Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (15): 2404-2411.doi: 10.12307/2023.362
Previous Articles Next Articles
Feasibility and advantages of mesenchymal stem cells for repairing Achilles tendon injury
Li Wei1, Lin Meina2, Lu Yongping2, Sui Yu2, Jiang Miao1, 2
- 1Shenyang Sport University, Shenyang 110115, Liaoning Province, China; 2NHC Key Laboratory of Reproductive Health and Medical Genetics (China Medical University) & Liaoning Key Laboratory of Reproductive Health, Liaoning Research Institute of Family Planning, Department of Eugenics and Genetics, Reproductive Hospital Affiliated to China Medical University, Shenyang 110031, Liaoning Province, China
-
Received:2022-05-05Accepted:2022-06-23Online:2023-05-28Published:2022-10-18 -
Contact:Jiang Miao, Master, Researcher, Shenyang Sport University, Shenyang 110115, Liaoning Province, China; NHC Key Laboratory of Reproductive Health and Medical Genetics (China Medical University) & Liaoning Key Laboratory of Reproductive Health, Liaoning Research Institute of Family Planning, Department of Eugenics and Genetics, Reproductive Hospital Affiliated to China Medical University, Shenyang 110031, Liaoning Province, China -
About author:Li Wei, Shenyang Sport University, Shenyang 110115, Liaoning Province, China -
Supported by:Liaoning Provincial Key Research & Development Program, No. 2017020199-201 (to JM)
CLC Number:
Cite this article
Li Wei, Lin Meina, Lu Yongping, Sui Yu, Jiang Miao. Feasibility and advantages of mesenchymal stem cells for repairing Achilles tendon injury[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(15): 2404-2411.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
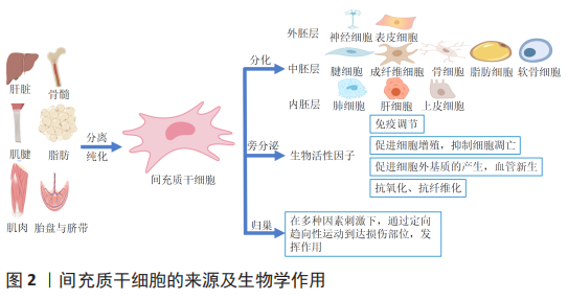
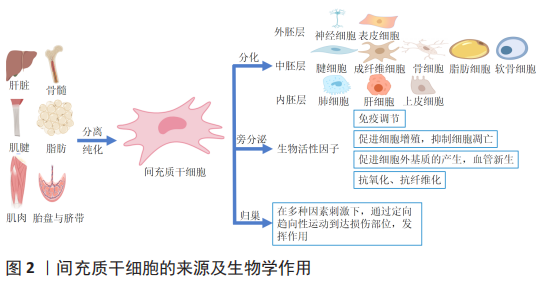
2.1 间充质干细胞在跟腱损伤修复中的应用 2.1.1 间充质干细胞概述 间充质干细胞是起源于中胚层的一类多能干细胞,主要存在于结缔组织和器官间质中,能分化为本胚层的腱细胞、成纤维细胞、成骨细胞、软骨细胞、脂肪细胞等,也可跨胚层分化为神经细胞、肺细胞、肝细胞等,是一类非常有前景的组织修复和再生的细胞来源。间充质干细胞可以从多种组织中获得,如骨髓、牙髓、骨骼肌、胎盘、脐带、羊水、滑膜、脂肪以及经血等。不同组织来源的间充质干细胞因为环境不同具有不同的增殖分化能力,例如经血来源间充质干细胞的分化能力要优于脐血来源间充质干细胞[4],骨髓和骨膜来源间充质干细胞在成骨方面更优越,脂肪组织来源间充质干细胞在成脂方面效果更佳[5]。 目前间充质干细胞还没有特异性的表面标志物,根据国际细胞治疗学会(International Society For Ccell Treatment)的标准,必须满足3个条件:贴壁生长;表面抗原CD105、CD73、CD90阳性,且CD45、CD34、CD14、CD11b、CD79α、CD19及HLA-DR阴性;在标准的体外分化条件下,细胞必须能分化为成骨细胞、成软骨细胞、脂肪细胞[6]。 间充质干细胞具有分化潜力大、增殖能力强、免疫原性低、易于制备等特征,因此是最具临床应用前景的多能干细胞[7]。现有的研究资料表明间充质干细胞作为损伤修复和疾病治疗的种子细胞主要通过以下途径实现:通过归巢效应到达损伤部位,发挥修复作用[8];分泌生物活性因子影响机体微环境,刺激患者体内残留的组织特异性干细胞再生组织[9];自身分化为损伤部位的细胞进行填充等[10],见图2。目前,间充质干细胞治疗跟腱损伤的方法主要包括支架负载、直接注射、制成细胞片、负载干细胞的缝线缝合及外泌体治疗等。间充质干细胞不会被免疫系统排斥,这为异体组织修复领域的发展提供了条件,并为其他干细胞的使用铺平了道路[11]。"
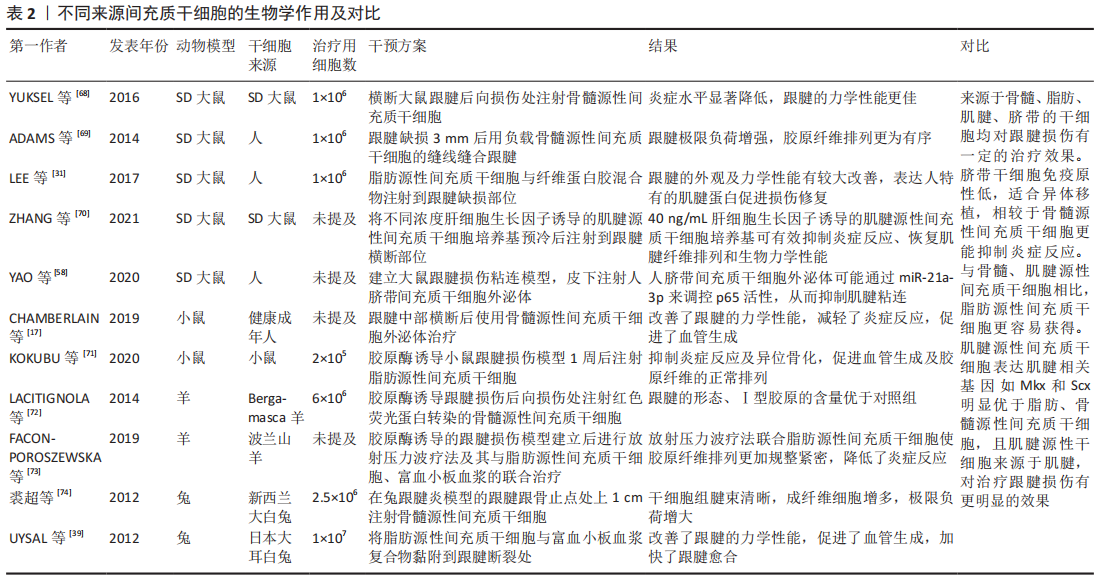
2.1.2 骨髓源性间充质干细胞与跟腱损伤修复 骨髓源性间充质干细胞是存在于骨髓基质内的非造血干细胞,占骨髓细胞的0.001%-0.01%,可从髂骨或长骨的骨髓抽吸物中获得[12]。目前,骨髓源性间充质干细胞是应用最广泛的干细胞,可通过多种形式参与疾病治疗[13-14]。存活时间是干细胞发挥生物学效应的一个关键因素,适当的存活时间为损伤修复提供了时效保障。体内注射骨髓源性间充质干细胞24 h后虽然仅有约24%的细胞残留,但这些细胞在植入后可存活长达5个月之久[15]。外泌体内的蛋白质、核酸、miRNA等可以在细胞间传递生物学信息,调控细胞的生物活动[16]。研究表明,骨髓源性间充质干细胞外泌体诱导的巨噬细胞会减轻跟腱损伤后的炎症反应,促进血管形成,改善跟腱的生物力学性能[17]。细胞代数是影响干细胞活性、安全性及治疗有效性的一个重要条件,代数越小,其干性会越强,促进跟腱恢复的效果越显著[18]。体外研究显示,低氧条件能促进骨髓源性间充质干细胞表达介导腱分化的基因MKx,分泌血管内皮生长因子、Ⅰ型胶原和Ⅲ型胶原[19-21];在力学因素刺激下,骨髓源性间充质干细胞可产生更多的饰胶蛋白聚糖及腱糖蛋白C,力学刺激与转化生长因子β1联合运用可进一步增强肌腱相关基因的表达[22];胰岛素样生长因子、转化生长因子β3、骨形态发生蛋白12和血管内皮生长因子等生长因子均可刺激骨髓源性间充质干细胞向腱细胞分化[23-25]。在跟腱损伤修复中使用此类生长因子会促进骨髓源性间充质干细胞修复损伤,减少炎症反应,加快跟腱损伤后的恢复。骨髓源性间充质干细胞治疗跟腱损伤效果显著,然而,骨髓源性间充质干细胞来源于骨髓,获取手段具有侵袭性,同时向骨、软骨分化的潜能更大,应用骨髓源性间充质干细胞治疗跟腱损伤时,可能产生异位骨化、加重损伤,因此限制了此类干细胞的应用。如何利用骨髓源性间充质干细胞的优势,发挥其积极作用是仍需解决的问题。 2.1.3 脂肪源性间充质干细胞与跟腱损伤修复 脂肪源性间充质干细胞是一类来自脂肪组织的间充质干细胞,其制备方法简单、可塑性强、致畸致瘤风险较低。与骨髓源性间充质干细胞相比,脂肪源性间充质干细胞的增殖速度快且表现出较低的成骨和成软骨分化潜能[26]。O BORTOLAZZO等[27]研究表明10 μA/min的微电流与脂肪源性间充质干细胞的联合运用增加了纤调蛋白基因的表达,使胶原组织增多,提高了跟腱的力学性能。γ-干扰素致敏的脂肪源性间充质干细胞外泌体可通过调节巨噬细胞减弱跟腱损伤后的早期炎症反应,降低组织修复后间隙的形成和二次断裂的发生率[28]。抑制人脂肪源性间充质干细胞中miR-34a表达,可减轻炎症反应,促进跟腱炎大鼠受损跟腱的修复[29]。干细胞归巢数量对损伤的恢复有至关重要的作用。YEROFEYEVA等[30]发现与0.25×106个脂肪源性间充质干细胞相比,0.5×106个脂肪源性间充质干细胞植入大鼠跟腱损伤处更有利于跟腱损伤的恢复。在异种干细胞移植过程中,干细胞是否会分化或者表达自身的蛋白质以促进损伤的愈合是值得探讨的问题。有学者将人脂肪源性间充质干细胞和具有3D结构的纤维蛋白胶混合注射到兔跟腱缺损部位,干细胞可通过表达人特异性肌腱标志基因腱糖蛋白C、Ⅰ型胶原来促进跟腱损伤愈合[31]。异种移植中免疫排斥对机体造成损伤的同时也会破坏植入体,影响治疗的效果。不同物种间干细胞的植入是否对机体无害仍需更深入的研究。生长因子是一种调节细胞生长和增殖的多肽类物质,可调节细胞的生长及其功能,诱导细胞分化、血管形成、胶原蛋白和基质的产生[32]。CHEN等[33]研究表明重组人血小板源性生长因子BB可通过miR-363/PI3K/Akt通路促进人脂肪源性间充质干细胞的增殖,显著促进大鼠跟腱炎的恢复。转化生长因子β3、骨形态发生蛋白12、胰岛素样生长因子等生长因子也可诱导脂肪源性间充质干细胞分化为腱细胞,改善跟腱损伤后的组织学、生物力学和分子特性[34-35]。骨形态发生蛋白14、抗坏血酸、转化生长因子β3和结缔组织生长因子的联合应用可更快速、更强烈诱导人脂肪源性间充质干细胞分化为肌腱样细胞[36]。PARK等[37]发现100 ng/mL生长分化因子5可显著促进脂肪源性间充质干细胞的增殖,增强肌腱标志性基因饰胶蛋白聚糖、SCX、腱糖蛋白C等的表达。富血小板血浆是一种血小板浓缩液,含有血小板源性生长因子、表皮生长因子、胰岛素样生长因子、血管内皮生长因子等细胞活性因子[38]。因此,富血小板血浆作为组织再生诱导因子引起了许多研究人员的关注。研究证实脂肪源性间充质干细胞与富血小板血浆的联合应用可抑制组织粘连,提高跟腱的抗拉强度[39]。 2.1.4 肌腱源性间充质干细胞与跟腱损伤修复 肌腱源性间充质干细胞是从肌腱组织中分离的具有多向分化潜能和自我更新能力的干细胞[40]。自2007年BI等[41]首次从肌腱中分离出肌腱源性间充质干细胞并证实其具有多向分化潜能、自我更新能力以及增殖以来,已有研究表明肌腱源性间充质干细胞是肌腱细胞的前体细胞,也是肌腱细胞的唯一来源。由于肌腱源性间充质干细胞来源于肌腱组织,所以其在治疗肌腱组织损伤时具有较大的优势。有学者将来源于SD大鼠的骨髓源性间充质干细胞和肌腱源性间充质干细胞治疗跟腱损伤进行了对比研究,结果表明肌腱源性间充质干细胞在跟腱愈合中表现出更好的治疗效果,跟腱具有更好的外观和纵向纤维结构[42]。SONG等[43]研究表明,相较于胚胎干细胞,种植于细胞外基质支架中的肌腱源性间充质干细胞显著改善了大鼠跟腱的结构和生物力学性能,肌腱源性间充质干细胞在治疗大鼠跟腱断裂方面有更佳的效果。肌腱的发育源于中胚层,发育过程中有很多关键基因参与调控,如Mkx、Scx、腱调蛋白等,在肌腱源性间充质干细胞中,Mkx和Scx的表达明显高于脂肪源性间充质干细胞和胚胎干细胞[44],这也反映了肌腱源性间充质干细胞治疗肌腱损伤的优势。在大鼠跟腱损伤模型中,肌腱源性间充质干细胞外泌体会抑制基质金属蛋白酶3的生成,促进腱调蛋白、Ⅰ型胶原的生成,显著促进跟腱损伤修复,对细胞外基质组分的维持有重要作用[45]。雌激素是一种类固醇类激素,可影响腱细胞的增殖和迁移速率,促进Ⅰ型胶原、蛋白聚糖等细胞外基质组分的分泌[46]。BIAN等[47]研究表明缺乏雌激素受体会激活过氧化物酶体增殖物激活受体γ导致肌腱源性间充质干细胞向脂肪细胞分化,造成跟腱损伤部位脂肪的堆积,而雌激素受体β激动剂LY3201可以部分逆转异常分化,这为增加修复跟腱损伤的效果、抑制肌腱源性间充质干细胞向脂肪分化提供了方法。雌激素及其受体在跟腱损伤中的积极作用为跟腱损伤的临床治疗提供了新的靶点,但雌激素及其受体联合肌腱源性间充质干细胞治疗跟腱损伤的疗效需要更多的研究以表征其在损伤治疗中的优势。张力是肌腱在体内环境的重要组成部分,能刺激腱细胞的增殖、细胞外基质的形成[48]。目前,关于机械载荷对肌腱源性间充质干细胞的影响主要集中在拉力方向及频率上。研究表明以0.5 Hz频率、4%振幅循环拉伸会刺激肌腱源性间充质干细胞增殖,促进腱糖蛋白C、SCX、腱调蛋白、Ⅰ型胶原的表达[49]。RUI等[50]发现以4%的频率对肌腱源性间充质干细胞进行重复拉伸可促进骨形态发生蛋白2的合成,而骨形态发生蛋白2会诱导肌腱源性间充质干细胞向成骨细胞分化,这为肌腱损伤或钙化性肌腱病提供了一种可能的解释。肌腱源性间充质干细胞在不同方向、频率及振幅的作用下会表现出具有差异性的分化特征,合理运用机械载荷使肌腱源性间充质干细胞向特定细胞分化有利于组织损伤后的再生与修复,为临床跟腱损伤后的康复治疗提供更好的治疗方案。除机械刺激外,一些生长因子也可促进肌腱源性间充质干细胞的增殖和腱性分化。LIU等[51]发现lncRNAH9通过抑制miR-140-5p促进血管内皮生长因子A的产生,诱导肌腱源性间充质干细胞的成腱分化。诱导肌腱源性间充质干细胞向腱细胞分化的生长因子还有成纤维细胞生长因子2、生长分化因子5、转化生长因子β3、结缔组织生长因子等[52-55]。 2.1.5 人脐带间充质干细胞与跟腱损伤修复 人脐带间充质干细胞是存在于新生儿脐带组织中的一种多功能干细胞,具有免疫原性低、增殖分化能力更强、取材方便、适合异体移植等优点[56]。CIARDULLI等[57]研究表明与骨髓源性间充质干细胞相比,人脐带间充质干细胞抑制跟腱损伤后的炎症反应更显著。近年来,人脐带间充质干细胞来源外泌体在组织修复和再生领域受到越来越多的关注。YAO等[58]研究证实来源于人脐带间充质干细胞的外泌体能优化跟腱的生物力学性能,通过抑制纤维化和炎症相关基因减轻肌腱粘连的程度。HAN等[59]发现人脐带间充质干细胞外泌体通过miR-27b-3p靶向抑制ARHGAP5表达,促进RhoA活化来提高细胞增殖能力,修复跟腱损伤。羟喜树碱是一种抑制DNA拓扑异构酶Ⅰ的生物碱,能通过降低Ⅲ型胶原蛋白α1链和α-平滑肌肌动蛋白基因的表达,抑制成纤维细胞增殖并诱导其凋亡,从而显著改善肌腱损伤后的组织粘连。由羟喜树碱致敏的人脐带间充质干细胞外泌体可通过激发成纤维细胞内质网应激反应提高治疗肌腱损伤的抗粘连潜能[60]。富血小板血浆的激活形式能释放多种有利于跟腱损伤修复的生长因子,与人脐带间充质干细胞共培养可以显著促进人脐带间充质干细胞中腱生蛋白C、SCX和Ⅰ型胶原基因的表达,使细胞外基质中胶原纤维排列更规整,炎症细胞更少,有效促进大鼠跟腱损伤的恢复[61]。 干细胞的生物安全性是应用干细胞治疗的关键问题。干细胞的多能性赋予了干细胞治疗疾病的可能,但也存在致瘤性、不稳定性等问题。有研究显示人脐带间充质干细胞端粒酶表达水平正常,遗传稳定,可将人脐带间充质干细胞作为一种安全有效的细胞治疗方式[62]。人脐带间充质干细胞取材方便、增殖速度快且免疫原性低于骨髓源性间充质干细胞,适合作为组织修复再生的理想干细胞来源[63]。 2.1.6 经血源性间充质干细胞与跟腱损伤修复 人类子宫内膜是一种高度再生的组织,在育龄妇女身上经历周期性的增殖、分化和脱落。已有研究表明在子宫内膜的基底层和功能层中存在少量的子宫内膜间充质干细胞[64]。子宫内膜间充质干细胞的脱落版本称为经血源性间充质干细胞,其与子宫内膜间充质干细胞具有相似的特征[65]。经血源性间充质干细胞的提取无侵袭性、倍增时间短、增殖能力强,经过长时间的培养仍具有稳定的染色体核型,是移植、治疗疾病的理想细胞来源[66]。已有研究证明经血源性干细胞能够在不同培养条件下分化为外胚层、中胚层和内胚层3个胚胎层的多种细胞系,如骨细胞、软骨细胞、脂肪细胞、肌肉细胞、心肌细胞、神经细胞、肝细胞、髓核样细胞、腱细胞等。ZHENG等[67]将经血源性间充质干细胞与跟腱细胞共培养,共培养组细胞的Ⅰ型胶原、Ⅲ型胶原、血小板反应蛋白4、SCX和腱糖蛋白C的基因表达水平显著增加,表明在治疗跟腱损伤中可以考虑使用经血源性间充质干细胞。将来源于骨髓、脂肪、肌腱及脐带的间充质干细胞进行对比,见表2[17,31,39,58,68-74]。 2.2 生物材料负载间充质干细胞与跟腱损伤修复 生物材料是与生命系统接触和发生相互作用并能对其细胞、组织和器官进行诊断治疗、替换修复或诱导再生的一类天然或人工合成的特殊功能材料。目前,常用的生物材料有生物衍生材料、合成聚合物材料等[75]。由生物材料制成的支架会为细胞生长提供稳定的外部环境,如果缺乏适当的支架来供给细胞合适的生存环境,细胞就会流失、死亡[76]。 2.2.1 聚合物材料 聚合物材料是指由许多单体通过化学键连接而成的高分子量化合物,由于其具有质量轻、成本低、力学性能良好等优点,在医疗、航天等领域均有广泛应用[77]。CAI等[78]将来源于兔的骨髓源性间充质干细胞负载到聚对苯二甲酸乙二酯支架上并植入兔跟腱缺损处,结果显示材料负载干细胞组有肌腱样组织形成,Ⅰ型胶原、Ⅲ型胶原含量及跟腱极限载荷和平均刚度均优于聚对苯二甲酸乙二酯组。随着年龄的增长,跟腱会逐渐发生退化,肌腱源性间充质干细胞的增殖与分化能力也会降低,这导致老年人跟腱损伤后更加难以恢复。RADA肽水凝胶的3D结构可使老化的肌腱源性干细胞恢复增殖和分化能力,通过3D微环境指导肌腱源性间充质干细胞的生物活动,克服了跟腱修复中肌腱源性间充质干细胞老化及退化的问题[79]。生长因子可通过传递信号诱导干细胞分化,而干细胞与生长因子、生物材料等的联合运用对损伤和疾病的治疗具有更突出的作用。MIN等[80]发现含有较高浓度血小板源性生长因子BB和较低浓度骨形态发生蛋白2的聚己内酯/普朗尼克F127膜为脂肪源性间充质干细胞成腱分化提供了更好的环境。生长分化因子5可以调节聚乙丙交酯纤维支架上的脂肪源性间充质干细胞表达肌腱标志性基因,满足肌腱再生的需要[81]。对种植于硅胶小室和胶原海绵3D支架上的骨髓源性间充质干细胞进行周期性的机械拉伸联合转化生长因子β1诱导可显著促进跟腱的再生[82]。通过聚丙交酯-乙交酯支架承载并递送经肿瘤坏死因子α处理的骨髓源性间充质干细胞可调节巨噬细胞极化从而加快跟腱的恢复[83]。将外源性的碱性成纤维细胞生长因子和骨髓源性间充质干细胞置入聚丙交酯-乙交酯支架中,对跟腱损伤也有显著的修复作用[84]。 循环干细胞是外周血中具有不同解剖学和发育起源的异质细胞群,其中造血干细胞和造血祖细胞构成了最丰富和最具特征的循环干细胞类型[85]。研究表明循环干细胞可通过旁分泌作用修复心血管损伤,通过分化为特定组织的细胞以填补组织缺损[86]。DAHER等[87]将循环干细胞负载在聚羟基乙酸支架上,干细胞与支架复合体显著改善了跟腱的组织结构水平,优化了跟腱的生物力学性能。外泌体有与其来源细胞相似的生物学效应,可恢复受损组织的正常功能。相较于细胞,外泌体有稳定性好、便于保存运输、免疫原性低等优势。ZHANG等[88]将来源于肌腱源性间充质干细胞的外泌体与明胶-甲基丙烯酰混合后注射到跟腱缺损处,经辐照后成为凝胶状,研究结果表明外泌体可通过激活PI3K/AKT和MAPK/ERK1/2信号通路促进腱细胞的增殖和迁移,外泌体与明胶-甲基丙烯酰混合后可更好地抑制炎症和细胞凋亡,平衡细胞外基质组分,减少瘢痕形成。 2.2.2 生物衍生材料 生物衍生材料的3D空间结构近似于细胞在人体内的生长环境,可影响细胞的生物学过程,促进组织再生。生物衍生材料可分为天然生物衍生材料和提纯生物衍生材料两大类[89]。天然生物衍生材料是对来源于动物的组织进行较小的处理,以用于负载、移植等;提纯生物衍生材料会重构原有的组织结构,改变其物理形态以适应治疗需要。 小肠黏膜下层是一种含有丰富胶原纤维、蛋白多糖和生长因子的细胞外基质,可以促进干细胞的黏附、增殖和分化[90]。将脂肪源性间充质干细胞种植在小肠黏膜下层支架上并进行缺氧预处理后移植到SD大鼠跟腱缺损处,缺氧处理的脂肪源性间充质干细胞进一步提高了Mkx、腱调蛋白的表达,增加了跟腱的极限断裂负荷[91]。李岱鹤等[92]将来源于Wistar大鼠的骨髓源性间充质干细胞负载到猪的小肠黏膜下层中,复合物桥接到跟腱缺损处增加了跟腱的力学性能,维持了跟腱内正常的胶原比例。富血小板纤维蛋白支架是一种自体纤维蛋白生物材料,富含多种细胞因子,能够局部且逐渐地输送生长因子以促进跟腱的愈合和再生[93]。肌腱细胞外基质中含有大量的胶原纤维,形成的网络结构对维持肌腱形态及促进细胞的增殖和分化有重要作用。与胶原水凝胶相比,脱细胞肌腱水凝胶有更好的组织相容性,可显著促进肌腱源性间充质干细胞的迁移与分化[94]。通过特殊方法制成的胶原海绵及脱细胞肌腱可以很好地保留三维结构,为骨髓源性间充质干细胞腱性分化提供了诱导性微环境[95]。在兔跟腱缺损修复中,负载骨髓源性间充质干细胞的胶原海绵提高了跟腱的力学强度,促进了血管生成[96]。除骨髓源性间充质干细胞外,脂肪源性间充质干细胞也可使脱细胞的支架再细胞化,这为肌腱组织重建提供了又一种细胞来源[97]。明胶微冰胶是一种可降解的生物材料,可以为干细胞的附着和增殖提供一个3D环境,有助于延长细胞存活时间,保持细胞功能[98]。负载脂肪源性间充质干细胞的明胶微冰胶在改善跟腱的外观、组织学形态、跟腱功能指数和生物力学性能方面有较好的效果[99]。CHEN等[100]将桑蚕丝脱丝胶蛋白后浸泡在Ⅰ型胶原中,经过脱水热处理后使支架交联化制成了蚕丝胶原海绵支架。将人胚胎干细胞植入编织的蚕丝胶原海绵支架中,在体外动态机械刺激下,呈腱细胞样形态,肌腱相关标志基因Epha4、Scx的表达呈阳性,在体内实验中,负载人胚胎干细胞的支架直接促进了跟腱的再生,而且在植入部位起到了改善环境的作用。将兔骨髓源性间充质干细胞负载到填充胶原蛋白和丝素蛋白的无丝胶蛋白支架上,植入兔跟腱缺损处20周后,组织学观察显示肌腱样组织已形成,生成了丰富的胶原纤维,这显示出了骨髓源性间充质干细胞-支架复合材料修复跟腱缺损的潜力[101]。 天然材料制成的支架本身含有疏松及多孔的结构,是一种丰富且优良的材料。虽然材料本身的力学性能较差,但与组织较好的相容性使得生物衍生材料有广阔的应用空间。在维持材料良好相容性的同时,提高材料各方面性能是目前仍需解决的问题。"
| [1] PARK S, LEE HS, YOUNG KW, et al. Treatment of Acute Achilles Tendon Rupture. Clin Orthop Surg. 2020;12(1):1-8. [2] ANDARAWIS-PURI N, FLATOW EL, SOSLOWSKY LJ. Tendon basic science: Development, repair, regeneration, and healing. J Orthop Res. 2015; 33(6):780-784. [3] GANESTAM A, KALLEMOSE T, TROELSEN A, et al. Increasing incidence of acute Achilles tendon rupture and a noticeable decline in surgical treatment from 1994 to 2013. A nationwide registry study of 33,160 patients. Knee Surg Sports Traumatol Arthrosc. 2016;24(12):3730-3737. [4] MENG X, ICHIM TE, ZHONG J, et al. Endometrial regenerative cells: a novel stem cell population. J Transl Med. 2007;5(1):57-67. [5] SAKAGUCHI Y, SEKIYA I, YAGISHITA K, et al. Comparison of human stem cells derived from various mesenchymal tissues: Superiority of synovium as a cell source. Arthritis Rheum. 2005;52(8):2521-2529. [6] DOMINICI M, LE BLANC K, MUELLER I, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8(4):315-317. [7] MCGUIRE JJ, FRIELING JS, LO CH, et al. Mesenchymal stem cell-derived interleukin-28 drives the selection of apoptosis resistant bone metastatic prostate cancer. Nat Commun. 2021;12(1):723. [8] LI D, XUE W, Li M, et al. VCAM-1+ macrophages guide the homing of HSPCs to a vascular niche. Nature. 2018;564(7734):119-124. [9] 陈津. 间充质(干)细胞的定义变迁[J].中华细胞与干细胞杂志(电子版),2017,7(4):247-250. [10] 刘子文,王文波.缺氧培养调控间充质干细胞的生理活性[J].中国组织工程研究,2022,26(13):2127-2132. [11] PITTENGER MF, MARTIN BJ, FINKEL T, et al. Mesenchymal Stem Cells and Their Potential as Cardiac Therapeutics. Circ Res. 2004;95(1): 9-20. [12] SCHNEIDER M, ANGELE P, JÄRVINEN TAH, et al. Rescue plan for Achilles Therapeutics steering the fate and functions of stem cells in tendon wound healing. Adv Drug Deliv Rev. 2018;129:352-375. [13] SQUILLARO T, PELUSO G, GALDERISI U. Clinical Trials with Mesenchymal Stem Cells: An Update. Cell Transplant. 2016;25(5):829-848. [14] MARUYAMA M, WEI L, THIO T, et al. The Effect of Mesenchymal Stem Cell Sheets on Early Healing of the Achilles Tendon in Rats. Tissue Eng Part A. 2020;26(3-4):206-213. [15] DUDHIA J, BECERRA P, VALDES MA, et al. In Vivo Imaging and Tracking of Technetium-99m Labeled Bone Marrow Mesenchymal Stem Cells in Equine Tendinopathy. J Vis Exp. 2015;(106):e52748. [16] 沈括,张锦鑫,王洪涛,等.脂肪间充质干细胞外泌体促进创面修复的研究进展[J].中华损伤与修复杂志(电子版), 2021,16(3): 260-264. [17] CHAMBERLAIN CS, CLEMENTS AEB, KINK JA, et al. Extracellular Vesicle-Educated Macrophages Promote Early Achilles Tendon Healing. Stem Cells. 2019;37(5):652-662. [18] HAYASHI Y, YIMITI D, SANADA Y, et al. The therapeutic capacity of bone marrow MSC-derived extracellular vesicles in Achilles tendon healing is passage-dependent and indicated by specific glycans. FEBS Lett. 2022;596(8):1047-1058. [19] CHEN G, FAN D, ZHANG W, et al. Mkx mediates tenogenic differentiation but incompletely inhibits the proliferation of hypoxic MSCs. Stem Cell Res Ther. 2021;2(1):1-15. [20] THEUS MH, WEI L, CUI L, et al. In vitro hypoxic preconditioning of embryonic stem cells as a strategy of promoting cell survival and functional benefits after transplantation into the ischemic rat brain. Exp Neurol. 2008;210(2):656-670. [21] HUANG TF, YEW TL, CHIANG ER, et al. Mesenchymal stem cells from a hypoxic culture improve and engraft Achilles tendon repair. Am J Sports Med. 2013;41(5):1117-1125. [22] YANG F, RICHARDSON DW. Comparative Analysis of Tenogenic Gene Expression in Tenocyte-Derived Induced Pluripotent Stem Cells and Bone Marrow-Derived Mesenchymal Stem Cells in Response to Biochemical and Biomechanical Stimuli. Stem Cells Int. 2021;2021: 8835576. [23] PERUCCA ORFEI C, VIGANÒ M, PEARSON JR, et al. In Vitro Induction of Tendon-Specific Markers in Tendon Cells, Adipose- and Bone Marrow-Derived Stem Cells is Dependent on TGFβ3, BMP-12 and Ascorbic Acid Stimulation. Int J Mol Sci. 2019;20(1):149. [24] LEE JY, ZHOU Z, TAUB PJ, et al. BMP-12 treatment of adult mesenchymal stem cells in vitro augments tendon-like tissue formation and defect repair in vivo. PLoS One. 2011;6(3):e17531. [25] BOTTAGISIO M, LOPA S, GRANATA V, et al. Different combinations of growth factors for the tenogenic differentiation of bone marrow mesenchymal stem cells in monolayer culture and in fibrin-based three-dimensional constructs. Differentiation. 2017;95:44-53. [26] FITZSIMMONS REB, MAZUREK MS, SOOS A, et al. Mesenchymal Stromal/Stem Cells in Regenerative Medicine and Tissue Engineering. Stem Cells Int. 2018;2018:8031718. [27] O BORTOLAZZO F, D LUCKE L, DE OLIVEIRA FUJII L, et al. Microcurrent and adipose-derived stem cells modulate genes expression involved in the structural recovery of transected tendon of rats. FASEB J. 2020; 34(8):10011-10026. [28] SHEN H, YONEDA S, ABU AMER Y, et al. Stem cell‐derived extracellular vesicles attenuate the early inflammatory response after tendon injury and repair. J Orthop Res. 2019;38(1):117-127. [29] 陈巧杰,武少坤,邬耀军,等.miR-34a对脂肪干细胞移植治疗跟腱炎的影响[J].中国病理杂志,2018,34(10):1884-1889. [30] YEROFEYEVA AM, ZHAVORONOK IP, ANTIPOVA OA, et al. Effects of adipose-derived mesenchymal stem cells on nociceptive sensitivity and repair processes at achilles tendon injury model in rats. Doklady of the National Academy of Sciences of Belarus. 2020;64(5):574-582. [31] LEE SY, KWON B, LEE K, et al. Therapeutic Mechanisms of Human Adipose-Derived Mesenchymal Stem Cells in a Rat Tendon Injury Model. Am J Sports Med. 2017;45(6):1429-1439. [32] SHARMA P, MAFFULLI N. Tendinopathy and tendon injury: the future. Disabil Rehabil. 2008;30(20-22):1733-1745. [33] CHEN QJ, CHEN L, WU SK, et al. rhPDGF-BB combined with ADSCs in the treatment of Achilles tendinitis via miR-363/PI3 K/Akt pathway. Mol Cell Biochem. 2018;438(1-2):175-182. [34] TITAN AL, FOSTER DS, CHANG J, et al. Flexor Tendon: Development, Healing, Adhesion Formation, and Contributing Growth Factors. Plast Reconstr Surg. 2019;144(4):639e-647e. [35] SCHNEIDER PR, BUHRMANN C, MOBASHERI A, et al. Three-dimensional high-density co-culture with primary tenocytes induces tenogenic differentiation in mesenchymal stem cells. J Orthop Res. 2011;29(9): 1351-1360. [36] STANCO D, CAPRARA C, CIARDELLI G, et al. Tenogenic differentiation protocol in xenogenic-free media enhances tendon-related marker expression in ASCs. PLoS One. 2019;14(2):e212192. [37] PARK A, HOGAN MV, KESTURU GS, et al. Adipose-Derived Mesenchymal Stem Cells Treated with Growth Differentiation Factor-5 Express Tendon-Specific Markers. Tissue Engineering Part A. 2010;16(9): 2941-2951. [38] FANG J, WANG X, JIANG W, et al. Platelet-Rich Plasma Therapy in the Treatment of Diseases Associated with Orthopedic Injuries. Tissue Eng Part B Rev. 2020;26(6):571-585. [39] UYSAL CA, TOBITA M, HYAKUSOKU H, et al. Adipose-derived stem cells enhance primary tendon repair: biomechanical and immunohistochemical evaluation. J Plast Reconstr Aesthet Surg. 2012; 65(12):1712-1719. [40] QI F, DENG Z, MA Y, et al. From the perspective of embryonic tendon development: various cells applied to tendon tissue engineering. Ann Transl Med. 2020;8(4):131. [41] BI Y, EHIRCHIOU D, KILTS TM, et al. Identification of tendon stem/progenitor cells and the role of the extracellular matrix in their niche. Nat Med. 2007;13(10):1219-1227. [42] AL-ANI MKH, XU K, SUN Y, et al. Study of Bone Marrow Mesenchymal and Tendon-Derived Stem Cells Transplantation on the Regenerating Effect of Achilles Tendon Ruptures in Rats. Stem Cells Int. 2015;2015: 984146. [43] SONG H, YIN Z, WU T, et al. Enhanced Effect of Tendon Stem/Progenitor Cells Combined With Tendon-Derived Decellularized Extracellular Matrix on Tendon Regeneration. Cell Transplant. 2018;27(11):1634-1643. [44] LIU H, ZHANG C, ZHU S, et al. Mohawk Promotes the Tenogenesis of Mesenchymal Stem Cells Through Activation of the TGFβ Signaling Pathway. Stem Cells. 2015;33(2):443-455. [45] WANG Y, HE G, GUO Y, et al. Exosomes from tendon stem cells promote injury tendon healing through balancing synthesis and degradation of the tendon extracellular matrix. J Cell Mol Med. 2019;23(8):5475-5485. [46] TORRICELLI P, VERONESI F, PAGANI S, et al. In vitro tenocyte metabolism in aging and oestrogen deficiency. Age (Dordr). 2013;35(6):2125-2136. [47] BIAN X, LIU T, ZHOU M, et al. Absence of estrogen receptor beta leads to abnormal adipogenesis during early tendon healing by an up‐regulation of PPARγ signalling. J Cell Mol Med. 2019;23(11):7406-7416. [48] DENG D, LIU W, XU F, et al. Engineering human neo-tendon tissue in vitro with human dermal fibroblasts under static mechanical strain. Biomaterials. 2009;30(35):6724-6730. [49] XU Y, WANG Q, LI Y, et al. Cyclic Tensile Strain Induces Tenogenic Differentiation of Tendon-Derived Stem Cells in Bioreactor Culture. Biomed Res Int. 2015;2015:790804. [50] RUI YF, LUI PP, NI M, et al. Mechanical loading increased BMP-2 expression which promoted osteogenic differentiation of tendon-derived stem cells. J Orthop Res. 2011;29(3):390-396. [51] LIU YJ, WANG HJ, XUE ZW, et al. Long noncoding RNA H19 accelerates tenogenic differentiation by modulating miR-140-5p/VEGFA signaling. Eur J Histochem. 2021;65(3):3297-3307. [52] 陈波,施又兴,郭宇鹏,等.不同浓度TGF-β3对体外肌腱干细胞分化的影响研究[J].中国修复重建外科杂志,2015,29(5):620-625. [53] HOLLADAY C, ABBAH SA, O’DOWD C, et al. Preferential tendon stem cell response to growth factor supplementation. J Tissue Eng Regen Med. 2016;10(9):783-798. [54] LUI PP, WONG OT, LEE YW. Transplantation of tendon-derived stem cells pre-treated with connective tissue growth factor and ascorbic acid in vitro promoted better tendon repair in a patellar tendon window injury rat model. Cytotherapy. 2016;18(1):99-112. [55] GUO D, LI H, LIU Y, et al. Fibroblast growth factor-2 promotes the function of tendon-derived stem cells in Achilles tendon restoration in an Achilles tendon injury rat model. Biochem Biophys Res Commun. 2020;521(1):91-97. [56] LI T, XIA M, GAO Y, et al. Human umbilical cord mesenchymal stem cells: an overview of their potential in cell-based therapy. Expert Opin Biol Ther. 2015;15(9):1293-1306. [57] CIARDULLI MC, MARINO L, LAMPARELLI EP, et al. Dose-Response Tendon-Specific Markers Induction by Growth Differentiation Factor-5 in Human Bone Marrow and Umbilical Cord Mesenchymal Stem Cells. Int J Mol Sci. 2020;21(16):5905. [58] YAO Z, LI J, WANG X, et al. MicroRNA-21-3p Engineered Umbilical Cord Stem Cell-Derived Exosomes Inhibit Tendon Adhesion. J Inflamm Res. 2020;13:303-316. [59] HAN Q, WANG S, CHEN D, et al. Exosomes derived from human umbilical cord mesenchymal stem cells reduce tendon injuries via the miR-27b-3p/ARHGAP5/RhoA signaling pathway. Acta Biochim Biophys Sin (Shanghai). 2022;54(2):232-242. [60] LI J, YAO Z, XIONG H, et al. Extracellular vesicles from hydroxycamptothecin primed umbilical cord stem cells enhance anti-adhesion potential for treatment of tendon injury. Stem Cell Res Ther. 2020;11(1):500. [61] 王位,付宇翀,周梅,等.人脐带间充质干细胞联合富含血小板血浆促进大鼠跟腱损伤的修复[J].第三军医大学学报,2017,39(2): 185-191. [62] 冯波,黎啸峰,彭海宁,等.人脐带间充质干细胞的安全性评价[J].激光生物学报,2022,31(1):43-49. [63] HE X, WANG Q, ZHAO Y, et al. Effect of Intramyocardial Grafting Collagen Scaffold With Mesenchymal Stromal Cells in Patients With Chronic Ischemic Heart Disease. JAMA Netw Open. 2020;3(9):e2016236. [64] LV Q, WANG L, LUO X, et al. Adult stem cells in endometrial regeneration: Molecular insights and clinical applications. Mol Reprod Dev. 2021;88(6):379-394. [65] TABATABAEI FS, AI J. Mesenchymal endometrial stem/stromal cells for hard tissue engineering: a review ofin vitro and in vivo evidence. Regen Med. 2017;12(8):983-995. [66] LV H, HU Y, CUI Z, et al. Human menstrual blood: a renewable and sustainable source of stem cells for regenerative medicine. Stem Cell Res Ther. 2018;9(1):325-336. [67] ZHENG Y, ZHOU Y, ZHANG X, et al. Effects of hypoxia on differentiation of menstrual blood stromal stem cells towards tenogenic cells in a co-culture system with Achilles tendon cells. Exp Ther Med. 2017; 13(6):3195-3202. [68] YUKSEL S, GULEC MA, GULTEKIN MZ, et al. Comparison of the early period effects of bone marrow-derived mesenchymal stem cells and platelet-rich plasma on the Achilles tendon ruptures in rats. Connect Tissue Res. 2016;57(5):360-373. [69] ADAMS SB JR, THORPE MA, PARKS BG, et al. Stem Cell-Bearing Suture Improves Achilles Tendon Healing in a Rat Model. Foot Ankle Int. 2014; 35(3):293-299. [70] ZHANG Z, LI Y, ZHANG T, et al. Hepatocyte Growth Factor-Induced Tendon Stem Cell Conditioned Medium Promotes Healing of Injured Achilles Tendon. Front Cell Dev Biol. 2021;9:654084. [71] KOKUBU S, INAKI R, HOSHI K, et al. Adipose-derived stem cells improve tendon repair and prevent ectopic ossification in tendinopathy by inhibiting inflammation and inducing neovascularization in the early stage of tendon healing. Regen Ther. 2020;14:103-110. [72] LACITIGNOLA L, STAFFIERI F, ROSSI G, et al. Survival of bone marrow mesenchymal stem cells labelled with red fluorescent protein in an ovine model of collagenase-induced tendinitis. Vet Comp Orthop Traumatol. 2014;27(3):204-209. [73] FACON-POROSZEWSKA M, KIEŁBOWICZ Z, PRZĄDKA P. Influence of Radial Pressure Wave Therapy (RPWT) on collagenase-induced Achilles tendinopathy treated with Platelet Rich Plasma and Autologous Adipose Derived Stem Cells. Pol J Vet Sci. 2019;22(4):743-751. [74] 裘超,洪建军,毛诚晃,等.局部注射骨髓间充质干细胞和曲安奈德治疗兔跟腱炎的效果对比[J].中国组织工程研究,2012,16(14): 2505-2508. [75] 冯鹏飞,王继宏,冀云涛,等.肌腱组织工程材料在肌腱损伤的应用特点及前景[J].中国组织工程研究,2017,21(18):2940-2945. [76] OUYANG HW, GOH JC, THAMBYAH A, et al. Knitted poly-lactide-co-glycolide scaffold loaded with bone marrow stromal cells in repair and regeneration of rabbit Achilles tendon. Tissue Eng. 2003;9(3):431-439. [77] GOUZMAN I, GROSSMAN E, VERKER R, et al. Advances in Polyimide‐Based Materials for Space Applications. Adv Mater. 2019;31(18): 1807738. [78] CAI J, YANG Y, AI C, et al. Bone Marrow Stem Cells‐Seeded Polyethylene Terephthalate Scaffold in Repair and Regeneration of Rabbit Achilles Tendon. Artif Organs. 2018;42(11):1086-1094. [79] YIN H, STRUNZ F, YAN Z, et al. Three-dimensional self-assembling nanofiber matrix rejuvenates aged/degenerative human tendon stem/progenitor cells. Biomaterials. 2020;236(2020):119802. [80] MIN HK, OH SH, LEE JM, et al. Porous membrane with reverse gradients of PDGF-BB and BMP-2 for tendon-to-bone repair: In vitro evaluation on adipose-derived stem cell differentiation. Acta Biomater. 2014;10(3):1272-1279. [81] JAMES R, KUMBAR SG, LAURENCIN CT, et al. Tendon tissue engineering: adipose-derived stem cell and GDF-5 mediated regeneration using electrospun matrix systems. Biomed Mater. 2011;6(2):25011. [82] ZHANG B, LUO Q, DENG B, et al. Construction of tendon replacement tissue based on collagen sponge and mesenchymal stem cells by coupled mechano-chemical induction and evaluation of its tendon repair abilities. Acta Biomater. 2018;74:247-259. [83] AKTAS E, CHAMBERLAIN CS, SAETHER EE, et al. Immune modulation with primed mesenchymal stem cells delivered via biodegradable scaffold to repair an Achilles tendon segmental defect. J Orthop Res. 2017;35(2):269-280. [84] ZHAO T, QI Y, XIAO S, et al. Integration of mesenchymal stem cell sheet and bFGF-loaded fibrin gel in knitted PLGA scaffolds favorable for tendon repair. J Mater Chem B. 2019;7(13):2201-2211. [85] FADINI GP, MEHTA A, DHINDSA DS, et al. Circulating stem cells and cardiovascular outcomes: from basic science to the clinic. Eur Heart J. 2020;41(44):4271-4282. [86] FADINI GP, BOSCARI F, CAPPELLARI R, et al. Effects of Hypoglycemia on Circulating Stem and Progenitor Cells in Diabetic Patients. J Clin Endocrinol Metab. 2018;103(3):1048-1055. [87] DAHER RJ, CHAHINE NO, RAZZANO P, et al. Tendon repair augmented with a novel circulating stem cell population. Int J Clin Exp Med. 2011; 4(3):214-219. [88] ZHANG M, LIU H, CUI Q, et al. Tendon stem cell-derived exosomes regulate inflammation and promote the high-quality healing of injured tendon. Stem Cell Res Ther. 2020;11(1):402. [89] 吕晶同,项舟.肌腱组织工程生物衍生支架材料[J].生物医学工程学杂志,2013,30(2):451-454. [90] BADYLAK SF, RECORD R, LINDBERG K, et al. Small intestinal submucosa: a substrate for in vitro cell growth. J Biomater Sci Polym Ed. 1998;9(8): 863-878. [91] GUO X, LV H, FAN Z, et al. Effects of hypoxia on Achilles tendon repair using adipose tissue-derived mesenchymal stem cells seeded small intestinal submucosa. J Orthop Surg Res. 2021;16(1):570. [92] 李岱鹤,温树正,王继宏,等.猪小肠黏膜下层复合骨髓间充质干细胞修复大鼠跟腱缺损的组织学和生物力学研究[J].中华手外科杂志,2016,32(3):217-220. [93] WONG C, HUANG Y, CHEN C, et al. Cytokine and Growth Factor Delivery from Implanted Platelet-Rich Fibrin Enhances Rabbit Achilles Tendon Healing. Int J Mol Sci. 2020;21(9):3221. [94] NING LJ, ZHANG YJ, ZHANG YJ, et al. Enhancement of Migration and Tenogenic Differentiation of Macaca Mulatta Tendon-Derived Stem Cells by Decellularized Tendon Hydrogel. Front Cell Dev Biol. 2021;9: 651583. [95] 刘洁,周永春,惠慧.脱细胞肌腱支架对骨髓间充质干细胞增殖分化的影响[J].中国骨与关节损伤杂志,2021,36(11):1156-1159. [96] CHAILAKHYAN RK, KON E, SHEKHTER AB, et al. Autologous bone marrow-derived mesenchymal stem cells provide complete regeneration in a rabbit model of the Achilles tendon bundle rupture. Int Orthop. 2021;45(12):3263-3276. [97] MARTINELLO T, BRONZINI I, VOLPIN A, et al. Successful recellularization of human tendon scaffolds using adipose-derived mesenchymal stem cells and collagen gel. J Tissue Eng Regen Med. 2014;8(8):612-619. [98] LI Y, LIU W, LIU F, et al. Primed 3D injectable microniches enabling low-dosage cell therapy for critical limb ischemia. Proc Natl Acad Sci U S A. 2014;111(37):13511-13516. [99] YANG X, MENG H, PENG J, et al. Construction of Microunits by Adipose-Derived Mesenchymal Stem Cells Laden with Porous Microcryogels for Repairing an Acute Achilles Tendon Rupture in a Rat Model. Int J Nanomedicine. 2020;15:7155-7171. [100] CHEN JL, YIN Z, SHEN WL, et al. Efficacy of hESC-MSCs in knitted silk-collagen scaffold for tendon tissue engineering and their roles. Biomaterials. 2010;31(36):9438-9451. [101] TANG L, YANG Y, LI Y, et al. Knitted silk mesh-like scaffold incorporated with sponge-like regenerated silk fibroin/collagen I and seeded with mesenchymal stem cells for repairing Achilles tendon in rabbits. Acta Bioeng Biomech. 2018;20(4):77-87. |
| [1] | Nong Fuxiang, Jiang Zhixiong, Li Yinghao, Xu Wencong, Shi Zhilan, Luo Hui, Zhang Qinglang, Zhong Shuang, Tang Meiwen. Bone cement augmented proximal femoral nail antirotation for type A3.3 intertrochanteric femoral fracturalysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-10. |
| [2] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [3] | Cai Zhihao, Xie Zhaoyong. Femoral neck anteversion measurement assessment: how to establish a unified method and standard [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1448-1454. |
| [4] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [5] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [6] | Gao Yu, Han Jiahui, Ge Xin. Immunoinflammatory microenvironment after spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1300-1305. |
| [7] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [8] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [9] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [10] | Chen Shisong, Liu Xiaohong, Xu Zhiyun. Current status and prospects of bioprosthetic heart valves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1096-1102. |
| [11] | Lu Di, Zhang Cheng, Duan Rongquan, Liu Zongxiang. Osteoinductive properties of calcium phosphate ceramic bone repair materials [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1103-1109. |
| [12] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [13] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [14] | Xue Ting, Zhang Xinri, Kong Xiaomei. Mesenchymal stem cell therapy for pneumoconiosis using nanomaterials combined with multi-modal molecular imaging [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1133-1140. |
| [15] | Li Qicheng, Deng Jin, Fu Xiaoyang, Han Na. Effects of bone marrow mesenchymal stem cells-derived exosomes on hypoxia-treated myoblasts [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 853-859. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||