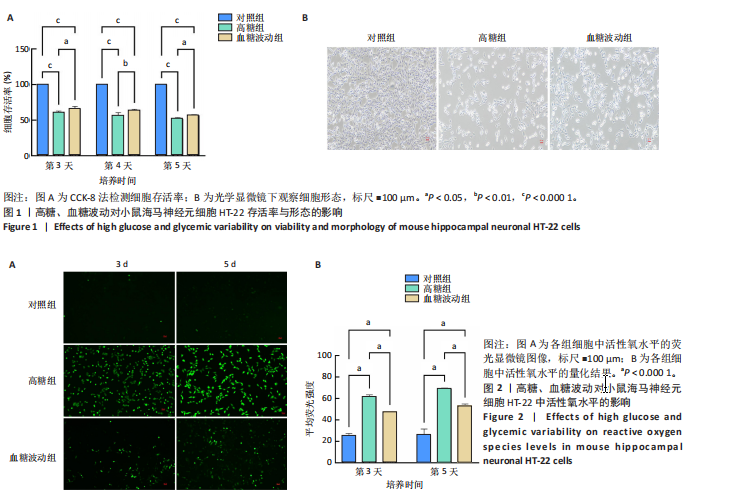
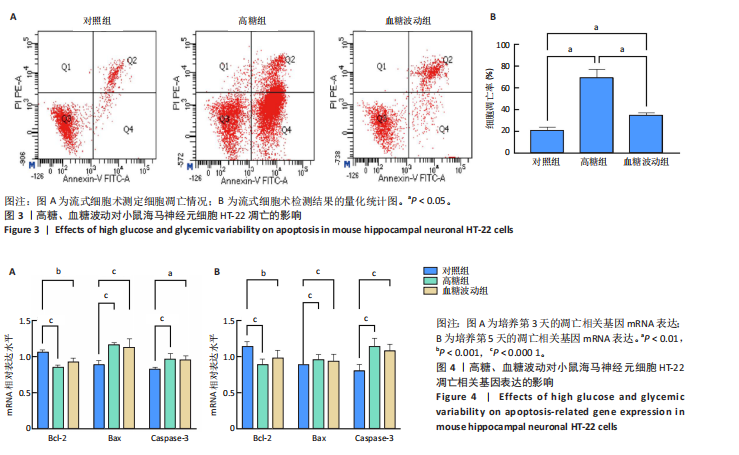
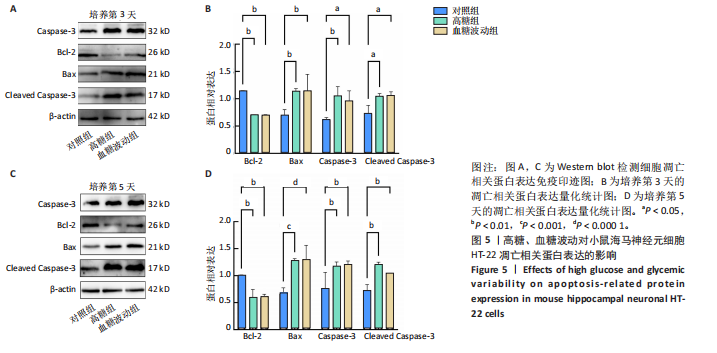
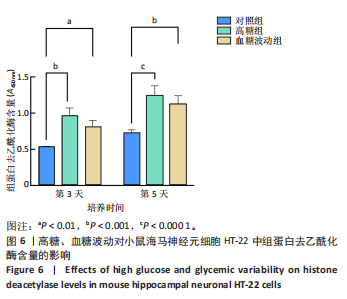
[1] HOSSAIN MJ, AL-MAMUN M, ISLAM MR. Diabetes mellitus, the fastest growing global public health concern: Early detection should be focused. Health Sci Rep. 2024;7(3):e2004.
[2] TAO S, YANG D, ZHANG L, et al. Knowledge domain and emerging trends in diabetic cardiomyopathy: A scientometric review based on CiteSpace analysis. Front Cardiovasc Med. 2022;9:891428.
[3] LUO Y, ZHU J, HU Z, et al. Progress in the Pathogenesis of Diabetic Encephalopathy: The Key Role of Neuroinflammation. Diabetes Metab Res Rev. 2024;40(6):e3841.
[4] LI Y, ZHANG H, LONG W, et al. Inhibition of NLRP3 and Golph3 ameliorates diabetes-induced neuroinflammation in vitro and in vivo. Aging (Albany NY). 2022;14(21):8745-8762.
[5] MA S, BI W, LIU X, et al. Single-Cell Sequencing Analysis of the db/db Mouse Hippocampus Reveals Cell-Type-Specific Insights Into the Pathobiology of Diabetes-Associated Cognitive Dysfunction. Front Endocrinol (Lausanne). 2022;13:891039.
[6] CAI J, CAI M, XIA W, et al. Explore the Mechanism of β-Asarone on Improving Cognitive Dysfunction in Rats with Diabetic Encephalopathy. J Alzheimers Dis Rep. 2022;6(1):195-206.
[7] XU S, GAO Z, JIANG L, et al. High glucose- or AGE-induced oxidative stress inhibits hippocampal neuronal mitophagy through the Keap1-Nrf2-PHB2 pathway in diabetic encephalopathy. Sci Rep. 2024;14(1):24044.
[8] LU M, YE X, DENG X, et al. Inhibition of LCP2 in T cells alleviated apoptosis and oxidative stress via PD-1/PD-L1 in diabetic retinopathy. Int Immunopharmacol. 2025;163:115240.
[9] YANG T, FENG Q, SHAO M, et al. The role of metabolic memory in diabetic kidney disease: identification of key genes and therapeutic targets. Front Pharmacol. 2024;15:1379821.
[10] DONG H, SUN Y, NIE L, et al. Metabolic memory: mechanisms and diseases. Signal Transduct Target Ther. 2024;9(1):38.
[11] TESTA R, BONFIGLI AR, PRATTICHIZZO F, et al. The “Metabolic Memory” Theory and the Early Treatment of Hyperglycemia in Prevention of Diabetic Complications. Nutrients. 2017;9(5):437.
[12] KOWLURU RA, MOHAMMAD G. Epigenetics and Mitochondrial Stability in the Metabolic Memory Phenomenon Associated with Continued Progression of Diabetic Retinopathy. Sci Rep. 2020;10(1):6655.
[13] HUANG L, PAN Y, ZHOU K, et al. Correlation Between Glycemic Variability and Diabetic Complications: A Narrative Review. Int J Gen Med. 2023;16:3083-3094.
[14] DANSINGER ML, GLEASON JA, MADDALENA J, et al. Lifestyle Modification in Prediabetes and Diabetes: A Large Population Analysis. Nutrients. 2025; 17(8):1333.
[15] FONSECA LM, HAWKS ZW, BEERI MS, et al. Cognitive vulnerability to glucose fluctuations: A digital phenotype of neurodegeneration. Alzheimers Dement. 2025;21(2):e70001.
[16] LAZAR S, REUREAN-PINTILEI DV, IONITA I, et al. Glycemic Variability and Its Association with Traditional Glycemic Control Biomarkers in Patients with Type 1 Diabetes: A Cross-Sectional, Multicenter Study. J Clin Med. 2025;14(7):2434.
[17] WANG H, DENG J, CHEN L, et al. Acute glucose fluctuation induces inflammation and neurons apoptosis in hippocampal tissues of diabetic rats. J Cell Biochem. 2021;122(9):1239-1247.
[18] YANG W, SI SC, LUO HY, et al. Cognitive impairment and hippocampal degeneration in aged rat models of type 2 diabetes with induced glycemic fluctuation: A pilot study. Brain Res. 2025;1850:149452.
[19] CHEN J, HUANG W, LIANG N. Blood glucose fluctuation and in-hospital mortality among patients with acute myocardial infarction: eICU collaborative research database. PLoS One. 2024;19(4):e0300323.
[20] LI W, SONG C, LI X, et al. Procalcitonin contributes to hippocampal neuronal damage and impairment of LTP: implications for cognitive dysfunction in LPS-induced neuroinflammation rat model. Arch Microbiol. 2025;207(6):127.
[21] CHU J, LIU CX, SONG R, et al. Ferrostatin-1 protects HT-22 cells from oxidative toxicity. Neural Regen Res. 2020;15(3):528-536.
[22] 段云峰,许永劼,杨婷婷,等.高糖诱导HT-22小鼠海马神经元代谢记忆细胞模型的构建及影响[J].天津医药,2024,52(1):44-50.
[23] SONTHALIA M, ROY BS, CHANDRAWANSHI D, et al. Histone deacetylase inhibitors as antidiabetic agents: Advances and opportunities. Eur J Pharmacol. 2022;935:175328.
[24] SPIRIN P, VEDERNIKOVA V, VOLKAVA T, et al. New and Effective Inhibitor of Class I HDACs, Eimbinostat, Reduces the Growth of Hematologic Cancer Cells and Triggers Apoptosis. Pharmaceutics. 2025;17(4):416.
[25] MCCLARTY BM, RODRIGUEZ G, DONG H. Class 1 histone deacetylases differentially modulate memory and synaptic genes in a spatial and temporal manner in aged and APP/PS1 mice. Brain Res. 2024;1837:148951.
[26] CAO W, JI Z, ZHU S, et al. Bioinformatic identification and experiment validation reveal 6 hub genes, promising diagnostic and therapeutic targets for Alzheimer’s disease. BMC Med Genomics. 2024;17(1):6.
[27] 许永劼,许雯,陈钢,等.2种高糖诱导海马神经元模型应用及优势比较[J].中国比较医学杂志,2021,31(8):1-8.
[28] LI Y, WANG G, ZHANG Y, et al. Biological effects of exosomes derived from 2D and 3D culture adipose stem cells on JEC Cell proliferation and migration. Front Bioeng Biotechnol. 2025;13:1541150.
[29] TELFORD WG, KOMORIYA A, PACKARD BZ, et al. Multiparametric analysis of apoptosis by flow cytometry. Methods Mol Biol. 2011;699:203-227.
[30] LI M, WANG Y, KE Q, et al. The inhibitory and anti-inflammatory effects of TMP269 on peste des petits ruminants virus replication. Virulence. 2025;16(1):2495838.
[31] ALPEROVICH NY, VASILYEVA OB, SCHAFFTER SW. Prevention of ribozyme catalysis through cDNA synthesis enables accurate RT-qPCR measurements of context-dependent ribozyme activity. RNA. 2025;31(5):633-645.
[32] LIN X, WANG S, LIU X, et al. Enhanced sensitivity in PCSK9 detection using binding-induced DNA walker-triggered Argonaute protein-based DNA circuit. Mikrochim Acta. 2025;192(8):509.
[33] LIU D, ZHENG Z, CHEN Z, et al. Ferroptosis in Müller cells under hyperglycemia: mechanisms and therapeutic implications for diabetic retinopathy-associated optic neuroinflammation. Int Ophthalmol. 2025; 45(1):302.
[34] HONGMEI H, MAOJUN Y, TING LI, et al. Neferine inhibits the progression of diabetic nephropathy by modulating the miR-17-5p/nuclear factor E2-related factor 2 axis. J Tradit Chin Med. 2024;44(1):44-53.
[35] DEN BRABER N, VOLLENBROEK-HUTTEN MMR, TEUNISSEN SEM, et al. The Contribution of Postprandial Glucose Levels to Hyperglycemia in Type 2 Diabetes Calculated from Continuous Glucose Monitoring Data: Real World Evidence from the DIALECT-2 Cohort. Nutrients. 2024;16(20):3557.
[36] KO J, JANG S, KWON W, et al. Protective Effect of GIP against Monosodium Glutamate-Induced Ferroptosis in Mouse Hippocampal HT-22 Cells through the MAPK Signaling Pathway. Antioxidants (Basel). 2022;11(2):189.
[37] SUN Y, LU Y, SAREDY J, et al. ROS systems are a new integrated network for sensing homeostasis and alarming stresses in organelle metabolic processes. Redox Biol. 2020;37:101696.
[38] GONZÁLEZ P, LOZANO P, ROS G, et al. Hyperglycemia and Oxidative Stress: An Integral, Updated and Critical Overview of Their Metabolic Interconnections. Int J Mol Sci. 2023;24(11):9352.
[39] KATO A, NIHEI W, YAKO H, et al. Imeglimin improves hyperglycemia and hypoglycemia-induced cell death and mitochondrial dysfunction in immortalized adult mouse Schwann IMS32 cells. J Diabetes Investig. 2025. doi: 10.1111/jdi.70092.
[40] DU W, SHI G, SHAN CM, et al. Mechanisms of chromatin-based epigenetic inheritance. Sci China Life Sci. 2022;65(11):2162-2190.
[41] MAKKAR R, BEHL T, ARORA S. Role of HDAC inhibitors in diabetes mellitus. Curr Res Transl Med. 2020;68(2):45-50.
[42] JIN T, WANG Z, FAN F, et al. HDAC1 Promotes Mitochondrial Pathway Apoptosis and Inhibits the Endoplasmic Reticulum Stress Response in High Glucose-Treated Schwann Cells via Decreased U4 Spliceosomal RNA. Neurochem Res. 2024;49(10):2699-2724.
[43] COLUSSI C, ACETO G, RIPOLI C, et al. Cytoplasmic HDAC4 recovers synaptic function in the 3×Tg mouse model of Alzheimer’s disease. Neuropathol Appl Neurobiol. 2023;49(1):e12861.
[44] KUMAR KK, ABURAWI EH, LJUBISAVLJEVIC M, et al. Exploring histone deacetylases in type 2 diabetes mellitus: pathophysiological insights and therapeutic avenues. Clin Epigenetics. 2024;16(1):78.
[45] LI M, SONG J, NIU X, MO F, et al. Baicalein ameliorates cognitive decline induced by chronic cerebral hypoperfusion through the SIRT1-mediated Notch1 pathway to improve angiogenesis and suppress neuroinflammation. Front Aging Neurosci. 2025;17:1521353.
[46] ZHAO Y, ZHANG AP, BAO BY, et al. Sirt1 protects lupus nephritis by inhibiting the NLRP3 signaling pathway in human glomerular mesangial cells. Open Life Sci. 2025;20(1):20221038.
[47] LI Y, YISHAJIANG S, CHEN Y, et al. TRPC5-mediated NLRP3 inflammasome activation contributes to myocardial cell pyroptosis in chronic intermittent hypoxia rats. Acta Cardiol. 2024;79(7):796-804.
[48] 许永劼,陈钢,林海容,等.慢病毒干扰HDAC4对HT-22小鼠海马神经元凋亡的影响及稳转株建立[J].中国老年学杂志,2024,44(16):3919-3923.
[49] 王安迪.妊娠期糖尿病中LPS通过HDAC1影响SIRT1相关胰岛素通路的机制研究[D].重庆:重庆医科大学,2022.
[50] HUANG X, SHI Y, CHEN H, et al. Isoliquiritigenin prevents hyperglycemia-induced renal injuries by inhibiting inflammation and oxidative stress via SIRT1-dependent mechanism. Cell Death Dis. 2020;11(12):1040.
[51] HUANG Z, LI Y, XIE Y, et al. Jiawei Erzhiwan Ameliorates Androgenetic Alopecia by Regulating the SIRT1/JNK/p38 MAPK Pathway. Drug Des Devel Ther. 2025;19:2393-2409.
[52] XU Y, LI H, CHEN G, et al. Radix polygoni multiflori protects against hippocampal neuronal apoptosis in diabetic encephalopathy by inhibiting the HDAC4/JNK pathway. Biomed Pharmacother. 2022;153:113427.
[53] WU LD, LIU Y, LI F, et al. Glucose fluctuation promotes cardiomyocyte apoptosis by triggering endoplasmic reticulum (ER) stress signaling pathway in vivo and in vitro. Bioengineered. 2022;13(5):13739-13751.
|