蛋白在关节炎中扮演的角色,并为下一步PIEZO离子通道蛋白作为靶点的治疗提供前提。
3.4 该综述的重要意义 该综述的重要意义在于阐明PIEZO离子通道在骨关节炎中的多重作用,为未来研究和治疗提供了新思路。PIEZO离子通道作为机械转导的关键中介,在调控钙信号和下游通路中的作用突出了它作为骨关节炎治疗靶点的潜力。通过抑制PIEZO离子通道,或可成为治疗关节炎的有效方法。该综述还强调PIEZO在整体关节稳态中的意义,其调控可能整合多细胞过程,如免疫和骨重塑。在疼痛管理方面,PIEZO2的靶向抑制可缓解疼痛,提供非阿片类镇痛选项。以上不仅为精准医学提供了基础,还为新型疗法的开发提供了前景。
3.5 课题专家组对未来的建议 未来研究应聚焦于PIEZO离子通道的临床转化,例如开发特异性调制剂并进行大型临床试验,以验证它在人类骨关节炎中的疗效。此外,专家建议整合PIEZO离子通道蛋白与压电材料的交叉研究,如可穿戴压电器件监测关节健康,或注入式压电水凝胶促进软骨再生。总体上,未来方向包括多学科合作,推动从基础机制到临床应用的转变。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
该综述系统阐述了机械应力通过压电型机械门控离子通道组件(piezo-type mechanosensitive ion channel component,Piezo)离子通道调控骨关节炎的分子机制与治疗靶点,填补了现有研究在多细胞整合与疼痛调控方面的空白。不同于以往聚焦单一细胞或机制的综述,该文从机械应力诱导的炎症反应入手,揭示Piezo通道作为核心机械感受器,如何感知环境压力变化,调控Ca²⁺内流并激活下游通路,驱动软骨细胞凋亡、内质网应激、铁死亡及衰老相关分泌表型,从而加速软骨退变和关节炎症。特别强调Piezo1在成骨细胞中的作用,通过叉头盒蛋白/Piezo1/Yes相关蛋白促进软骨下骨重塑和骨赘形成;而在滑膜细胞中,Piezo1介导ERK1/2信号诱导纤维化;在免疫细胞中,Piezo1调控间充质干细胞-Th17轴,促进炎症因子释放。这些机制形成恶性正反馈循环,解释了骨关节炎的进展动态。同时,该文评估了Piezo抑制剂的治疗潜力,如GsMTx4、青蒿素和G-1,提示Piezo作为新兴靶点的临床转化前景。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||