Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (8): 1272-1277.doi: 10.12307/2023.057
Previous Articles Next Articles
A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia
Wang Ji1, Zhang Min2, Yang Zhongya1, Zhang Long1
- 1School of Physical Education, Liupanshui Normal University, Liupanshui 553000, Guizhou Province, China; 2Liupanshui Preschool Education College, Liupanshui 553000, Guizhou Province, China
-
Received:2022-01-24Accepted:2022-03-07Online:2023-03-18Published:2022-07-29 -
About author:Wang Ji, Master, Lecturer, School of Physical Education, Liupanshui Normal University, Liupanshui 553000, Guizhou Province, China -
Supported by:Youth Science and Technology Talent Growth Project of Guizhou Provincial Education Department, No. KY[2020]113 (to WJ)
CLC Number:
Cite this article
Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.2 非闲暇体力活动对2型糖尿病肌少症的影响 体力活动(physical activity,PA)指任何由骨骼肌收缩引起的导致能量消耗的身体运动,一般将体力活动分为非闲暇的体力活动(包括工作、家庭、交通中的体力活动)和闲暇的体力活动(包括参与各种体育活动和体育锻炼),其中非闲暇的体力活动指在工作、家庭和交通中的体力活动。《中国2型糖尿病防治指南(2020版)》(简称《指南》)建议将增加日常体力活动(非闲暇体力活动)和规律的体育锻炼(闲暇体力活动)有效结合[20]。虽然《指南》明确了体力活动在防治2型糖尿病中的重要作用,但主要是基于体力活动的直接能量消耗对2型糖尿病患者糖脂代谢和胰岛素敏感性的改善作用[21-22]。《指南》也并未提及和明确肌少症作为老年2型糖尿病的慢性并发症,事实上,2型糖尿病肌少症的发生将显著降低患者下肢肌力和步态稳定性,增加跌倒和骨折风险,应得到足够的重视[23]。 随着社会经济的快速发展尤其是人工智能技术的突飞猛进,人们在工作、家庭和交通中的非闲暇体力活动显著降低,并普遍形成了静坐少动的生活方式。非闲暇体力活动的降低直接减少了人类的日常能量消耗,导致过多的能量储存和脂肪积累,增加了肥胖、2型糖尿病等代谢疾病的发病风险。HOUSTON等[24]的一项大型前瞻性研究表明,通过健康教育改变2型糖尿患者的生活方式、增加日常非闲暇体力活动可使超重和肥胖患者以及中老年2型糖尿病患者(年龄45-76岁)在11年后仍表现出较对照组更好的下肢运动功能,提示增加非闲暇体力活动可以对2型糖尿病及其肌少症的发生发展起到长效干预效果。 近期DE FREITAS等[25]的一项横向研究也显示,改变静坐少动生活方式、增加非闲暇体力活动可以有效降低老年2型糖尿病患者罹患肌少症的风险。该研究通过在糖尿病门诊护理中心招募242例2型糖尿病老年患者[(68.3±5.6)岁]为研究对象,分析显示2型糖尿病肌少症的总体患病率为21%,其中女性约占88%;该研究进一步的分析还显示增加非闲暇体力活动(日常步数> 5 401步/d)可以对2型糖尿病肌少症患病率起到70%的保护作用。限制于非闲暇体力活动难以量化的实际研究困难,目前关于非闲暇体力活动改善2型糖尿病肌少症的研究相对较少,但现有研究结果依旧表明了非闲暇体力活动的重要性;与此同时,研究者们也正集中关注多样化的闲暇体力活动对2型糖尿病肌少症的影响。 2.3 闲暇体力活动对2型糖尿病肌少症的影响 闲暇体力活动包括参与各种体育活动和为提高体质健康水平而进行的体育锻炼等,也称为运动。美国一项随访研究对3 234名2型糖尿病前期患者进行3年的随访显示,通过二甲双胍(2次/d,每次850 mg)和闲暇体力活动干预(每周至少150 min的体育锻炼)均可降低2型糖尿病前期患者的发病率,而体力活动干预比二甲双胍治疗更为有效(其中二甲双胍治疗将2型糖尿病前期发病率低了31%,而生活方式干预将发病率降低了58%)[26]。除了降低2型糖尿病的发病率外,生活方式干预还能有效降低2型糖尿病肌少症的发生率。SAZLINA等[27]最新的研究对506名60岁以上2型糖尿病患者的社会人口统计学、临床和功能状态、饮食和身体活动水平数据进行纵向分析显示,低体质量指数、低水平的体力活动与肌少症发病率息息相关,体力活动水平可作为预测肌少症的因子之一,而增加体力活动和保持健康的体质量则有助于预防2型糖尿病老年人肌少症的发生。此外,STEFFL 等[28]一项最新的Meta分析也显示,规律地参与任何体育锻炼活动均能有效降低40岁以上成年人发生肌少症的概率。由于体育锻炼项目和运动方式的多样性,此次研究进一步以运动生理学为依据,根据体育锻炼过程中肌肉的收缩形式和运动能量代谢特点将体育锻炼分为抗阻运动和有氧运动分别进行探讨。 2.3.1 抗阻运动对2型糖尿病肌少症的影响 (1)抗阻运动干预2型糖尿病肌少症的方案和效果评价:抗阻运动指肌肉通过收缩来克服阻力做功的运动,因此又称为“力量练习”。抗阻运动是提高肌少症患者肌肉质量的有效方法已经得到广泛认识,《指南》建议无急性和严重慢性并发症的2型糖尿病成人患者在执行运动疗法时每周应该坚持两三次抗阻运动,但却并未指明适宜的运动方案和运动强度,而这也正是研究者们目前关注的重点(表2)[29-33]。 "
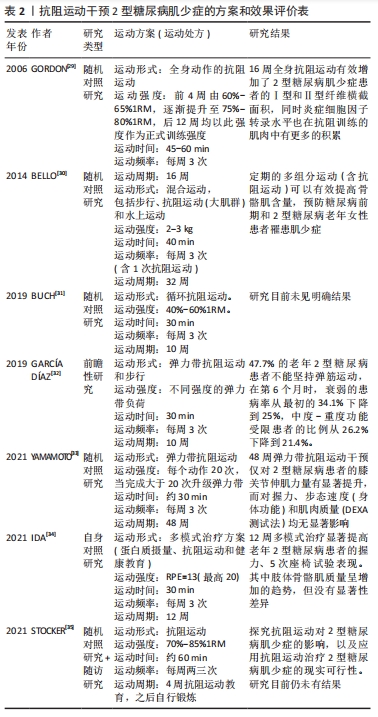
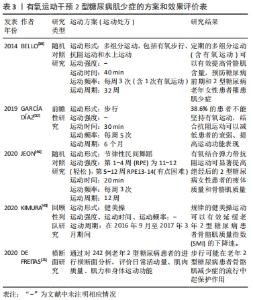
研究显示,长期坚持弹力带抗阻运动可有效改善2型糖尿病的虚弱状况、提高骨骼肌力量,但对肌肉质量无显著影响[32-33]。而16周中等强度(每周3次,75%-80%1RM)抗阻运动则可有效增加2型糖尿病肌少症患者的Ⅰ型和Ⅱ型纤维横截面积,提高骨骼肌质量[29]。此外,更多的研究还集中在抗阻运动联合有氧运动、抗阻运动联合营养干预和健康教育,也均对2型糖尿病肌少症起到了较好的防治效果[30,34]。 (2)抗阻运动干预2型糖尿病肌少症的机制:研究显示,与增龄相关的2型糖尿病、恶病质和去负荷等刺激均可引发骨骼肌流失,尽管不同的疾病或环境刺激所引发的骨骼肌流失机制不尽相同,但导致肌少症的最主要原因仍是与年龄和代谢相关的骨骼肌收缩蛋白的合成与分解失衡[36]。在健康机体中,未折叠或受损蛋白质不断被泛素蛋白酶体分解,新的蛋白质不断由雷帕霉素靶蛋白(mammalian target of rapamycin,mTOR)为核心介导的各种途径不断合成,从而维持了细胞内环境的相对稳态。但在2型糖尿病肌少症的发展进程中,胰岛素抵抗和长期慢性炎症反应导致机体蛋白质合成和分解失衡,加剧骨骼肌质量的流失,并导致骨骼肌功能的下降[37]。 正是由于抗阻运动的促蛋白合成效应有利于增加肌肉质量和肌肉力量,抗阻运动也被认为是在2型糖尿病肌少症状态下促进肌肉肥大的有效方法。尽管抗阻运动有效改善2型糖尿病肌少症的治疗效果已经得到普遍认可,但不同研究者对抗阻运动诱导2型糖尿病肌肉肥大的机制仍持不同观点。IBANEZ等[38]一项对老年男性糖尿病患者进行为期16周的渐进式抗阻运动干预研究显示,2型糖尿病患者对抗阻运动诱导的肌肉力量增长和骨骼肌肥大效应均要比健康人群更低,研究者认为这可能与2型糖尿病患者基础状态的下丘脑-垂体-肾上腺轴功能受损有关,主要表现为2型糖尿病患者的总睾酮水平较普通人群更低,而低睾酮水平限制了老年2型糖尿病患者骨骼肌力量的发展。而GEIRSDOTTIR等[39]针对237名老年2型糖尿病患者的一项回顾性研究显示,为期12周的抗阻运动(每周3次,75%-80%1RM)可以有效提高健康受试者、2型糖尿病前期患者和2型糖尿病患者的上肢握力、下肢膝关节伸肌力量和运动表现,且不论是2型糖尿病还是2型糖尿病前期患者,运动导致的骨骼肌功能改善作用都没有被抑制。GEIRSDOTTIR等的这项结论也在动物实验中得到进一步验证。ATO等[40]通过对老年2型糖尿病大鼠进行为期6周的抗阻运动干预研究显示,2型糖尿病显著降低了骨骼肌中mTORC1(mechanistic target of rapamycin complex 1)活性机制靶点(p70S6KThr389和4EBP1Thr37/46)的磷酸化水平,而仅需3周的抗阻运动即可显著激活mTORC1信号通路、提高2型糖尿病大鼠腓肠肌的合成效应和肌肉质量;更可观的是,尽管在2型糖尿病条件下骨骼肌蛋白质合成系统受损,但抗阻运动诱导的骨骼肌细胞mTORC1信号通路激活和骨骼肌肥大效应并没有受损。 Apró等[41]认为这可是能是由于抗阻运动的机械刺激能使mTOR独立于胰岛素或胰岛素样生长因子1(IGF-1)而激活;MAZO等[42]的研究则从另一方面证实了抗阻运动还可以通过激活IRS-PI3K-Akt信号途径提高2型糖尿病的胰岛素敏感性,进而提高其下游的mTOR信号通路活性,上调p70S6K、4EBP1的磷酸化水平。 此外研究还发现,2型糖尿病大鼠骨骼肌肌卫星细胞标志物Pax7的表达均显著降低,同时伴随肌核的萎缩和丢失,而超负荷或持续的抗阻运动则可以通过诱导肌卫星细胞增殖和分化,增加肌核数量来实现肌肉肥大效应,提示2型糖尿病会损害肌卫星细胞功能和数量,而抗阻运动也可通过刺激骨骼肌的增殖分化来实现肌肉增长[40,43]。 2.3.2 有氧运动对2型糖尿病肌少症的影响 (1)有氧运动干预2型糖尿病肌少症的方案和效果评价:有氧运动是指主要以有氧代谢供能的运动方式,如慢跑、游泳、健美操、广播体操等。虽然大量研究已经报道了抗阻运动可以有效改善2型糖尿病肌少症,但在实践操作中却发现大部分2型糖尿病患者很难坚持进行规律的抗阻运动;而有氧运动因其项目的多样性和趣味性,可以不分年龄和性别的覆盖不同群体而具有广泛的群众基础[44]。因此《指南》也将2型糖尿病成人患者每周至少坚持150 min中等强度的有氧运动放在了运动疗法的首位。 研究者常常选择用易于实施、趣味性高和依从性较好的有氧运动项目(如健美操、舞蹈和步行等)来研究有氧运动对2型糖尿病肌少症的影响,现有研究显示规律的有氧运动可以较好地延缓2型糖尿病患者骨骼肌的流失速度,从而抑制2型糖尿病肌少症的发病进程[25,45],但要实现骨骼肌的有效增长则需要通过联合抗阻运动来实现[30,32,46],见表3。 "
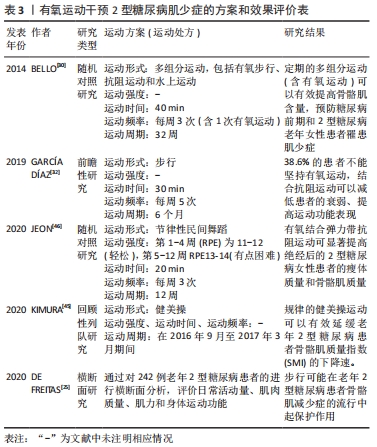

(2)有氧运动干预2型糖尿病肌少症的机制:有氧运动对健康人群或啮齿类实验动物的骨骼肌质量无显著影响的主要原因是有氧运动后的蛋白合成效应明显低于抗阻运动,而在分解代谢旺盛的2型糖尿病环境下,有氧运动对骨骼肌质量的维持则发挥了较好作用,但人们对这种机制的理解在很大程度上仍依赖于啮齿类糖尿病动物模型。大量的研究已经表明,在2型糖尿大鼠模型中,泛素蛋白酶体系存在过度激活现象[47-51];而有氧跑台运动干预能显著提高2型糖尿病大鼠骨骼肌肌纤维横截面积、增加骨骼肌质量,这与有氧运动降低骨骼肌中泛素蛋白连接酶Atrogin-1和MuRF1的蛋白表达以及抑制骨骼肌自噬作用密切相关。 TAKAMURA等[52]针对2型糖尿病小鼠模型的研究同样显示,有氧运动可有效降低Atrogin-1和MuRF-1的表达,同时还可显著增加2型糖尿病小鼠比目鱼肌中p70S6K的表达,提示有氧运动在抑制2型糖尿病骨骼肌分解系统的同时还可能通过提高骨骼肌合成系统来共同实现骨骼肌质量增长,研究者认为这主要与有氧运动提升Akt的磷酸化水平有关,Akt的磷酸化激活将进一步激活mTORC1和p70S6K的蛋白合成效应,最终导致机体对营养摄入和运动刺激的蛋白合成效应增加。但KIM等[53]的研究却显示,尽管有氧运动能显著提高肥胖大鼠骨骼肌中AMPK蛋白Thr172位点的磷酸化水平、降低Atrogin-1水平,但对肌肉中mTOR及其下游S6K1的蛋白磷酸化水平并无明显影响,研究者认为有氧运动主要通过抑制蛋白质分解系统来实现对2型糖尿病肌肉流失的抑制,而对蛋白质合成系统影响较小。 "

| [1] JAMES SL, CASTLE CD, DINGELS ZV, et al. Global injury morbidity and mortality from 1990 to 2017: results from the Global Burden of Disease Study 2017. Inj Prev. 2020;26(1):96-114. [2] OGURTSOVA K, DA ROCHA FERNANDES JD, HUANG Y, et al. IDF Diabetes Atlas: Global estimates for the prevalence of diabetes for 2015 and 2040. Diabetes Res Clin Pract. 2017;128:40-50. [3] AMERICAN DIABETES ASSOCIATION. 12. Older Adults: Standards of Medical Care in Diabetes-2019. Diabetes Care. 2019;42(1):139-147. [4] ROSENBERG IH. Sarcopenia: origins and clinical relevance. J Nutr. 1997; 127(5l):990-991. [5] 姜珊,康琳,刘晓红.2019亚洲肌少症诊断及治疗共识解读[J].中华老年医学杂志,2020,39(4):373-376. [6] IZZO A, MASSIMINO E, RICCARDI G, et al. A Narrative Review on Sarcopenia in Type 2 Diabetes Mellitus: Prevalence and Associated Factors. Nutrients. 2021;13(1):183. [7] 白友菊,康霜,李颖,等.基于简易指标的综合指数对老年2型糖尿病患者肌少症的诊断价值[J].中国实用内科杂志,2020,40(8):662-667. [8] 陆玉凤,刘富甜,朱小红,等.住院2型糖尿病患者肌少症相关因素影响分析[J].中国糖尿病杂志,2021,29(4):284-287. [9] KIM YK, LEE HS, RYU JJ, et al. Sarcopenia increases the risk for mortality in patients who undergo amputation for diabetic foot. J Foot Ankle Res. 2018;19(11):32. [10] BAUMGARTNER RN, WAYNE SJ, WATERS DL, et al. Sarcopenic obesity predicts instrumental activities of daily living disability in the elderly. Obes Res. 2004;12(12):1995-2004. [11] No authors listed. Weekly clinicopathological exercises. Case 21-1987. A 68-year-old man with diabetes mellitus and progressive muscle weakness. N Engl J Med. 1987;316(21):1326-1335. [12] No authors listed. Weekly clinicopathological exercises. Case 50-1991. A 71-year-old woman with a sensorimotor neuropathy and radiographically demonstrable abnormalities. N Engl J Med. 1991;325(24):1723-1735. [13] BAUMGARTNER RN, KOEHLER KM, GALLAGHER D, et al. Epidemiology of sarcopenia among the elderly in New Mexico. Am J Epidemiol. 1998; 147(8):755-763. [14] SAADEDDINE D, ITANI L, KREIDIEH D, et al. Association between Levels of Physical Activity, Sarcopenia, Type 2 Diabetes and the Quality of Life of Elderly People in Community Dwellings in Lebanon. Geriatrics (Basel). 2021;6(1):28. [15] CRUZ-JENTOFT AJ, BAEYENS JP, BAUER JM, et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing. 2010;39(4):412-423. [16] ANBALAGAN VP, VENKATARAMAN V, PRADEEPA R, et al. The prevalence of presarcopenia in Asian Indian individuals with and without type 2 diabetes. Diabetes Technol Ther. 2013;15(9):768-775. [17] CHEN LK, LIU LK, WOO J, et al. Sarcopenia in Asia: consensus report of the Asian Working Group for Sarcopenia. J Am Med Dir Assoc. 2014; 15(2):95-101. [18] WANG T, FENG X, ZHOU J, et al. Type 2 diabetes mellitus is associated with increased risks of sarcopenia and pre-sarcopenia in Chinese elderly. Sci Rep. 2016;13(6):38937. [19] CRUZ-JENTOFT AJ, BAHAT G, BAUER J, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 2019;48(1):16-31. [20] 中华医学会糖尿病学分会.中国2型糖尿病防治指南(2020年版)[J].中华糖尿病杂志,2021,13(4):315-409. [21] 李颖,林文弢,翁锡全.不同运动强度干预2型糖尿病模型大鼠的内脂素及糖代谢变化[J].中国组织工程研究,2020,24(26):4196-4200. [22] 张建伟,吕邵钧,王继,等.太极拳对不同性别2型糖尿病患者治疗效果的比较研究[J]. 北京师范大学学报(自然科学版),2019,55(1): 545-550. [23] 何清华,常鑫淼,孙明晓,等.2型糖尿病患者肌肉质量和力量的变化特点[J].中华老年医学杂志,2020,39(5):550-554. [24] HOUSTON DK, NEIBERG RH, MILLER ME, et al. Physical Function Following a Long-Term Lifestyle Intervention Among Middle Aged and Older Adults With Type 2 Diabetes: The Look AHEAD Study. J Gerontol A Biol Sci Med Sci. 2018;73(11):1552-1559. [25] DE FREITAS MM, DE OLIVEIRA VLP, GRASSI T, et al. Difference in sarcopenia prevalence and associated factors according to 2010 and 2018 European consensus (EWGSOP) in elderly patients with type 2 diabetes mellitus. Exp Gerontol. 2020;132:110835. [26] KNOWLER WC, BARRETT-CONNOR E, FOWLER SE, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;46(6):393-403. [27] SAZLINA SG, LEE PY, CHAN YM, et al. The prevalence and factors associated with sarcopenia among community living elderly with type 2 diabetes mellitus in primary care clinics in Malaysia. PLoS One. 2020; 15(5):e0233299. [28] STEFFL M, BOHANNON RW, SONTAKOVA L, et al. Relationship between sarcopenia and physical activity in older people: a systematic review and meta-analysis. Clin Interv Aging. 2017;12:835-845. [29] GORDON PL, VANNIER E, HAMADA K, et al. Resistance training alters cytokine gene expression in skeletal muscle of adults with type 2 diabetes. Int J Immunopathol Pharmacol. 2006;19(4):739-749. [30] BELLO M, SOUSA MC, NETO G, et al. The effect of a long-term, community-based exercise program on bone mineral density in postmenopausal women with pre-diabetes and type 2 diabetes. J Hum Kinet. 2014;43:43-48. [31] BUCH A, ELDOR R, KIS O, et al. The effect of circuit resistance training, empagliflozin or “vegeterranean diet” on physical and metabolic function in older subjects with type 2 diabetes: a study protocol for a randomized control trial (CEV-65 trial). BMC Geriatr. 2019;19(1):228. [32] GARCÍA DÍAZ E, ALONSO RAMÍREZ J, HERRERA FERNÁNDEZ N, et al. Effect of strength exercise with elastic bands and aerobic exercise in the treatment of frailty of the elderly patient with type 2 diabetes mellitus. Endocrinol Diabetes Nutr (Engl Ed). 2019;66(9):563-570. [33] YAMAMOTO Y, NAGAI Y, KAWANABE S, et al. Effects of resistance training using elastic bands on muscle strength with or without a leucine supplement for 48 weeks in elderly patients with type 2 diabetes. Endocr J. 2021;68(3):291-298. [34] IDA S, KANEKO R, IMATAKA K, et al. Multimodal treatment program for elderly diabetic patients with sarcopenia improves the muscle strength and physical function. Nihon Ronen Igakkai Zasshi. 2021;58(1):143-151. [35] STOCKER R, SHAW J, TAYLOR GS, et al. EXercise to Prevent frailty and Loss Of independence in insulin treated older people with DiabetEs (EXPLODE): protocol for a feasibility randomised controlled trial (RCT). BMJ Open. 2021;11(12):e048932. [36] AL-OZAIRI E, ALSAEED D, ALROUDHAN D, et al. Skeletal Muscle and Metabolic Health: How Do We Increase Muscle Mass and Function in People with Type 2 Diabetes?. J Clin Endocrinol Metab. 2021;106(2):309-317. [37] 王继,周越.2型糖尿病与肌萎缩研究进展[J].中国运动医学杂志, 2017,36(7):645-650. [38] IBANEZ J, GOROSTIAGA EM, ALONSO AM, et al. Lower muscle strength gains in older men with type 2 diabetes after resistance training . Journal of diabetes and its complications. 2008;22(2):112-118. [39] GEIRSDOTTIR OG, ARNARSON A, BRIEM K, et al. Effect of 12-week resistance exercise program on body composition, muscle strength, physical function, and glucose metabolism in healthy, insulin-resistant, and diabetic elderly Icelanders. J Gerontol A Biol Sci Med Sci. 2012; 67(11):1259-1265. [40] ATO S , KIDO K, SATO K, et al. Type 2 diabetes causes skeletal muscle atrophy but does not impair resistance training‐mediated myonuclear accretion and muscle mass gain in rats. Exp Physiol. 2019;104(10):1518-1531. [41] APRÓ W, MOBERG M, HAMILTON DL, et al. Resistance exercise-induced S6K1 kinase activity is not inhibited in human skeletal muscle despite prior activation of AMPK by high-intensity interval cycling. Am J Physiol Endocrinol Metab. 2015;308(6):470-581. [42] MAZO CE, D’LUGOS AC, SWEENEY KR, et al. The effects of acute aerobic and resistance exercise on mTOR signaling and autophagy markers in untrained human skeletal muscle. Eur J Appl Physiol. 2021;121(10): 2913-2924. [43] SNIJDERS T, SMEETS JS, VAN KRANENBURG J, et al. Changes in myonuclear domain size do not precede muscle hypertrophy during prolonged resistance-type exercise training. Acta Physiol (Oxf). 2016; 216(2):231-239. [44] DARYABEYGI-KHOTBEHSARA R, WHITE KM, DJAFARIAN K, et al. Short-term effectiveness of a theory-based intervention to promote diabetes management behaviours among adults with type 2 diabetes in Iran: A randomised control trial. Int J Clin Pract. 2021;75(5):e13994. [45] KIMURA T, OKAMURA T, IWAI K, et al. Japanese radio calisthenics prevents the reduction of skeletal muscle mass volume in people with type 2 diabetes. BMJ Open Diabetes Res Care. 2020;8(1):e001027. [46] JEON YK, KIM SS, KIM JH, et al. Combined Aerobic and Resistance Exercise Training Reduces Circulating Apolipoprotein J Levels and Improves Insulin Resistance in Postmenopausal Diabetic Women. Diabetes Metab J. 2020;44(1):103-112. [47] CHEN GQ, MOU CY, YANG YQ, et al. Exercise training has beneficial anti-atrophy effects by inhibiting oxidative stress-induced MuRF1 upregulation in rats with diabetes. Life Sci. 2011;89(2):44-49. [48] AKAGAWA M, MIYAKOSHI N, KASUKAWA Y, et al. Effects of activated vitamin D, alfacalcidol, and low-intensity aerobic exercise on osteopenia and muscle atrophy in type 2 diabetes mellitus model rats. PLoS One. 2018;13(10):e0204857. [49] LIU HW, CHANG SJ. Moderate Exercise Suppresses NF-kappaB Signaling and Activates the SIRT1-AMPK-PGC1alpha Axis to Attenuate Muscle Loss in Diabetic db/db Mice. Front Physiol. 2018;9(6):36-46. [50] 王继,杨中亚,张龙,等.AMPK/PGC-1α在有氧运动改善2型糖尿病大鼠骨骼肌萎缩中的作用[J].中国组织工程研究,2020,24(20):3180-3185. [51] 高前进,柏海平,王彦伟.有氧运动对糖尿病大鼠自噬性肌萎缩的影响[J].中国康复医学杂志,2016,31(7):739-745. [52] TAKAMURA Y, NOMURA M, UCHIYAMA A, et al. Effects of Aerobic Exercise Combined with Panaxatriol Derived from Ginseng on Insulin Resistance and Skeletal Muscle Mass in Type 2 Diabetic Mice. J Nutr Sci Vitaminol (Tokyo). 2017;63(5):339-348. [53] KIM HJ, LEE WJ. Low-intensity aerobic exercise training: inhibition of skeletal muscle atrophy in high-fat-diet-induced ovariectomized rats. J Exerc Nutrition Biochem. 2017;21(3):19-25. |
| [1] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [2] | Cai Zhihao, Xie Zhaoyong. Femoral neck anteversion measurement assessment: how to establish a unified method and standard [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1448-1454. |
| [3] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [4] | Sun Jiajia, Zhu Haidi, Lu Yun, Zhang Kai. Comparison of bone metabolism markers between type 2 diabetes mellitus and non-type 2 diabetes mellitus patients with hip fracture [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1156-1160. |
| [5] | Zhang Yan, He Ruibo, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Ma Gang, Peng Peng. Effects of aerobic exercises with different load volumes on inflammatory response and insulin signaling pathway of skeletal muscle in obese rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1237-1244. |
| [6] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [7] | Gao Yu, Han Jiahui, Ge Xin. Immunoinflammatory microenvironment after spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1300-1305. |
| [8] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [9] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [10] | Chen Shisong, Liu Xiaohong, Xu Zhiyun. Current status and prospects of bioprosthetic heart valves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1096-1102. |
| [11] | Lu Di, Zhang Cheng, Duan Rongquan, Liu Zongxiang. Osteoinductive properties of calcium phosphate ceramic bone repair materials [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1103-1109. |
| [12] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [13] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [14] | Zhou Yinan, Yang Kun, Jiang Jiayi, Zhang Sizhuo, Zhu Yingwen. The relationship between glucose transporter 1/4 gene polymorphisms and athletic ability [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 745-750. |
| [15] | Xiong Bohan, Yu Yang, Lu Xiaojun, Wang Xu, Yang Tengyun, Zhang Yaozhang, Liao Xinyu, Zhou Xiaoxiang, He Lu, Li Yanlin. Research progress in promoting tendon to bone healing during anterior cruciate ligament reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 779-786. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

