Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (15): 2400-2405.doi: 10.12307/2022.598
Previous Articles Next Articles
Angiogenesis regulation in bone repair: new ideas and new methods
Peng Huizhen1, 2, 3, Cai Mingxiang3, 2, 1, Liu Xiangning1, 2, 3
- 1College of Stomatology, Jinan University, Guangzhou 510632, Guangdong Province, China; 2Clinical Research Platform for Interdiscipline of Stomatology, Jinan University, Guangzhou 510630, Guangdong Province, China; 3First Affiliated Hospital of Jinan University & Department of Stomatology, Guangzhou 510630, Guangdong Province, China
-
Received:2021-09-14Revised:2021-10-15Accepted:2021-11-23Online:2022-05-28Published:2022-01-06 -
Contact:Liu Xiangning, PhD, Chief physician, Associate professor, College of Stomatology, Jinan University, Guangzhou 510632, Guangdong Province, China; Clinical Research Platform for Interdiscipline of Stomatology, Jinan University, Guangzhou 510630, Guangdong Province, China; First Affiliated Hospital of Jinan University & Department of Stomatology, Guangzhou 510630, Guangdong Province, China -
About author:Peng Huizhen, Master candidate, College of Stomatology, Jinan University, Guangzhou 510632, Guangdong Province, China; Clinical Research Platform for Interdiscipline of Stomatology, Jinan University, Guangzhou 510630, Guangdong Province, China; First Affiliated Hospital of Jinan University & Department of Stomatology, Guangzhou 510630, Guangdong Province, China -
Supported by:Natural Science Foundation of Guangdong Province, No. 2021A1515010882 (to LXN); Guangzhou Science and Technology Plan Project, No. 202102010040 (to LXN)
CLC Number:
Cite this article
Peng Huizhen, Cai Mingxiang, Liu Xiangning. Angiogenesis regulation in bone repair: new ideas and new methods[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(15): 2400-2405.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
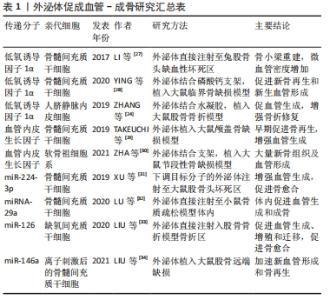
2.1.1 成血管-成骨生长因子 骨缺损修复中血管生成生长因子有血管内皮生长因子(vascular endothelial growth factor,VEGF)、 成纤维细胞生长因子、促红细胞生成素和血小板衍生生长因子等,其中最关键的是VEGF,它的研究也最为深入。VEGF家族包括多个成员,通常指VEGFA,它与受体VEGFR-1和VEGFR-2结合,诱导受体二聚化,从而刺激内皮细胞有丝分裂,诱导其迁移和增殖,促进血管的形成[1]。VEGF不仅是血管生成的关键生长因子,也调节成骨过程。VEGF可以增强血管生成进而增加成骨;成骨细胞衍生的VEGF可刺激间充质干细胞向成骨细胞分化,并抑制其向脂肪细胞分化,对维持骨稳态起着至关重要的作用[5]。 成骨生长因子也有许多,其中,骨形态发生蛋白属于转化生长因子超家族,是最著名的成骨生长因子。许多骨形态发生蛋白家族成员,如骨形态发生蛋白2、骨形态发生蛋白4、骨形态发生蛋白6、骨形态发生蛋白7 和骨形态发生蛋白9,都表现出很强的骨诱导性,可诱导非骨部位的新生骨形成[6]。VEGF虽然具有成血管-成骨功能,但单独递送VEGF不足以成骨[7],因此多与成骨生长因子骨形态发生蛋白联合应用。 2.1.2 VEGF和骨形态发生蛋白共递送系统 由于VEGF出色的成血管能力与骨形态发生蛋白的成骨特性,研究最多的是VEGF与骨形态发生蛋白的双重递送。从纳米纤维到水凝胶[8-9],再到3D打印的骨移植物[10],都表明其在促成血管-成骨耦合中优异的效果。但生长因子的双递送相对复杂,如果双分子间的物理或化学性质相似,很难对双分子进行定量以及控制释放。 最近与生长因子相关的骨组织工程研究仍致力于借助生物材料来设计双模块控释系统,以增强递送效率,并实现长期利用。TANG等[11]开发了一种双模块支架,该支架由具有分级多孔结构的介孔生物活性玻璃(模块Ⅰ)和原位固定在模块Ⅰ中空通道中的水凝胶柱(模块Ⅱ)组成,通过将重组人骨形态发生蛋白2固定在模块Ⅰ中并将VEGF嵌入模块Ⅱ中,实现了由最初高浓度,随时间降低的VEGF和较慢、可持续的重组人骨形态发生蛋白2的组合释放模式。所用的双模块支架不仅增强了生长因子的生物活性,而且模仿其体内供应模式[11]。LEE等[12]开发了由明胶/壳聚糖冷冻凝胶和明胶/肝素冷冻凝胶包围的双重冷冻凝胶系统,同样初始释放VEGF,诱导血管生成,同时持续释放骨形态发生蛋白4以连续诱导成骨,增强临界尺寸的颅骨缺损模型的骨再生。 除了VEGF与骨形态发生蛋白2,还研究了其他生长因子的双递送:VEGF与成纤维细胞生长因子2共递送[13],骨形态发生蛋白2与促红细胞生成素或血小板衍生生长因子共递送[14-15];生长因子与其他促成血管分子双递送:骨形态发生蛋白2与钴离子[16]、QK肽等共递送[17],都表现出优异的成骨成血管效果。 2.1.3 单递送系统 递送单个促进成血管成骨的生长因子或重组生长因子可以避免双递送带来的缺陷。LIM等[18]将血管生成素1的重组嵌合体应用于大鼠颅骨缺损,术后4周和8周,实验组显示出比对照组更多的新骨形成和成熟;此外,该嵌合体增加了VEGF的基因表达,说明增强的骨生成与血管生成有关。其他几个分子,如血小板衍生生长因子[19]、基质细胞衍生因子1和胰岛素等[20-21],也已被证明在骨再生期间既可促进血管生成又可促进成骨。 尽管如此,递送生长因子依旧存在许多挑战:①半衰期短,不稳定,传统的向靶区快速注射后经体液扩散易于失活,不能达到原位局部表达;②难以在最佳时间控制释放,突然释放可能对邻近组织产生系统毒性;③生产昂贵。因此,除了向缓释技术方面的努力,目前已从生长因子治疗技术向基因治疗技术发展。 2.2 外泌体基因传递与成血管-成骨 递送生长因子成本高且效率低,而基因治疗利用天然细胞机制,传递生理水平的治疗性因子,可作为生长因子的替代疗法。基因治疗是一种将小的DNA或RNA序列传递至细胞或组织以纠正遗传缺陷或治疗疾病的技术。基因传递可以收集患者的细胞进行体外转染,也可以将携带治疗性基因的载体直接转移到患者体内。 为了促进骨骼血管化,病毒和非病毒载体均已被用于修饰细胞、传递基因[22]。与病毒载体相比,非病毒载体制造简单、成本更低并且毒性更小,但是转染效率显著降低;因此,大多数实验室致力于开发病毒转染法。但长久以来,生物安全、免疫反应等敏感问题限制了其临床应用。 最近,利用细胞外囊泡(外泌体)作为非病毒载体进行递送是基因治疗的热点。外泌体是具有蛋白质、核酸和脂质等复杂组成的纳米级(30-150 nm)细胞外囊泡[23]。与病毒载体相比,外泌体具有优异的生物相容性;与其他非病毒载体相比,外泌体具有有效的细胞内化作用,这些特点使外泌体成为再生医学领域中药物及基因递送的优良载体。已经证明,人脐带间充质干细胞和骨髓间充质干细胞产生的外泌体都可以促进大鼠骨折修复中的血管生成[24-25]。而且,结合骨髓间充质干细胞来源外泌体的支架,体外能加速血管再生,在体内促进骨再生[26]。更重要的是,可以基因调控亲代细胞,增加外泌体中的促血管生成分子,进一步增强这些外泌体的促血管生成及其促骨组织工程血管化的能力,见表1。"
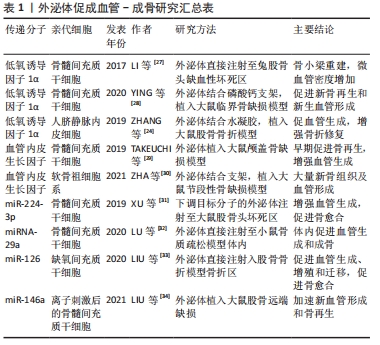

2.2.1 成血管基因 首先,在外泌体中过表达低氧诱导因子1α与VEGF,可以促进骨组织工程血管化。LI等[27]通过使骨髓间充质干细胞衍生的外泌体携带突变低氧诱导因子1α,减轻了兔股骨头缺血性坏死。该外泌体联合β-三磷酸钙支架可促进新骨再生和新生血管形成,效果优于单纯外泌体与支架相结合[28]。并且,最近的研究报道,来自人脐带间充质干细胞的外泌体也通过低氧诱导因子1α介导大鼠骨折模型的血管生成,并促进骨折愈合[24]。 另外,TAKEUCHI等[29]证明了在间充质干细胞外泌体诱导的血管生成中,VEGF起重要作用。他们发现,在大鼠颅盖骨缺损中,用外泌体处理的大鼠新生骨量增加,而用外泌体和血管生成抑制剂(抗VEGFA抗体)处理的大鼠的新生骨量明显减少,甚至低于PBS处理组[29]。ZHA等[30]使用小鼠软骨生成祖细胞系衍生的外泌体来包裹VEGF基因,并与3D打印的多孔骨支架有效结合,该支架植入大鼠节段性骨缺损12周后,有大量新骨组织及血管形成,成功诱导了血管化成骨。总之,在外泌体中过表达成血管因子,并结合成骨支架,可能是一种诱导骨骼血管化的可行方法。 2.2.2 非编码RNA 除了过表达成血管因子,外泌体中递送微小RNA(microRNA)也可以促进成血管与成骨。装载miR-29a的外泌体,可以靶向血管生成抑制蛋白1,在骨质疏松小鼠模型中促进血管生成和成骨[32]。类似地,骨髓间充质干细胞衍生的外泌体中的miR-224-3p[31]、miR-126和miR-146a也可以调节成骨和血管生成[33-34]。此外,长非编码RNA一直在癌症的血管生成方面应用广泛,最近发现,来自间充质干细胞的外泌体产生的长非编码RNA能增强血管生成[35],促进伤口愈合。但是,外泌体中长非编码RNA是否能促进成血管-成骨,以及携带成血管microRNA的外泌体在骨组织工程血管化中的作用,有待进一步研究。 外泌体作为一种新兴载体,还具有很多局限:外泌体包装非编码RNA的方法十分有限,目前多采用病毒转染法[23];此外,纳米级外泌体产量低,严重限制其潜在应用。最近,ZHA等[36]通过渐进式机械挤压从软骨生成祖细胞系获得一种特异的外泌体模拟物,可以实现大规模生产纳米级类似物。他们同时使用这种外泌体模拟物设计了一个基因激活基质,可以包裹VEGF质粒并灵活结合到核壳纳米纤维膜上,持续传递VEGF基因,显著增强体内血管化成骨[37]。这种外体模拟物,似乎是大量生产纳米外体的潜在方式,但更有效且生物安全的装载方式,仍需要大量探索。 2.3 生物活性离子与成血管-成骨 "

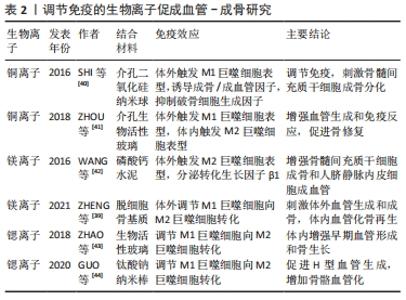
骨骼修复和再生中,工程设计功能性植入物的血管化至关重要。已经提出了不同的生物材料用于骨再生的合成骨移植物,但是缺损较大时,生物材料促进血管生成的能力有限,这限制了其在骨缺损修复中的应用[37]。装载生物活性物质,例如VEGF,可以改善生物材料的血管生成,但其作用时间很短,价格高昂。近年来,基于骨骼中羟基磷灰石的仿生成分,将生物活性离子掺入生物材料中,是一种增强血管生成与骨生成的有效方法[38]。 离子掺杂剂结合生物材料能促进骨骼修复,部分原因是它能改造免疫微环境,引发免疫反应,与成骨、成血管反应相互作用。其中,巨噬细胞的促炎M1表型和抗炎M2表型发挥了重要作用。M1巨噬细胞参与急性炎症,它在骨骼修复的早期释放促炎分子,诱导血管周围细胞的募集和尖端细胞的内皮细胞分化;而后M1巨噬细胞复极化形成抗炎M2巨噬细胞。M2巨噬细胞在骨骼愈合的后期发挥作用,有助于血管重塑,导致主动修复或产生纤维组织。巨噬细胞促炎M1表型和抗炎M2表型的协调转换,很可能决定了活跃的骨再生和植入物的成功[39]。已经探索了一些无机元素的控制释放,能调节免疫细胞功能和极化。在此,将讨论离子掺杂剂结合生物材料促进骨组织工程血管化的最新进展,尤其集中在免疫反应相关,见表2。"
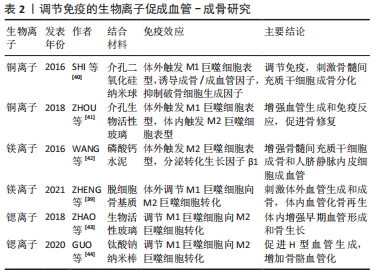
2.3.1 铜离子 据报道,铜离子(Cu2+)具有促血管生成和促成骨特性[45]。Cu2+通过低氧诱导因子1依赖性途径上调VEGF,增强血管形成和成骨分化[46]。引入Cu2+的中孔二氧化硅纳米球和中孔生物活性玻璃都可以通过免疫反应[40-41],提高这些材料的血管生成和骨形成。 SHI等[40]发现掺杂铜的中孔二氧化硅纳米球,在体外诱导巨噬细胞表达促血管生成分子和M1促炎分子,并促进骨髓间充质干细胞成骨。ZHOU等[41]发现将铜掺入中孔生物活性玻璃中,会产生一种低氧样微环境,诱导骨髓间充质干细胞分泌VEGF,刺激人脐静脉内皮细胞迁移与血管形成,并且触发组织的M2巨噬细胞浸润,有助于骨组织再生。与传统的中孔生物活性玻璃相比,铜掺杂能诱导体外巨噬细胞表达M1促炎分子,而在体内大鼠皮下的植入则导致更多的M2细胞浸润[41]。体内和体外结果的差异可能是由于体内微环境的复杂性所致,宿主的间充质干细胞在免疫反应中也起着一定作用,还需进一步探究。 2.3.2 镁离子 镁离子(Mg2+)除了诱导血管生成与骨形成[47-48],在免疫炎症过程中也能起重要作用。WANG等[42]发现镁-磷酸钙水泥能抑制巨噬细胞分泌促炎因子,并增加骨修复相关因子,即转化生长因子β1的分泌,创造的免疫微环境在体外增强了骨髓间充质干细胞成骨和人脐静脉内皮细胞的成血管潜力。与此相一致的是,ZHENG等[39]发现Mg2+能协调炎症,促进M1巨噬细胞复极化为促愈合的M2表型,刺激血管生成和成骨。具体来讲,他们构建了一种富镁氧化石墨烯纳米平台,其中氧化石墨烯激活了炎性M1巨噬细胞,而Mg2+促进了M1巨噬细胞复极化为促愈合的M2表型,两者协调刺激体外血管生成和成骨;在大鼠颅骨缺损模型中,通过纳米平台沉积的脱细胞骨基质支架也可以实现明显的血管化骨再生[39]。 2.3.3 锶离子 锶离子(Sr4+)能促进成骨细胞活性,抑制破骨细胞活性,已用作治疗骨质疏松症的临床药物[49]。支架引入Sr4+被证明具有强大血管再生能力。例如,锶-氧化石墨烯修饰的胶原蛋白支架,通过氧化石墨烯和Sr4+协同激活MAPK信号传导途径,促进成骨性骨形态发生蛋白2和血管生成性VEGF的分泌[50]。 此外,Sr4+对免疫反应有积极作用,Sr4+整合植入物会引发更多的促再生M2巨噬细胞。将Sr4+掺入亚微米生物活性玻璃颗粒中,可抑制巨噬细胞的促炎反应和破骨细胞生成[51],并增强大鼠股骨缺损模型中的早期血管形成和骨生长[43]。具体是,Sr4+调节M1巨噬细胞向M2巨噬细胞转化,M2亚型巨噬细胞表达高水平的血小板衍生生长因子BB,增强含锶生物活性玻璃微球的早期血管形成能力[43]。最近,GUO等[44]发现了Sr4+促进早期骨骼血管化的可能机制。他们制造了一个由Sr4+负载的钛酸钠纳米持续释放系统,释放的Sr4+同样调节巨噬细胞向M2型转化并表达高水平的血小板衍生生长因子BB,进而促进H型血管的生成,增加骨骼血管化[44]。然而,Sr4+对巨噬细胞行为的影响是浓度依赖性的[44],仍然需要进一步的研究来了解Sr4+影响巨噬细胞行为的潜在机制。 2.4 小分子活性肽与成血管-成骨 小分子活性肽(短肽)是人工合成的短肽序列,其氨基酸序列来源于天然功能蛋白,合成成本远低于全长蛋白,在靶向性、安全性及摄取效率等方面,具有明显优势。"

2.4.1 QK肽 VEGF具有优越的成血管成骨功能、高免疫原性和低稳定性,已经开发了许多VEGF模拟肽。其中,具有15个氨基酸的QK肽,它模拟VEGF的17-25螺旋区域,可以结合VEGF受体,拥有显著的成血管活性[52]。此外,CHEN等[53]首次证明了QK肽的成骨活性。 QK肽结合骨组织工程的研究正在进行。SUN等[37]将QK肽加载入介孔二氧化硅中,促进了大鼠临界尺寸颅骨缺损模型中人内皮细胞管状结构的形成和体内血管生成,并改善碱性磷酸酶的活性、矿化结节的形成以及骨髓间充质干细胞成骨相关基因和蛋白质的表达,增强了骨修复和血管生成的能力。QK肽和骨形态发生蛋白2衍生肽交联的纳米羟基磷灰石/聚酰胺支架,可以增强血管形成,提高骨髓间充质干细胞的成骨分化能力,预防SD大鼠股骨不愈合[17]。 2.4.2 Exendin-4 Exendin-4是胰高血糖素样肽1受体的完全激动剂,广泛用于2型糖尿病的临床治疗[54]。Exendin-4能促进血管生成。KANG等[55]发现exendin-4可以通过VEGF血管生成相关途径诱导血管生成,从而预防小鼠后肢缺血性损伤。SEO等[54]发现EX-4可以加速糖尿病小鼠伤口愈合,并且能改善体外人内皮细胞的迁移、侵袭和增殖,增加VEGF的表达。关于其促成血管机制,有研究证明exendin-4通过激活PI3K/AKT途径,参与人冠状动脉内皮细胞的血管生成[56]。此外,Exendin-4还可以促进成骨。已报道它可以促进间充质干细胞的增殖和迁移,PI3K/AKT-SDF-1/CXCR4信号传导途径在其中起到重要的作用[57]。最近,研究已经证实,Exendin-4具有促进成骨细胞分化的能力,抑制多种干细胞/前体细胞的成脂分化和促进骨形成以修复骨缺损[58]。因此,Exendin-4可能促进了成血管-成骨耦合,还需结合具体实验,探索其应用于骨组织工程的可能。 2.4.3 Prominin-1 衍生肽 Prominin-1 衍生肽是ADINI等[59]设计的一种新型的12个氨基酸的肽,其序列源自促血管生成糖蛋白prominin-1的胞外VEGF结合域。该肽直接结合VEGF,并增强VEGF与内皮细胞以及VEGF受体VEGFR2和神经纤毛蛋白1的结合。但Prominin-1 衍生肽增强成血管-成骨作用是通过VEGF实现的,仅当与VEGF结合使用时,Prominin-1 衍生肽可增加血管生成,不添加VEGF则无活性,因而其应用受到极大限制。 2.4.4 骨形成肽1 骨形成肽1源自骨形成蛋白7的未成熟区域,其成骨作用显著[60]。研究报道,骨形成肽1也可以通过上调内皮细胞中VEGF受体的表达水平来增强血管形成[61],此外,还能激活ALK-1/Smad 通路来促进内皮祖细胞的血管生成[62]。骨形成肽1与支架结合,能促进β-磷酸三钙支架血管化,诱导比格犬的下颌骨缺损中新骨形成,增加下颌骨的高度[61]。WANG等[62]发现与单独的β-磷酸三钙支架相比,结合骨形成肽1与内皮祖细胞的支架改善了支架血管化,促进了兔股骨节段性缺损的血管形成与骨再生。目前骨形成肽1在骨组织工程中多应用于成骨,关于其成血管-成骨效应,还需要进一步的研究。 2.5 中药单体与成血管-成骨 复方中药,如益气活血药[63],可以促进骨修复中的血管生成。复方中药机制复杂,在此主要讨论促进成血管-成骨耦合的中药单体。红景天苷(SAL)是玫瑰红景天的主要生物活性化合物,能通过低氧诱导因子1α与VEGF信号传导,促进血管生成,加速骨折愈合[64]。西洋参皂苷能加速人微血管内皮细胞的迁移,促进VEGF和头蛋白的分泌,并增加后肢卸载小鼠的成骨细胞活性和H型血管数量[65]。骨碎补总黄酮是干燥骨碎补根茎中提取的有效成分,通过 Notch 信号通路促进成血管-成骨耦联,加速了骨缺损愈合[66]。三七总皂苷是一种活血化瘀药,其促骨生成机制不明,但已被证明能通过血管生成改善绝经后骨质疏松[67]。柚皮苷及淫羊藿苷可以促进大鼠骨折早期愈合过程中VEGF的表达,并增强骨折愈合强度[68-69]。 综上,该文探讨了近年来促进血管生成,修复骨骼缺损的几种前沿方式与进展,生长因子、递送基因、生物活性离子、小分子活性肽和中药单体等都可以促进骨骼血管化,各有其优势,同时也面临不同的挑战。"

| [1] DIOMEDE F, MARCONI GD, FONTICOLI L, et al. Functional relationship between osteogenesis and angiogenesis in tissue regeneration. Int J Mol Sci. 2020;21(9): 3242. [2] HANKENSON KD, DISHOWITZ M, GRAY C, et al. Angiogenesis in bone regeneration. Injury. 2011;42(6):556-561. [3] KUSUMBE AP, RAMASAMY SK, ITKIN T, et al. Age-dependent modulation of vascular niches for haematopoietic stem cells. Nature. 2016;532(7599):380-384. [4] DOS SANTOS BP, GARBAY B, FENELON M, et al. Development of a cell-free and growth factor-free hydrogel capable of inducing angiogenesis and innervation after subcutaneous implantation. Acta Biomater. 2019;99:154-167. [5] LIU Y, OLSEN BR. Distinct vegf functions during bone development and homeostasis. Arch Immunol Ther Exp (Warsz). 2014;62(5):363-368. [6] BESSA PC, CASAL M, REIS RL. Bone morphogenetic proteins in tissue engineering: The road from laboratory to clinic, part ii (bmp delivery). J Tissue Eng Regen Med. 2008;2(2-3):81-96. [7] SHARMA S, SAPKOTA D, XUE Y, et al. Delivery of vegfa in bone marrow stromal cells seeded in copolymer scaffold enhances angiogenesis, but is inadequate for osteogenesis as compared with the dual delivery of vegfa and bmp2 in a subcutaneous mouse model. Stem Cell Res Ther. 2018;9(1):23. [8] SUKUL M, NGUYEN TB, MIN YK, et al. Effect of local sustainable release of bmp2-vegf from nano-cellulose loaded in sponge biphasic calcium phosphate on bone regeneration. Tissue Eng Part A. 2015;21(11-12):1822-1836. [9] BARATI D, SHARIATI SRP, MOEINZADEH S, et al. Spatiotemporal release of bmp-2 and vegf enhances osteogenic and vasculogenic differentiation of human mesenchymal stem cells and endothelial colony-forming cells co-encapsulated in a patterned hydrogel. J Control Release. 2016;223:126-136. [10] BAO X, ZHU L, HUANG X, et al. 3d biomimetic artificial bone scaffolds with dual-cytokines spatiotemporal delivery for large weight-bearing bone defect repair. Sci Rep. 2017;7(1):7814. [11] TANG W, YU Y, WANG J, et al. Enhancement and orchestration of osteogenesis and angiogenesis by a dual-modular design of growth factors delivery scaffolds and 26scs decoration. Biomaterials. 2020;232:119645. [12] LEE SS, KIM JH, JEONG J, et al. Sequential growth factor releasing double cryogel system for enhanced bone regeneration. Biomaterials. 2020;257:120223. [13] LARSEN M, WILLEMS WF, PELZER M, et al. Fibroblast growth factor-2 and vascular endothelial growth factor mediated augmentation of angiogenesis and bone formation in vascularized bone allotransplants. Microsurgery. 2014;34(4):301-307. [14] PATEL JJ, MODES JE, FLANAGAN CL, et al. Dual delivery of epo and bmp2 from a novel modular poly-ɛ-caprolactone construct to increase the bone formation in prefabricated bone flaps. Tissue Eng Part C Methods. 2015;21(9):889-897. [15] BAYER EA, JORDAN J, ROY A, et al. (*) programmed platelet-derived growth factor-bb and bone morphogenetic protein-2 delivery from a hybrid calcium phosphate/alginate scaffold. Tissue Eng Part A. 2017;23(23-24):1382-1393. [16] PEREZ RA, KIM JH, BUITRAGO JO, et al. Novel therapeutic core-shell hydrogel scaffolds with sequential delivery of cobalt and bone morphogenetic protein-2 for synergistic bone regeneration. Acta Biomater. 2015;23:295-308. [17] LI A, LI J, ZHANG Z, et al. Nanohydroxyapatite/polyamide 66 crosslinked with qk and bmp-2-derived peptide prevented femur nonunion in rats. J Mater Chem B. 2021;9(9):2249-2265. [18] LIM SS, KOOK SH, BHATTARAI G, et al. Local delivery of comp-angiopoietin 1 accelerates new bone formation in rat calvarial defects. J Biomed Mater Res A. 2015;103(9):2942-2951. [19] PHIPPS MC, XU Y, BELLIS SL. Delivery of platelet-derived growth factor as a chemotactic factor for mesenchymal stem cells by bone-mimetic electrospun scaffolds. PLoS One. 2012;7(7):e40831. [20] LIU H, LI M, DU L, et al. Local administration of stromal cell-derived factor-1 promotes stem cell recruitment and bone regeneration in a rat periodontal bone defect model. Mater Sci Eng C Mater Biol Appl. 2015;53:83-94. [21] PAGLIA DN, WEY A, BREITBART EA, et al. Effects of local insulin delivery on subperiosteal angiogenesis and mineralized tissue formation during fracture healing. J Orthop Res. 2013;31(5):783-791. [22] 冯一梅,徐辉.间充质干细胞基因转染的相关研究现状[J].中国组织工程研究与临床康复,2007,11(11):2118-2121. [23] ELSHARKASY OM, NORDIN JZ, HAGEY DW, et al. Extracellular vesicles as drug delivery systems: Why and how? Adv Drug Deliv Rev. 2020;159:332-343. [24] ZHANG Y, HAO Z, WANG P, et al. Exosomes from human umbilical cord mesenchymal stem cells enhance fracture healing through hif-1α-mediated promotion of angiogenesis in a rat model of stabilized fracture. Cell Prolif. 2019; 52(2):e12570. [25] ZHANG L, JIAO G, REN S, et al. Exosomes from bone marrow mesenchymal stem cells enhance fracture healing through the promotion of osteogenesis and angiogenesis in a rat model of nonunion. Stem Cell Res Ther. 2020;11(1):38. [26] XIE H, WANG Z, ZHANG L, et al. Extracellular vesicle-functionalized decalcified bone matrix scaffolds with enhanced pro-angiogenic and pro-bone regeneration activities. Sci Rep. 2017;7:45622. [27] LI H, LIU D, LI C, et al. Exosomes secreted from mutant-hif-1α-modified bone-marrow-derived mesenchymal stem cells attenuate early steroid-induced avascular necrosis of femoral head in rabbit. Cell Biol Int. 2017;41(12):1379-1390. [28] YING C, WANG R, WANG Z, et al. Bmsc-exosomes carry mutant hif-1α for improving angiogenesis and osteogenesis in critical-sized calvarial defects. Front Bioeng Biotechnol. 2020;8:565561. [29] TAKEUCHI R, KATAGIRI W, ENDO S, et al. Exosomes from conditioned media of bone marrow-derived mesenchymal stem cells promote bone regeneration by enhancing angiogenesis. PLoS One. 2019;14(11):e0225472. [30] ZHA Y, LI Y, LIN T, et al. Progenitor cell-derived exosomes endowed with vegf plasmids enhance osteogenic induction and vascular remodeling in large segmental bone defects. Theranostics. 2021;11(1):397-409. [31] XU HJ, LIAO W, LIU XZ, et al. Down-regulation of exosomal microrna-224-3p derived from bone marrow-derived mesenchymal stem cells potentiates angiogenesis in traumatic osteonecrosis of the femoral head. FASEB J. 2019;33(7): 8055-8068. [32] LU GD, CHENG P, LIU T, et al. Bmsc-derived exosomal mir-29a promotes angiogenesis and osteogenesis. Front Cell Dev Biol. 2020;8:608521. [33] LIU W, LI L, RONG Y, et al. Hypoxic mesenchymal stem cell-derived exosomes promote bone fracture healing by the transfer of mir-126. Acta Biomater. 2020; 103:196-212. [34] LIU L, YU F, LI L, et al. Bone marrow stromal cells stimulated by strontium-substituted calcium silicate ceramics: Release of exosomal mir-146a regulates osteogenesis and angiogenesis. Acta Biomater. 2021;119:444-457. [35] BORN LJ, CHANG KH, SHOURESHI P, et al. Hotair-loaded mesenchymal stem/stromal cell extracellular vesicles enhance angiogenesis and wound healing. Adv Healthc Mater. 2021:e2002070. [36] ZHA Y, LIN T, LI Y, et al. Exosome-mimetics as an engineered gene-activated matrix induces in-situ vascularized osteogenesis. Biomaterials. 2020;247:119985. [37] SUN P, ZHANG Q, NIE W, et al. Biodegradable mesoporous silica nanocarrier bearing angiogenic qk peptide and dexamethasone for accelerating angiogenesis in bone regeneration. ACS Biomater Sci Eng. 2019;5(12):6766-6778. [38] BOSE S, FIELDING G, TARAFDER S, et al. Understanding of dopant-induced osteogenesis and angiogenesis in calcium phosphate ceramics. Trends Biotechnol. 2013;31(10):594-605. [39] ZHENG Z, CHEN Y, HONG H, et al. The “yin and yang” of immunomodulatory magnesium-enriched graphene oxide nanoscrolls decorated biomimetic scaffolds in promoting bone regeneration. Adv Healthc Mater. 2021;10(2):e2000631. [40] SHI M, CHEN Z, FARNAGHI S, et al. Copper-doped mesoporous silica nanospheres, a promising immunomodulatory agent for inducing osteogenesis. Acta Biomater. 2016;30:334-344. [41] ZHOU Y, HAN S, XIAO L, et al. Accelerated host angiogenesis and immune responses by ion release from mesoporous bioactive glass. J Mater Chem B. 2018;6(20):3274-3284. [42] WANG M, YU Y, DAI K, et al. Improved osteogenesis and angiogenesis of magnesium-doped calcium phosphate cement via macrophage immunomodulation. Biomater Sci. 2016;4(11):1574-1583. [43] ZHAO F, LEI B, LI X, et al. Promoting in vivo early angiogenesis with sub-micrometer strontium-contained bioactive microspheres through modulating macrophage phenotypes. Biomaterials. 2018;178:36-47. [44] GUO S, YU D, XIAO X, et al. A vessel subtype beneficial for osteogenesis enhanced by strontium-doped sodium titanate nanorods by modulating macrophage polarization. J Mater Chem B. 2020;8(28):6048-6058. [45] WU C, ZHOU Y, XU M, et al. Copper-containing mesoporous bioactive glass scaffolds with multifunctional properties of angiogenesis capacity, osteostimulation and antibacterial activity. Biomaterials. 2013;34(2):422-433. [46] ZHANG W, CHANG Q, XU L, et al. Graphene oxide-copper nanocomposite-coated porous cap scaffold for vascularized bone regeneration via activation of hif-1α. Adv Healthc Mater. 2016;5(11):1299-1309. [47] MAIER JA, BERNARDINI D, RAYSSIGUIER Y, et al. High concentrations of magnesium modulate vascular endothelial cell behaviour in vitro. Biochim Biophys Acta. 2004;1689(1):6-12. [48] ZHANG Y, XU J, RUAN YC, et al. Implant-derived magnesium induces local neuronal production of cgrp to improve bone-fracture healing in rats. Nat Med. 2016;22(10):1160-1169. [49] PENG S, LIU X S, HUANG S, et al. The cross-talk between osteoclasts and osteoblasts in response to strontium treatment: Involvement of osteoprotegerin. Bone. 2011;49(6):1290-1298. [50] CHEN Y, ZHENG Z, ZHOU R, et al. Developing a strontium-releasing graphene oxide-/collagen-based organic-inorganic nanobiocomposite for large bone defect regeneration via mapk signaling pathway. ACS Appl Mater Interfaces. 2019;11(17):15986-15997. [51] ZHANG W, ZHAO F, HUANG D, et al. Strontium-substituted submicrometer bioactive glasses modulate macrophage responses for improved bone regeneration. ACS Appl Mater Interfaces. 2016;8(45):30747-30758. [52] D’ANDREA LD, IACCARINO G, FATTORUSSO R, et al. Targeting angiogenesis: Structural characterization and biological properties of a de novo engineered vegf mimicking peptide. Proc Natl Acad Sci U S A. 2005;102(40):14215-14220. [53] CHEN J, HU G, LI T, et al. Fusion peptide engineered “statically-versatile” titanium implant simultaneously enhancing anti-infection, vascularization and osseointegration. Biomaterials. 2021;264:120446. [54] SEO E, LIM J S, JUN JB, et al. Exendin-4 in combination with adipose-derived stem cells promotes angiogenesis and improves diabetic wound healing. J Transl Med. 2017;15(1):35. [55] KANG HM, SOHN I, JUNG J, et al. Exendin-4 protects hindlimb ischemic injury by inducing angiogenesis. Biochem Biophys Res Commun. 2015;465(4):758-763. [56] QI X, LIU H, MAO L, et al. Combination of exendin-4 and dpp-4 silencing promoted angiogenesis of human coronary artery endothelial cells via activation of pi3k/akt pathway. Pak J Pharm Sci. 2017;30(2(Suppl.)):555-560. [57] ZHOU H, LI D, SHI C, et al. Effects of exendin-4 on bone marrow mesenchymal stem cell proliferation, migration and apoptosis in vitro. Sci Rep. 2015;5:12898. [58] MENG J, MA X, WANG N, et al. Activation of glp-1 receptor promotes bone marrow stromal cell osteogenic differentiation through β-catenin. Stem Cell Reports. 2016;6(4):579-591. [59] ADINI A, ADINI I, CHI ZL, et al. A novel strategy to enhance angiogenesis in vivo using the small vegf-binding peptide pr1p. Angiogenesis. 2017;20(3):399-408. [60] KIM HK, KIM JH, PARK DS, et al. Osteogenesis induced by a bone forming peptide from the prodomain region of bmp-7. Biomaterials. 2012;33(29):7057-7063. [61] YU T, LIU Q, JIANG T, et al. Channeled β-tcp scaffolds promoted vascularization and bone augmentation in mandible of beagle dogs. Advanced Functional Materials. 2016;26(37):6719-6727. [62] WANG H, CHENG H, TANG X, et al. The synergistic effect of bone forming peptide-1 and endothelial progenitor cells to promote vascularization of tissue engineered bone. J Biomed Mater Res A. 2018;106(4):1008-1021. [63] 周毅,陈跃平,章晓云,等.复方中药圣愈汤治疗股骨头坏死机制的网络药理学分析[J]. 中国组织工程研究,2021,25(17):2687-2696. [64] GUO Q, YANG J, CHEN Y, et al. Salidroside improves angiogenesis-osteogenesis coupling by regulating the hif-1α/vegf signalling pathway in the bone environment. Eur J Pharmacol. 2020;884:173394. [65] LIANG S, LING S, DU R, et al. The coupling of reduced type h vessels with unloading-induced bone loss and the protection role of panax quinquefolium saponin in the male mice. Bone. 2021;143:115712. [66] 黄敏玲,卢赵琦,申震,等.骨碎补总黄酮干预notch信号通路影响骨重建过程中成血管-成骨耦联[J].中国组织工程研究,2021,25(32):5116-5122. [67] 王晓燕,李冠武,常时新.三七总皂苷通过血管生成改善绝经后骨质疏松机制探析[J].中国骨质疏松杂志,2014,20(8):964-967+977. [68] 赵志虎,孟新民,孙晓雷,等.柚皮苷对去势大鼠骨折骨痂血管发生的影响及其机制[J].中华骨科杂志, 2016,36(3):177-183. [69] 武通,陈东宁,许占江.基于vegf/vegfr-2信号通路研究淫羊藿苷促进大鼠胫骨干骨折愈合的作用机制[J].浙江中医药大学学报,2021,45(3):282-288,293. |
| [1] | Xue Yadong, Zhou Xinshe, Pei Lijia, Meng Fanyu, Li Jian, Wang Jinzi . Reconstruction of Paprosky III type acetabular defect by autogenous iliac bone block combined with titanium plate: providing a strong initial fixation for the prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1424-1428. |
| [2] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [3] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [4] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [5] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [6] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [7] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [8] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [9] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [10] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [11] | Huang Chenwei, Fei Yankang, Zhu Mengmei, Li Penghao, Yu Bing. Important role of glutathione in stemness and regulation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1119-1124. |
| [12] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [13] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1005-1011. |
| [14] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| [15] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

