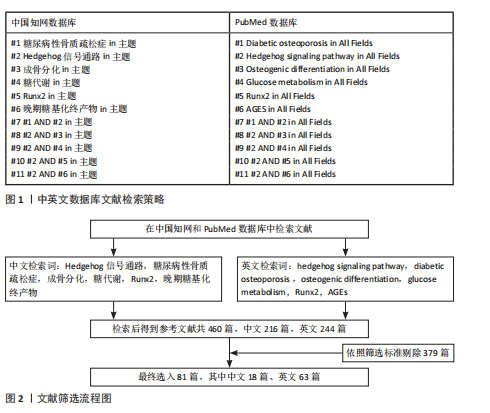
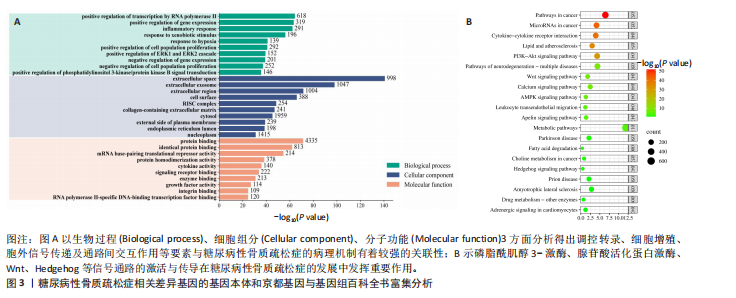
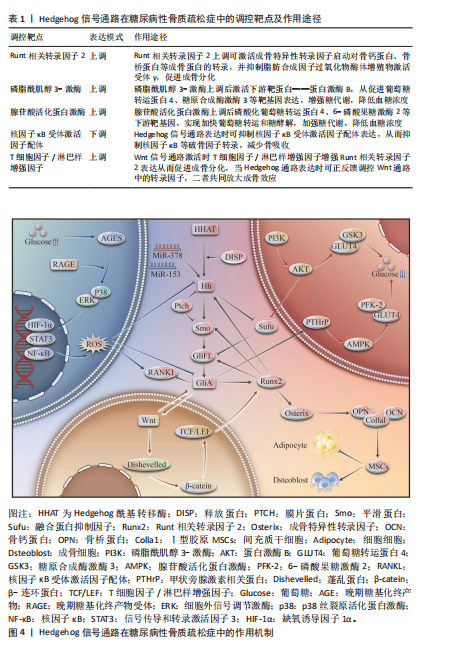
[1] 郭曼, 黄炜, 伍棋, 等. 铁死亡在2型糖尿病性骨质疏松症中的作用机制和研究进展[J].中华骨质疏松和骨矿盐疾病杂志,2024,17(5):498-505.
[2] 阮泽松, 陈云丰. 间充质干细胞源性外泌体在糖尿病性骨质疏松中的研究进展[J]. 中国骨质疏松杂志,2022,28(1):104-108.
[3] 文晓晨, 修静, 刘子英, 等. 糖尿病性骨质疏松症与糖尿病肾病机制及异病同治探析[J]. 辽宁中医药大学学报,2025,27(7):162-170.
[4] 柴家超, 杨卫芳, 扈艳雯, 等. SGLT2i对2型糖尿病患者骨代谢影响的研究进展[J]. 中国骨质疏松杂志,2023,29(12):1866-1872.
[5] 高浩. 降糖药对糖尿病患者骨代谢的影响[J]. 黑龙江医学,2023,47(22):2815-2817.
[6] 马玉萍. 二甲双胍对1型糖尿病患者骨代谢标志物及骨密度影响的研究[D]. 兰州:兰州大学,2021.
[7] SUCHORS C, KIM J. Canonical Hedgehog Pathway and Noncanonical GLI Transcription Factor Activation in Cancer. Cells. 2022;11(16):2523.
[8] JING J, WU Z, WANG J, et al. Hedgehog signaling in tissue homeostasis, cancers, and targeted therapies. Signal Transduct Target Ther. 2023;8(1):315.
[9] ZHOU H, ZHANG L, CHEN Y, et al. Research progress on the hedgehog signalling pathway in regulating bone formation and homeostasis. Cell Prolif. 2022;55(1):e13162.
[10] 王雨荷, 刘红, 史婧儒, 等. 由Hedgehog信号通路探讨左归丸对卵巢切除大鼠骨质疏松症的影响[J]. 中国骨质疏松杂志,2023,29(6):781-785+791.
[11] 阿布都克热木·达吾提, 柯义兵, 金裕林, 等. 骨质疏松症相关信号通路研究进展[J]. 国际骨科学杂志,2025,46(3):178-182.
[12] NGUYEN NM, CHO J. Hedgehog Pathway Inhibitors as Targeted Cancer Therapy and Strategies to Overcome Drug Resistance. Int J Mol Sci. 2022;23(3):1733.
[13] JIANG J. Hedgehog signaling mechanism and role in cancer. Semin Cancer Biol. 2022;85:107-122.
[14] ZARZOSA P, GARCIA-GILABERT L, HLADUN R, et al. Targeting the Hedgehog Pathway in Rhabdomyosarcoma. Cancers (Basel). 2023;15(3):727.
[15] FENG Z, ZHU S, LI W, et al. Current approaches and strategies to identify Hedgehog signaling pathway inhibitors for cancer therapy. Eur J Med Chem. 2022;244:114867.
[16] MORSCZECK C, RECK A, BECK HC. The hedgehog-signaling pathway is repressed during the osteogenic differentiation of dental follicle cells. Mol Cell Biochem. 2017;428(1-2):79-86.
[17] ZHANG L, FU X, NI L, et al. Hedgehog Signaling Controls Bone Homeostasis by Regulating Osteogenic/Adipogenic Fate of Skeletal Stem/Progenitor Cells in Mice. J Bone Miner Res. 2022; 37(3):559-576.
[18] HU Z, CHEN B, ZHAO Q. Hedgehog signaling regulates osteoblast differentiation in zebrafish larvae through modulation of autophagy. Biol Open. 2019;8(5):bio040840.
[19] NIRALA BK, YAMAMICHI T, YUSTEIN JT. Deciphering the Signaling Mechanisms of Osteosarcoma Tumorigenesis. Int J Mol Sci. 2023;24(14):11367.
[20] LU W, ZHENG C, ZHANG H, et al. Hedgehog signaling regulates bone homeostasis through orchestrating osteoclast differentiation and osteoclast-osteoblast coupling. Cell Mol Life Sci. 2023;80(6):171.
[21] 杨仁慧, 王怡璇, 甘胜露, 等. 基于Hedgehog信号通路探讨Shh和Ihh蛋白在原发性骨质疏松症患者血清中的表达[J]. 贵州中医药大学学报,2024,46(1):82-86.
[22] FAN W, MENG Y, ZHANG J, et al. To investigate the mechanism of Yiwei Decoction in the treatment of premature ovarian insufficiency-related osteoporosis using transcriptomics, network pharmacology and molecular docking techniques. Sci Rep. 2023;13(1):19016.
[23] LIN FX, DU SX, LIU DZ, et al. Naringin promotes osteogenic differentiation of bone marrow stromal cells by up-regulating Foxc2 expression via the IHH signaling pathway. Am J Transl Res. 2016;8(11): 5098-5107.
[24] ZHANG C, LI H, LI J, et al. Oxidative stress: A common pathological state in a high-risk population for osteoporosis. Biomed Pharmacother. 2023;163:114834.
[25] WANG B, VASHISHTH D. Advanced glycation and glycoxidation end products in bone. Bone. 2023; 176:116880.
[26] 文晓晨, 刘若实, 朱克, 等. 补肾健脾法调控自噬相关PINK1/Parkin信号通路治疗糖尿病性骨质疏松症[J]. 中国中医基础医学杂志,2025, 31(4):649-654.
[27] GARG C, KHAN H, KAUR A, et al. Therapeutic implications of sonic hedgehog pathway in metabolic disorders: Novel target for effective treatment. Pharmacol Res. 2022;179:106194.
[28] BRAUNE J, WEYER U, MATZ-SOJA M, et al. Hedgehog signalling in myeloid cells impacts on body weight, adipose tissue inflammation and glucose metabolism. Diabetologia. 2017;60(5):889-899.
[29] KOMORI T. Regulation of Skeletal Development and Maintenance by Runx2 and Sp7. Int J Mol Sci. 2024;25(18):10102.
[30] ZHU S, CHEN W, MASSON A, et al. Cell signaling and transcriptional regulation of osteoblast lineage commitment, differentiation, bone formation, and homeostasis. Cell Discov. 2024;10(1):71.
[31] SUN H, WANG S, YANG Z, et al. Chordin Like-1 Regulates Osteoblast and Adipocyte Differentiation Through Stabilizing Insulin-Like Growth Factor Binding Protein 3. Stem Cells. 2023;41(4):400-414.
[32] PEI Y, LIU F, ZHAO Y, et al. Role of hedgehog signaling in the pathogenesis and therapy of heterotopic ossification. Front Cell Dev Biol. 2024; 12:1454058.
[33] WANG Y, DONG Z, YANG R, et al. Inactivation of Ihh in Sp7-Expressing Cells Inhibits Osteoblast Proliferation, Differentiation, and Bone Formation, Resulting in a Dwarfism Phenotype with Severe Skeletal Dysplasia in Mice. Calcif Tissue Int. 2022; 111(5):519-534.
[34] HUO S, TANG X, CHEN W, et al. Epigenetic regulations of cellular senescence in osteoporosis. Ageing Res Rev. 2024;99:102235.
[35] ZHANG YX, SUN HL, LIANG H, et al. Dynamic and distinct histone modifications of osteogenic genes during osteogenic differentiation. J Biochem. 2015;158(6):445-457.
[36] CHI B, LIU G, XING L, et al. [Research progress of hedgehog signaling pathway in regulating bone formation and osteogenic differentiation of bone mesenchymal stem cells]. Chin J Repar Reconstr Surg. 2016;30(12):1545-1550.
[37] NAN K, ZHANG Y, ZHANG X, et al. Exosomes from miRNA-378-modified adipose-derived stem cells prevent glucocorticoid-induced osteonecrosis of the femoral head by enhancing angiogenesis and osteogenesis via targeting miR-378 negatively regulated suppressor of fused (Sufu). Stem Cell Res Ther. 2021;12(1):331.
[38] GAO X, ZHOU J, WANG J, et al. Mechanism of exosomal miR-155 derived from bone marrow mesenchymal stem cells on stemness maintenance and drug resistance in myeloma cells. J Orthop Surg Res. 2021;16(1):637.
[39] JANKAUSKAS SS, GAMBARDELLA J, SARDU C, et al. Functional Role of miR-155 in the Pathogenesis of Diabetes Mellitus and Its Complications. Noncoding RNA. 2021;7(3):39.
[40] FONTANA F, GIANNITTI G, MARCHESI S, et al. The PI3K/Akt Pathway and Glucose Metabolism: A Dangerous Liaison in Cancer. Int J Biol Sci. 2024; 20(8):3113-3125.
[41] BENCHOULA K, PARHAR IS, WONG EH. The crosstalk of hedgehog, PI3K and Wnt pathways in diabetes. Arch Biochem Biophys. 2021;698:108743.
[42] LI M, WANG Y, WU X, et al. KIAA0753 enhances osteoblast differentiation suppressed by diabetes. J Cell Mol Med. 2024;28(17):e70035.
[43] HERMAN R, KRAVOS NA, JENSTERLE M, et al. Metformin and Insulin Resistance: A Review of the Underlying Mechanisms behind Changes in GLUT4-Mediated Glucose Transport. Int J Mol Sci. 2022;23(3):1264.
[44] ZHANG L, ZHOU X, CHEN H, et al. Mulberry extract ameliorates T2DM-related symptoms via AMPK pathway in STZ-HFD-induced C57BL/6J mice. J Ethnopharmacol. 2023;313:116475.
[45] MERA P, FERRON M, MOSIALOU I. Regulation of Energy Metabolism by Bone-Derived Hormones. Cold Spring Harb Perspect Med. 2018; 8(6):a031666.
[46] ZHANG X, CHENG Q, WANG Y, et al. Hedgehog signaling in bone regulates whole-body energy metabolism through a bone-adipose endocrine relay mediated by PTHrP and adiponectin. Cell Death Differ. 2017;24(2):225-237.
[47] XIE Z, WANG F, LIN L, et al. An SGLT2 inhibitor modulates SHH expression by activating AMPK to inhibit the migration and induce the apoptosis of cervical carcinoma cells. Cancer Lett. 2020;495: 200-210.
[48] SHEN CY, LU CH, WU CH, et al. The Development of Maillard Reaction, and Advanced Glycation End Product (AGE)-Receptor for AGE (RAGE) Signaling Inhibitors as Novel Therapeutic Strategies for Patients with AGE-Related Diseases. Molecules. 2020;25(23):5591.
[49] PASSARELLI M, MACHADO UFF. AGEs-Induced and Endoplasmic Reticulum Stress/Inflammation-Mediated Regulation of GLUT4 Expression and Atherogenesis in Diabetes Mellitus. Cells. 2021; 11(1):104.
[50] DEWANJEE S, DAS S, DAS AK, et al. Molecular mechanism of diabetic neuropathy and its pharmacotherapeutic targets. Eur J Pharmacol. 2018;833:472-523.
[51] NIU C, CHEN Z, KIM KT, et al. Metformin alleviates hyperglycemia-induced endothelial impairment by downregulating autophagy via the Hedgehog pathway. Autophagy. 2019;15(5):843-870.
[52] KHALID M, PETROIANU G, ADEM A. Advanced Glycation End Products and Diabetes Mellitus: Mechanisms and Perspectives. Biomolecules. 2022;12(4):542.
[53] GASIOROWSKI K, BROKOS B, ECHEVERRIA V, et al. RAGE-TLR Crosstalk Sustains Chronic Inflammation in Neurodegeneration. Mol Neurobiol. 2018;55(2): 1463-1476.
[54] DU H, MA Y, WANG X, et al. Advanced glycation end products induce skeletal muscle atrophy and insulin resistance via activating ROS-mediated ER stress PERK/FOXO1 signaling. Am J Physiol Endocrinol Metab. 2023;324(3):E279-E287.
[55] WYSHAM C, SHUBROOK J. Beta-cell failure in type 2 diabetes: mechanisms, markers, and clinical implications. Postgrad Med. 2020;132(8):676-686.
[56] GE W, JIE J, YAO J, et al. Advanced glycation end products promote osteoporosis by inducing ferroptosis in osteoblasts. Mol Med Rep. 2022; 25(4):140.
[57] DING X, HU Y, LUO D, et al. [Effects of advanced glycation end products on osteoclasts at different stages of differentiation]. J South Med Univ. 2020; 40(4):573-579.
[58] MAIESE K. Cornerstone Cellular Pathways for Metabolic Disorders and Diabetes Mellitus: Non-Coding RNAs, Wnt Signaling, and AMPK. Cells. 2023;12(22):2595.
[59] RONG X, KOU Y, ZHANG Y, et al. ED-71 Prevents Glucocorticoid-Induced Osteoporosis by Regulating Osteoblast Differentiation via Notch and Wnt/β-Catenin Pathways. Drug Des Devel Ther. 2022;16:3929-3946.
[60] KOMORI T. Bone development by Hedgehog and Wnt signaling, Runx2, and Sp7. J Bone Miner Metab. 2025;43(1):33-38.
[61] XUE C, CHU Q, SHI Q, et al. Wnt signaling pathways in biology and disease: mechanisms and therapeutic advances. Signal Transduct Target Ther. 2025;10(1)106.
[62] LI SY, XUE ST, LI ZR. Osteoporosis: Emerging targets on the classical signaling pathways of bone formation. Eur J Pharmacol. 2024;973:176574.
[63] LI J, YAN R, SHI S, et al. Recent progress and application of the tetrahedral framework nucleic acid materials on drug delivery. Expert Opin Drug Deliv. 2023;20(11):1511-1530.
[64] ZHAO Y, LI S, FENG M, et al. Effects of Puerarin-Loaded Tetrahedral Framework Nucleic Acids on Osteonecrosis of the Femoral Head. Small. 2023; 19(41):e2302326.
[65] LI Y, TANG Y, SHI S, et al. Tetrahedral Framework Nucleic Acids Ameliorate Insulin Resistance in Type 2 Diabetes Mellitus via the PI3K/Akt Pathway. ACS Appl Mater Interfaces. 2021;13(34):40354-40364.
[66] DENG TT, DING WY, LU XX, et al. Pharmacological and mechanistic aspects of quercetin in osteoporosis. Front Pharmacol. 2024;15:1338951.
[67] DHANYA R. Quercetin for managing type 2 diabetes and its complications, an insight into multitarget therapy. Biomed Pharmacother. 2022; 146:112560.
[68] CHEN Y, WEI Z, SHI H, et al. BushenHuoxue formula promotes osteogenic differentiation via affecting Hedgehog signaling pathway in bone marrow stem cells to improve osteoporosis symptoms. PLoS One. 2023;18(11):e0289912.
[69] CUI Z, ZHAO X, AMEVOR FK, et al. Therapeutic application of quercetin in aging-related diseases: SIRT1 as a potential mechanism. Front Immunol. 2022;13:943321.
[70] WANG N, WANG L, YANG J, et al. Quercetin promotes osteogenic differentiation and antioxidant responses of mouse bone mesenchymal stem cells through activation of the AMPK/SIRT1 signaling pathway. Phytother Res. 2021;35(5):2639-2650.
[71] 徐嵩昊, 董德龙. 有氧与抗阻运动对老年人慢性病的影响[J]. 中国老年学杂志,2025,45(11): 2798-2802.
[72] 代新宇, 李斌, 靳丹, 等. 运动通过调节骨自噬途径防治骨质疏松症的作用及可能机制 [J]. 生物化学与生物物理进展,2024,51(7):1589-1603.
[73] 张帅军, 张锦, 杜旭辉, 等. 跑台运动对切除卵巢大鼠骨质疏松症的保护机制[J]. 中国骨质疏松杂志,2024,30(6):824-828.
[74] ZHANG M, LI Y, LIU L, et al. The effects on type 2 diabetes mellitus mouse femoral bone achieved by anti-osteoporosis exercise interventions. Front Endocrinol (Lausanne). 2022;13:914872.
[75] 文武, 侯铁奇. 不同运动强度对去势大鼠骨质疏松软骨形态的影响[J]. 中国骨质疏松杂志, 2020,26(6):876-880.
[76] 侯慧卿, 王晓曦, 叶天鸣, 等. 八段锦对中年人群血脂、血糖及血尿酸水平的影响[J]. 现代中西医结合杂志,2025,34(5):685-689.
[77] 邵霞, 王宁, 王花欣, 等. 中医导引术在骨质疏松症中的临床应用概述[J]. 山东中医药大学学报,2025,49(2):249-255.
[78] 董子萱, 马占科. Piezo1信号通路在运动干预改善2型糖尿病中的作用及其机制[J]. 生物化学与生物物理进展,2025,52(2):290-298.
[79] ZHANG J, TAM WWS, HOUNSRI K, et al. Effectiveness of Combined Aerobic and Resistance Exercise on Cognition, Metabolic Health, Physical Function, and Health-related Quality of Life in Middle-aged and Older Adults With Type 2 Diabetes Mellitus: A Systematic Review and Meta-analysis. Arch Phys Med Rehabil. 2024;105(8):1585-1599.
[80] 王惠国, 杨高源, 谢仙燕, 等. 运动诱导的代谢重塑对免疫功能的调控作用[J]. 生物化学与生物物理进展,2025,52(6):1574-1586.
[81] STOCKS B, ZIERATH JR. Post-translational Modifications: The Signals at the Intersection of Exercise, Glucose Uptake, and Insulin Sensitivity. Endocr Rev. 2022;43(4):654-677.
|