Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (15): 2379-2384.doi: 10.12307/2023.376
Previous Articles Next Articles
Cell co-culture system of dental pulp regenerative tissue engineering
Zhang Chuhan1, Zhang Dongmin2, Xu Wenan1
- 1Shenzhen Stomatology Hospital (Pingshan), Southern Medical University, Shenzhen 510811, Guangdong Province, China; 2School of Stomatology, Southern Medical University, Guangzhou 510515, Guangdong Province, China
-
Received:2022-04-14Accepted:2022-07-07Online:2023-05-28Published:2022-10-18 -
Contact:Xu Wenan, Associate professor, Associate chief physician, Shenzhen Stomatology Hospital (Pingshan), Southern Medical University, Shenzhen 510811, Guangdong Province, China -
About author:Zhang Chuhan, Master candidate, Shenzhen Stomatology Hospital (Pingshan), Southern Medical University, Shenzhen 510811, Guangdong Province, China -
Supported by:National Natural Science Foundation of China (Youth Project), No. 81700956 (to XWA); Shenzhen Stomatological Hospital (Pingshan) Dean Fund of Southern Medical University, No. 2021A001 (to XWA)
CLC Number:
Cite this article
Zhang Chuhan, Zhang Dongmin, Xu Wenan. Cell co-culture system of dental pulp regenerative tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(15): 2379-2384.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
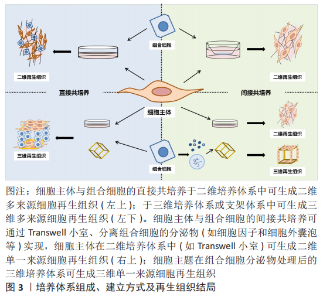
2.1 共培养体系的组成 2.1.1 细胞主体 目前用于构建牙髓再生组织工程共培养体系的细胞主体为口腔来源的间充质干细胞,如牙髓干细胞、脱落乳牙牙髓干细胞、牙周膜干细胞、牙囊干细胞和根尖牙乳头干细胞等。牙源性间充质干细胞在体内和体外均具有牙髓组织再生能力,而且具有易获取、易培养的优点,无论是牙髓组织再生领域还是口腔外组织再生均有广泛运用,但由于单一细胞的生成组织的结构与功能与天然组织有差异,因此,引入其他细胞共同培养能提供更好的细胞微环境[4]。 2.1.2 组合细胞 组合细胞的来源主要包括终末分化成熟的成体细胞,如人脐静脉内皮细胞;成体干/祖细胞,如间充质干细胞和内皮祖细胞;胚胎干细胞;诱导多能干细胞。组合细胞要求具有一定的调控增殖分化能力,并且本身细胞源容易获取或可以在细胞主体和组合细胞的相互作用下可分化为组织必需的结构。体外构建细胞共培养系统的方式通过引入组合细胞,可更好地模拟体内细胞生长环境,研究细胞间及细胞与环境之间的相互作用和信号通路[5]。 牙髓再生组织工程中的细胞共培养组合类型众多,多数为牙源性干细胞和内皮细胞共培养,其中牙髓干细胞和人脐静脉内皮细胞联合培养体系应用最为广泛[6]。已知内皮细胞和牙源性干细胞可分泌一系列有利于细胞生长和分化的生长因子,当二者进行共培养时,一方面内皮细胞提供的细胞外基质环境可以促进牙源性干细胞的增殖及分化能力,另一方面,牙源性干细胞作为周细胞样细胞,可为内皮细胞的血管化提供局部微环境,增强新生血管的稳定性[7-9]。 2.2 共培养体系的模型和维度 细胞共培养根据模型一般可分为直接接触共培养、间接接触共培养,而根据形成组织的维度,又可分为二维细胞培养和三维细胞共培养,二者可交叉使用,如直接接触的二维细胞培养等,见图3。目前牙髓再生的共培养模型多为间接接触的二维细胞培养。"

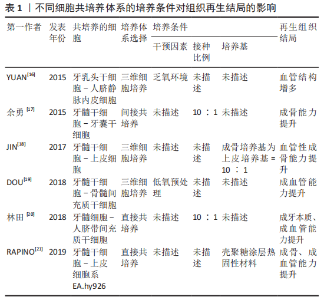
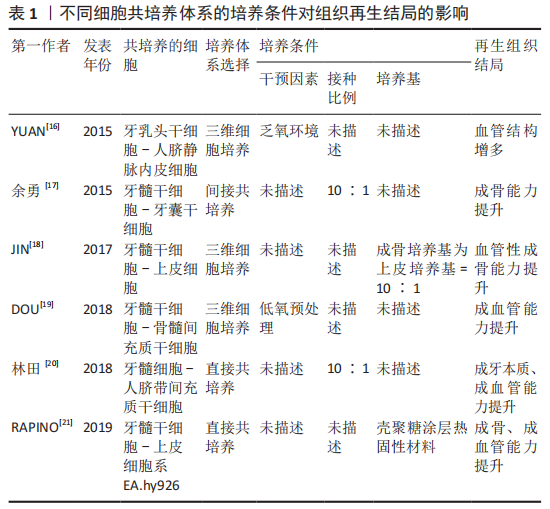
2.2.1 直接接触培养 是指将不同细胞放在同一培养体系中,细胞之间直接接触。直接接触共培养中细胞主体和组合细胞间通过细胞外基质或细胞接触相互通信,既促进细胞主体形成目标组织,又使组合细胞形成目标组织必须的周围支持组织,从而实现组织再生。DISSANAYAKA等[10]研究发现将牙髓干细胞与人脐静脉内皮细胞进行直接共培养,可提高牙髓干细胞成牙/成骨潜能和人脐静脉内皮细胞的血管生成能力。 直接接触共培养可形成有整体性的组织,但是也有其短板:因为细胞处于同种环境,无法完全区分不同种细胞分泌的生长因子等物质,无法对相互作用的机制进行更深入的研究。由于培养手段的影响,若无生物支架介入,形成的再生组织则无生理性组织的三维结构,同时直接接触培养体系中不同细胞的接种比例对组织再生的结局有决定性影响[11]。 2.2.2 间接接触培养 是将不同细胞置于同一培养体系中,但不同细胞之间无直接接触,如嵌入式细胞共培养(Transwell小室)、细胞衍生物共培养等,细胞之间通过旁分泌途径产生单向作用或相互作用,从而促进组织再生。MERCKX等[12]通过向牙髓干细胞培养基中添加骨髓间充质干细胞的条件培养基以及细胞外小囊泡,构建了牙髓干细胞-骨髓间充质干细胞间接共培养体系,发现牙髓干细胞的组织再生能力不但有所增强,还可向内皮损伤部位迁移。 相比直接接触培养,间接接触培养不但可以准确说明细胞间通讯作用机制的影响,同时还可以对细胞的趋化、迁移能力进行观察,而细胞衍生物共培养作为一种脱细胞介入手段可以避免免疫排异反应并提高干细胞的利用率,与此同时,对如细胞条件培养基、细胞外囊泡等细胞衍生物的分离、成分分析以及作用机制也成为研究热点之一。但是由于细胞间无直接接触,无法分析细胞直接接触产生的细胞通讯作用,也无法分析复杂组织中细胞外基质介导的细胞间信号传递。 2.2.3 三维细胞共培养 三维细胞共培养即将细胞接种于具有三维结构的生物支架材料上或细胞三维成团培养,为细胞提供近似于体内的生长环境,其优点是能最大限度地有序还原组织原有形态,缺点则是生物支架材料和三维成团培养的成本和技术敏感性过高,并且研究者还在进一步探索最优材料。 安莉等[13]将牙髓干细胞和人脐静脉内皮细胞接种于基质胶上,并且在共培养体系中加入肿瘤坏死因子α,结果表明肿瘤坏死因子α可促进共培养系统体外成血管能力,同时在共培养体系中,牙髓干细胞可分泌血管内皮生长因子、成纤维细胞生长因子及血小板衍生生长因子等多种促血管生长因子,促进内皮细胞的存活和分化;人脐静脉内皮细胞同样分泌信号分子和趋化因子来影响牙髓干细胞的生长和分化能力。体内实验包括免疫缺陷小鼠皮下移植和组织工程牙齿种植。HONDA等[14]将间充质细胞和人口腔上皮细胞以1∶1比例接种于胶原蛋白海绵上,并将其移植到裸鼠皮下,最终发现这种细胞接种技术可以有效地调控再生牙的形态。LIU等[15]将牙周膜干细胞和牙髓干细胞接种到体外制备的羟基磷灰石-磷酸三钙支架材料(HA/TCP)上,将其移入生物根并种植于小型猪的下颌骨中,6个月后观察到牙根样结构和牙本质样结构形成。 2.3 牙髓再生组织工程共培养体系的培养条件 细胞共培养的条件包括:细胞的接种密度与比例、培养基的选择、培养时间和干预因素等。如安莉等[13]还在实验过程中发现的共培养体系中以1∶5的比例进行共培养,其血管的形成优于其他实验组且结构最为稳定,2种细胞的比例是共培养结果的关键,不同比例下细胞之间的交流产生不同的实验效应。 除了细胞主体及组合细胞的选择、共培养体系的选择外,细胞共培养体系的培养条件对组织再生结局也有重要影响[16-21],详见表1。"
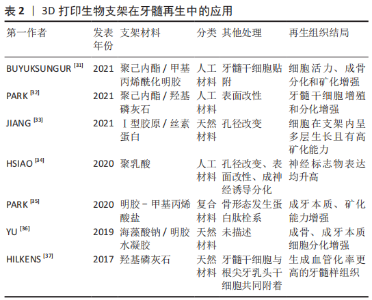
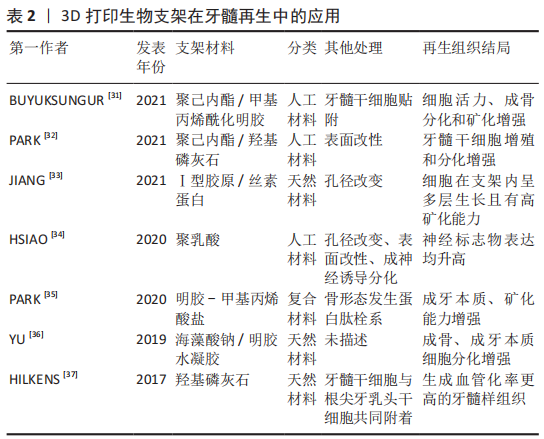
2.4 生物支架材料的应用 牙髓再生中三维支架材料的应用不仅能为细胞提供黏附、增殖和迁移的场所,还有构建组织三维结构的功能以及通过不同材料特性达到控释细胞因子的作用,有助于细胞的成血管化和成牙本质分化[22]。常用的支架材料可分为天然材料、人工合成材料和复合材料。天然材料是指由动植物组织中提取的高分子可降解材料,具有良好的生物相容性,如胶原、水凝胶和脱细胞支架等。人工合成材料主要包括聚乳酸、聚乙醇酸等,无机材料主要以钙盐为基础,如羟基磷灰石和聚磷酸钙等。由于任何单一支架材料均难以满足牙髓组织对生长环境的要求,而复合材料是将天然材料与人工合成材料结合,以此不同材料的特点和优势来实现某些特定的功能,已成为目前组织工程生物支架材料研究的热点[23-24]。在牙髓再生研究中首次应用支架材料,是由YOUNG等[25]将牙髓单细胞悬液接种在聚乳酸-聚乙醇酸支架上,将其移植到免疫缺陷大鼠宿主体内生长20-30周后,观察到牙齿结构的形成。目前多数用于牙髓再生研究的材料都具有一定的可塑性和流动性,以适应根管的解剖形态[26],前文所提DISSANAYAKA等[27-28]采用琼脂糖微模具制备牙髓干细胞-人脐带血管内皮细胞共培养的三维微组织球体,结果发现经过预血管化的组织可形成更为稳定的毛细血管结构,在体内体外都表现出更高的骨/牙源性基因表达水平和矿化水平。 3D打印生物材料已在组织工程学中广泛运用,其高个性化、便捷、可设计等性能为未来的组织工程再生提供诸多可能性。随着材料技术发展,3D打印已可以涉猎水凝胶一类形变能力极强、能适应不同解剖结构的材料,因而3D打印生物支架逐步进入到牙髓再生领域中[29-30],因此3D打印生物材料在近5年也在不同研究中发挥着诱导细胞形成三维组织形态、促进细胞增殖分化的重要作用[31-37],见表2。"
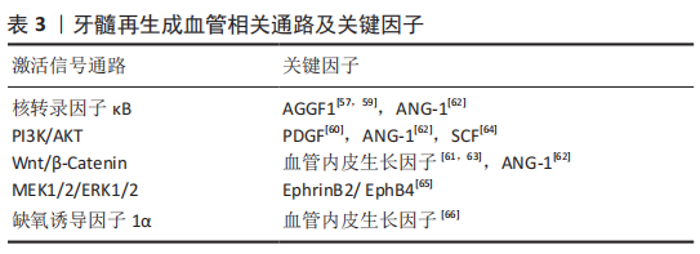
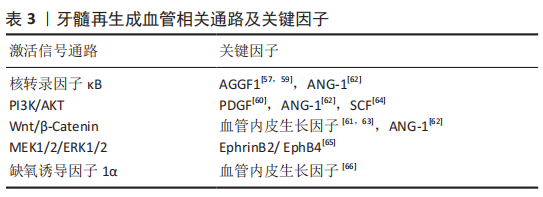
2.5 牙髓再生组织工程共培养体系的作用机制 细胞之间的交互作用通过以下3种机制实现:①细胞表面分子的直接接触;②邻近细胞间的缝隙连接;③分泌到细胞外环境中的信号分子与目标细胞的特异受体相互作用:细胞之间释放扩散性分泌因子,如细胞因子、生长因子等[38-39]。无论是细胞表面分子的直接接触还是缝隙连接,由于细胞间有直接接触且细胞交流时不向外分泌信号分子等介质,可将二者归类为细胞间的直接通讯,故文章根据细胞间的直接作用和间接作用展开阐述牙髓再生组织工程共培养体系中的细胞交互机制。 2.5.1 共培养细胞间的直接作用 细胞表面分子的直接接触多见于免疫反应中的抗原呈递过程,在共培养中细胞主体与组合细胞间鲜有涉及。而缝隙连接是一种细胞间直接通讯方式,相邻细胞间通过细胞膜上的连接子相互锚定,允许相对分子质量小于 1 500的离子、小分子代谢物 (如氨基酸、核苷酸、维生素和糖类等)及第二信使(如cAMP,Ca2+,IP3)通过[40-41]。缝隙连接蛋白(connexin,Cx)是构成缝隙连接的基本单位,在哺乳动物体内分布广泛。牙髓组织中主要表达Cx26,Cx32和Cx43[42-43],成骨细胞的主要连接蛋白为Cx43[44],内皮细胞为Cx37,Cx40和Cx4[45]。在牙髓再生组织工程共培养体系中关于细胞间直接通讯作用的研究较少,但在其他学科领域的细胞共培养体系中,细胞间的信号通讯作用主要通过Cx43介导实现,如成骨细胞与血管内皮细胞[46]、单核细胞与内皮细胞[47-48]。同样地,亦有研究表明成牙本质细胞间的缝隙连接是由Cx43所介导的[49],LI等[50]研究发现牙髓干细胞通过Cx43介导的Ca2+在细胞间相互交流,促进牙本质涎磷蛋白的表达和成牙分化。因此在牙髓再生共培养体系中,细胞间的直接作用很有可能是通过Cx43介导实现,但仍需更多证据支持。 2.5.2 共培养细胞间的间接作用 共培养体系中细胞之间通过分泌到细胞外环境中的信号分子与目标细胞的特异受体相互作用,即通过旁分泌/自分泌的生长因子/细胞因子发挥间接接触作用[51-66]。 健康牙髓由血管、神经和胶原纤维等部分组成,对于牙髓再生组织工程而言,需要形成与之形态功能相似的牙髓样组织。在此过程中,不同方向的分化与组织形成具有不同机制,其中成血管是牙髓组织再生的关键,健康组织中血管提供细胞和组织生命活动所必须的营养物质,而在组织再生相关组织工程研究中,血管不仅是物质运输的通道,还是富含趋化因子的细胞归巢支架,同时还承担血管化骨再生的功能[51],因此文章主要对牙髓再生中成血管机制进行总结,见表3。 IWAYA等[52]在2001年的病例报道中首次引用牙髓血运重建这一概念,而与此同时血管内皮生长因子的发现改变了研究者对血管生成和发育过程中血管生成以及生理稳态的理解[53],在牙髓再生过程中,血管内皮生长因子通过与细胞膜上的受体结合,发生一系列的生理过程从而影响细胞增殖、新生血管生成,具体过程如下:血管内皮生长因子A与细胞膜上的受体及跨膜蛋白结合,受体自身磷酸化或二聚化,经过胞内一系列底物及蛋白的活化水解,引起信号级联反应,使细胞大量钙离子通道开放,引发钙内流,进而释放一氧化氮和前列腺素,从而调节血管的生成[54-56]。在牙髓干细胞-人脐静脉内皮细胞共培养体系中,肿瘤坏死因子α和脂多糖可激活血管内皮生长因子-SIRT1信号通路,并激活下游核转录因子κB信号从而增强人脐静脉内皮细胞和牙髓干细胞的迁移能力和新形成血管结构的稳定性[57]。此外,其他的研究表明血管内皮生长因子调节血管生成还有可能与PI3K/AKT信号通路以及MEK1/2/ERK1/2信号通路[58]。随着细胞间交流关键生物因子的提纯以及信号通路的发现,研究者揭示了越来越多与牙髓再生成血管相关的关键因子以及被关键因子激活的信号通路,且时至今日牙髓再生的成血管机制依旧是研究热点。"
| [1] SUI B, CHEN C, KOU X, et al. Pulp stem cell-mediated functional pulp regeneration. J Dent Res. 2019;98(1):27-35. [2] GRONTHOS S, MANKANI M, BRAHIM J, et al. Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc Natl Acad Sci U S A. 2000; 97(25):13625-13630. [3] CHEUNG VC, PENG CY, MARINIĆ M, et al. Pluripotent stem cell-derived endometrial stromal fibroblasts in a cyclic, hormone-responsive, coculture model of human decidua. Cell Rep. 2021;35(7):109138. [4] ANITUA E, TROYA M, ZALDUENDO M. Progress in the use of dental pulp stem cells in regenerative medicine. Cytotherapy. 2018;20(4):479-498. [5] BATTISTON KG, CHEUNG JW, JAIN D, et al. Biomaterials in co-culture systems: towards optimizing tissue integration and cell signaling within scaffolds. Biomaterials. 2014;35(15):4465-4476. [6] TRAN-HUNG L, LAURENT P, CAMPS J, et al. Quantification of angiogenic growth factors released by human dental cells after injury. Arch Oral Biol. 2008;53(1):9-13. [7] DISSANAYAKA WL, ZHU L, HARGREAVES KM, et al. In vitro analysis of scaffold-free prevascularized microtissue spheroids containing human dental pulp cells and endothelial cells. J Endod. 2015;41(5):663-670. [8] 黄雨亭,沈帅,蒋鹏飞,等.EphrinB2对TNF-α作用下的脐静脉内皮细胞与牙髓干细胞血管生成的影响[J].实用口腔医学杂志,2021,37(6): 773-777. [9] 吴一梦,张爽,潘爽,等.脐静脉内皮细胞与牙本质浸提液诱导的牙髓干细胞共培养成血管的研究[J].口腔医学研究,2017,33(5):504-508. [10] DISSANAYAKA WL, ZHAN X, ZHANG C, et al. Coculture of dental pulp stem cells with endothelial cells enhances osteo-/odontogenic and angiogenic potential in vitro. J Endod. 2012;38(4):454-463. [11] LI M, WANG Q, HAN Q, et al. Novel molecule nell-1 promotes the angiogenic differentiation of dental pulp stem cells. Front Physiol. 2021;12:703593. [12] MERCKX G, HOSSEINKHANI B, KUYPERS S, et al. Angiogenic effects of human dental pulp and bone marrow-derived mesenchymal stromal cells and their extracellular vesicles. Cells. 2020;9(2):312. [13] 安莉,沈帅,王璐瑶,等.TNF-α增强牙髓干细胞与脐静脉内皮细胞共培养血管生成能力[J].口腔医学研究,2019,35(10):966-969. [14] HONDA MJ, TSUCHIYA S, SUMITA Y, et al. The sequential seeding of epithelial and mesenchymal cells for tissue-engineered tooth regeneration. Biomaterials. 2007;28(4):680-689. [15] LIU M, ZHAO L, HU J, et al. Endothelial cells and endothelin 1 promote the odontogenic differentiation of dental pulp stem cells. Mol Med Rep. 2018; 18(1):893-901. [16] YUAN C, WANG P, ZHU L, et al. Coculture of stem cells from apical papilla and human umbilical vein endothelial cell under hypoxia increases the formation of three-dimensional vessel-like structures in vitro. Tissue Eng Part A. 2015;21(5-6):1163-1172. [17] 余勇,陶昱,邓锋,王睿,等.体外共培养人牙髓干细胞对人牙囊干细胞增殖、成骨分化的作用[J].第三军医大学学报,2015,37(22):2267-2272. [18] JIN GZ, KIM HW. Co-culture of human dental pulp stem cells and endothelial cells using porous biopolymer microcarriers: a feasibility study for bone tissue engineering. Tissue Eng Regen Med. 2017;14(4):393-401. [19] DOU L, YAN Q, LIANG P, et al. iTRAQ-based proteomic analysis exploring the influence of hypoxia on the proteome of dental pulp stem cells under 3D culture. Proteomics. 2018. doi: 10.1002/pmic.201700215. [20] 林田,赵文青,陆彦玲,等.人脐带间充质干细胞与牙髓细胞体外共培养对细胞生物学的影响[J].上海口腔医学,2018,27(4):365-369. [21] RAPINO M, DI VALERIO V, ZARA S, et al. Chitlac-coated thermosets enhance osteogenesis and angiogenesis in a co-culture of dental pulp stem cells and endothelial cells. Nanomaterials (Basel). 2019;9(7):928. [22] RADDALL G, MELLO I, LEUNG BM. Biomaterials and scaffold design strategies for regenerative endodontic therapy. Front Bioeng Biotechnol. 2019;7:317. [23] 刘阳,莫春香,贺艳,等.组织工程支架材料研究进展[J].化工新型材料,2019,47(12):37-40. [24] BADYLAK SF, FREYTES DO, GILBERT TW. Extracellular matrix as a biological scaffold material: Structure and function. Acta Biomater. 2009;5(1):1-13. [25] YOUNG CS, TERADA S, VACANTI JP, et al. Tissue engineering of complex tooth structures on biodegradable polymer scaffolds. J Dent Res. 2002; 81(10):695-700. [26] MOUSSA DG, APARICIO C. Present and future of tissue engineering scaffolds for dentin-pulp complex regeneration. J Tissue Eng Regen Med. 2019;13(1): 58-75. [27] DISSANAYAKA WL, ZHU L, HARGREAVES KM, et al. Scaffold-free prevascularized microtissue spheroids for pulp regeneration. J Dent Res. 2014;93(12):1296-1303. [28] DISSANAYAKA WL, ZHU L, HARGREAVES KM, et al. In vitro analysis of scaffold-free prevascularized microtissue spheroids containing human dental pulp cells and endothelial cells. J Endod. 2015;41(5):663-670. [29] TURNBULL G, CLARKE J, PICARD F, et al. 3D bioactive composite scaffolds for bone tissue engineering. Bioact Mater. 2017;3(3):278-314. [30] MA Y, XIE L, YANG B, et al. Three-dimensional printing biotechnology for the regeneration of the tooth and tooth-supporting tissues. Biotechnol Bioeng. 2019;116(2):452-468. [31] BUYUKSUNGUR S, HASIRCI V, HASIRCI N. 3D printed hybrid bone constructs of PCL and dental pulp stem cells loaded GelMA. J Biomed Mater Res A. 2021;109(12):2425-2437. [32] PARK S, KIM JE, HAN J, et al. 3D-printed poly (ε-caprolactone)/hydroxyapatite scaffolds modified with alkaline hydrolysis enhance osteogenesis in vitro. Polymers (Basel). 2021;13(2):257. [33] JIANG S, YU Z, ZHANG L, et al. Effects of different aperture-sized type I collagen/silk fibroin scaffolds on the proliferation and differentiation of human dental pulp cells. Regen Biomater. 2021;8(4):rbab028. [34] HSIAO D, HSU SH, CHEN RS, et al. Characterization of designed directional polylactic acid 3D scaffolds for neural differentiation of human dental pulp stem cells. J Formos Med Assoc. 2020;119(1 Pt 2):268-275. [35] PARK JH, GILLISPIE GJ, COPUS JS, et al. The effect of BMP-mimetic peptide tethering bioinks on the differentiation of dental pulp stem cells (DPSCs) in 3D bioprinted dental constructs. Biofabrication. 2020;12(3):035029. [36] YU H, ZHANG X, SONG W, et al. Effects of 3-dimensional bioprinting alginate/gelatin hydrogel scaffold extract on proliferation and differentiation of human dental pulp stem cells. J Endod. 2019;45(6):706-715. [37] HILKENS P, BRONCKAERS A, RATAJCZAK J, et al. The angiogenic potential of dpscs and scaps in an in vivo model of dental pulp regeneration. Stem Cells Int. 2017;2017:2582080. [38] THURLEY K, WU LF, ALTSCHULER SJ. Modeling cell-to-cell communication networks using response-time distributions. Cell Syst. 2018;6(3):355-367.e5. [39] CORDERO CERVANTES D, ZURZOLO C. Peering into tunneling nanotubes-The path forward. EMBO J. 2021;40(8):e105789. [40] BEYER EC, BERTHOUD VM. Gap junction gene and protein families: connexins, innexins, and pannexins. Biochim Biophys Acta Biomembr. 2018; 1860(1):5-8. [41] VINKEN M, VANHAECKE T, PAPELEU P, et al. Connexins and their channels in cell growth and cell death. Cell Signal. 2006;18(5):592-600. [42] YIN J, XU J, CHENG R, et al. Role of connexin 43 in odontoblastic differentiation and structural maintenance in pulp damage repair. Int J Oral Sci. 2021;13(1):1. [43] LIU W, CUI Y, WEI J, et al. Gap junction-mediated cell-to-cell communication in oral development and oral diseases: a concise review of research progress. Int J Oral Sci. 2020;12(1):17. [44] QIN L, LIU W, CAO H, et al. Molecular mechanosensors in osteocytes. Bone Res. 2020;8:23. [45] SIMON AM, MCWHORTER AR. Decreased intercellular dye-transfer and downregulation of non-ablated connexins in aortic endothelium deficient in connexin 37 or connexin 40. J Cell Sci. 2003;116(Pt 11):2223-2236. [46] LI M, ZHANG A, LI J, et al. Osteoblast/fibroblast coculture derived bioactive ECM with unique matrisome profile facilitates bone regeneration. Bioact Mater. 2020;5(4):938-948. [47] CHAI Y, YU R, LIU Y, et al. Dexmedetomidine attenuates monocyte-endothelial adherence via inhibiting connexin43 on vascular endothelial cells. Mediators Inflamm. 2020;2020:7039854. [48] JI H, QIU R, GAO X, et al. Propofol attenuates monocyte-endothelial adhesion via modulating connexin43 expression in monocytes. Life Sci. 2019;232:116624. [49] MURAMATSU T, HASHIMOTO S, SHIBUKAWA Y, et al. Immunoelectron microscopic observation of connexin43 in rat odontoblasts. Microsc Res Tech. 2013;76(10):988-991. [50] LI S, HU J, ZHANG G, et al. Extracellular Ca2+ promotes odontoblastic differentiation of dental pulp stem cells via bmp2-mediated Smad1/5/8 and Erk1/2 pathways. J Cell Physiol. 2015;230(9):2164-2173. [51] MATSUBARA T, IGA T, SUGIURA Y, et al. Coupling of angiogenesis and odontogenesis orchestrates tooth mineralization in mice. J Exp Med. 2022; 219(4):e20211789. [52] IWAYA SI, IKAWA M, KUBOTA M. Revascularization of an immature permanent tooth with apical periodontitis and sinus tract. Dent Traumatol. 2001;17(4):185-187. [53] APTE RS, CHEN DS, FERRARA N. VEGF in signaling and disease: beyond discovery and development. Cell. 2019;176(6):1248-1264. [54] SIMONS M, GORDON E, CLAESSON-WELSH L. Mechanisms and regulation of endothelial VEGF receptor signalling. Nat Rev Mol Cell Biol. 2016;17(10): 611-625. [55] CHENG HW, JAMES AF, FOSTER RR, et al. VEGF activates receptor-operated cation channels in human microvascular endothelial cells. Arterioscler Thromb Vasc Biol. 2006;26(8):1768-1776. [56] HEINOLAINEN K, KARAMAN S, D’AMICO G, et al. VEGFR3 modulates vascular permeability by controlling VEGF/VEGFR2 signaling. Circ Res. 2017; 120(9):1414-1425. [57] SHIN MR, KANG SK, KIM YS, et al. TNF-α and LPS activate angiogenesis via VEGF and SIRT1 signalling in human dental pulp cells. Int Endod J. 2015; 48(7):705-716. [58] WANG HJ, RAN HF, YIN Y, et al. Catalpol improves impaired neurovascular unit in ischemic stroke rats via enhancing VEGF-PI3K/AKT and VEGF-MEK1/2/ERK1/2 signaling. Acta Pharmacol Sin. 2022;43(7):1670-1685. [59] SHEN S, SHANG L, LIU H, et al. AGGF1 inhibits the expression of inflammatory mediators and promotes angiogenesis in dental pulp cells. Clin Oral Investig. 2021;25(2):581-592. [60] ZHANG M, JIANG F, ZHANG X, et al. The effects of platelet-derived growth factor-bb on human dental pulp stem cells mediated dentin-pulp complex regeneration. Stem Cells Transl Med. 2017;6(12):2126-2134. [61] ZHANG Z, NÖR F, OH M, et al. Wnt/β-catenin signaling determines the vasculogenic fate of postnatal mesenchymal stem cells. Stem Cells. 2016; 34(6):1576-1587. [62] YUN HM, KANG SK, SINGH RK, et al. Magnetic nanofiber scaffold-induced stimulation of odontogenesis and pro-angiogenesis of human dental pulp cells through Wnt/MAPK/NF-κB pathways. Dent Mater. 2016;32(11): 1301-1311. [63] LEE SI, KIM SY, PARK KR, et al. Baicalein promotes angiogenesis and odontoblastic differentiation via the BMP and Wnt pathways in human dental pulp cells. Am J Chin Med. 2016;44(7):1457-1472. [64] PAN S, DANGARIA S, GOPINATHAN G, et al. SCF promotes dental pulp progenitor migration, neovascularization, and collagen remodeling - potential applications as a homing factor in dental pulp regeneration. Stem Cell Rev Rep. 2013;9(5):655-667. [65] GONG T, XU J, HENG B, et al. EphrinB2/EphB4 signaling regulates DPSCs to induce sprouting angiogenesis of endothelial cells. J Dent Res. 2019; 98(7):803-812. [66] YUAN C, WANG P, ZHU L, et al. Coculture of stem cells from apical papilla and human umbilical vein endothelial cell under hypoxia increases the formation of three-dimensional vessel-like structures in vitro. Tissue Eng Part A. 2015;21(5-6):1163-1172. [67] HUANG GT. Dental pulp and dentin tissue engineering and regeneration: advancement and challenge. Front Biosci (Elite Ed). 2011;3(2):788-800. |
| [1] | Sun Kexin, Zeng Jinshi, Li Jia, Jiang Haiyue, Liu Xia. Mechanical stimulation enhances matrix formation of three-dimensional bioprinted cartilage constructs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [2] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [3] | Yang Yitian, Wang Lu, Yao Wei, Zhao Bin. Application of the interaction between biological scaffolds and macrophages in bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1071-1079. |
| [4] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [5] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [6] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [7] | Xu Yan, Li Ping, Lai Chunhua, Zhu Peijun, Yang Shuo, Xu Shulan. Piezoelectric materials for vascularized bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1126-1132. |
| [8] | Li Xinyue, Li Xiheng, Mao Tianjiao, Tang Liang, Li Jiang. Three-dimensional culture affects morphology, activity and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 846-852. |
| [9] | Li Xiaoyin, Yang Xiaoqing, Chen Shulian, Li Zhengchao, Wang Ziqi, Song Zhen, Zhu Daren, Chen Xuyi. Collagen/silk fibroin scaffold combined with neural stem cells in the treatment of traumatic spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 890-896. |
| [10] | Yuan Bo, Xie Lide, Fu Xiumei. Schwann cell-derived exosomes promote the repair and regeneration of injured peripheral nerves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 935-940. |
| [11] | Qin Yuxing, Ren Qiangui, Li Zilong, Quan Jiaxing, Shen Peifeng, Sun Tao, Wang Haoyu. Action mechanism and prospect of bone microvascular endothelial cells for treating femoral head necrosis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 955-961. |
| [12] | Xiong Bohan, Yu Yang, Lu Xiaojun, Wang Xu, Yang Tengyun, Zhang Yaozhang, Liao Xinyu, Zhou Xiaoxiang, He Lu, Li Yanlin. Research progress in promoting tendon to bone healing during anterior cruciate ligament reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 779-786. |
| [13] | Zhang Min, Zhang Xiaoming, Liu Tongbin. Application potential of naringin in bone tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 787-792. |
| [14] | Ning Ziwen, Wang Xu, Shi Zhengliang, Qin Yihua, Wang Guoliang, Jia Di, Wang Yang, Li Yanlin. Meniscal injury repair methods for non-blood supply area [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 420-426. |
| [15] | Zong Mingrui, Liu Haiyan, Li Bing, Wu Xiuping. Application of carboxymethyl chitosan in tissue engineering of stomatology [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 447-452. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||