Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (5): 780-785.doi: 10.12307/2022.127
Previous Articles Next Articles
Molecular mechanism by which icariin regulates osteogenic signaling pathways in the treatment of steroid-induced avascular necrosis of the femoral head
Li Jiajun1, Xia Tian1, Liu Jiamin1, Chen Feng1, Chen Haote2, Zhuo Yinghong1, Wu Weifeng1
- 1Guangxi University of Traditional Chinese Medicine, Nanning 530001, Guangxi Zhuang Autonomous Region, China; 2China Academy of Chinese Medical Sciences, Beijing 100000, China
-
Received:2020-11-11Revised:2020-11-14Accepted:2020-12-18Online:2022-02-18Published:2021-12-02 -
Contact:Xia Tian, MD candidate, Associate professor, Guangxi University of Traditional Chinese Medicine, Nanning 530001, Guangxi Zhuang Autonomous Region, China -
About author:Li Jiajun, Guangxi University of Traditional Chinese Medicine, Nanning 530001, Guangxi Zhuang Autonomous Region, China -
Supported by:the Natural Science Foundation of Guangxi Zhuang Autonomous Region for the Youth, No. 2020GXNSFBA159053; Young Teachers' Basic Ability Improvement Project in Guangxi Universities, No. 2019KY0352; 2019 School-level Scientific Research Project of Guangxi University of Traditional Chinese Medicine, No. 2019QN027; First-class Subject Project of Guangxi University of Traditional Chinese Medicine, No. 2019XK029; 2016 National Famous Traditional Chinese Medicine Heritage Studio Construction Project, No. {2016}11; National College Student Innovation and Entrepreneurship Training Program, No. 202010600025 (to LJJ)
CLC Number:
Cite this article
Li Jiajun, Xia Tian, Liu Jiamin, Chen Feng, Chen Haote, Zhuo Yinghong, Wu Weifeng. Molecular mechanism by which icariin regulates osteogenic signaling pathways in the treatment of steroid-induced avascular necrosis of the femoral head[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 780-785.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
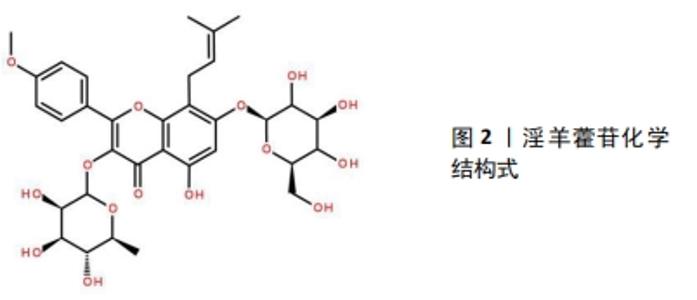
2.1 淫羊藿苷在骨代谢中的作用 在中医里SANFH归属为“骨蚀”“骨痿”“骨痹”“骨痰”等范畴。中医认为肾“主骨生髓”,为先天之本。激素有“纯阳”之性,激素的使用提前激活和耗竭肾脏封藏的肾阳之气,一方面易出现“阳胜劫阴”,阴精耗损而无以濡养、滋润骨骼,另一方面肾阳提前调动激活,使本身存储的肾阳过度耗损而匮乏。又因SANFH多久病,故肾精亏虚,肾阳虚损;就“阴阳互化”“少火生气”理论,针对肾阳虚损过多,提出“温阳补肾”的治法;淫羊藿能温补虚损之肾阳,以求补养肾阳而肾阴得养,实现温阳补肾,填髓强骨的目的[8]。 淫羊藿别称仙灵脾,药用历史悠久,最开始记载于《神农本草经》,性味辛、甘、温,归肾、肝经,具有壮阳补肾、强筋骨、除湿祛风的功效,主治筋骨痿软、腰酸腿痛等病症。历代医学典籍中的相关记载:如《别录》述云:淫羊藿“坚筋骨……”;《本经》记载淫羊藿“主阴痿瘘绝伤,莲中痛。利小便,益气力,强志”;《医学入门》描述其云:淫羊藿“补肾虚,助阳。治偏风手足不遂,四肢皮肤不仁”等。根据中医“治病求本”“虚则补之”的治则,治疗SANFH当温肾助阳,而淫羊藿是温补肾阳的代表中药之一,故选其作为单味中药治疗SANFH的研究。从淫羊藿干燥茎叶中提取的淫羊藿苷为8-异戊烯基黄酮苷类化合物,见图2,是传统中草药淫羊藿的重要活性成分,其提高了淫羊藿中药制剂的内在质量,使淫羊藿的临床治疗效果得以最大限度地发挥,改善骨内血循环和造血功能、促进骨细胞代谢及提高机体免疫能力等。"
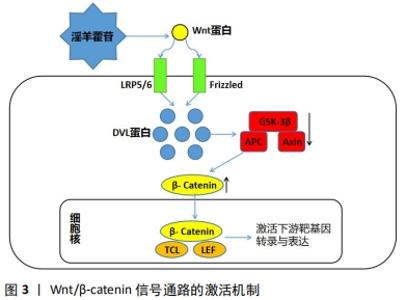
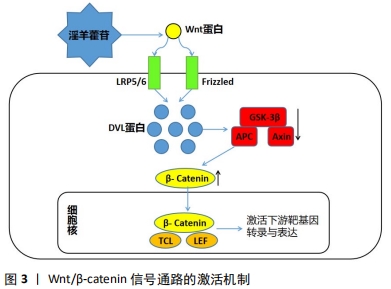
2.1.1 淫羊藿苷促进成骨细胞形成 淫羊藿苷可通过刺激骨形成来预防骨质流失,增强骨的生物力学性能。实验证实淫羊藿可通过降低骨代谢、改善骨矿含量、提高骨生物力学性能,显著减轻骨质疏松症对大鼠的影响[9]。ZHANG 等[10]学者证实淫羊藿苷可作用于雌激素受体介导的通路促进和刺激成骨细胞的分化与增殖。MA等[11]研究证实淫羊藿苷可减轻氧化应激和细胞凋亡,并且能保留体外缺氧的成骨细胞活力和成骨潜能,提示其作用可归因于其抗缺氧活性和植物雌激素特性。在淫羊藿苷对MAPK信号通路活化的研究中,淫羊藿苷能快速促进ERK1/2的磷酸化及胞内钙离子浓度的增加[12]。当给予内质网应激拮抗剂和MEK1特异性抑制剂预处理成骨细胞后,淫羊藿苷对ERK1/2的活化及升高胞内钙离子浓度作用均被抑制,表明淫羊藿苷通过MAPK信号通路提高胞内钙离子浓度促进成骨。以上细胞实验说明淫羊藿苷不仅能通过其自身的抗缺氧活性和植物雌激素特性来促进成骨细胞的形成,而且还能通过信号通路和调节细胞体内的钙离子浓度促进成骨作用。 在动物实验中,WANG等[13]研究表明,淫羊藿苷能够通过竞争细胞铁离子来抑制脯氨酸羟化酶活性,激活软骨细胞中缺氧诱导因子1的表达,通过调节软骨细胞的增殖、分化与软骨下骨形成的整合来促进关节软骨修复。为了进一步确定淫羊藿苷在体内关节软骨修复中的作用,通过构建海藻酸三维复合物小鼠股骨远端软骨间切迹骨软骨缺损模型,进一步表明淫羊藿苷通过增强软骨下骨形成和透明软骨覆盖的恢复,促进关节软骨缺损的修复和软骨细胞的增殖。以淫羊藿苷为基础的新型治疗方法在临床上已经被研究用于预防和治疗骨质疏松症。在健康的晚期绝经妇女中进行了24个月的随机双盲安慰剂对照临床试验,以探讨淫羊藿苷预防骨丢失的疗效,结果表明,相比安慰剂组(n=50),干预组(n=50)有显著减少骨质流失、促进成骨作用,增加骨密度;且长期(长达12-24个月)服用淫羊藿苷产品可改善腰椎和股骨颈的骨密度,并呈时间依赖性[14]。 2.1.2 淫羊藿苷抑制骨吸收 骨是一种动态组织,破骨细胞是骨组织中唯一具有介导骨吸收作用的细胞,破骨细胞的过度增殖会导致骨质大量被吸收,造成股骨头的坏死和塌陷。大量研究表明,淫羊藿苷不仅能够通过上调成骨通路的表达诱导干细胞成骨分化,提高成骨细胞活性,增强骨强度,同时它也能通过调节OPG/RANK/RANKL等细胞通路降低破骨细胞活性,从而抑制骨的吸收[15]。YANG等[16]研究发现淫羊藿总黄酮可以有效促进骨形成,抑制骨吸收,其机制可能与其对TGF-β1/Smads通路的调控有关。陈喆[17]通过实验发现单核细胞的破骨分化过程能被淫羊藿苷稳定且有效地抑制,且其作用效果能随着淫羊藿苷浓度的升高而逐渐增强,12 h达到稳定抑制水平。也有研究应用淫羊藿苷口服给药骨质疏松的牙周炎小鼠,发现口服淫羊藿苷的小鼠不仅有效减少了牙周炎引起的牙槽骨吸收,也可预防小鼠骨质疏松的发生[18]。通过国内外对淫羊藿苷抑制破骨分化的研究中,不难看出淫羊藿苷不仅能通过细胞水平上调节基因的表达来控制破骨细胞的分化,而且在动物个体实验上淫羊藿苷能可通过口服给药的方式使产生的代谢产物抑制骨吸收,发挥着抗骨质疏松、抗激素性股骨头坏死的作用。 综上,作者认为淫羊藿苷既能促进骨细胞的形成和分化又可抑制骨吸收,调节破骨作用,具有双重性。目前淫羊藿苷对成骨细胞具有的这种双重作用在治疗骨质疏松、股骨头坏死和骨性关节炎等方面具有很好的临床疗效,现已逐渐被大量临床医学工作者所接受。 2.2 淫羊藿苷抗SANFH的相关通路 近年来大量实验和研究表明有多条成骨信号通路在调控SANFH过程中发挥着重要作用,主要涉及的成骨信号通路有Wnt、MAPK、PI3K/AKT、mTOR、Notch、RANK/RANKL、miRNA-335、TRAF6 /NF-κB/ ERK、雌激素信号通路等,其中关于Wnt、MAPK和Notch等信号通路现研究多集中于对成骨细胞的增殖分化的影响,在PI3K/AKT信号通路相关实验中主要关注于其对骨代谢的调控,在mTOR、RANK/RANKL等信号通路中则聚焦于其对成骨细胞自噬作用的探讨等。现依据SANFH的发病机制特点,将淫羊藿苷治疗SANFH相对应具有代表性的分子机制进行阐述。 2.2.1 淫羊藿苷调控 Wnt/β-catenin信号通路促进成骨作用提高骨密度 Wnt信号通路与细胞的增殖、分化与迁移过程具有紧密联系。Wnt/β-catenin信号通路属于Wnt信号通路中的一种,该途径会导致β-连环蛋白在细胞质中积累并最终会作为属于TCF的转录因子的转录共激活因子/LEF家族易位至细胞核。没有Wnt信号通路的介导,β-连环蛋白会被破坏复合物降解从而不会在细胞质中积累。经典的Wnt途径(Wnt/β-catenin信号通路)能导致基因转录的调节,并且被认为部分由SPATS1基因负调节。SANFH的早期病理变化可能是由于骨髓间充质干细胞的异常增殖分化导致成骨细胞的凋亡,而Wnt/β-catenin信号通路在调节骨髓间充质干细胞的成骨分化进程中发挥着关键作用。 已有实验证实淫羊藿苷可通过Wnt/β-catenin 通路促进成骨,其作用机制为细胞内的散乱蛋白DVL通过细胞膜上的LRP5/6、Fz复合体与细胞外的Wnt蛋白结合后被激活,进而抑制APC、Axin和GSK-3β复合体,解除β-Catenin的降解作用,使细胞内β-Catenin 浓度升高,在细胞核内与转录因子(TCL/LEF)结合,从而诱导靶基因(C-myc、Runx-2、OSX)转录和表达[19],见图3。"
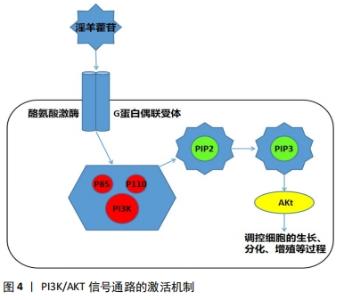
研究表明,低剂量的淫羊藿苷具有激活Wnt/β-catenin信号通路促进骨髓间充质干细胞增殖的作用[20]。淫羊藿苷可强化Wnt信号通路,刺激骨髓间充质干细胞向成骨细胞分化,同时能够抑制破骨细胞作用且抑制骨吸收活性,从而促进骨形成[21-22]。WEI等 [23]进行实验的结果显示,一定浓度的淫羊藿苷可以显著促进骨髓间充质干细胞的增殖,而且能够提高Wnt/β-catenin 通路相关蛋白和成骨相关基因的表达。LIU等[24]通过小鼠MC3T3-E1细胞置于四点弯曲装置的机械拉伸应变下,建立超负荷损伤模型,采用MTT法测定淫羊藿苷对MC3T3-E1细胞增殖的影响,发现淫羊藿苷可促进β-catenin和RUNX2基因的表达水平,增强超载下成骨细胞的增殖,并促进小鼠胚胎成骨细胞前体细胞MC3T3-E1的分化。WANG等[25]在研究钛(Ti)颗粒是否影响间充质干细胞的分化实验中,也证实了淫羊藿苷可通过稳定连接β-catenin蛋白和激活Wnt/β-catenin信号通路来减弱钛颗粒对成骨细胞的抑制作用。综上,作者认为,淫羊藿苷诱导成骨细胞增殖和分化进而提高骨质、防止股骨头塌陷的作用依赖于Wnt信号通路,而且主要是激活Wnt经典通路,从而能达到抗SANFH的目的。 2.2.2 淫羊藿苷激活PI3K/AKT信号通路调控血运及成骨分化影响股骨头缺血性坏死 SANFH两大主要损伤机制分别为血管损伤或股骨头周围血运障碍和股骨头局部骨质疏松,因此促进骨细胞的增殖生长延缓凋亡和找到相关机制改善血运障碍调控血管的修复,对防治激素性股骨头坏死至关重要。 PI3K/Akt通路是与细胞的增殖、生长与分化等基本过程密切相关的关键通路,PI3K/Akt通路已多次被证实参与成骨细胞分化过程[26]。其激活过程为外来的配体与细胞膜上的酪氨酸激酶受体或G蛋白偶联受体相结合,通过磷酸化酪氨酸残基,从而使细胞质内的调控亚基p85激活[27-29],与此同时,其相邻的催化亚基p110也被激活,使得磷脂酰肌醇(3,4)-二磷酸(PIP2)被磷酸化,转化为磷脂酰肌醇(3,4,5)-三磷酸(PIP3)[29-30],激活处在下游的AKT信号,从而调控细胞的增殖、生长与分化等过程[31]。淫羊藿苷可作为配体通过此机制激活PI3K/AKT 信号通路,见图4。"
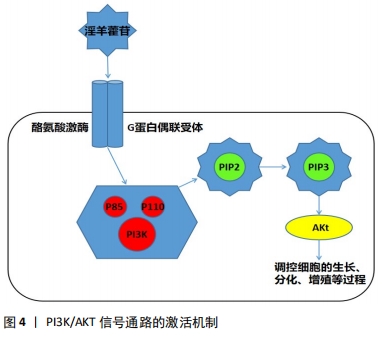
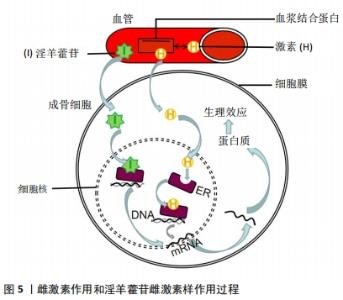
目前发现PI3K/AKT信号通路可以对一氧化氮、血管内皮生长因子、血管生成素、缺氧诱导因子等血管修复再生的关键因子进行调控[27],而且先前已有研究表明增强PI3K/AKT信号可以刺激成骨细胞的生长增殖、延缓其凋亡进程,这对治疗骨质疏松极为有利[32]。因此作者认为淫羊藿苷通过刺激PI3K/AKT信号通路调控一氧化氮的表达与产生、促进成骨细胞生长增殖两方面影响SANFH的发生与发展。一氧化氮具有扩张血管和增加血管通透性的能力,PI3K/AKT信号通路具有调节一氧化氮在体内产生与表达。低氧诱导因子可被PI3K/AKT诱导表达,促进机体内血管内皮生长因子增殖,刺激一氧化氮的分泌。且AKT在调控一氧化氮表达与产生的同时还能够促进血管内皮细胞的迁移[33]。实验研究表明,PI3K/AKT/eNOS信号通路可有效调控一氧化氮的表达与产生过程[34-35]。 ZHOU等[26]通过研究发现原硅酸通过促进体外PI3K-Akt-mTOR信号通路的表达积极调节成骨过程,证实PI3K-Akt-mTOR通路在成骨过程的上游起着重要作用。也有研究发现,淫羊藿苷通过激活PI3K/AKT信号通路促进成骨细胞分化,预防糖皮质激素引起的骨质疏松症[36]。淫羊藿苷通过PI3K/Akt信号通路不仅能调节一氧化氮改善血运、提高骨代谢,而且还能激活骨形态发生蛋白、缺氧诱导因子1α、GSK-3β等下游细胞因子促进血管修复,对治疗股骨头缺血性坏死具有重大意义[37]。上述各项实验研究均表明淫羊藿苷通过PI3K/AKT信号通路对成骨细胞及血运功能机制的影响,提示淫羊藿苷相关药物的研发将对SANFH的临床治疗具有重大帮助。 2.2.3 淫羊藿苷通过mTOR信号通路对多条信号调控进而影响骨重建 磷脂酰肌醇-4,5-二磷酸3-激酶(PI3K)-蛋白激酶B(PKB/Akt)-哺乳动物雷帕霉素靶点(mTOR)通路首次在肿瘤细胞中被发现,并被发现与多种功能的此类细胞有关[38]。mTOR是Akt的下游效应因子,是一种丝氨酸/苏氨酸蛋白激酶,由两种不同的复合物(mTORC1和mTORC2)组成。mTOR的激活通过促进mRNA转位导致糖、脂肪和蛋白质的代谢。PI3K-Akt-mTOR通路在多种细胞功能调控中发挥重要作用,包括细胞存活、增殖、分化和血管生成[39-42]。而且 JIANG 等[43]研究发现,淫羊藿苷还可以通过激活mTOR信号通路抑制卵巢癌耐药细胞自噬,促进细胞凋亡。 目前已有研究发现,PI3K-Akt-mTOR通路参与了骨形成过程。LUO等[44]研究淫羊藿次苷Ⅱ(ICSII)对犬骨髓间充质干细胞的成骨作用实验发现,淫羊藿次苷Ⅱ可以通过调节PI3K/AKT/mTOR/S6K1信号通路促进犬骨髓间充质干细胞的成骨分化。LI等[45]研究发现牙釉质基质衍生物对在钛表面生长的牙周膜干细胞增殖和成骨分化的促进作用可能与Akt/ mTOR信号通路的激活有关。TONG等[46]研究发现,机械生长因子主要是通过磷酸化AKT从而促进骨髓间充质干细胞的生长和成骨分化。YAO等[47]的研究也表明,淫羊藿苷可以通过调节PI3K/AKT/mTOR和MAPK通路,从而减轻铁超载对骨髓间充质干细胞的有害影响。综上所述,淫羊藿苷可以通过调节PI3K/AKT/mTOR信号通路,促进骨髓间充质干细胞的生长和成骨分化,抑制细胞自噬,促进细胞凋亡,加快新骨重建,提高骨密度。因此,具有调控PI3K/AKT/mTOR信号通路作用的药物将成为骨质疏松症、SANFH等疾病临床治疗新的研究方向,而淫羊藿苷将是此研究方向的热点和重点药物之一。 2.2.4 淫羊藿苷作用雌激素受体介导激素信号表达影响激素性股骨头坏死 雌激素受体位于细胞膜、细胞质或细胞核,是一种蛋白质分子,可与激素特异性结合发挥生物学效应。其中位于细胞内的雌激素受体已经被证实存在于破骨细胞、破骨细胞前体细胞和成骨细胞中,且分为雌激素受体α和雌激素受体β两类。在经典通路“核”途径中,雌激素结合雌激素受体α或雌激素受体β受体,从而受体被激活,雌激素受体β移位至细胞核后启动下游靶基因进行转录,且调控着成骨细胞的分化[48],见图5。"
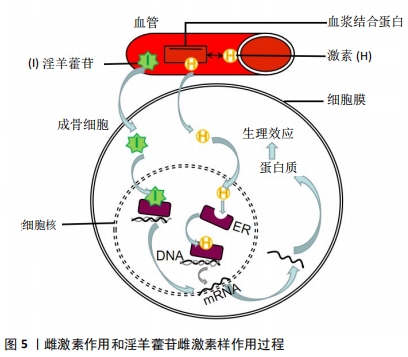

现有研究表明,淫羊藿苷表现出雌激素样作用,能够激活成骨细胞中雌激素受体的磷酸化,显著促进骨髓间质干细胞的成骨性分化,抑制破骨细胞的骨吸收与成骨细胞的成脂转化,增加骨密度,降低破骨细胞数[49-51],但其具体机制尚未明确。ZHANG 等[52]在淫羊藿苷促进成骨细胞和破骨细胞的分化和抑制机制的研究中,发现淫羊藿苷在 0.1-10 μmol/L促进成骨细胞分化的同时也可以上调表达雌激素受体,通过细胞化学分析、实时定量PCR、Western blotting和cDNA微阵列分析等方法评估成骨细胞分化过程中表达的细胞,得出淫羊藿苷在原代成骨细胞中的基因表达谱,提示淫羊藿苷可能通过雌激素受体发挥作用,通过雌激素受体介导的途径增加成骨细胞向成熟成骨细胞分化的数量。通过以上的研究和实验,不难发现淫羊藿苷可以通过雌激素介导的信号通路来调节成骨细胞的分化,进而促进骨形成。而且临床上显示淫羊藿苷对绝经后晚期妇女具有预防骨质流失的有益作用。因此作者提出一个大胆的想法,是否能用淫羊藿苷代替激素来治疗骨质疏松,减少激素的应用,从而防止因激素过量引起的SANFH。然而对于淫羊藿苷能否替代激素及其二者之间的联系机制尚未见有相关明确的报道,相关进展还待研究。 淫羊藿苷作为淫羊藿的主要有效单体成分,对于调控上述相关信号通路有效治疗SANFH已通过实验和临床研究得到充分有力的证实,但对于淫羊藿苷作用于信号通路影响成骨的作用及不同的淫羊藿苷药物浓度梯度对实验结果是否造成偏倚目前仍存有争议。比如邓宇等[53]研究表明通过淫羊藿苷激活Notch信号通路能促进成骨细胞的分化,提高了成骨分化相关因子的表达,但目前也有多项研究证实淫羊藿苷通过抑制Notch信号通路促进成骨细胞的分化作用[54-55],两种研究结论相互矛盾。在李智奎等[56]研究淫羊藿苷调控Wnt/β-catenin信号通路影响成脂成骨双向分化的实验中,发现10 μg/L淫羊藿苷能促进成骨细胞分化,显著提高成骨基因表达,而在40 μg/L淫羊藿苷作用下其结果则与之相反,且在10 μg/L和40 μg/L淫羊藿苷浓度时均显示Wnt/β-catenin信号通路呈显著上调状态,淫羊藿苷是通过调控Wnt/β-catenin信号通路发挥成骨作用,还是另通过其他形式促进成骨,后期仍需进行大量的实验和深入的研究。淫羊藿苷作用于成骨信号通路促进成骨是普遍认可的,将来的信号通路的实验研究也需进行更多药物浓度梯度的实验操作来防止实验数据的偏差和遗漏,保证实验结论准确,使临床上药物防治SANFH更加安全有效。"

| [1] 汪亮,刘耀升,刘蜀彬.糖皮质激素诱导股骨头坏死发病机制的研究进展[J].中华损伤与修复杂志(电子版),2015,10(5):439-445. [2] 陈镇秋,何伟,魏秋实,等.激素性股骨头坏死患者骨组织中骨代谢相关因子的表达[J].中华关节外科杂志(电子版),2015,9(2):183-188. [3] FANG SH, LI YF, JIANG JR, et al. Relationship of α2-Macroglobulin with Steroid-Induced Femoral Head Necrosis: A Chinese Population-Based Association Study in Southeast China. Orthop Surg. 2019;11(3):481-486. [4] WANG A, REN M, WANG J. The pathogenesis of steroid-induced osteonecrosis of the femoral head: A systematic review of the literature. Gene. 2018;671:103-109. [5] LI H, LIU D, LI C, et al. Exosomes secreted from mutant-HIF-1α-modified bone-marrow-derived mesenchymal stem cells attenuate early steroid-induced avascular necrosis of femoral head in rabbit. Cell Biol Int. 2017; 41(12):1379-1390. [6] 章晓云,李华南,陈跃平.淫羊藿苷防治股骨头坏死分子机制研究进展[J].中国骨质疏松杂志,2020,26(11):1694-1699. [7] XIE X, PEI F, WANG H,et al. Icariin: A promising osteoinductive compound for repairing bone defect and osteonecrosis. J Biomater Appl. 2015;30(3):290-299. [8] 万亚宁,李双蕾,蒋云霞,等.淫羊藿及其复方制剂治疗糖皮质激素性骨质疏松症的研究进展[J].中国骨质疏松杂志,2019,25(5): 713-716. [9] LIU RH, KANG X, XU LP, et al. Effects of the Combined Extracts of Herba Epimedii and Fructus Ligustri Lucidi On Bone Mineral Content and Bone Turnover in Osteoporotic Rats. BMC Complement Altern Med. 2015;15:112. [10] ZHANG D, FONG C, JIA Z, et al. Icariin Stimulates Differentiation and Suppresses Adipocytic Transdifferentiation of Primary Osteoblasts through Estrogen Receptor-Mediated Pathway. Calcif Tissue Int. 2016; 99(2):187-198. [11] MA HP, MA XN, GE BF, et al. Icariin Attenuates Hypoxia-Induced Oxidative Stress and Apoptosis in Osteoblasts and Preserves their Osteogenic Differentiation Potential in Vitro. Cell Prolif. 2014;47(6):527-539. [12] 钱卫庆. 淫羊藿苷促成骨细胞增殖中非核效应及机制研究[D]. 苏州:苏州大学, 2016. [13] WANG P, ZHANG F, HE Q, et al.Flavonoid Compound Icariin Activates Hypoxia Inducible Factor-1α in Chondrocytes and Promotes Articular Cartilage Repair. PLoS One. 2016;11(2):e0148372. [14] WANG Z, WANG D, YANG D, et al. The effect of icariin on bone metabolism and its potential clinical application. Osteoporos Int. 2018;29(3):535-544. [15] 秦爽,刘红,刘称称,等.淫羊藿苷影响骨代谢的研究[J].现代口腔医学杂志,2018,32(5):303-306. [16] YANG Y, NIAN H, TANG X, et al. Effects of the Combined Herba Epimedii and Fructus Ligustri Lucidi On Bone Turnover and TGF-beta1/Smads Pathway in GIOP Rats. J Ethnopharmacol. 2017;201:91-99. [17] 陈喆.淫羊藿苷对RAW264.7细胞破骨分化中MAPK信号通路的影响探究[D].北京:首都医科大学,2017. [18] 杨卫,赵品,陶美玲,等.淫羊藿苷口服给药对骨质疏松牙周炎小鼠牙槽骨吸收的影响[J].上海口腔医学,2019,28(1):30-35. [19] KAHN M.Can we safely target the WNT pathway.Nat Rev Drug Discov. 2014; 13(7):513-532. [20] FU S, YANG L, HONG H, et al. Wnt/β-catenin signaling is involved in the Icariin induced proliferation of bone marrow mesenchymal stem cells. J Tradit Chin Med. 2016;36(3):360-368. [21] WANG Z, WANG D, YANG D, et al. The effect of icariin on bone metabolism and its potential clinical application. Osteoporos Int. 2018;29(3):535-544. [22] YANG A, YU C, LU Q, et al. Mechanism of Action of Icariin in Bone Marrow Mesenchymal Stem Cells. Stem Cells Int. 2019;2019:5747298. [23] WEI Q, ZHANG J, HONG G, et al. Icariin promotes osteogenic differentiation of rat bone marrow stromal cells by activating the ERα- Wnt/β- catenin signaling pathway. Biomed Pharmacother. 2016;84: 931-939. [24] LIU Y, HUANG L, HAO B, et al. Use of an Osteoblast Overload Damage Model to Probe the Effect of Icariin on the Proliferation, Differentiation and Mineralization of MC3T3-E1 Cells through the Wnt/β-Catenin Signalling Pathway. Cell Physiol Biochem. 2017;41(4):1605-1615. [25] WANG J, TAO Y, PING Z, et al. Icariin attenuates titanium-particle inhibition of bone formation by activating the Wnt/β-catenin signaling pathway in vivo and in vitro. Sci Rep. 2016;6:23827. [26] ZHOU H, JIAO G, DONG M, et al. Orthosilicic Acid Accelerates Bone Formation in Human Osteoblast-Like Cells Through the PI3K-Akt-mTOR Pathway. Biol Trace Elem Res. 2019;190(2):327-335. [27] DIBBLE C, CANTLEY L. Regulation of mTORC1 by PI3K signaling. Trends Cell Biol. 2015;25(9):545-555. [28] FRUMAN D, ROMMEL C. PI3K and cancer: lessons, challenges and opportunities. Nat Rev Drug Discov. 2014;13(2): 140-156. [29] THORPE L, YUZUGULLU H, ZHAO J. PI3K in cancer: divergent roles of isoforms, modes of activation and therapeutic targeting. Nat Rev Cancer. 2015;15(1):7-24. [30] ZOU H, LI L, GARCIA CARCEDO I, et al. Synergistic inhibition of colon cancer cell growth with nanoemulsion-loaded paclitaxel and PI3K/mTOR dual inhibitor BEZ235 through apoptosis. Int J Nanomedicine. 2016;11:1947-1958. [31] MANNING BD, TOKER A. AKT/PKB Signaling: Navigating the Network.Cell. 2017;169(3):381-405. [32] XUE XH, FENG ZH, LI ZX, et al. Salidroside inhibits steroid-induced avascular necrosis of the femoral head via the PI3K/Akt signaling pathway: In vitro and in vivo studies. Mol Med Rep. 2018;17(3):3751-3757. [33] LU JM, ZHANG ZZ, MA X, et al. Repression of microRNA-21 inhibits retinal vascular endothelial cell growth and angiogenesis via PTEN dependent-PI3K/Akt/VEGF signaling pathway in diabetic retinopathy. Exp Eye Res. 2020;190:107886. [34] AHMAD KA, ZE H, CHEN J, et al. The protective effects of a novel synthetic β-elemene derivative on human umbilical vein endothelial cells against oxidative stress-induced injury: Involvement of antioxidation and PI3k/Akt/eNOS/NO signaling pathways. Biomed Pharmacother. 2018;106:1734-1741. [35] 王雅婧,张学志,刁力,等.PI3K/AKt/eNOS信号通路在硫化氢抑制ET-1诱导的心肌肥大中的作用[J].中国循证心血管医学杂志, 2014,6(5):551-554+557. [36] HU J, MAO Z, HE S, et al. Icariin protects against glucocorticoid induced osteoporosis, increases the expression of the bone enhancer DEC1 and modulates the PI3K/Akt/GSK3β/β-catenin integrated signaling pathway. Biochem Pharmacol. 2017;136:109-121. [37] 宋世雷,陈跃平,章晓云.PI3K/AKT信号通路调控股骨头坏死的相关机制[J].中国组织工程研究,2020,24(3):408-415. [38] ZHOU H, JIAO G, DONG M, et al. Orthosilicic Acid Accelerates Bone Formation in Human Osteoblast-Like Cells Through the PI3K-Akt-mTOR Pathway. Biol Trace Elem Res. 2019;190(2):327-335. [39] BERTACCHINI J, HEIDARI N, MEDIANI L, et al. Targeting PI3K/AKT/mTOR network for treatment of leukemia. Cell Mol Life Sci. 2015;72(12): 2337-2347. [40] FOLLO MY, MANZOLI L, POLI A, et al. PLC and PI3K/Akt/mTOR signalling in disease and cancer. Adv Biol Regull. 2015;57:10-16. [41] MANFREDI GI, DICITORE A, GAUDENZI G, et al. PI3K/Akt/mTOR signaling in medullary thyroid cancer: a promising molecular target for cancer therapy. Endocrine. 2015;48(2):363-370. [42] GUO X, WEI S, LU M, et al. Dose-dependent effects of strontium ranelate on ovariectomy rat bone marrow mesenchymal stem cells and human umbilical vein endothelial cells. Int J Biol Sci. 2016;12(12):1511-1522. [43] JIANG S, CHANG H, DENG S, et al. Icariin inhibits autophagy and promotes apoptosis in SKVCR cells through mTOR signal pathway. Cell Mol Biol (Noisy-le-grand). 2018;64(6):4-10. [44] LUO G, XU B, HUANG Y. Icariside II promotes the osteogenic differentiation of canine bone marrow mesenchymal stem cells via the PI3K/AKT/mTOR/S6K1 signaling pathways. Am J Transl Res. 2017;9(5):2077-2087. [45] LI G, HU J, CHEN H, et al. Enamel matrix derivative enhances the proliferation and osteogenic differentiation of human periodontal ligament stem cells on the titanium implant surface. Organogenesis. 2017;13(3):103-113. [46] TONG Y, FENG W, WU Y, et al. Mechano-growth factor accelerates the proliferation and osteogenic differentiation of rabbit mesenchymal stem cells through the PI3K/AKT pathway. BMC Biochem. 2015;16:1. [47] YAO X, JING X, GUO J, et al. Icariin Protects Bone Marrow Mesenchymal Stem Cells Against Iron Overload Induced Dysfunction Through Mitochondrial Fusion and Fission,PI3K/AKT/mTOR and MAPK Pathways. Front Pharmacol. 2019;10:163. [48] CIPOLLETTI M, MONTALESI E, NUZZO MT, et al. Potentiation of paclitaxel effect by resveratrol in human breast cancer cells by counteracting the 17β-estradiol/estrogen receptor α/neuroglobin pathway. J Cell Physiol. 2019;234(4):3147-3157. [49] XU YX, XU B, WU CL, et al. Dynamic expression of DKK1 protein in the process whereby Epimedium-derived flflavonoids up-regulate osteogenic and down-regulate adipogenic differentiation of bone marrow stromal cells in ovariecto mized rats. Orthop Surg. 2011;3:119-126. [50] ZHOU L, POON CC, WONG KY, et al. Prenylflavonoid Icariin Induces Estrogen Response Element-Independent Estrogenic Responses in a Tissue-Selective Manner. J Endocr Soc. 2019;4(2):bvz025. [51] ZHAO R, BU W, CHEN Y.Icariin Treatment Enhanced the Skeletal Response to Exercise in Estrogen-Deficient Rats. Int J Environ Res Public Health. 2019; 16(19):3779. [52] ZHANG D, FONG C, JIA Z, et al. Icariin Stimulates Differentiation and Suppresses Adipocytic Transdifferentiation of Primary Osteoblasts Through Estrogen Receptor-Mediated Pathway. Calcif Tissue Int. 2016;99(2):187-198. [53] 邓宇,陈廖斌.淫羊藿苷通过激活Notch信号通路促进骨髓间充质干细胞向成骨细胞分化的实验研究[J].中医学报,2017,32(12): 2393-2398,2403. [54] YANG M, LIU H, WANG Y, et al. Hypoxia reduces the osteogenic differentiation of peripheral blood mesenchymal stem cells by upregulating Notch-1 expression. Connect Tissue Res. 2019;60(6):583-596. [55] XU Y, LI L, TANG Y, et al. Icariin promotes osteogenic differentiation by suppressing Notch signaling. Eur J Pharmacol. 2019;865:172794. [56] 李智奎,孔俊博,赵王林.淫羊藿苷调控Wnt/β-catenin信号通路干预大鼠MSCs成脂成骨双向分化实验研究[J].中国免疫学杂志, 2019,35(24):2985-2990. |
| [1] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [2] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [3] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [4] | Li Huo, Wang Peng, Gao Jianming, Jiang Haoran, Lu Xiaobo, Peng Jiang. Relationship between revascularization and internal microstructure changes in osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1323-1328. |
| [5] | Wang Baojuan, Zheng Shuguang, Zhang Qi, Li Tianyang. Miao medicine fumigation can delay extracellular matrix destruction in a rabbit model of knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1180-1186. |
| [6] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [7] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [8] | Gao Yujin, Peng Shuanglin, Ma Zhichao, Lu Shi, Cao Huayue, Wang Lang, Xiao Jingang. Osteogenic ability of adipose stem cells in diabetic osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 999-1004. |
| [9] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [10] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [11] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| [12] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [13] | Zhou Hongqin, Wu Dandan, Yang Kun, Liu Qi. Exosomes that deliver specific miRNAs can regulate osteogenesis and promote angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1107-1112. |
| [14] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [15] | Huang Chenwei, Fei Yankang, Zhu Mengmei, Li Penghao, Yu Bing. Important role of glutathione in stemness and regulation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1119-1124. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||