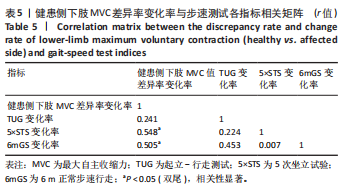
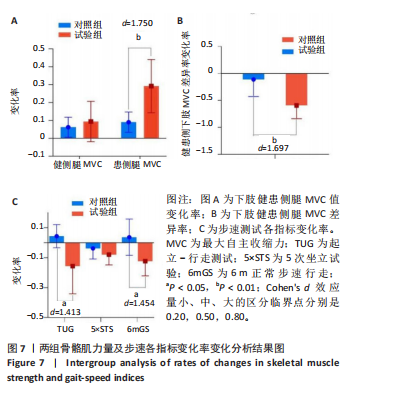
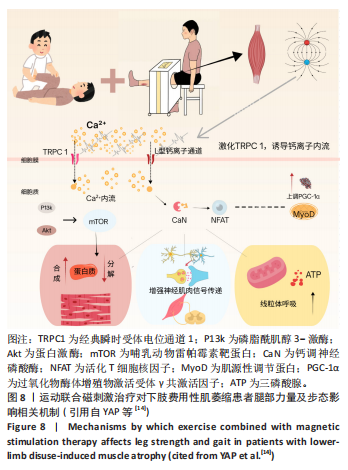
[1] DEANE CS, PIASECKI M, ATHERTON PJ. Skeletal muscle immobilisation-induced atrophy: mechanistic insights from human studies. Clin Sci. 2024;138(12):741-756.
[2] MCKENDRY J, COLETTA G, NUNES EA, et al. Mitigating disuse-induced skeletal muscle atrophy in ageing: resistance exercise as a critical countermeasure. Exp Physiol. 2024;109(10):1650-1662.
[3] 冯丽,来芹美,周公民,等.肠内营养制剂联合维生素D对老年失用性肌萎缩症患者肌肉力量及骨折事件的影响[J].浙江临床医学,2018,20(9):1559-1561.
[4] NUNES EA, STOKES T, MCKENDRY J, et al. Disuse-induced skeletal muscle atrophy in disease and nondisease states in humans: mechanisms, prevention, and recovery strategies. Am J Physiol Cell Physiol. 2022;322(6):C1068-1084.
[5] GORDON BS, KELLEHER AR, KIMBALL SR. Regulation of muscle protein synthesis and the effects of catabolic states. Int J Biochem Cell Biol. 2013;45(10):2147-2157.
[6] VALENTIM MA, BRAHMBHATT AN, TUPLING AR. Skeletal and cardiac muscle calcium transport regulation in health and disease. Biosci Rep. 2022;42(12): BSR20211997.
[7] GAO Y, ARFAT Y, WANG H,et al. Muscle atrophy induced by mechanical unloading: mechanisms and potential countermeasures. Front Physiol. 2018;9:235.
[8] 周永战,陈佩杰,郑莉芳,等.失用性肌萎缩的发生机制及治疗策略[J].中国康复医学杂志,2017,32(11):1307-1313.
[9] KOUZAKI M, MASANI K, AKIMA H, et al. Effects of 20-day bed rest with and without strength training on postural sway during quiet standing. Acta Physiol. 2007;189(3):279-292.
[10] SHINOHARA M, YOSHITAKE Y, KOUZAKI M, et al. Strength training counteracts motor performance losses during bed rest. J Appl Physiol. 2003;95(4):1485-1492.
[11] LIM JY. Therapeutic potential of eccentric exercises for age-related muscle atrophy. Integr Med Res. 2016;5(3):176-181.
[12] DESHMUKH AS. Proteomics of skeletal muscle: focus on insulin resistance and exercise biology. Proteomes. 2016;4(1):6.
[13] FRANCO-OBREGÓN A, TAI YK, WU KY, et al. The developmental implications of muscle-targeted magnetic mitohormesis: a human health and longevity perspective. Bioengineering (Basel). 2023;10(8):956.
[14] YAP JLY, TAI YK, FRÖHLICH J, et al. Ambient and supplemental magnetic fields promote myogenesis via a TRPC1‐mitochondrial axis: evidence of a magnetic mitohormetic mechanism. FASEB J. 2019;33(11):12853-12872.
[15] FRANCO-OBREGÓN A. Magnetic mitohormesis: a non-invasive therapy for inflammatory disorders? Biocell. 2022;47(2):239-244.
[16] WONG CJK, TAI YK, YAP JLY, et al. Brief exposure to directionally-specific pulsed electromagnetic fields stimulates extracellular vesicle release and is antagonized by streptomycin: a potential regenerative medicine and food industry paradigm. Biomaterials. 2022;287:121658.
[17] 史仍飞,胡莉莉,李志刚,等.雷帕霉素和LY294002对游泳训练大鼠骨骼肌生长及PI3K/Akt/mTOR信号通路的影响[J].体育科学,2011,31(7):62-66.
[18] 李伟,厉中山,白石.TRPC1钙离子通道在骨骼肌收缩功能中的作用及磁诱导激活效应在训练中的应用探索方向[J].体育科技文献通报,2023,31(2):257-261.
[19] 杨笑莹. 低频脉冲电磁场促进运动性骨骼肌肥大的效果及分子机制研究[D].西安:陕西师范大学,2016.
[20] 厉中山,白石,刘洁,等.短期低频脉冲磁场诱导经典瞬时感受器电位通道1对局部肌肉肌力提升后的保持与衰减变化轨迹[J].中国组织工程研究, 2023,27(23):3721-3727.
[21] 厉中山,包义君,刘洁,等.低频脉冲磁场诱导TRPC1改善COVID-19患者康复期下肢的肌肉无力症状[J].中国组织工程研究,2024,28(16):2605-2612.
[22] 王选强,张文洋,李阳,等.低频脉冲磁场慢性暴露对健康成年股四头肌收缩力及形态的影响[J].中国组织工程研究,2025,29(8):1634-1642.
[23] LI Z, LI W, BAI S, et al. Effects of chronic low-frequency pulsed magnetic fields exposure on the contractility and morphology of biceps brachii in healthy adults–a randomized controlled, double-blind trial. Front Med. 2025;12:1614054.
[24] AZHARUDDIN M, ZIA NU. Correlation between sit-to-stand ability, dynamic balance, gait speed, and quality of life in stroke population: a non-randomized pilot study. Bull Faculty Phys Ther. 2021. doi: 10.1186/s43161-021-00043-x.
[25] PODSIADLO D, RICHARDSON S. The timed “Up & Go”: a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc. 1991;39(2):142-148.
[26] BÅRDSTU HB, ANDERSEN V, FIMLAND MS, et al. Muscle strength is associated with physical function in community-dwelling older adults receiving home care. a cross-sectional study. Front Public Health. 2022;10:856632.
[27] KO V, NAYLOR JM, HARRIS IA, et al. The six-minute walk test is an excellent predictor of functional ambulation after total knee arthroplasty. BMC Musculoskelet Disord. 2013;14:145.
[28] JACKMAN RW, KANDARIAN SC. The molecular basis of skeletal muscle atrophy. Am J Physiol Cell Physiol. 2004;287(4):C834-843.
[29] LEI Y, GAN M, QIU Y, et al. The role of mitochondrial dynamics and mitophagy in skeletal muscle atrophy: from molecular mechanisms to therapeutic insights. Cell Mol Biol Lett. 2024;29(1):59.
[30] POWERS SK, MORTON AB, AHN B, et al. Redox control of skeletal muscle atrophy. Free Radic Biol Med. 2016;98:208-217.
[31] SANDRI M, SANDRI C, GILBERT A, et al. Foxo transcription factors induce the atrophy-related ubiquitin ligase atrogin-1 and cause skeletal muscle atrophy. Cell. 2004;117(3):399-412.
[32] BROCCA L, PELLEGRINO MA, DESAPHY JF, et al. Is oxidative stress a cause or consequence of disuse muscle atrophy in mice? A proteomic approach in hindlimb-unloaded mice. Exp Physiol. 2010;95(2):331-350.
[33] GAO Y, ARFAT Y, WANG H, et al. Muscle atrophy induced by mechanical unloading: mechanisms and potential countermeasures. Front Physiol. 2018;9:235.
[34] TREVINO MB, ZHANG X, STANDLEY RA, et al. Loss of mitochondrial energetics is associated with poor recovery of muscle function but not mass following disuse atrophy. Am J Physiol Endocrinol Metab. 2019;317(5): E899-910.
[35] BROOK MS, STOKES T, GORISSEN SHM, et al. Declines in muscle protein synthesis account for short-term muscle disuse atrophy in humans in the absence of increased muscle protein breakdown. J Cachexia Sarcopenia Muscle. 2022;13(4):2005-2016.
[36] STÄUDLE B, SEYNNES O, LAPS G, et al. Altered gastrocnemius contractile behavior in former achilles tendon rupture patients during walking. Front Physiol. 2022;13: 792576.
[37] 厉中山,王春露,刘洁,等.短期低频脉冲磁场诱导经典瞬时感受器电位通道1对肱二头肌最大自主收缩力与力量耐力的影响[J].中国组织工程研究, 2023,27(11):1796-1804.
[38] POWERS SK, WIGGS MP, DUARTE JA, et al. Mitochondrial signaling contributes to disuse muscle atrophy. Am J Physiol Endocrinol Metab. 2012;303(1):E31-39.
[39] PIETRANGELO L, MICHELUCCI A, AMBROGINI P, et al. Muscle activity prevents the uncoupling of mitochondria from Ca2+ release units induced by ageing and disuse. Arch Biochem Biophys. 2019;663:22-33.
[40] JI LL, YEO D. Mitochondrial dysregulation and muscle disuse atrophy. F1000Res. 2019;8:F1000 Faculty Rev-1621.
[41] CANNAVINO J, BROCCA L, SANDRI M, et al. PGC1-α over-expression prevents metabolic alterations and soleus muscle atrophy in hindlimb unloaded mice. J Physiol. 2014;592(20):4575-4589. doi: 10.1113/jphysiol.2014.275545.
[42] ZHANG Y, WANG Y, LU S, et al. Nicotinamide phosphoribosyltransferase-elevated NAD+ biosynthesis prevents muscle disuse atrophy by reversing mitochondrial dysfunction. J Cachexia Sarcopenia Muscle. 2023;14(2): 1003-1018.
[43] TAI Y K, NG C, PURNAMAWATI K, et al. Magnetic fields modulate metabolism and gut microbiome in correlation with pgc-1α expression: follow-up to an in vitro magnetic mitohormetic study. FASEB J. 2020;34(8):11143-11167.
[44] HANDSCHIN C, SPIEGELMAN BM. Peroxisome proliferator-activated receptor gamma coactivator 1 coactivators, energy homeostasis, and metabolism. Endocr Rev. 2006;27(7):728-735.
[45] HARGREAVES M, SPRIET LL. Skeletal muscle energy metabolism during exercise. Nat Metab. 2020;2(9):817-828.
[46] YAP JLY, TAI YK, FRÖHLICH J, et al. Ambient and supplemental magnetic fields promote myogenesis via a TRPC1‐mitochondrial axis: evidence of a magnetic mitohormetic mechanism. FASEB J. 2019;33(11):12853-12872.
[47] DINKOVA-KOSTOVA AT, ABRAMOV AY. The emerging role of Nrf2 in mitochondrial function. Free Radic Biol Med. 2015;88(Pt B):179-188.
[48] FRANCO-OBREGÓN A, TAI YK, WU KY, et al. The developmental implications of muscle-targeted magnetic mitohormesis: a human health and longevity perspective. Bioengineering (Basel). 2023;10(8):956.
[49] ROSA-CALDWELL ME, LIM S, HAYNIE WS, et al. Mitochondrial aberrations during the progression of disuse atrophy differentially affect male and female mice. J Cachexia Sarcopenia Muscle. 2021;12(6):2056-2068.
[50] SCHIAFFINO S, REGGIANI C. Fiber types in mammalian skeletal muscles. Physiol Rev. 2011;91(4):1447-1531.
[51] 邓树勋.运动生理学[M].3版.北京:高等教育出版社,2015.
[52] VENUGOBAL S, TAI YK, GOH J, et al . Brief, weekly magnetic muscle therapy improves mobility and lean body mass in older adults: a southeast Asia community case study. Aging. 2023;15(6):1768-1790.
[53] 韩东生,梁擎宇,石晓峰.抗阻运动激活Piezo1/AMPK/PGC-1α缓解小鼠废用性骨骼肌萎缩[J].中国生物化学与分子生物学报,2025,41(1):136-146.
[54] DOWLING P, GARGAN S, SWANDULLA D, et al. Fiber-type shifting in sarcopenia of old age: proteomic profiling of the contractile apparatus of skeletal muscles. Int J Mol Sci. 2023;24(3):2415.
[55] LEXELL J. Human aging, muscle mass, and fiber type composition. J Gerontol A Biol Sci Med Sci. 1995;50 Spec No:11-16.
[56] LIM JY, FRONTERA WR. Skeletal muscle aging and sarcopenia: perspectives from mechanical studies of single permeabilized muscle fibers. J Biomech. 2023; 152:111559.
[57] 白志坤,汤豪杰,黄凌凌,等.基于网络药理学探讨芒果叶干预肌肉减少症的活性成分和分子机制[J].东北农业大学学报,2023,54(7):49-57.
[58] DEANE CS, PIASECKI M, ATHERTON PJ. Skeletal muscle immobilisation-induced atrophy: mechanistic insights from human studies. Clin Sci. 2024; 138(12):741-756.
[59] HOWARD EE, PASIAKOS SM, FUSSELL MA, et al. Skeletal muscle disuse atrophy and the rehabilitative role of protein in recovery from musculoskeletal injury. Adv Nutr. 2020;11(4):989-1001.
[60] SAYED RKA, HIBBERT JE, JORGENSON KW, et al. The structural adaptations that mediate disuse-induced atrophy of skeletal muscle. Cells. 2023;12(24):2811.
[61] MAIULLARI S, CICIRELLI A, PICERNO A, et al. Pulsed electromagnetic fields induce skeletal muscle cell repair by sustaining the expression of proteins involved in the response to cellular damage and oxidative stress. Int J Mol Sci. 2023;24(23):16631.
[62] BI J, JING H, ZHOU C, et al. Regulation of skeletal myogenesis in C2C12 cells through modulation of Pax7, MyoD, and myogenin via different low-frequency electromagnetic field energies. Technol Health Care. 2022;30(S1):371-382.
[63] MIRZOEV TM. Skeletal muscle recovery from disuse atrophy: protein turnover signaling and strategies for accelerating muscle regrowth. Int J Mol Sci. 2020; 21(21):7940.
[64] WINGERTZAHN MA, OCHS RS. Control of calcium in skeletal muscle excitation-contraction coupling: implications for malignant hyperthermia. Mol Genet Metab. 1998;65(2):113-120.
[65] AGRAWAL A, SURYAKUMAR G, RATHOR R. Role of defective Ca2+ signaling in skeletal muscle weakness: pharmacological implications. J Cell Commun Signal. 2018;12(4):645-659.
[66] GEHLERT S, BLOCH W, SUHR F. Ca2+-dependent regulations and signaling in skeletal muscle: from electro-mechanical coupling to adaptation. Int J Mol Sci. 2015;16(1):1066-1095.
[67] LAMBOLEY CR, WYCKELSMA VL, PERRY BD, et al. Effect of 23-day muscle disuse on sarcoplasmic reticulum Ca2+ properties and contractility in human type I and type II skeletal muscle fibers. J Appl Physiol. 2016;121(2):483-492.
[68] VARANI K, VINCENZI F, PASQUINI S, et al. Pulsed electromagnetic field stimulation in osteogenesis and chondrogenesis: signaling pathways and therapeutic implications. Int J Mol Sci. 2021;22(2):809.
[69] LIPSCOMBE D, HELTON TD, XU W. L-type calcium channels: the low down. J Neurophysiol. 2004;92(5):2633-2641.
[70] KUO IY, EHRLICH BE. Signaling in muscle contraction. Cold Spring Harb Perspect Biol. 2015;7(2):a006023.
|