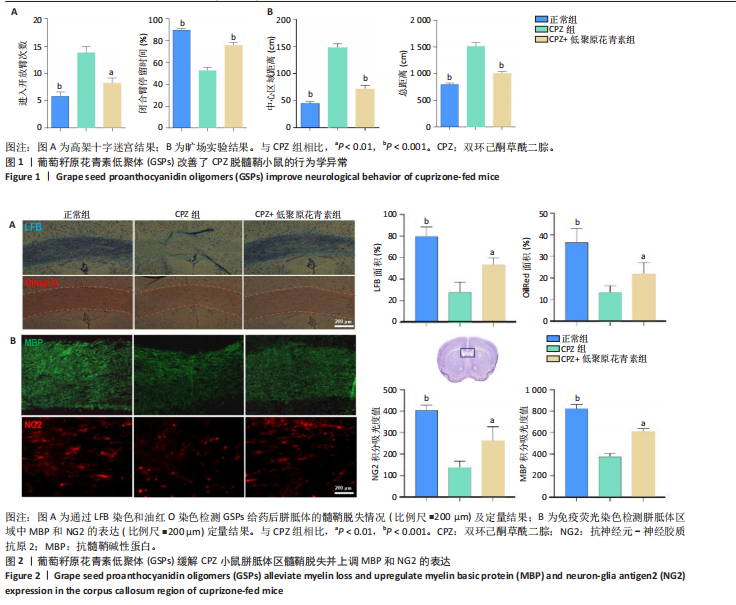
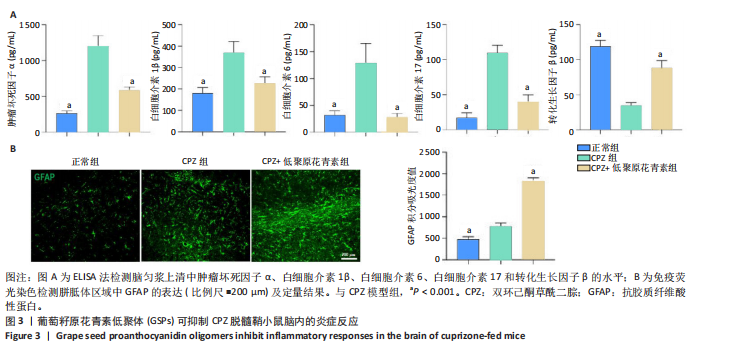
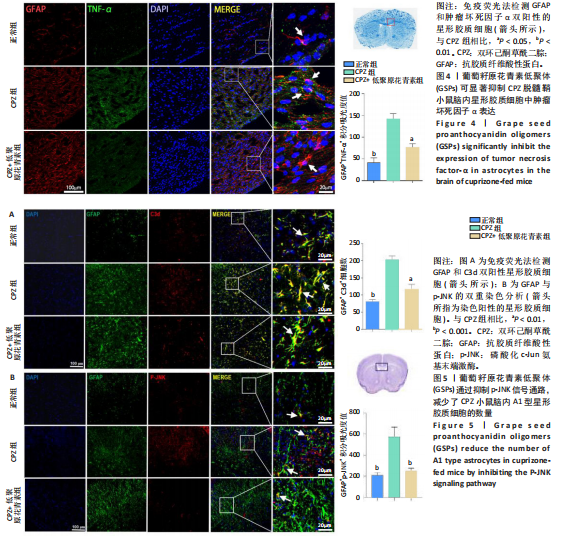
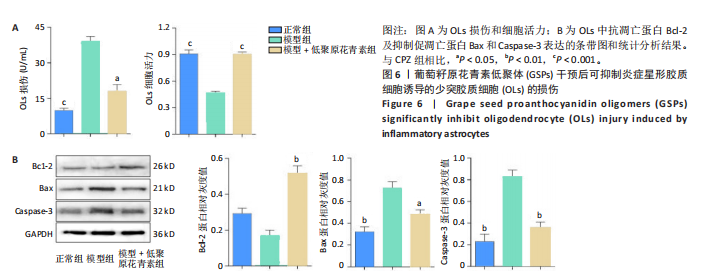
[1] HAKI M, AL-BIATI HA, AL-TAMEEMI ZS, et al. Review of multiple sclerosis: Epidemiology, etiology, pathophysiology, and treatment. Medicine (Baltimore). 2024;103(8):e37297.
[2] WARD M, GOLDMAN MD. Epidemiology and Pathophysiology of Multiple Sclerosis. Continuum (Minneap Minn). 2022;28(4):988-1005.
[3] 王柳清, 朱峰, 薛群, 等. 多发性硬化疾病修饰治疗药物的临床应用现状[J].临床药物治疗杂志,2023,21(10):6-11.
[4] LIU R, DU S, ZHAO L, et al.Autoreactive lymphocytes in multiple sclerosis: Pathogenesis and treatment target. Front Immunol. 2022;13:996469.
[5] ABSINTA M, MARIC D, GHARAGOZLOO M, et al. A lymphocyte-microglia-astrocyte axis in chronic active multiple sclerosis. Nature. 2021;597(7878): 709-714.
[6] ZHANG C, LIU W, WANG L, et al. Prevalence and Burden of Multiple Sclerosis in China, 1990-2019: Findings From the Global Burden of Disease Study 2019. Neurology. 2024;102(11):e209351.
[7] WANG M, LIU C, ZOU M, et al. Recent progress in epidemiology, clinical features, and therapy of multiple sclerosis in China. Ther Adv Neurol Disord. 2023;16:17562864231193816.
[8] AMIN M, HERSH CM. Updates and advances in multiple sclerosis neurotherapeutics. Neurodegener Dis Manag. 2023;13(1):47-70.
[9] BAECHER-ALLAN C, KASKOW BJ, WEINER HL. Multiple sclerosis: mechanisms and immunotherapy. Neuron. 2018;97:742-768.
[10] SEN MK, MAHNS DA, COORSSEN JR, et al. The roles of microglia and astrocytes in phagocytosis and myelination: Insights from the cuprizone model of multiple sclerosis. Glia. 2022;70(7):1215-1250.
[11] ANGELINO D, CARREGOSA D, DOMENECH-COCA C, et al. 5-(Hydroxyphenyl)-γ-Valerolactone-Sulfate, a Key Microbial Metabolite of Flavan-3-ols, Is Able to Reach the Brain: Evidence from Different in Silico, In Vitro and In Vivo Experimental Models. Nutrients. 2019;11(11):2678.
[12] GAO WL, LI XH, DUN XP, et al. Grape Seed Proanthocyanidin Extract Ameliorates Streptozotocin-induced Cognitive and Synaptic Plasticity Deficits by Inhibiting Oxidative Stress and Preserving AKT and ERK Activities. Curr Med Sci. 2020;40(3):434-443.
[13] WANG Q, WANG J, Y ZC, et al. Therapeutic effect of oligomeric proanthocyanidin in cuprizone-induced demyelination. Exp Physiol. 2019; 104(6):876-886.
[14] 王青, 杨智超, 董艺薇, 等. 葡萄籽原花青素低聚体抑制A1型星形胶质细胞极化机制[J].中国药理学通报,2023,39(1):77-82.
[15] TORKILDSEN O, BRUNBORG LA, MYHR KM, et al. The cuprizone model for demyelination. Acta Neurol Scand Suppl. 2008;188:72-76.
[16] YOO DY, KIM W, YOO KY, et al. Grape seed extract enhances neurogenesis in the hippocampal dentate gyrus in C57BL/6 mice. Phytother Res. 2011; 25(5):668-674.
[17] GONG XD, XU LZ, FANG X, et al. Protective effects of grape seed procyanidin on isoflurane-induced cognitive impairment inmice. Pharm Biol. 2020;58(1): 200-207.
[18] 王芸, 李永德, 彭超, 等. CPZ诱导精神分裂症样模型小鼠海马脱髓鞘改变与行为学变化及其相关性的研究[J]. 重庆医科大学学报,2014, 39(2):129-136.
[19] LIU HX, ZHAI JG, WANG B, et al. Olig2 Silence Ameliorates Cuprizone-Induced Schizophrenia-Like Symptoms in Mice. Med Sci Monit. 2017; 3:4834-4840.
[20] 陈玉宁, 蒋颖, 廖翔宇, 等. 补气活血合剂干预脑缺血再灌注模型大鼠相关因子及自噬蛋白的表达 [J]. 中国组织工程研究,2024,29(6):1252-1258.
[21] CONG H, LIANG M, WANG Y, et al. Icariin ameliorates the cuprizone-induced acute brain demyelination and modulates the number of oligodendrocytes, microglia and astrocytes in the brain of C57BL/6J mice. Brain Res Bull. 2021; 175:37-47.
[22] 谢媛, 梅晓峰, 陶柳颖, 等. 香烟烟雾联合Poly I:C诱导COPD模型小鼠气道上皮屏障损伤的机制 [J].中国病理生理杂志,2024,40(7):1222-1229.
[23] KERKERING J, MUINJONOV B, ROSIEWICZ KS, et al. iPSC-derived reactive astrocytes from patients with multiple sclerosis protect cocultured neurons in inflammatory conditions. J Clin Invest. 2023;133(13):e164637.
[24] SEN MK, MAHNS DA, COORSSEN JR, et al. The roles of microglia and astrocytes in phagocytosis and myelination: Insights from the cuprizone model of multiple sclerosis. Glia. 2022;70(7):1215-1250.
[25] SHI J, XIAO Y, ZHANG N, et al. HMGB1 from Astrocytes Promotes EAE by Influencing the Immune Cell Infiltration-Associated Functions of BMECs in Mice. Neurosci Bull. 2022;38(11):1303-1314.
[26] ABULABAN AA, AL-KURAISHY HM, AL-GAREEB AI, et al. The janus face of astrocytes in multiple sclerosis: Balancing protection and pathology. Brain Res Bull. 2025;226:111356.
[27] LEE HG, RONE JM, LI Z, et al. Disease-associated astrocyte epigenetic memory promotes CNS pathology. Nature. 2024;627(8005):865-872.
[28] ZHA Z, LIU YJ, LIU SS, et al. Bu Shen Yi Sui Capsule Promotes Myelin Repair by Modulating the Transformation of A1/A2 Reactive Astrocytes In Vivo and In Vitro. Oxid Med Cell Longev. 2022;2022:3800004.
[29] ABSINTA M, MARIC D, GHARAGOZLOO M, et al. A lymphocyte-microglia-astrocyte axis in chronic active multiple sclerosis. Nature. 2021;597(7878): 709-714.
[30] TIZIANA V, ANDRONIKI VK, MARIELLA E, et al. Mesenchymal stem cells instruct a beneficial phenotype in reactive astrocytes. Glia. 2021;69(5):1204-1215.
[31] SUTTER PA, WILLIS CM, MENORET A, et al. Astrocytic TIMP-1 regulates production of Anastellin, an inhibitor of oligodendrocyte differentiation and FTY720 responses. Proc Natl Acad Sci U S A. 2024; 121(5):e2306816121.
[32] HE Y, AN J, YIN JJ, et al. Ethyl Pyruvate-Derived Transdifferentiation of Astrocytes to Oligodendrogenesis in Cuprizone-Induced Demyelinating Model. Neurotherapeutics. 2021;18(1):488-502.
[33] CHEN C, SHU Y, YAN C, et al. Astrocyte-derived clusterin disrupts glial physiology to obstruct remyelination in mouse models of demyelinating diseases. Nat Commun. 2024;15(1):7791.
[34] LI X, DING Z, LIU K, et al. Astrocytic phagocytosis of myelin debris and reactive characteristics in vivo and in vitro. Biol Cell. 2023;115(12): e202300057.
[35] SANTIAGO-BALMASEDA A, AGUIRRE-OROZCO A, VALENZUELA-ARZETA IE, et al. Neurodegenerative Diseases: Unraveling the Heterogeneity of Astrocytes. Cells. 2024;13(11):921.
[36] SAMTLEBEN S, MINA L, YAP MC, et al. Astrocytes show increased levels of Ero1alpha in multiple sclerosis and its experimental autoimmune encephalomyelitis animal model. Eur J Neurosci. 2022;56(8):5177-5190.
[37] KEMPURAJ D, THANGAVEL R, NATTERU PA, et al. Neuroinflammation Induces Neurodegeneration. J Neurol Neurosurg Spine. 2016;1(1):1003.
[38] LUO J. TGF-beta as a Key Modulator of Astrocyte Reactivity: Disease Relevance and Therapeutic Implications. Biomedicines. 2022;10(5):1206.
[39] KHAN Z, MEHAN S, GUPTA GD, et al. Immune System Dysregulation in the Progression of Multiple Sclerosis: Molecular Insights and Therapeutic Implications. Neuroscience. 2024;548:9-26.
[40] CHENG YJ, WANG F, FENG J, et al. Prolonged myelin deficits contribute to neuron loss and functional impairments after ischaemic stroke. Brain. 2024;147(4):1294-1311.
[41] DEDONI S, SCHERMA M, CAMOGLIO C, et al. An overall view of the most common experimental models for multiple sclerosis. Neurobiol Dis. 2023; 184:106230.
[42] TORKILDSEN O, BRUNBORG LA, MYHR KM, et al. The cuprizone model for demyelination. Acta Neurol Scand Suppl. 2008;188:72-76.
[43] MI Y, QI G, VITALI F, et al. Loss of fatty acid degradation by astrocytic mitochondria triggers neuroinflammation and neurodegeneration. Nat Metab. 2023;5(3):445-465.
[44] SHINOZAKI Y, SHIBATA K, YOSHIDA K, et al. Transformation of Astrocytes to a Neuroprotective Phenotype by Microglia via P2Y(1) Receptor Downregulation. Cell Rep. 2017;19(6):1151-1164.
[45] WU L, LIU YC, HE QG, et al. PEDF-34 attenuates neurological deficit and suppresses astrocyte-dependent neuroinflammation by modulating astrocyte polarization via 67LR/JNK/STAT1 signaling pathway after subarachnoid hemorrhage in rats. J Neuroinflammation. 2024;21(1):178.
[46] LI X, DING Z, LIU K, et al. Astrocytic phagocytosis of myelin debris and reactive characteristics in vivo and in vitro. Biol Cell. 2023;115(12): e202300057.
[47] CHEN C, SHU Y, YAN C, et al. Astrocyte-derived clusterin disrupts glial physiology to obstruct remyelination in mouse models of demyelinating diseases. Nat Commun. 2024;15(1):7791.
[48] SCHIRMER L, SCHAFER DP, BARTELS T, et al. Diversity and Function of Glial Cell Types in Multiple Sclerosis. Trends Immunol. 2021;42(3):228-247.
[49] GUPTA S, SIMIC M, SAGAN SA, et al. CAR-T Cell-Mediated B-Cell Depletion in Central Nervous System Autoimmunity. Neurol Neuroimmunol Neuroinflamm. 2023;10(2):e200080.
[50] ROBINSON AP, HARP CT, NORONHA A, et al. The experimental autoimmune encephalomyelitis (EAE) model of MS: utility for understanding disease pathophysiology and treatment. Handb Clin Neurol. 2014;122:173-189.
|