Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (6): 921-927.doi: 10.12307/2023.301
Previous Articles Next Articles
Application potential of adipose-derived stem cells in female pelvic floor dysfunction diseases
Bai Siqi, Xiao Zhen, Liu Jing
- First Affiliated Hospital of Dalian Medical University, Dalian 116011, Liaoning Province, China
-
Received:2022-03-26Accepted:2022-06-02Online:2023-02-28Published:2022-08-12 -
Contact:Xiao Zhen, MD, Associate professor, First Affiliated Hospital of Dalian Medical University, Dalian 116011, Liaoning Province, China Liu Jing, MD, Professor, First Affiliated Hospital of Dalian Medical University, Dalian 116011, Liaoning Province, China -
About author:Bai Siqi, Master candidate, First Affiliated Hospital of Dalian Medical University, Dalian 116011, Liaoning Province, China
CLC Number:
Cite this article
Bai Siqi, Xiao Zhen, Liu Jing. Application potential of adipose-derived stem cells in female pelvic floor dysfunction diseases[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 921-927.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
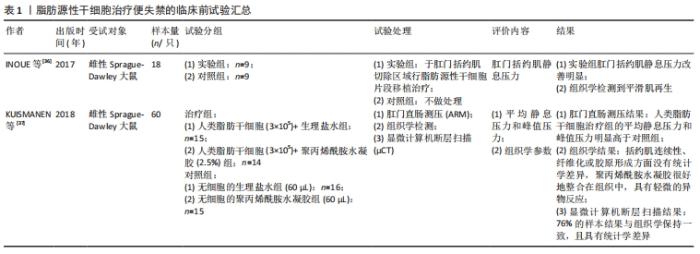
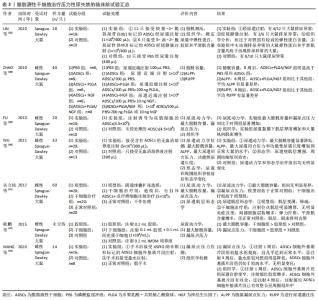
2.1 脂肪源性干细胞的来源、培养及特点 脂肪组织中的基质血管细胞成分(stromal-vascular cell fraction,SVF)是多能干细胞的丰富来源,脂肪源性干细胞是指从脂肪组织中获取的基质血管细胞成分内具有多向分化潜能的细胞,这些干细胞被称为脂肪来源的干细胞或脂肪组织来源的基质细胞[10]。脂肪源性干细胞主要来源于脂肪组织,而脂肪组织遍布于身体各处,最常见的获取部位为皮下。由于白色脂肪中基质血管细胞成分的获取率较高[11],通常认为皮下白色脂肪组织是脂肪源性干细胞的主要来源。有研究发现,从200 mL经过处理的人类皮下脂肪组织中,可获取大约750万个干细胞[12],这提示脂肪源性干细胞的来源丰富,在人体中存储量巨大,使得分离和提取足够数量的干细胞难度显著降低,采集脂肪组织的外科过程本身相对收集其他来源干细胞对人体的影响也更小[13]。 细胞外基质是细胞生长的天然材料,其中盆底支持组织细胞外基质的主要成分是弹性蛋白、胶原蛋白、蛋白聚糖、层粘连蛋白和纤连蛋白[14]。在组织工程研究中,干细胞需要与支架材料结合形成细胞-材料复合体,在体外或体内适宜条件下培养,使干细胞在三维空间中生长、分化,最终形成所需的功能组织。相比细胞外基质,理想的支架材料应该具备良好的生物降解性、组织相容性和三维空间结构。水凝胶是一种很好的模拟天然细胞外基质的三维材料,用水凝胶搭载干细胞或细胞因子等用于盆底疾病的治疗也成为了近年来的研究热点[15]。聚丙烯酰胺作为目前最为常见的水凝胶材料,其化学性能稳定,无细胞毒性、无胚胎毒性、无致癌性,进入人体组织后不受催化剂影响,不易降解[16-17]。有研究显示,人脱细胞脂肪组织水凝胶能够携带脂肪源性干细胞到达相应部位,且脂肪源性干细胞包埋的人类脱细胞脂肪组织水凝胶在体温下即可形成,为脂肪源性干细胞提供了培养平台[18]。除此之外,以壳聚糖、β-甘油磷酸酯和胶原为原料制备的杂化水凝胶支架,能与有机物组织具有良好的组织相容性[17],其在低温态下为液状,体温下则糊化。而热敏可注射水凝胶能够将细胞、药物等生物活性物质包埋后注入生物体内发挥作用。例如:将脂源性干细胞包埋于凝胶内,再将凝胶注射到膀胱壁和尿道后,脂肪源性干细胞大量成活并形成具有收缩性的肌纤维[16-17];而且脂肪源性干细胞在体外培养过程能够长时间保持扩增能力,其增殖能力也不会因为传代次数的增加而有所减弱。由此可见,脂肪源性干细胞能够在体外培养,现有的培养材料和培养环境能够满足其增殖和分化。 在应用于临床之前,有必要对新鲜获取的脂肪源性干细胞进行体外扩增以确保在治疗期间给予临床上需要的细胞数量。因此,在早期传代时通过冷冻保存,可以延长新鲜分离细胞的储存时间。目前,冷冻保存被视为一种能够安全贮存脂肪源性干细胞的常规方法。尽管有人担心冷冻保存处理会对细胞特性造成影响。对此,DURANDT等[10]的研究显示,冷冻保存不会影响脂肪源性干细胞分化为脂肪细胞的能力,连续几轮的扩增并没有改变细胞表面标记的表达。KUMAR等 [19]评估了长期冷冻保存脂肪源性干细胞的特性及其在平均冷冻12年后的再生潜力,结果表明脂肪源性干细胞在长期冷冻保存后仍能保持分化能力,只是不同供者之间存在差异。而且一些冷冻保存溶液已经用于临床研究,且不会对细胞存活率产生较大影响,例如细胞因子系列、丝胶和麦芽糖、甲基纤维素、动物血清的替代品等[20]。 通过流式细胞技术可以鉴定出脂肪源性干细胞独特的表型。与其他间充质干细胞一样,脂肪源性干细胞表达 CD90、CD73、CD105、CD44,不表达 CD45 和 CD31,通过表面标记 CD36 阳性和 CD106 阴性能够将其与骨髓间充质干细胞进行鉴别[21]。脂肪源性干细胞属于成体干细胞的一种,研究表明其具有向中胚层细胞分化的巨大潜力,如脂肪细胞、成骨细胞、软骨细胞和心肌细胞[22]。体外条件下的研究显示,它们具有很高的可塑性和分化成外胚层来源细胞和内胚层来源细胞的能力[23]。因此,将脂肪源性干细胞分化为目标细胞,有望替换受损、患病和有缺陷的细胞和组织。而相比于同样具有多向分化能力的其他来源干细胞,脂肪源性干细胞具有储备量大、获取方便、获取过程中较少的痛苦性和培养条件的非苛刻性、可观的增殖分化性等特点,已成为干细胞领域一种可供选择的种子细胞来源。 2.2 脂肪源性干细胞治疗盆底功能障碍性疾病的可能机制 虽然盆底功能障碍性疾病的具体机制目前并不清楚,但是衰老和各种原因导致的盆底组织损伤是造成此病可能的主要危险因素。生理状态下,肌肉组织受损后主要由卫星细胞发挥修复作用,这些细胞被激活、增殖,并分化成肌纤维,从而重建受损的组织。而病理和衰老状态下,卫星细胞群发生功能障碍或衰竭,严重影响肌肉再生,从而影响其功能[24]。脂肪源性干细胞可能通过以下机制发挥促进组织修复作用,进而有助于盆底功能障碍性疾病的治疗。 2.2.1 通过旁分泌作用参与机体免疫调节 脂肪源性干细胞能够促进肌肉再生与其旁分泌能力参与机体免疫调节过程有关。研究表明,脂肪源性干细胞通过2种主要机制调节免疫系统,一种是直接通过细胞间通讯,另一种是间接通过分泌可溶性递质、生长因子和血管外小泡[25],其中脂肪源性干细胞的旁分泌活动是参与组织修复的主要机制。研究表明,脂肪源性干细胞能够在特定的区域进行特异性分化以确保细胞修复,而且还通过分泌多种类型的趋化因子、细胞因子和蛋白质生长因子等[23],从而能够减轻免疫反应,支持肌肉重建[24]。此外,脂肪源性干细胞还通过分泌白细胞介素2干预T细胞和B细胞的增殖、细胞毒作用和自然杀伤细胞的激活[26],并且支持T细胞活化、巨噬细胞相互作用、白细胞介素10分泌和Th1细胞增殖下调[27],降低了干细胞移植治疗后发生免疫排斥反应的风险。 2.2.2 分泌体的组织损伤修复作用 人脂肪来源干细胞中有一种名叫分泌体的结构,是细胞释放到胞外空间的囊泡[28],因此又称外泌体、胞外囊泡[29]。分泌体中富含积极参与再生过程的趋化因子、细胞因子和蛋白质生长因子[30]。外切体作为分泌体的一部分,被认为是一种多功能的、丰富的组织损伤修复工具,可抑制或激活信号通路,参与细胞外基质重塑、免疫调节、血管再生,以及细胞的迁移、增殖和分化过程[30],主要由蛋白质、mRNA、miRNA和细胞器共同构成[31]。人脂肪源性干细胞的分泌体具有强大的生物学功能和组织再生修复作用,它能够参与机体免疫和抗炎活动,影响细胞的生长、增殖和分化,发挥促进血管生成和细胞基质重建的作用[30]。MITCHELL等[32]研究表明,脂肪源性干细胞分泌体内的可溶性分子能够发挥促进肌肉生成、加速骨骼肌损伤的修复作用,并证实了脂肪源性干细胞分泌体能够刺激细胞向肌管分化,但对细胞的迁移没有影响。此外,分泌体可以避开干细胞治疗的相关不良反应,增加了干细胞治疗的安全性,降低了免疫原性[28]。由此可见,脂肪源性干细胞的分泌体在肌肉组织再生中具有一定作用。 正常盆底组织结构的维持主要与盆底结缔组织中的细胞外基质成分能否保持合成和降解的动态平衡有关,如果平衡破坏就可能产生以盆底结构改变为特征的女性盆底功能障碍性疾病[14]。有研究证实,脂肪干细胞来源分泌体通过增加压力性尿失禁患者阴道成纤维细胞胶原合成和减少胶原降解来增加Ⅰ型胶原含量,从而保持盆底组织细胞外基质的动态平衡[33]。这些关于干细胞分泌体作用的研究,将有助于扩大基于干细胞治疗组织缺陷相关盆底疾病的潜力,也为盆底功能障碍性疾病的治疗提供了合理解释的可能性。 2.3 脂肪源性干细胞治疗盆底功能障碍性疾病的临床前和临床试验 2.3.1 临床前试验 近年来,许多研究表明脂肪源性干细胞能在动物模型上改善盆底功能,其应用主要集中在尿失禁和便失禁方面。从病因学考虑,干细胞治疗失禁有很多优势,传统手术治疗失禁更多关注改善症状,较少根治具体的病因[34]。2010年LIN等[35]第1次探讨脂肪源性干细胞治疗压力性尿失禁的可行性,结果显示,经尿道和静脉注射脂肪源性干细胞的治疗组无论是膀胱计量学还是组织学分析,均显示疗效优于对照组。自此有关脂肪源性干细胞用于盆底功能障碍性疾病的研究逐渐开展,特别是针对失禁的研究备受关注。因此,针对脂肪源性干细胞对便失禁和压力性尿失禁的临床前试验做了整理,见表1和表2。 "

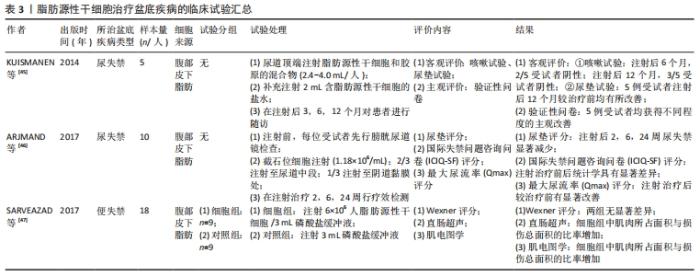
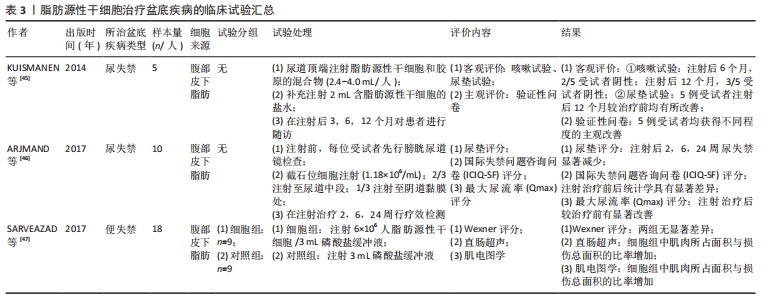
2.3.2 临床试验 一项自体脂肪来源间充质干细胞治疗尿失禁的两期临床试验中,招募了9名男性(8名治疗)和10名女性,行尿道镜下细胞腔内注射自体脂肪间充质干细胞用于尿失禁治疗,试验结果用于评价脂肪源性干细胞的可行性和安全性。在随访过程中,行尿道镜检查、尿动力学等客观检查和尿垫试验、生活质量、尿便失禁问卷等主观评价的检测,结果显示,在3/8的男性和5/10的女性中,客观改善率在50%,主观改善率达到了70%-80%,可见应用脂肪源性干细胞治疗尿失禁是一种安全可行的方法[44]。脂肪源性干细胞对于失禁的疗效,已经在很多动物实验中得到了验证,此外相关临床试验也在逐步开展,为了解其可行性并为临床应用提供进一步的理论支持,现将临床试验汇总至表3。 "
| [1] LANE FL, JACOBS S. Stem cells in gynecology. Am J Obstet Gynecol. 2012;207(3):149-156. [2] POLYKANDRIOTIS E, POPESCU LM, HORCH RE. Regenerative medicine: Then and now--an update of recent history into future possibilities. J Cell Mol Med. 2010;14(10):2350-2358. [3] NI H, ZHAO Y, JI Y, et al. Adipose-derived stem cells contribute to cardiovascular remodeling. Aging (Albany NY). 2019;11(23):11756-11769. [4] LELEK J, ZUBA-SURMA EK. Perspectives for Future Use of Extracellular Vesicles from Umbilical Cord- and Adipose Tissue-Derived Mesenchymal Stem/Stromal Cells in Regenerative Therapies-Synthetic Review. Int J Mol Sci. 2020;21(3):799. [5] SIMA Y, CHEN Y. MSC-based therapy in female pelvic floor disorders. Cell Biosci. 2020;10:104. [6] GOOD MM, SOLOMON ER. Pelvic floor disorders. Obstet Gynecol Clin North Am. 2019;46(3):527-540. [7] HASTINGS J, MACHEK M. Pelvic floor dysfunction in women. Curr Phys Med Rehabil Rep. 2020;8(2):64-75. [8] 孟晓静,李旻,王少为.中老年女性盆底功能障碍性疾病的临床研究进展[J].中国老年保健医学,2021,19(2):94-97. [9] ALVAREZ J, CVACH K, DWYER P. Complications in pelvic floor surgery. Minerva Ginecol. 2013;65(1):53-67. [10] DURANDT C, DESSELS C, DA SILVA C, et al. The effect of early rounds of ex vivo expansion and cryopreservation on the adipogenic differentiation capacity of adipose-derived stromal/stem cells. Sci Rep. 2019;9(1):15943. [11] MELIEF SM, ZWAGINGA JJ, FIBBE WE, et al. Adipose tissue-derived multipotent stromal cells have a higher immunomodulatory capacity than their bone marrow-derived counterparts. Stem Cells Transl Med. 2013;2(6):455-463. [12] YU G, WU X, DIETRICH MA, et al. Yield and characterization of subcutaneous human adipose-derived stem cells by flow cytometric and adipogenic mrna analyzes. Cytotherapy. 2010; 12(4):538-546. [13] 杨洋,肖云翔,金杰. 压力性尿失禁的干细胞治疗进展[J].中华泌尿外科杂志,2012,33(6):471-473. [14] 单倩倩,陶丽,颜士杰.女性压力性尿失禁的发病机制及治疗进展[J].安徽医药,2013,17(3):364-367. [15] 刘剑锋,阳莲,洪莉.水凝胶治疗盆底功能障碍性疾病的研究进展[J].医学研究杂志,2021,50(10):155-157,196. [16] SONG K, LI L, YAN X, et al. Characterization of human adipose tissue-derived stem cells in vitro culture and in vivo differentiation in a temperature-sensitive chitosan/β- glycerophosphate/collagen hybrid hydrogel. Mater Sci Eng C Mater Biol Appl. 2017;70(Pt 1):231-240. [17] 王琳琳,洪莉.可注射水凝胶治疗压力性尿失禁研究进展[J].中国实用妇科与产科杂志,2018,34(2):228-231. [18] PU W, HAN Y, YANG M. Human decellularized adipose tissue hydrogels as a culture platform for human adipose-derived stem cell delivery. J Appl Biomater Funct Mater. 2021;19:2280800020988141. [19] KUMAR A, XU Y, YANG E, et al. Fidelity of long-term cryopreserved adipose-derived stem cells for differentiation into cells of ocular and other lineages. Exp Eye Res. 2019;189:107860. [20] MIYAGI-SHIOHIRA C, KURIMA K, KOBAYASHI N, et al. Cryopreservation of Adipose-Derived Mesenchymal Stem Cells. Cell Med. 2015;8(1-2):3-7. [21] 金琦,彭程,王志平.脂肪来源干细胞在泌尿外科疾病中的研究进展[J].医学综述,2021,27(21):4204-4209. [22] HARASYMIAK-KRZYŻANOWSKA I, NIEDOJADŁO A, KARWAT J, et al. Adipose tissue-derived stem cells show considerable promise for regenerative medicine applications. Cell Mol Biol Lett. 2013;18(4):479-493. [23] MAZINI L, ROCHETTE L, AMINE M, et al. Regenerative Capacity of Adipose Derived Stem Cells (ADSCs), Comparison with Mesenchymal Stem Cells (MSCs). Int J Mol Sci. 2019;20(10):2523. [24] ARCHACKA K, BEM J, BRZOSKA E, et al. Beneficial Effect of IL-4 and SDF-1 on Myogenic Potential of Mouse and Human Adipose Tissue-Derived Stromal Cells. Cells. 2020;9(6):1479. [25] TABATABAEI QOMI R, SHEYKHHASAN M. Adipose-derived stromal cell in regenerative medicine: A review. World J Stem Cells. 2017;9(8):107-117. [26] DOMENIS R, CIFÙ A, QUAGLIA S, et al. Pro inflammatory stimuli enhance the immunosuppressive functions of adipose mesenchymal stem cells-derived exosomes. Sci Rep. 2018;8(1):13325. [27] AL-GHADBAN S, BUNNELL BA. Adipose Tissue-Derived Stem Cells: Immunomodulatory Effects and Therapeutic Potential. Physiology (Bethesda). 2020;35(2):125-133. [28] SANDONÀ M, DI PIETRO L, ESPOSITO F, et al. Mesenchymal Stromal Cells and Their Secretome: New Therapeutic Perspectives for Skeletal Muscle Regeneration. Front Bioeng Biotechnol. 2021;9:652970. [29] ZHUANG G, WEN Y, BRIGGS M, et al. Secretomes of human pluripotent stem cell-derived smooth muscle cell progenitors upregulate extracellular matrix metabolism in the lower urinary tract and vagina. Stem Cell Res Ther. 2021;12(1):228. [30] DINESCU S, DOBRANICI A, TECUCIANU R, et al. Exosomes as Part of the Human Adipose-Derived Stem Cells Secretome- Opening New Perspectives for Cell-Free Regenerative Applications. Adv Exp Med Biol. 2021;1312:139-163. [31] XING X, HAN S, CHENG G, et al. Proteomic Analysis of Exosomes from Adipose-Derived Mesenchymal Stem Cells: A Novel Therapeutic Strategy for Tissue Injury. Biomed Res Int. 2020;2020:6094562. [32] Mitchell R, Mellows B, Sheard J, et al. Secretome of adipose-derived mesenchymal stem cells promotes skeletal muscle regeneration through synergistic action of extracellular vesicle cargo and soluble proteins. Stem Cell Res Ther. 2019;10(1):116. [33] LIU X, WANG S, WU S, et al. Exosomes secreted by adipose-derived mesenchymal stem cells regulate type I collagen metabolism in fibroblasts from women with stress urinary incontinence. Stem Cell Res Ther. 2018;9(1):159. [34] 汪成合,陈忠.干细胞治疗女性压力性尿失禁的研究进展[J].临床泌尿外科杂志,2014,29(5):457-460. [35] LIN G, WANG G, BANIE L, et al. Treatment of stress urinary incontinence with adipose tissue-derived stem cells. Cytotherapy. 2010;12(1):88-95. [36] INOUE Y, FUJITA F, YAMAGUCHI I, et al. Improvement of Anal Function by Adipose-Derived Stem Cell Sheets. Dig Surg. 2018;35(1):64-69. [37] KUISMANEN K, JUNTUNEN M, NARRA GIRISH N, et al. Functional Outcome of Human Adipose Stem Cell Injections in Rat Anal Sphincter Acute Injury Model. Stem Cells Transl Med. 2018;7(3):295-304. [38] ZHAO W, ZHANG C, JIN C, et al. Periurethral injection of autologous adipose-derived stem cells with controlled-release nerve growth factor for the treatment of stress urinary incontinence in a rat model. Eur Urol. 2011;59(1):155-163. [39] FU Q, SONG XF, LIAO GL, et al. Myoblasts differentiated from adipose-derived stem cells to treat stress urinary incontinence. Urology. 2010; 75(3):718-723. [40] WU G, SONG Y, ZHENG X,et al. Adipose-derived stromal cell transplantation for treatment of stress urinary incontinence. Tissue Cell. 2011;43(4):246-253. [41] 关立铭,李秀娟,陈亚萍,等.自体脂肪源性干细胞与成纤维细胞联合尿道周围注射治疗大鼠压力性尿失禁[J].中国组织工程研究, 2012,16(14):2530-2533. [42] 耿鹏,宋红娟,刘亚玲,等.脂肪源成体干细胞移植治疗压力性尿失禁的实验研究[J].中华细胞与干细胞杂志(电子版),2015,5(3): 197-201. [43] WANG Y, DUAN M, RAHMAN M, et al. Use of bioactive extracellular matrix fragments as a urethral bulking agent to treat stress urinary incontinence. Acta Biomater. 2020;117:156-166. [44] GARCIA-ARRANZ M, ALONSO-GREGORIO S, FONTANA-PORTELLA P, et al. Two phase I/II clinical trials for the treatment of urinary incontinence with autologous mesenchymal stem cells. Stem Cells Transl Med. 2020; 9(12):1500-1508. [45] KUISMANEN K, SARTONEVA R, HAIMI S, et al. Autologous adipose stem cells in treatment of female stress urinary incontinence: results of a pilot study. Stem Cells Transl Med. 2014;3(8):936-941. [46] ARJMAND B, SAFAVI M, HEIDARI R, et al. Concomitant Transurethral and Transvaginal-Periurethral Injection of Autologous Adipose Derived Stem Cells for Treatment of Female Stress Urinary Incontinence: A Phase One Clinical Trial. Acta Med Iran. 2017;55(6):368-374. [47] SARVEAZAD A, NEWSTEAD GL, MIRZAEI R, et al. A new method for treating fecal incontinence by implanting stem cells derived from human adipose tissue: preliminary findings of a randomized double-blind clinical trial. Stem Cell Res Ther. 2017;8(1):40. [48] 李露,洪莉.成体干细胞治疗压力性尿失禁的应用[J].现代妇产科进展,2019,28(5):385-387. [49] 江长琴. 女性压力性尿失禁的治疗进展[J].国际泌尿系统杂志, 2021,41(4):753-755. [50] DE LA GARZA-RODEA AS, VAN DER VELDE-VAN DIJKE I, BOERSMA H, et al. Myogenic properties of human mesenchymal stem cells derived from three different sources. Cell Transplant. 2012;21(1):153-173. [51] 常悦,刘海峰,王建六. 组织工程及再生医学在盆底功能障碍性疾病中的应用进展[J].中华妇产科杂志,2015,50(6):470-472. [52] MANODORO S, FRIGERIO M, BARBA M, et al. Stem Cells in Clinical Trials for Pelvic Floor Disorders: a Systematic Literature Review. Reprod Sci. 2022;29(6):1710-1720. [53] 唐翔,朱兰.干细胞在妇科泌尿领域的应用[J].现代妇产科进展, 2017,26(10):788-790. [54] HONG P, YANG H, WU Y, et al. The functions and clinical application potential of exosomes derived from adipose mesenchymal stem cells: a comprehensive review. Stem Cell Res Ther. 2019;10(1):242. [55] SHUKLA L, YUAN Y, SHAYAN R, et al. Fat Therapeutics: The Clinical Capacity of Adipose-Derived Stem Cells and Exosomes for Human Disease and Tissue Regeneration. Front Pharmacol. 2020;11:158. |
| [1] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [2] | Cai Zhihao, Xie Zhaoyong. Femoral neck anteversion measurement assessment: how to establish a unified method and standard [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1448-1454. |
| [3] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [4] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [5] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [6] | Gao Yu, Han Jiahui, Ge Xin. Immunoinflammatory microenvironment after spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1300-1305. |
| [7] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [8] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [9] | Xu Cong, Zhao He, Sun Yan. Regeneration of facial nerve injury repaired by biomaterial nerve conduits [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1089-1095. |
| [10] | Chen Shisong, Liu Xiaohong, Xu Zhiyun. Current status and prospects of bioprosthetic heart valves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1096-1102. |
| [11] | Lu Di, Zhang Cheng, Duan Rongquan, Liu Zongxiang. Osteoinductive properties of calcium phosphate ceramic bone repair materials [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1103-1109. |
| [12] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [13] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [14] | Xue Ting, Zhang Xinri, Kong Xiaomei. Mesenchymal stem cell therapy for pneumoconiosis using nanomaterials combined with multi-modal molecular imaging [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1133-1140. |
| [15] | Yuan Wei, Liu Jingdong, Xu Guanghui, Kang Jian, Li Fuping, Wang Yingjie, Zhi Zhongzheng, Li Guanwu. Osteogenic differentiation of human perivascular stem cells and its regulation based on Wnt/beta-catenin signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 866-871. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||