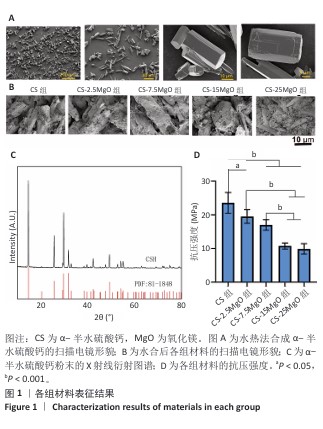
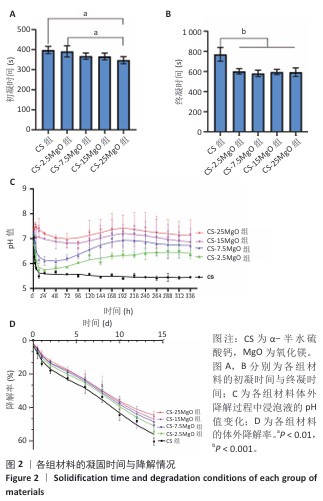
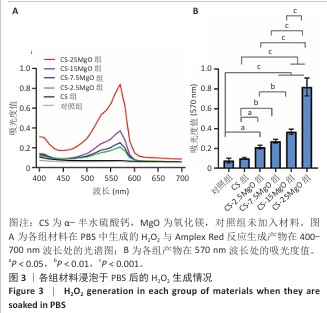
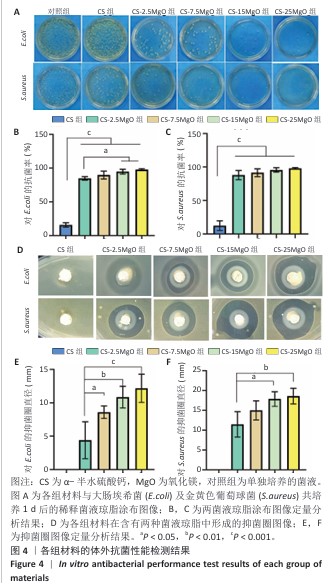
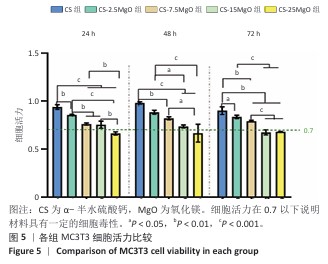
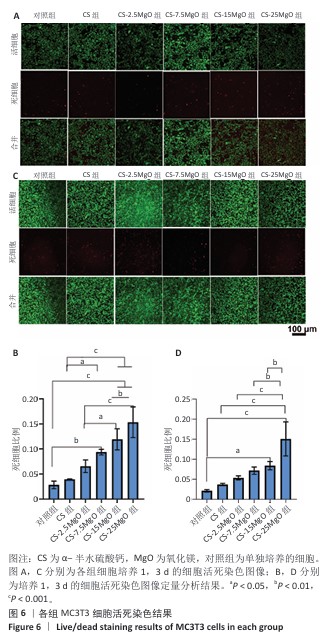
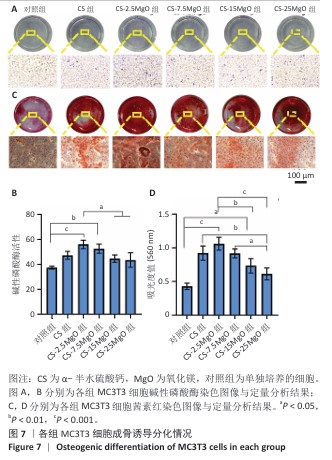
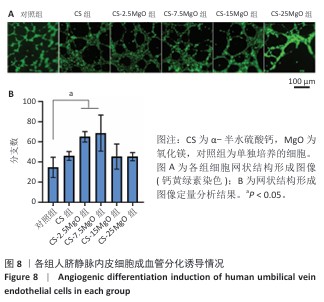
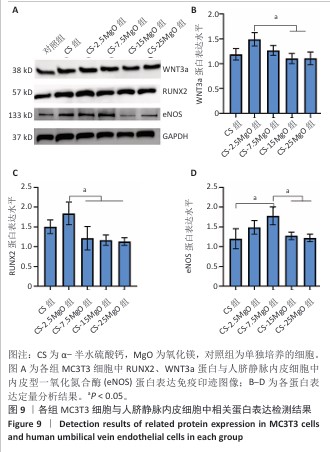
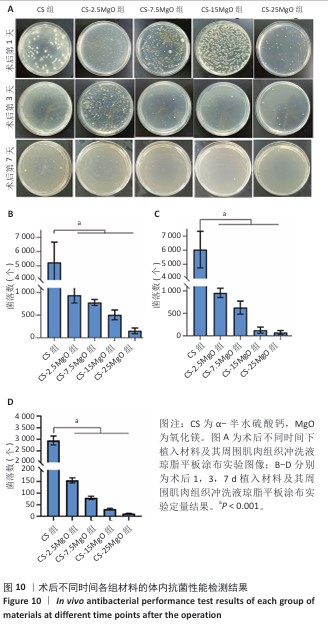
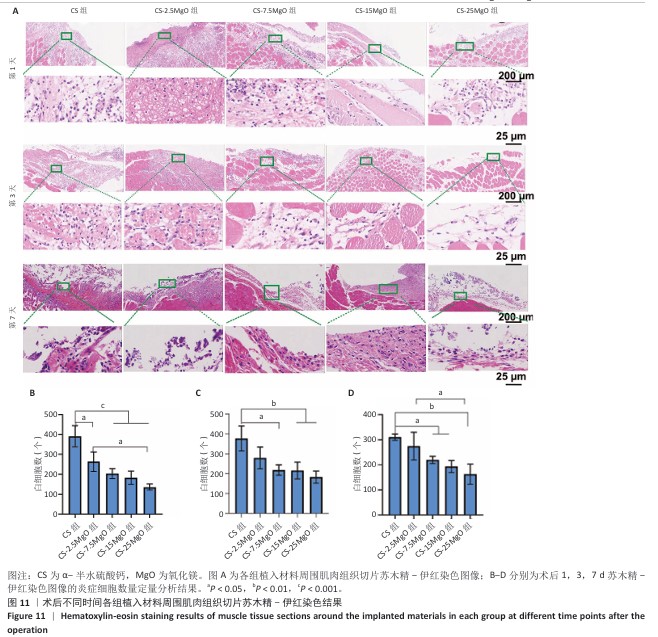
[1] LI C, LV H, DU Y, et al. Biologically modified implantation as therapeutic bioabsorbable materials for bone defect repair. Regen Ther. 2021;19:9-23.
[2] KURIEN T, PEARSON RG, SCAMMELL BE. Bone graft substitutes currentlyavailable in orthopaedic practice: the evidence for their use. Bone Joint J. 2013;95(5): 583-597.
[3] 程玮璐,张译丹,刘英慧.骨填充材料的临床应用进展[J].中国医疗器械信息,2023,29(21):43-47.
[4] PUTRA NE, MIRZAALI MJ, APACHITEI I, et al. Multi-material additive manufacturing technologies for Ti-,Mg-,and Fe-based biomaterials for bone substitution. Acta Biomater. 2020;16(109):1-20.
[5] 谢广渊,潘伟城,谭志斌,等.硫酸钙人工骨在良性骨肿瘤刮除术后骨缺损填充中的愈合情况及其重建作用研究[J].生物骨科材料与临床研究,2017, 14(2):45-48.
[6] 杨国敬,林勉,张雷,等.骨损伤修复用硫酸钙及其无机复合材料的研究进展[J].无机材料学报,2013,28(8):795-803.
[7] CHAMANSARA A, BEHNAMGHADER A, ZAMANIAN A. Preparation and characterization of injectable gelatin/alginate/chondroitin sulfate/alphacalcium sulfate hemihydrate composite paste for bone repair application. J Biomater Appl. 2022;36(10):1758-1774.
[8] ZIRAN BH, SMITH WR, MORGAN SJ. Use of calcium-based demineralized bone matrix/allograft for nonunions and posttraumatic reconstruction of the appendicular skeleton: preliminary results and complications. J Trauma. 2007; 63(6):1324-1328.
[9] MORLEY R, ROTHWELL M, STEPHENSON J, et al. Complex foot infection treated with surgical debridement and antibiotic loaded calcium sulfate—a retrospective cohort study of 137 cases. J Foot Ankle Surg. 2022;61(2):239-247.
[10] LIU H, LI P, TANG Z, et al. Study on injectable silver-incorporated calcium phosphate composite with enhanced antibacterial and biomechanical properties for fighting bone cement-associated infections. Colloids Surf B Biointerfaces. 2023;227:113382.
[11] 倪方方,王博林,宋腾蛟,等.纳米银颗粒的毒性效应及作用机制研究进展[J].中国药理学通报,2016,32(5):593-597.
[12] WEI S, MA JX, XU L, et al. Biodegradable materials for bone defect repair. Mil Med Res. 2020;7:54.
[13] SAWAI J, IGARASHI H, HASHIMOTO A, et al. Evaluation of growth inhibitory effect of ceramics powder slurry on bacteria by conductance method. J Chem Eng Japan. 1995;28(3):288-293.
[14] SAWAI J, YOSHIKAWA T. Quantitative evaluation of antifungal activity of metallic oxide powders (MgO, CaO and ZnO) by an indirect conductimetric assay. J Appl Microbiol. 2004;96(4):803-809.
[15] WU X, JIAO Y, JIA C, et al. Joule heating-induced active Mg0 into nano-Mg composites for boosted oxidation and antiviral performance. ACS EST Eng. 2024;4(6):1302-1311.
[16] HAYAT S, MUZAMMIL S, RASOOL MH, et al. In vitro antibiofilm and anti‐adhesion effects of magnesium oxide nanoparticles against antibiotic resistant bacteria. Microbiol Immunol. 2018;62(4):211-220.
[17] SAWAI J, IGARASHI H, HASHIMOTO A, et al. Evaluation of growth inhibitory effect of ceramics powder slurry on bacteria by conductance method. J Chem Eng Japan. 1995;28(3):288-293.
[18] JIN T, HE Y. Antibacterial activities of magnesium oxide (MgO) nanoparticles against foodborne pathogens. J Nanopart Res. 2011;13:6877-6885.
[19] LI X, LIU H, HE Y, et al. Enhanced antibacterial effect with MgO nanoplates: Role of oxygen vacancy and alkalinity. Ceram Int. 2024;50(21):42877-42885.
[20] LEUNG YH, NG AMC, XU X, et al. Mechanisms of antibacterial activity of MgO: non‐ROS mediated toxicity of MgO nanoparticles towards Escherichia coli. Small. 2014;10(6):1171-1183.
[21] YIN Z, LI S, LI X, et al. Effect of pH on the microstructure and antibacterial properties of MgO nanoparticles by microwave-assisted solution combustion. J Alloys Compd. 2024;1009:176858.
[22] NIE X, ZHANG X, LEI B, et al. Regulation of magnesium matrix composites materials on bone immune microenvironment and osteogenic mechanism. Front Bioeng Biotechnol. 2022;10:842706.
[23] MONFOULET LE, BECQUART P, MARCHAT D, et al. The pH in the microenvironment of human mesenchymal stem cells is a critical factor for optimal osteogenesis in tissue-engineered constructs. Tissue Eng Part A. 2014;20(13-14):1827-1840.
[24] ZHANG Y, XU J, RUAN Y, et al. Implant-derived magnesium induces local neuronal production of CGRP to improve bone-fracture healing in rats. Nat Med. 2016;22:1160-1169.
[25] CHEN L, ZHU J, GE N, et al. A biodegradable magnesium alloy promotes subperiosteal osteogenesis via interleukin-10-dependent macrophage immunomodulation. Biomaterials. 2025;318:122992.
[26] SUN L, LI X, XU M, et al. In vitro immunomodulation of magnesium on monocytic cell toward anti-inflammatory macrophages. Regen Biomater. 2020;7(4):391-401.
[27] 中国居民膳食常量元素参考摄入量[C]//中国营养学会微量元素营养分会.中国营养学会微量元素营养第十二次学术会议暨第六届微量元素营养分会会员大会论文集,2014:164.
[28] 赵颖,曾利兰,梁涛.可降解镁基金属的生物相容性研究进展[J].金属学报, 2017,53(10):1181-1196.
[29] 张丹.Mg-ZnO-HA可降解镁合金生物材料的生物相容性研究[D].长沙:中南大学,2011.
[30] GANDOLFI MG, IACONO F, AGEE K, et al. Setting time and expansion in different soaking media of experimental accelerated calcium-silicate cements and ProRoot MTA. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;108(6):e39-e45.
[31] ZHANG X, WANG Y, GUO J, et al. Comparing two functions for optical density and cell numbers in bacterial exponential growth phase. J Pure Appl Microbiol. 2015;9(1):299-305.
[32] ISO 10993-12:2021. Biological evaluation of medical devices - Part 12: Sample preparation and reference materials. International Organization for Standardization, Geneva, Switzerland, 2021.
[33] YUAN K, CHAN YJ, KUNG KC, et al. Comparison of osseointegration on various implant surfaces after bacterial contamination and cleaning: a rabbit study. Int J Oral Maxillofac Implants. 2014;29(1):32-40.
[34] STRAUB J, SEWING A, WALTER N, et al. Calcium Sulfate Bone Substitutes in Clinical Use: History, Material Properties, Application, and Outlook for the Future. J Biomed Mater Res B Appl Biomater. 2025;113(4):e35555.
[35] PETTAUER M, BALDERMANN A, EDER S, et al. Hydration of MgO: reaction kinetics and pH control on brucite crystal morphology. Crystal Growth Design. 2024;24(7):3085-3092.
[36] GALOW AM, REBL A, KOCZAN D, et al. Increased osteoblast viability at alkaline pH in vitro provides a new perspective on bone regeneration. Biochem Biophys Rep. 2017;10:17-25.
[37] HARADA M, UDAGAWA N, FUKASAWA K, et al. Inorganic pyrophosphatase activity of purified bovine pulp alkaline phosphatase at physiological pH. J Dent Res. 1986;65(2):125-127.
[38] 李政垚,刘洁颖,吴狄,等.高镁离子浓度对人骨髓间充质干细胞增殖与成骨分化的影响[J].中华骨与关节外科杂志,2020,13(5): 419-426.
[39] QIAO W, WONG KHM, SHEN J, et al. TRPM7 kinase-mediated immunomodulation in macrophage plays a central role in magnesium ion-induced bone regeneration. Nat Commun. 2021;12(1):2885.
[40] NYGREN H, CHAUDHRY M, GUSTAFSSON S, et al. Increase of compact bone thickness in rat tibia after implanting MgO into the bone marrow cavity. J Funct Biomater. 2014;5(3):158-166.
[41] MATHEW AA, PANONNUMMAL R. ‘Magnesium’-the master cation-as a drug—possibilities and evidences. Biometals. 2021;34(5):955-986.
[42] WANG J, MA XY, FENG YF, et al. Magnesium ions promote the biological behaviour of rat calvarial osteoblasts by activating the PI3K/Akt signalling pathway. Biol Trace Elem Res. 2017;179:284-293.
[43] RESNICK LM. Cellular calcium and magnesium metabolism in the pathophysiology and treatment of hypertension and related metabolic disorders. Am J Med. 1992;93(2):S11-S20.
[44] BURBRIDGE MF, WEST DC, ATASSI G, et al. The effect of extracellular pH on angiogenesis in vitro. Angiogenesis. 1999;3:281-288.
[45] LIU W, GUO S, TANG Z, et al. Magnesium promotes bone formation and angiogenesis by enhancing MC3T3-E1 secretion of PDGF-BB. Biochem Biophys Res Commun. 2020;528(4):664-670.
[46] SARIS NEL, MERVAALA E, KARPPANEN H, et al. Magnesium: an update on physiological, clinical and analytical aspects. Clin Chim Acta. 2000;294(1-2):1-26.
[47] HANG R, TIAN X, QU G, et al. Exosomes derived from magnesium ion—stimulated macrophages inhibit angiogenesis. Biomed Mater. 2022;17(4):045008. |