Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (13): 2060-2069.doi: 10.3969/j.issn.2095-4344.3505
Previous Articles Next Articles
Effect and mechanism of traditional Chinese medicine for replenishing qi and promoting blood combined with bone marrow mesenchymal stem cells in promoting angiogenesis in ischemic stroke
Fan Feiyan, Zhang Yunke
- Henan University of Chinese Medicine, Zhengzhou 450046, Henan Province, China
-
Received:2020-03-28Revised:2020-04-09Accepted:2020-05-30Online:2021-05-08Published:2020-12-28 -
Contact:Zhang Yunke, MD, Professor, Chief physician, Doctoral supervisor, Post-doctoral cooperative supervisor, Henan University of Chinese Medicine, Zhengzhou 450046, Henan Province, China -
About author:Fan Feiyan, Master candidate, Henan University of Chinese Medicine, Zhengzhou 450046, Henan Province, China -
Supported by:the National Natural Science Foundation of China, No. 81974564 (to ZYK); the Science and Technology Innovation Outstanding Talent Project of Henan Province, No. 2018JR0006 (to ZYK)
CLC Number:
Cite this article
Fan Feiyan, Zhang Yunke. Effect and mechanism of traditional Chinese medicine for replenishing qi and promoting blood combined with bone marrow mesenchymal stem cells in promoting angiogenesis in ischemic stroke[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2060-2069.
share this article
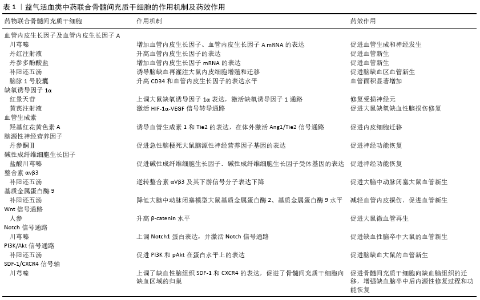
2.1 血管新生的概念和影响因素 血管新生是指在原有血管基础上通过血管内皮细胞增殖、芽生、游走、血管分裂和分支而形成新的毛细血管网,使其功能与局部需要相适应的生物学过程[21],该过程涉及诸多生长因子和信号通路分子[22], 如血管内皮生长因子、脑源性神经营养因子、碱性成纤维细胞生长因子、缺氧诱导因子1α、血管生成素、整合素αvβ3、基质金属蛋白酶9及Wnt信号通路、Notch信号通路、PI3K/Akt信号通路和SDF-1/CXCR4信号轴等。 在脑梗死中,梗死周边血管密度增加主要是通过血管新生的方式[23],脑梗死后血管新生及成熟过程存在着复杂的病理生理变化[24]。中药在促进血管新生方面存在一定疗效,根据祛瘀促生的治疗原则,活血化瘀药和益气补血药可能是候选药物[20],研究发现骨髓间充质干细胞与益气活血类中药联合治疗在促进血管新生方面优于单独治疗[25]。 2.2 益气活血类中药联合骨髓间充质干细胞对缺血性脑卒中血管新生的影响 见表1。 2.2.1 益气活血类中药联合骨髓间充质干细胞对血管内皮生长因子及血管内皮生长因子A的影响 血管内皮生长因子是一种高度特异性的促血管内皮细胞生长因子[26-27],是目前最常见的血管生成因子,其主要功能是诱导内皮细胞增殖、分化和迁移,是血管新生的标志性指标[28]。血管内皮生长因子具有酪氨酸激酶活性,能够与血管内皮细胞上的酪氨酸受体结合,产生一系列信号传递效应,促进血管内皮细胞增殖、迁移,参与血管的新生[29]。血管新生效应主要与2个酪氨酸激酶结合有关,即VEGFR-1(fms-like tyrosine kinase,Flt-1) 和VEGFR-2(kinase insert domain containing reptor/fetal liver kinase-1,KDR/flk-1)[30]。血管内皮生长因子A是血管内皮生长因子家族中血管新生的主要参与者,并激活VEGFR1(Flt-1)和VEGFR2(KDR/flk-1),VEGFR1对血管内皮生长因子A具有非常紧密的配体结合域,而VEGFR2具有较强的酪氨酸激酶活性,两者通过独特的分子机制在血管生成中发挥着不同的作用[31]。血管内皮生长因子是血管新生的关键调控因子,在中风模型大鼠神经元、星形胶质细胞、小胶质细胞和血管中的表达均明显上调[32]。 川芎具有活血行气的作用,为血中之气药,川芎嗪是中药川芎的一种主要有效成分,川芎嗪在中国已广泛应用于脑血管疾病的临床治疗[33]。川芎嗪可促进大鼠血管新生相关蛋白血管内皮生长因子的表达增加[34]。JIN等[35]研究发现动脉灌注骨髓间充质干细胞可促进血管内皮生长因子 mRNA的表达及微血管密度增加。LI等[36]研究发现骨髓间充质干细胞与川芎嗪联合组脑缺血大鼠微血管密度较单纯骨髓间充质干细胞组或川芎嗪治疗组明显增加,且联合组血管内皮生长因子的mRNA和蛋白表达水平较骨髓间充质干细胞组和川芎嗪组显著升高,提示骨髓间充质干细胞联合川芎嗪可增加缺血区血管内皮生长因子的分泌。 丹红注射液是从具有活血功效的中药丹参、红花中提取精制而成的,具有促进血管新生、缩小梗死面积、改善梗死边缘区微环境等作用[37]。研究发现,丹红注射液入血后的5个主要活性成分(羟基红花黄色素A、丹酚酸B、丹参素、原儿茶醛和迷迭香酸)均可使血管内皮生长因子A的表达显著升高[38]。丹红注射液联合骨髓间充质干细胞移植治疗脑梗死大鼠疗效显著,可以对脑细胞起到保护作用[39]。CHEN等[40]研究发现间充质干细胞与丹红注射液联合应用与二者单独应用相比,梗死边缘区血管内皮生长因子表达明显升高。 丹参具有活血作用,丹参及其提取物目前已广泛用于缺血性脑血管病的治疗[41]。丹参粗提物和丹酚酸B通过上调血管内皮生长因子和VEGFR2基因表达促进血管新生[42]。丹参多酚酸盐为丹参的水提物,王晓娟[43]探讨丹参多酚酸盐对大鼠骨髓间充质干细胞增殖及血管内皮生长因子表达的影响,实验组为丹参多酚酸盐与大鼠骨髓间充质干细胞共培养,对照组为无药物单独培养的大鼠骨髓间充质干细胞,共培养48 h后实验组骨髓间充质干细胞生长密集程度明显优于对照组;实验组血管内皮生长因子mRNA表达高于对照组,提示丹参多酚酸盐能促进大鼠骨髓间充质干细胞增殖,且二者联合能提高血管内皮生长因子的表达。 补阳还五汤由当归、川芎、黄芪、赤芍、地龙、红花、桃仁组成,具有补气活血通络之功效,作为治疗中风的经典中药方剂,具有显著的神经保护、促血管新生和功能改善作用[44]。课题组之前的研究表明,补阳还五汤可增加脑缺血再灌注大鼠缺血侧大脑皮质区血管内皮生长因子的表达量和微血管密度,促进血管新生[45]。补阳还五汤联合骨髓间充质干细胞移植主要经VEGFR2所介导的信号通路及PI3K/AKT信号途径,诱导脑缺血再灌注大鼠内皮细胞增殖和迁移,促进脑缺血区血管新生[46]。 脑脉1号胶囊由黄芪、川芎、益母草、炙天麻、水蛭、制南星、半夏组成,是以调节脑血运为目的的中药制剂,是治疗缺血性脑卒中的有效药物[47]。陈朝等[48]发现脑脉1号胶囊可以提高骨髓间充质干细胞移植后的大脑中动脉闭塞大鼠的微血管再生作用,增加移植细胞区域的血供,促进骨髓间充质干细胞移植后的存活。GUO等[49]研究表明脑脉1号胶囊联合骨髓间充质干细胞移植组CD34和血管内皮生长因子的表达水平升高,联合组的血管面积显著增加;脑脉1号胶囊促进骨髓间充质干细胞移植大脑中动脉闭塞大鼠血管新生,行为评分显著改善,提示血管新生机制可能与血管内皮生长因子表达上调有关。 2.2.2 益气活血类中药联合骨髓间充质干细胞对缺氧诱导因子1α的影响 组织缺血缺氧时,机体可通过多种途径产生多种细胞因子作用于缺血组织,促进新生血管的形成,最终达到改善缺血组织供血的目的。缺氧诱导因子1是缺氧和局部缺血反应中基因表达的关键调节剂[50],缺氧诱导因子1包含2个基本蛋白,即缺氧诱导因子1α和缺氧诱导因子1β[51]。缺氧诱导因子1α在低氧应答反应中起关键性作用,在缺氧条件下缺氧诱导因子1α与缺氧反应元件结合,能够显著提高细胞的生存活力,对血管新生和血管新生相关因子如成纤维细胞生长因子、血管内皮生长因子、表皮生长因子、血管生成素等表达起着重要的调控作用[52]。缺氧诱导因子1α通过调节一系列基因,参与了血管新生、葡萄糖代谢、铁代谢及细胞增殖与存活等。脑缺血缺氧时,缺氧诱导因子1α表达增多可以增加血液、氧及营养的供应[53]。NORITADA等[54]研究发现抑制缺氧条件下缺氧诱导因子1α和血管内皮生长因子的表达,可达到抑制血管新生的作用。 红景天具有益气活血之功效,可用于治疗气虚血瘀之中风偏瘫。红景天苷是从红景天中提取的活性成分,具有体内外抗缺氧作用,可增加缺氧诱导因子1α的转录活性,从而上调血管内皮生长因子的表达,可通过HIF-1α-VEGF信号途径部分改善缺氧[55]。韩国雄等[56]通过研究低氧对骨髓间充质干细胞中缺氧诱导因子1基因表达水平及其分泌的血管生长因子的影响,发现低氧条件下缺氧诱导因子1α和缺氧诱导因子1β基因表达水平均增高,骨髓间充质干细胞分泌的血管内皮生长因子水平高于常氧培养组。HA等[57]研究发现红景天苷可延长移植骨髓间充质干细胞的存活时间,红景天苷联合骨髓间充质干细胞可以暂时性上调大鼠缺氧诱导因子1α表达,激活缺氧诱导因子1通路,修复受损神经元。 黄芪注射液中的主要有效成分为黄芪,具有益气扶正的作用,黄芪注射液在低氧环境、低质量浓度下可促进体外培养的人骨髓间充质干细胞增殖[58]。黄芪注射液可能通过激活HIF-1α-VEGF信号转导通路促进脑缺血再灌注损伤大鼠脑内血管新生[59]。余勤等[60]实验发现大鼠骨髓间充质干细胞联合黄芪注射液组大鼠的学习、记忆能力改善更显著,脑组织内Brdu阳性细胞数更多,黄芪注射液在体内能够促进大鼠骨髓间充质干细胞向损伤脑组织迁移,协同骨髓间充质干细胞促进大鼠缺氧缺血性脑损伤修复。 2.2.3 益气活血类中药联合骨髓间充质干细胞对血管生成素的影响 血管生成素是一类糖蛋白,可作用于脉管系统,控制血管生长发育和保证新生成血管的稳定性[61]。血管生成素主要由血管生成素1、血管生成素2、血管生成素3、血管生成素4及其两种受体Tie1和Tie2组成[62]。血管生成素1、血管生成素2与血管新生密切相关[63]。血管生成素1/2及其受体Tie2代表主要的酪氨酸激酶-配体系统,对调节血管新生至关重要[64]。血管生成素1与其在内皮细胞上表达的酪氨酸激酶受体Tie2结合,在内皮细胞的存活、迁移、发芽和小管形成中起重要作用[65],可调节涉及新血管形成的促血管生成途径[66]。血管生成素1促进内皮细胞存活和血管成熟,血管生成素2由内皮细胞表达[67],血管生成素2主要是让血管稳定性降低,另一方面还可以活化内皮细胞[68],血管生成素2作为血管生成素1的拮抗剂,可以结合Tie2受体,通过这一作用使血管通透性增加[69],抑制血管稳定和成熟,松解内皮细胞和周细胞间的紧密接触,从而使血管更加可塑[70]。 红花具有养血活血的作用,广泛应用于脑血管疾病的治疗。羟基红花黄色素A是红花中主要的生物活性和水溶性化合物之一,具有多种生物活性。羟基红花黄色素A能促进人骨髓间充质干细胞的增殖[71],而且可诱导血管生成素1和Tie2的表达,在体外激活Ang1/Tie2信号通路,羟基红花黄色素A通过Ang1/Tie2信号通路在体外促进内皮细胞迁移和管形成[72]。 三七总皂甙(total saponins of Panax notoginseng,tPNS)是从具有活血补血作用的三七中提取的主要成分,其主要成分包括人参皂苷Rg1、人参皂苷Rb1和三七皂苷R1[73]。HUI等[74]发现三七总皂甙可通过激活Shh信号通路,上调血管生成素1的表达,增加大脑中动脉闭塞大鼠缺血边缘区毛细血管密度,促进血管新生。MOISAN等[75]在一项单独的体内纵向MRI研究中,观察到注射人骨髓间充质干细胞后2周和3周缺血区血管密度增强,组织学和定量聚合酶链反应显示人骨髓间充质干细胞处理的大脑中动脉闭塞大鼠血管生成素1表达增加。三七皂苷联合骨髓间充质干细胞移植能够促进大鼠急性脑损伤后的神经功能恢复[76],BAO等[77]发现人参皂苷Rg1联合骨髓间充质干细胞移植可减少脑缺血再灌注损伤大鼠细胞凋亡,改善脑功能。 2.2.4 益气活血类中药联合骨髓间充质干细胞对脑源性神经营养因子的影响 脑源性神经营养因子是神经营养因子家族成员之一,通过其特异性受体TrkB促进血管新生[78]。脑源性神经营养因子主要由血管内皮细胞分泌,是脑内广泛分布于中枢神经系统的重要蛋白[79]。脑源性神经营养因子通过发挥神经保护、神经生成和血管新生的作用,促进中枢神经系统损伤的修复[80]。脑源性神经营养因子不仅具有神经营养因子活性,并且作为血管新生调节因子,参与促进内皮细胞生存,诱导缺血组织血管新生[81]。PIKULA等[82]发现血清脑源性神经营养因子水平降低与人类中风风险增加相关。实验证明,脑源性神经营养因子参与脑卒中后血管新生、突触形成及功能恢复[83]。为了确定脑源性神经营养因子是否具有促进体内血管新生的作用,SUN等[84]进行了小鼠基质凝胶塞实验,结果表明脑源性神经营养因子组移行到基质凝胶中的内皮细胞数、血管密度和血管结构面积均显著高于对照组;含脑源性神经营养因子明胶海绵周围可见放射状新生血管,对照组未见新生血管,结果提示脑源性神经营养因子显著促进小鼠体内血管新生。 丹参具有活血的作用,丹参乙酸镁、丹参酮Ⅱ均是从丹参中提取的水溶性有效成分,生物利用率较高。丹参乙酸镁可增加大鼠脑缺血损伤后缺血半暗带区脑源性神经营养因子的表达[85],卢乙众等[86]研究发现丹参酮Ⅱ联合骨髓间充质干细胞移植可促进急性脑梗死大鼠脑源性神经营养因子基因的表达,并且脑源性神经营养因子基因表达:联合组>骨髓间充质干细胞移植组>模型组。 2.2.5 益气活血类中药联合骨髓间充质干细胞对碱性成纤维细胞生长因子的影响 碱性成纤维细胞生长因子是成纤维细胞生长因子蛋白家族中的一员,是中胚层来源的对细胞有强烈促增殖和分化作用的细胞生长调节因子,具有调节血管新生、神经网络发育和轴突生长的功能[87]。碱性成纤维细胞生长因子是一种积极的血管新生调节因子,通过募集炎性细胞,如单核细胞、巨噬细胞和中性粒细胞,从而上调多种趋化因子的表达[88]。碱性成纤维细胞生长因子能通过促进内皮细胞的增殖诱导血管新生,并作为一种化学引诱剂,促进成纤维细胞和上皮细胞的增殖[89]。碱性成纤维细胞生长因子参与脑梗死后的病理生理变化,脑梗死患者血清碱性成纤维细胞生长因子水平明显升高,其在脑内上调有助于脑梗死后的血管新生[90]。 中药川芎具有活血行气的作用,盐酸川芎嗪是川芎中的主要活性成分,目前广泛应用于缺血性脑血管病的治疗[91]。川芎嗪通过促进骨髓组织中碱性成纤维细胞生长因子、碱性成纤维细胞生长因子受体的表达,加速骨髓造血微环境的修 复[92]。ZHANG等[93]研究发现表达人碱性成纤维细胞生长因子的骨髓间充质干细胞通过增加移植细胞的存活率促进大鼠血管新生,进而分化为血管内皮细胞。吴晓明等[94]研究发现盐酸川芎嗪联合骨髓间充质干细胞移植可使大鼠碱性成纤维细胞生长因子基因的表达显著升高,从而促进神经细胞再生,改善肢体运动功能。 2.2.6 益气活血类中药联合骨髓间充质干细胞对整合素αvβ3的影响 整合素是介导细胞-细胞或细胞-基质相互作用的细胞表面异二聚体受体[95]。目前已知有24种人整合素亚型和8种整合素二聚体:αvβ1、αvβ3、αvβ5、αvβ6、αvβ8、α5β1、α8β1和αIIbβ3,在8种整合素二聚体中,整合素αvβ3最早被鉴定,并被证实参与了细胞收缩性、细胞/细胞外基质刚性和细胞运动的调节[96]。整合素αvβ3参与内皮细胞的活化和迁移与血管内皮生长因子诱导的血管新生过 程[95]。研究表明,阻断整合素αvβ3黏附功能的抗体、肽或拟肽类药物能够成功抑制多种动物模型中的血管新生[97]。脑缺血动物模型缺血区血管内皮细胞可见整合素αvβ3的表达,且整合素αvβ3 mRNA表达水平明显增高。缺血区血管内皮细胞上整合素αvβ3的表达与血管内皮生长因子的表达密切相关[98]。 补阳还五汤是一种传统的中草药方剂,在临床上广泛用于治疗中风已有数百年的历史,通过益气活血作用,能上调新生血管整合素αVβ3和β3亚基的表达,参与血管新生,影响微血管系统重建[99]。课题组研究发现骨髓间充质干细胞移植联合补阳还五汤可逆转抗CD34+CD45抗体引起的血管内皮细胞数量减少、整合素αVβ3及其下游信号分子表达下降,促进大脑中动脉闭塞大鼠血管新生[100]。 2.2.7 益气活血类中药联合骨髓间充质干细胞对基质金属蛋白酶9的影响 基质金属蛋白酶是一种内源性Ca2+、Zn2+依赖性蛋白酶超家族。根据功能不同将基质金属蛋白酶分为胶原酶、明胶酶、基质水解酶、膜型基质金属蛋白酶、其他基质金属蛋白酶[101]。基质金属蛋白酶9是基质金属蛋白酶的主要成员,其主要功能是降解和重塑细胞外基质[102]。基质金属蛋白酶9是Ⅳ型胶原酶,又称为明胶酶B,其作用底物包括明胶Ⅳ、Ⅴ型明胶原、弹力蛋白等,可以降解细胞外基质中的Ⅳ型胶原[103]。研究发现,基质金属蛋白酶9能通过降解内皮和间质中基质,并激活其生长因子,从而启动血管新生[104]。基质金属蛋白酶9在正常人的脑组织及血浆中表达较低,而在炎症、新生血管形成等多种生理和病理过程中,其表达量明显增加[105]。在生理条件下,基质金属蛋白酶可以参与组织重塑、血管新生,但是在病理条件下却可以参与血管的损伤[106]。基质金属蛋白酶9在脑梗死的发生发展中起着重要作用[107]。脑缺血灌注损伤后体内基质金属蛋白酶9表达明显增强,且随着脑缺血再灌注时间延长呈递增变 化[108],在脑梗死后抑制基质金属蛋白酶9的表达会刺激血管生成[109]。 补阳还五汤出自《医林改错》,是治疗气虚血瘀型中风的经典方剂。蓝艳[110]研究发现补阳还五汤能明显降低气虚血瘀型缺血性脑卒中患者基质金属蛋白酶3、基质金属蛋白酶9水平,改善患者的缺血缺氧和缺损神经功能。骨髓间充质干细胞移植可以降低心肌梗死家兔基质金属蛋白酶9 mRNA表达[111]。补阳还五汤联合骨髓间充质干细胞移植能够与基质金属蛋白酶抑制剂发挥协同作用,降低大脑中动脉闭塞模型大鼠基质金属蛋白酶2、基质金属蛋白酶9的含量,达到减轻血管内皮损伤、促进血管新生的作用[112]。 2.2.8 益气活血类中药联合骨髓间充质干细胞对Wnt信号通路的影响 Wnt信号通路是一类以分泌型Wnt蛋白为配体激活的信号通路,分为经典Wnt/β-catenin信号通路和非经典Wnt/β-catenin信号通路。脑缺血可启动血管新生,Wnt信号通路是调控细胞增殖分化的关键环节,以经典Wnt途径的研究最为透彻[113]。经典Wnt途径也称为 Wnt/β-catenin信号通路,主要激活细胞核中靶基因的表达。许多研究表明,间充质干细胞表达多种Wnt配体(Wnt2、Wnt 4、Wnt5a、Wnt11和Wnt16)和Wnt受体,Wnt信号转导的激活参与了骨髓间充质干细胞等干细胞的自我更新和增殖过程,Wnt信号转导在骨髓间充质干细胞增殖中起重要作用[114]。Wnt/β-catenin信号传导在诱导新血管形成中起重要作用[115],DU等[116]认为Wnt信号通路的激活可诱导相关炎症细胞因子的表达,具有促进血管新生的作用。LI等[117]认为通过抑制Wnt/β-Catenin信号通路诱导自噬,可抑制血管新生和诱导细胞凋亡,GUO等[118]通过增强Wnt/β-catenin诱导的FGF18表达可促进血管新生。 人参具有益气的功效,人参皂苷Rg1是从人参的根或茎中提取和纯化而来,是人参中最常见的活性成分,具有促进血管新生的作用[119]。LEUNG等[120]研究发现人参皂苷Rg1是血管内皮细胞的有效促血管生成因子,人参皂苷Rg1刺激导致β-catenin水平升高,最终达到其核蓄积,随后激活了血管内皮生长因子表达。MCBRIDE等[121]研究发现高Wnt环境中骨髓间充质干细胞衍生的CD63+外泌体是Wnt3a外显子的重要载体,可在体外诱导成纤维细胞的增殖、迁移和血管新生。人参皂苷Rg1联合骨髓间充质干细胞移植治疗大鼠脑缺血再灌注损伤,联合组治疗效果最佳,细胞凋亡减少,脑功能恢复较好[77]。王祺[122]研究发现由单味人参组成的独参汤联合骨髓间充质干细胞能改善大鼠血管内皮功能,促进微血管再生,改善梗死区及缺血区的血供。 2.2.9 益气活血类中药联合骨髓间充质干细胞对Notch信号通路的影响 Notch信号通路是一条进化过程中高度保守的信号转导系统,调控多细胞机体的细胞增殖、分化和凋亡[123]。Notch信号通路由Notch受体、Notch配体(DSL蛋白)及细胞内效应器分子(CSL-DNA结合蛋白)组成,Notch通路激活是血管新生和功能血管最终形成的重要调控因素[124]。目前,哺乳动物体内已发现4种类型的Notch受体(Notch 1-4)以及5种类型的配体(DLL-1,DLL-3,DLL-4和JAG-1,JAG-2)[125]。当配体与受体结合后,Notch分子活化,释放受体胞内段活性结构域,启动Notch下游靶基因Hes1等的转录而发挥调控作用,Notch通路激活是血管新生和功能血管最终形成的重要调控因素[124]。Notch信号通路可以决定血管的起源和调节血管的形成过程,影响血管新生的细胞增殖、分化、迁移、凋亡等多个方面[126]。 刘琦等[127]研究发现骨髓间充质干细胞移植能通过上调Notch1蛋白表达,并可通过激活Notch信号通路促进大脑中动脉闭塞大鼠缺血皮质区血管内皮生长因子165的表达,进而促进缺血区的血管新生。ZHAO等[128]研究证实通过抑制Notch信号通路可抑制肿瘤血管新生。SHI等[129]认为川芎和赤芍通过Notch信号和干细胞向梗死边缘区的动员而发挥促血管新生作用,而川芎的主要有效成分川芎嗪与骨髓间充质干细胞联合治疗能显著促进骨髓间充质干细胞向脑缺血区迁移,从而协同促进缺血性脑卒中大鼠的血管新生[42]。 2.2.10 益气活血类中药联合骨髓间充质干细胞对PI3K/Akt 信号通路的影响 PI3K/AKT信号通路是调节细胞存活、生长和增殖的重要信号通路[130],PI3K/Akt信号通路和血管新生紧密关联,它整合了血管新生全过程的各种信号[131]。PI3K/Akt信号通路通过作用于内皮细胞中的血管内皮生长因子和内皮型一氧化氮合酶来促进血管新生[132]。血管内皮生长因子与其受体VEGFR2的结合可诱导下游促细胞存活相关通路激活,进而促进细胞增殖,如PI3K/Akt信号通路[133]。PI3K/Akt信号通路的激活能调控血管内皮生长因子的表达,抑制PI3K/Akt信号通路激活能下调血管内皮生长因子表达,抑制缺氧后血管内皮细胞增殖及迁移[134],提示PI3K/Akt信号通路能通过诱导血管内皮生长因子表达促进血管新生。研究发现,抑制PI3K和AKT的磷酸化,阻断PI3K/AKT途径可抑制血管新生[135]。 补阳还五汤主治气虚血瘀之中风,临床上多用于脑血管意外后遗症及其他原因引起的偏瘫、截瘫等属于气虚血瘀 者[45]。补阳还五汤通过PI3K/Akt信号途径促进VEGFR2磷酸化,PI3K和pAkt在蛋白水平上的表达升高,进而促进神经功能恢复和血管新生[136]。补阳还五汤联合骨髓间充质干细胞移植主要经VEGFR2所介导的信号通路及PI3K/AKT信号途径,诱导内皮细胞增殖和迁移,促进脑缺血大鼠血管新生[46]。 2.2.11 益气活血类中药联合骨髓间充质干细胞对SDF-1/ CXCR4信号轴的影响 SDF-1属于CXC类趋化因子,其在造血干细胞/祖细胞的动员及归巢中起关键作用[137],其受体CXCR4是一种有7个跨膜区域的G蛋白耦联受体,可与SDF-1特异性结合构成SDF-1/CXCR4生物轴,不仅参与造血作用、免疫反应、器官发育,还参与血管生成的调节[138]。SDF-1/CXCR4可通过引导内皮细胞迁移和黏附,使缺血血管向血供充足的区域延伸[139]。SDF-1/CXCR4轴至少可以通过2条途径促进缺血损伤区域血管新生:①募集骨髓源细胞并诱导其分泌血管生成因子,从而促进血管新生;②募集血管内皮细胞及其前体细胞参加血管新生[140]。SDF-1/CXCR4信号轴对脑缺血后血管新生有着潜在的作用[141-142],脑缺血后内皮祖细胞被SDF-1/CXCR4信号轴动员:损伤局部产生大量 SDF-1, 进而通过囊泡运输系统-胞转过程,从外周血中易位到骨髓腔内,在骨髓腔中激活基质金属蛋白酶9,激活释放可溶性Kit配体,可溶性Kit配体诱导释放更多的SDF-1,促进内皮祖细胞随SDF-1浓度梯度向外周血动员,到达损伤部位参与血管新生[143]。 益气活血类中药可通过SDF-1/CXCR4通路影响血管生 成[144]。川芎嗪是具有活血行气的中药川芎的有效成分,戴儒丽[145]研究表明川芎嗪可促进骨髓间质干细胞表达SDF-1 mRNA;LI等[36]研究发现川芎嗪与骨髓间充质干细胞联合治疗显著上调了缺血性脑组织SDF-1和CXCR4的表达,并且比骨髓间充质干细胞单一疗法进一步促进了骨髓间充质干细胞向缺血区域的归巢,表明川芎嗪与骨髓间充质干细胞联合治疗不仅可以促进骨髓间充质干细胞向缺血脑组织迁移,而且可以协同作用,增强缺血脑卒中后内源性修复过程和功能恢复。 "
| [1] WANG J, ZHANG XH, XU X, et al. Pro-angiogenic activity of Tongnao decoction on HUVECs in vitro and zebrafish in vivo. J Ethnopharmacol. 2020;254:112737. [2] ZOU Y, HU J, HUANG W, et al. Non-Mitogenic Fibroblast Growth Factor 1 Enhanced Angiogenesis Following Ischemic Stroke by Regulating the Sphingosine-1-Phosphate 1 Pathway. Front Pharmacol. 2020;11:59. [3] KIM HY, KIM TJ, KANG L, et al. Mesenchymal stem cell-derived magnetic extracellular nanovesicles for targeting and treatment of ischemic stroke. Biomaterials. 2020;243:119942. [4] HEYDARI E, ALISHAHI M, GHAEDRAHMATI F, et al. The role of non-coding RNAs in neuroprotection and angiogenesis following ischemic stroke. Metab Brain Dis. 2020;35(1):31-43. [5] CARMELIET P, JAIN RK. Molecular mechanisms and clinical applications of angiogenesis. Nature. 2011;473(7347):298-307. [6] LIU L, WANG D, WONG KS, et al. Stroke and stroke care in China: huge burden, significant workload, and a national priority. Stroke. 2011; 42(12):3651-3654. [7] 李祥,杨文明.缺血性中风气虚血瘀证理论探讨[J].中医药临床杂志,2015,27(5):601-603. [8] 张凯亚,任媛媛.益气活血法在缺血性中风各期应用的研究进展[J].世界最新医学信息文摘,2019,19(37):55-56. [9] VAHIDINIA Z, AZAMI TAMEH A, NEJATI M, et al. The protective effect of bone marrow mesenchymal stem cells in a rat model of ischemic stroke via reducing the C-Jun N-terminal kinase expression. Pathol Res Pract. 2019;215(9):152519. [10] TSAI MJ, TSAI SK, HU BR, et al. Recovery of neurological function of ischemic stroke by application of conditioned medium of bone marrow mesenchymal stem cells derived from normal and cerebral ischemia rats. J Biomed Sci. 2014;21(1):5. [11] LV W, LI WY, XU XY, et al. Bone marrow mesenchymal stem cells transplantation promotes the release of endogenous erythropoietin after ischemic stroke. Neural Regen Res. 2015;10(8):1265-1270. [12] ONODERA Y, TERAMURA T, TAKEHARA T, et al. Transforming Growth Factor β-Activated Kinase 1 Regulates Mesenchymal Stem Cell Proliferation Through Stabilization of Yap1/Taz Proteins. Stem Cells. 2019;37(12):1595-1605. [13] SADAN O, SHEMESH N, COHEN Y, et al. Adult neurotrophic factor-secreting stem cells: a potential novel therapy for neurodegenerative diseases. Isr Med Assoc J. 2009;11(4):201-204. [14] LI N, LI XR, YUAN JQ. Effects of bone-marrow mesenchymal stem cells transplanted into vitreous cavity of rat injured by ischemia/reperfusion. Graefes Arch Clin Exp Ophthalmol. 2009;247(4):503-514. [15] NASSIRI SM, RAHBARGHAZI R. Interactions of mesenchymal stem cells with endothelial cells. Stem Cells Dev. 2014;23(4):319-332. [16] SETO SW, CHANG D, JENKINS A, et al. Angiogenesis in Ischemic Stroke and Angiogenic Effects of Chinese Herbal Medicine. J Clin Med. 2016; 5(6):56. [17] ERGUL A, ALHUSBAN A, FAGAN SC. Angiogenesis: a harmonized target for recovery after stroke. Stroke. 2012;43(8):2270-2274. [18] SAIJO Y, FURUMOTO H, YOSHIDA K, et al. Clinical Significance of Vascular Endothelial Growth Factor Expression and Microvessel Density in Invasive Cervical Cancer. J Med Invest. 2015;62(3-4):154-160. [19] 叶淑静.中药联合骨髓间充质干细胞移植治疗缺血性脑血管病实验研究进展[J].浙江中西医结合杂志,2015,25(10):984-987. [20] ZHAO YH. Essential Role of Chinese Medicines in Mesenchymal Stem Cells Transplantation for Treatment of Ischemic Stroke. Chin J Integr Med. 2019;25(10):723-727. [21] SEET RC, WIJDICKS EF, RABINSTEIN AA. Stroke from acute cervical internal carotid artery occlusion: treatment results and predictors of outcome. Arch Neurol. 2012;69(12):1615-1620. [22] ZIMNA A, KURPISZ M. Hypoxia-Inducible Factor-1 in Physiological and Pathophysiological Angiogenesis: Applications and Therapies. Biomed Res Int. 2015;2015:549412. [23] 魏珊珊.正丁基苯酞对实验性脑缺血小鼠脑梗死后血管新生的研究[D].石家庄:河北医科大学,2017. [24] 周逸凡.Sema3E/PlexinD1和Sema4D/PlexinB1信号在脑梗死后血管新生和血脑屏障破坏中的作用研究[D].武汉:华中科技大学,2018. [25] 王燃冰,胡红艳,刘颖,等.益气活血类中药及骨髓间充质干细胞对脑血管新生相关血管内皮生长因子及其受体影响的研究进展[J].中医临床研究,2015,7(4):144-145,148. [26] ŁAWICKI S, ZAJKOWSKA M, GŁAŻEWSKA EK, et al. Plasma levels and diagnostic utility of VEGF, MMP-2 and TIMP-2 in the diagnostics of breast cancer patients. Biomarkers. 2017;22(2):157-164. [27] ÖZKAYA A, KARABAŞ L, ALAGÖZ C, et al. Real-World Outcomes of Anti-VEGF Treatment for Neovascular Age-Related Macular Degeneration in Turkey: A Multicenter Retrospective Study, Bosphorus Retina Study Group Report No: 1. Turk J Ophthalmol. 2018;48(5):232-237. [28] HERMANN DM, ZECHARIAH A. Implications of vascular endothelial growth factor for postischemic neurovascular remodeling. J Cereb Blood Flow Metab. 2009;29(10):1620-1643. [29] 付曼妮,石年.活血消银片对银屑病患者VEGF水平和PASI指数的影响以及其临床疗效分析[J].中国中西医结合皮肤性病学杂志, 2017,16(1):38-40. [30] TALWAR T, SRIVASTAVA MV. Role of vascular endothelial growth factor and other growth factors in post-stroke recovery. Ann Indian Acad Neurol. 2014;17(1):1-6. [31] ZHENG H, LIU C, OU Y, et al. Total saponins of Panax notoginseng enhance VEGF and relative receptors signals and promote angiogenesis derived from rat bone marrow mesenchymal stem cells. J Ethnopharmacol. 2013;147(3):595-602. [32] OH TW, PARK KH, JUNG HW, et al. Neuroprotective effect of the hairy root extract of Angelica gigas NAKAI on transient focal cerebral ischemia in rats through the regulation of angiogenesis. BMC Complement Altern Med. 2015;15:101. [33] LIN JB, ZHENG CJ, ZHANG X, et al. Effects of Tetramethylpyrazine on Functional Recovery and Neuronal Dendritic Plasticity after Experimental Stroke. Evid Based Complement Alternat Med. 2015; 2015:394926. [34] QING L, WU P, ZHOU Z, et al. Tetramethylpyrazine improved the survival of multiterritory perforator flaps by inducing angiogenesis and suppressing apoptosis via the Akt/Nrf2 pathway. Drug Des Devel Ther. 2019;13:1437-1447. [35] JIN H, XIA B, YU N, et al. The effects of autologous bone marrow mesenchymal stem cell arterial perfusion on vascular repair and angiogenesis in osteonecrosis of the femoral head in dogs. Int Orthop. 2012;36(12):2589-2596. [36] LI L, CHU L, REN C, et al. Enhanced Migration of Bone Marrow-Derived Mesenchymal Stem Cells with Tetramethylpyrazine and Its Synergistic Effect on Angiogenesis and Neurogenesis After Cerebral Ischemia in Rats. Stem Cells Dev. 2019;28(13):871-881. [37] CHEN J, CAO W, ASARE PF, et al. Amelioration of cardiac dysfunction and ventricular remodeling after myocardial infarction by danhong injection are critically contributed by anti-TGF-β-mediated fibrosis and angiogenesis mechanisms. J Ethnopharmacol. 2016;194:559-570. [38] 卫国,殷英,段佳林,等.丹红注射液中5种主要活性成分促血管新生作用研究[J].现代生物医学进展,2017,17(17):3230-3233,3249. [39] 张鹏,周国庆,孙晶晶.丹红注射液联合骨髓间充质干细胞移植治疗大鼠脑梗死[J].中国组织工程研究,2013,17(23):4286-4291. [40] CHEN J, WEI J, HUANG Y, et al. Danhong Injection Enhances the Therapeutic Efficacy of Mesenchymal Stem Cells in Myocardial Infarction by Promoting Angiogenesis. Front Physiol. 2018;9:991. [41] FEI YX, WANG SQ, YANG LJ, et al. Salvia miltiorrhiza Bunge (Danshen) extract attenuates permanent cerebral ischemia through inhibiting platelet activation in rats. J Ethnopharmacol. 2017;207:57-66. [42] YANG Y, CHIN A, ZHANG L, et al. The role of traditional Chinese medicines in osteogenesis and angiogenesis. Phytother Res. 2014; 28(1):1-8. [43] 王晓娟.丹参多酚酸盐对大鼠骨髓间充质干细胞增殖及血管内皮生长因子表达的影响[J].环球中医药,2013,6(7):492-495. [44] WEI RL, TENG HJ, YIN B, et al. A systematic review and meta-analysis of buyang huanwu decoction in animal model of focal cerebral ischemia. Evid Based Complement Alternat Med. 2013;2013:138484. [45] 张振强,贾亚泉,宋军营,等.补阳还五汤对脑缺血大鼠模型ITGαvβ3表达及脑皮质区新生血管的影响[J].时珍国医国药,2013, 24(6):1308-1310. [46] 张运克,张振强,宋军喜.补阳还五汤联合MSCs移植对脑缺血再灌注模型大鼠VEGF及信号通路的影响[J].时珍国医国药,2016, 27(4):806-809. [47] HUANG Y, PEI J, WANG X. Outcome and safety assessment of YIN pattern of acute ischemic stroke treated with an herbal medicine protocol. Cerebrovasc Dis. 2007;24(6):483-494. [48] 陈朝,黄一挚,伍小燕,等.脑脉1号胶囊对移植骨髓间充质干细胞存活的影响[J].医学研究生学报,2015,28(1):32-36. [49] GUO JW, CHEN C, HUANG Y, et al. Combinatorial effects of naomai yihao capsules and vascular endothelial growth factor gene-transfected bone marrow mesenchymal stem cells on angiogenesis in cerebral ischemic tissues in rats. J Tradit Chin Med. 2012;32(1):87-92. [50] HEIL M, EITENMÜLLER I, SCHMITZ-RIXEN T, et al. Arteriogenesis versus angiogenesis: similarities and differences. J Cell Mol Med. 2006;10(1): 45-55. [51] WANG GL, JIANG BH, RUE EA, et al. Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular O2 tension. Proc Natl Acad Sci U S A. 1995;92(12):5510-5514. [52] 谢佳佳,李瑜辉,唐其东,等.三突变型低氧诱导因子-1α基因对老年小鼠缺血下肢血管的新生效应[J].广东医学,2016,37(14): 2065-2068. [53] 马进,甘雨,袁媛,等.川芎赤芍对脑缺血大鼠血管生成素及低氧诱导因子表达的影响[J].中国医药导报,2016,13(17):8-11,15. [54] YOSHIKAWA N, SHIMIZU N, OJIMA H, et al. Down-regulation of hypoxia-inducible factor-1 alpha and vascular endothelial growth factor by HEXIM1 attenuates myocardial angiogenesis in hypoxic mice. Biochem Biophys Res Commun. 2014;453(3):600-605. [55] LI L, QU Y, JIN X, et al. Protective effect of salidroside against bone loss via hypoxia-inducible factor-1α pathway-induced angiogenesis. Sci Rep. 2016;6:32131. [56] 韩国雄,李建平,严文琪,等.低氧对骨髓间充质干细胞缺氧诱导因子-1基因表达水平及其分泌的血管生长因子的影响[J].高原医学杂志,2016,26(4):12-16. [57] HA XQ, YANG B, HOU HJ, et al. Protective effect of rhodioloside and bone marrow mesenchymal stem cells infected with HIF-1-expressing adenovirus on acute spinal cord injury. Neural Regen Res. 2020;15(4): 690-696. [58] 侯茜,张秋菊,刘永琦,等.低氧环境下黄芪注射液对人骨髓间充质干细胞体外增殖的影响[J].中成药,2015,37(4):873-875. [59] 余晴晴,柏建峰,王江军.黄芪注射液对脑缺血再灌注损伤大鼠脑内血管新生及HIF-1α/VEGF信号转导通路的影响[J].蚌埠医学院学报,2017,42(10):1309-1313. [60] 余勤,李佩佩,周丽萍,等.黄芪联合鞘内移植间充质干细胞对大鼠缺氧缺血性脑损伤修复作用的研究[J].中华中医药学刊,2011, 29(7):1455-1458. [61] KORHONEN EA, LAMPINEN A, GIRI H, et al. Tie1 controls angiopoietin function in vascular remodeling and inflammation. J Clin Invest. 2016; 126(9):3495-3510. [62] SAHARINEN P, LEPPÄNEN VM, ALITALO K. SnapShot: Angiopoietins and Their Functions. Cell. 2017;171(3):724. [63] 宋祯彦,周瑜,王珊珊,等.中药脑泰方对大鼠脑缺血后VEGF和Angiopoietins表达的实验研究[J].湖南中医药大学学报,2016, 36(1):19-22. [64] KRÜGER-GENGE A, BLOCKI A, FRANKE RP, et al. Vascular Endothelial Cell Biology: An Update. Int J Mol Sci. 2019;20(18):4411. [65] GAONAC’H-LOVEJOY V, BOSCHER C, DELISLE C, et al. Rap1 is Involved in Angiopoietin-1-Induced Cell-Cell Junction Stabilization and Endothelial Cell Sprouting. Cells. 2020;9(1):155. [66] HWANG-BO J, PARK JH, CHUNG IS. 3-O-Acetyloleanolic acid inhibits angiopoietin-1-induced angiogenesis and lymphangiogenesis via suppression of angiopoietin-1/Tie-2 signaling. Phytother Res. 2020; 34(2):359-367. [67] THOMAS M, AUGUSTIN HG. The role of the Angiopoietins in vascular morphogenesis. Angiogenesis. 2009;12(2):125-137. [68] PARIKH SM. The Angiopoietin-Tie2 Signaling Axis in Systemic Inflammation. J Am Soc Nephrol. 2017;28(7):1973-1982. [69] PARIKH SM. Angiopoietins and Tie2 in vascular inflammation. Curr Opin Hematol. 2017;24(5):432-438. [70] 胡夏晓,张文倩,刘炳男,等.Ang-1和Tie-2在血管生成中的作用及中医药干预研究进展[J].中医药临床杂志,2018,30(8):1391-1394. [71] 梁晓伟,李阳飞,李琥,等.羟基红花黄色素A对人骨髓间充质干细胞成骨分化的影响[J].口腔生物医学,2017,8(2):90-94. [72] CHEN T, CHEN N, PANG N, et al. Hydroxysafflor Yellow A Promotes Angiogenesis via the Angiopoietin 1/ Tie-2 Signaling Pathway. J Vasc Res. 2016;53(5-6):245-254. [73] YAO H, SHI P, SHAO Q, et al. Chemical fingerprinting and quantitative analysis of a Panax notoginseng preparation using HPLC-UV and HPLC-MS. Chin Med. 2011;6:9. [74] HUI Z, SHA DJ, WANG SL, et al. Panaxatriol saponins promotes angiogenesis and enhances cerebral perfusion after ischemic stroke in rats. BMC Complement Altern Med. 2017;17(1):70. [75] MOISAN A, FAVRE I, ROME C, et al. Intravenous Injection of Clinical Grade Human MSCs After Experimental Stroke: Functional Benefit and Microvascular Effect. Cell Transplant. 2016;25(12):2157-2171. [76] 李雷兵,王皎月,孙彩虹.三七皂苷联合骨髓间充质干细胞移植促进急性脑损伤的恢复[J].中国组织工程研究,2016,20(41): 6138-6144. [77] BAO C, WANG Y, MIN H, et al. Combination of ginsenoside Rg1 and bone marrow mesenchymal stem cell transplantation in the treatment of cerebral ischemia reperfusion injury in rats. Cell Physiol Biochem. 2015;37(3):901-910. [78] KERMANI P, HEMPSTEAD B. Brain-derived neurotrophic factor: a newly described mediator of angiogenesis. Trends Cardiovasc Med. 2007;17(4):140-143. [79] WANG Y, YANG J, DU H, et al. Yangyin Tongnao granules enhance neurogenesis in the peri-infarct area and upregulate brain-derived neurotrophic factor and vascular endothelial growth factor after focal cerebral ischemic infarction in rats. Mol Biol Rep. 2019;46(4): 3817-3826. [80] NAGAHARA AH, TUSZYNSKI MH. Potential therapeutic uses of BDNF in neurological and psychiatric disorders. Nat Rev Drug Discov. 2011; 10(3):209-219. [81] 张璐.多发性骨髓瘤骨髓微环境中脑源性神经营养因子促进血管新生的作用及调控机制的研究[D].武汉:华中科技大学,2010. [82] PIKULA A, BEISER AS, CHEN TC, et al. Serum brain-derived neurotrophic factor and vascular endothelial growth factor levels are associated with risk of stroke and vascular brain injury: Framingham Study. Stroke. 2013;44(10):2768-2775. [83] FOUDA AY, ALHUSBAN A, ISHRAT T, et al. Brain-Derived Neurotrophic Factor Knockdown Blocks the Angiogenic and Protective Effects of Angiotensin Modulation After Experimental Stroke. Mol Neurobiol. 2017;54(1):661-670. [84] SUN C, HU Y, CHU Z, et al. The effect of brain-derived neurotrophic factor on angiogenesis. J Huazhong Univ Sci Technolog Med Sci. 2009; 29(2):139-143. [85] 刘鹏,王瑾,苗常青,等.丹参乙酸镁对大鼠局灶性脑缺血损伤后神经营养因子表达的影响[J].脑与神经疾病杂志,2015,23(3): 165-169. [86] 卢乙众,李合华,卢奕帆.丹参酮Ⅱ联合骨髓间充质干细胞移植促进脑梗死后的神经再生[J].中国组织工程研究,2016,20(23): 3425-3431. [87] CHEN P, ZHANG H, ZHANG Q, et al. Basic Fibroblast Growth Factor Reduces Permeability and Apoptosis of Human Brain Microvascular Endothelial Cells in Response to Oxygen and Glucose Deprivation Followed by Reoxygenation via the Fibroblast Growth Factor Receptor 1 (FGFR1)/ERK Pathway. Med Sci Monit. 2019;25:7191-7201. [88] NAKAMICHI M, AKISHIMA-FUKASAWA Y, FUJISAWA C, et al. Basic Fibroblast Growth Factor Induces Angiogenic Properties of Fibrocytes to Stimulate Vascular Formation during Wound Healing. Am J Pathol. 2016;186(12):3203-3216. [89] HORIKOSHI-ISHIHARA H, TOBITA M, TAJIMA S, et al. Coadministration of adipose-derived stem cells and control-released basic fibroblast growth factor facilitates angiogenesis in a murine ischemic hind limb model. J Vasc Surg. 2016;64(6):1825-1834. [90] GUO H, HUANG L, CHENG M, et al. Serial measurement of serum basic fibroblast growth factor in patients with acute cerebral infarction. Neurosci Lett. 2006;393(1):56-59. [91] MAO D, LI F, MA Q, et al. Intraocular administration of tetramethylpyrazine hydrochloride to rats: a direct delivery pathway for brain targeting? Drug Deliv. 2019;26(1):841-848. [92] 吴宁,孙汉英,刘文励,等.川芎嗪对急性放射损伤小鼠骨髓碱性成纤维细胞生长因子及其受体表达水平的影响[J].中国中西医结合杂志,2004,24(5):439-441. [93] ZHANG JC, ZHENG GF, WU L, et al. Bone marrow mesenchymal stem cells overexpressing human basic fibroblast growth factor increase vasculogenesis in ischemic rats. Braz J Med Biol Res. 2014;47(10): 886-894. [94] 吴晓明,高文山,王静,等.盐酸川芎嗪联合骨髓间充质干细胞移植对脊髓损伤模型大鼠的神经保护[J].中国组织工程研究,2016, 20(1):95-101. [95] BI JJ, YI L. Effects of integrins and integrin αvβ3 inhibitor on angiogenesis in cerebral ischemic stroke. J Huazhong Univ Sci Technolog Med Sci. 2014;34(3):299-305. [96] YUAN HX, WANG WP, WEN JX, et al. Dual-Targeted Microbubbles Specific to Integrin αVβ3 and Vascular Endothelial Growth Factor Receptor 2 for Ultrasonography Evaluation of Tumor Angiogenesis. Ultrasound Med Biol. 2018;44(7):1460-1467. [97] SOMANATH PR, MALININ NL, BYZOVA TV. Cooperation between integrin alphavbeta3 and VEGFR2 in angiogenesis. Angiogenesis. 2009; 12(2):177-185. [98] 段枫阳,刘颖,张运克.益气活血类中药及骨髓间充质干细胞对脑缺血组织血管新生相关因子整合素影响的研究进展[J].中医临床研究,2015,7(4):146-148. [99] 吴季,唐涛,薛俐,等. 补阳还五汤对脑出血大鼠血肿周围新生血管整合素αvβ3和β3亚基表达的影响[J].中药新药与临床药理, 2011,22(4):402-406. [100] 张运克,杨俊红,高峰,等.补阳还五汤联合骨髓间充质干细胞移植对大脑中动脉栓塞模型整合素相关因子的影响[J].中国组织工程研究,2017,21(5):724-729. [101] 王有科,覃兴乐.脑源性神经营养因子和血管内皮生长因子及基质金属蛋白酶-9与脑梗死关系研究进展[J].中华实用诊断与治疗杂志, 2017,31(1):95-97. [102] 侯凌波.灯盏细辛对血瘀型急性脑梗死VEGF、MMP-9、EPCs血清学水平的影响[D].广州:广州中医药大学,2014. [103] 袁加文,赵玉武.基质金属蛋白酶-9及血管性血友病因子与急性脑梗死的关系[J].河北医学,2013,19(5):756-759. [104] 庄钦,毛威.丹参多种活性成分调节血管新生机制的研究概述[J].浙江中医药大学学报,2014,38(4):506-510. [105] 俞鸣.急性脑梗死患者血清血管内皮生长因子和基质金属蛋白酶-9水平的动态变化及其意义[J].中国全科医学,2010,13(18): 1983-1985. [106] HULKKONEN J, PERTOVAARA M, ANTONEN J, et al. Matrix metalloproteinase 9 (MMP-9) gene polymorphism and MMP-9 plasma levels in primary Sjogren’s syndrome. Rheumatology (Oxford). 2004;43(12):1476-1479. [107] 余猛进,卫波,顾桂国.血浆基质金属蛋白酶-9与血管假性血友病因子浓度在急性脑梗死早期诊断中的意义[J].临床医学,2010, 30(5):6-8. [108] 李建生,刘敬霞,孙捷,等.大黄苷元对骨髓间充质干细胞移植脑缺血大鼠基质金属蛋白酶的影响[J].中西医结合学报,2008,6(8): 810-816. [109] 胡红艳,王燃冰,刘颖,等.益气活血类方药及骨髓间充质干细胞(BMSCs)在脑缺血再灌注损伤中对基质金属蛋白酶(MMPs)的影响的研究进展[J].中医临床研究,2015,7(4):137-139. [110] 蓝艳.补阳还五汤对气虚血瘀型缺血性脑卒中患者血浆基质金属蛋白酶影响[J].福建中医药,2013,44(1):21-22. [111] 李树仁,王天红,刘东霞,等.自体骨髓间充质干细胞移植对急性心肌梗死后心功能不全家兔基质金属蛋白酶-9的影响[J].中国老年学杂志,2013,33(23):5947-5950. [112] 张运克,车志英,梅雪蕊.补阳还五汤联合骨髓间充质干细胞移植对大鼠脑缺血再灌注模型神经细胞凋亡的影响[J].中华中医药刊,2009,27(8):1660-1662. [113] 陈海丽.经典Wnt信号通路在大鼠缺血性脑卒中后血管新生中的作用研究[D].济南:山东大学,2015. [114] FU S, YANG L, HONG H, et al. Wnt/β-catenin signaling is involved in the Icariin induced proliferation of bone marrow mesenchymal stem cells. J Tradit Chin Med. 2016;36(3):360-368. [115] WANG Y, WU Z, TIAN J, et al. Intermedin protects HUVECs from ischemia reperfusion injury via Wnt/β-catenin signaling pathway. Ren Fail. 2019;41(1):159-166. [116] DU J, LI J. The role of Wnt signaling pathway in atherosclerosis and its relationship with angiogenesis. Exp Ther Med. 2018;16(3):1975-1981. [117] LI X, LU Q, XIE W, et al. Anti-tumor effects of triptolide on angiogenesis and cell apoptosis in osteosarcoma cells by inducing autophagy via repressing Wnt/β-Catenin signaling. Biochem Biophys Res Commun. 2018;496(2):443-449. [118] GUO P, WANG Y, DAI C, et al. Ribosomal protein S15a promotes tumor angiogenesis via enhancing Wnt/β-catenin-induced FGF18 expression in hepatocellular carcinoma. Oncogene. 2018;37(9):1220-1236. [119] LI X,HONG S,TAN C,et al. Combined study on the action and mechanism of g-rg1/sr-cas bone substitute material for ossification and pro-vascularization.Materials Express.2020;10(2):177-189. [120] LEUNG KW, PON YL, WONG RN, et al. Ginsenoside-Rg1 induces vascular endothelial growth factor expression through the glucocorticoid receptor-related phosphatidylinositol 3-kinase/Akt and beta-catenin/T-cell factor-dependent pathway in human endothelial cells. J Biol Chem. 2006;281(47):36280-36288. [121] MCBRIDE JD, RODRIGUEZ-MENOCAL L, GUZMAN W, et al. Bone Marrow Mesenchymal Stem Cell-Derived CD63+ Exosomes Transport Wnt3a Exteriorly and Enhance Dermal Fibroblast Proliferation, Migration, and Angiogenesis In Vitro. Stem Cells Dev. 2017;26(19):1384-1398. [122] 王祺.独参汤联合骨髓间充质干细胞治疗急性心肌梗死大鼠的相关研究[D].沈阳:辽宁中医药大学,2011. [123] YAMAMOTO S, SCHULZE KL, BELLEN HJ. Introduction to Notch signaling. Methods Mol Biol. 2014;1187:1-14. [124] GRIDLEY T. Notch signaling in the vasculature. Curr Top Dev Biol. 2010; 92:277-309. [125] FIÚZA UM, ARIAS AM. Cell and molecular biology of Notch. J Endocrinol. 2007;194(3):459-474. [126] 刘思彤.姜黄素通过Notch信号通路抑制角膜新生血管的体内、体外研究[D].锦州:锦州医科大学,2018. [127] 刘琦,魏璐珊,王颖,等.Notch信号通路在大鼠骨髓间充质干细胞移植后促进脑缺血区血管新生过程中的作用[J].中国老年学杂志, 2017,37(17):4185-4187. [128] ZHAO J, LIN W, CAO Z, et al. Total alkaloids of Rubus alceifolius Poir inhibit tumor angiogenesis through suppression of the Notch signaling pathway in a mouse model of hepatocellular carcinoma. Mol Med Rep. 2015;11(1):357-361. [129] SHI WL, ZHAO J, YUAN R, et al. Combination of Ligusticum Chuanxiong and Radix Paeonia Promotes Angiogenesis in Ischemic Myocardium through Notch Signalling and Mobilization of Stem Cells. Evid Based Complement Alternat Med. 2019;2019:7912402. [130] SONG D, LIU X, DIAO Y, et al. Hydrogen‑rich solution against myocardial injury and aquaporin expression via the PI3K/Akt signaling pathway during cardiopulmonary bypass in rats. Mol Med Rep. 2018;18(2): 1925-1938. [131] DAVIES EM, GURUNG R, LE KQ, et al. Effective angiogenesis requires regulation of phosphoinositide signaling. Adv Biol Regul. 2019;71: 69-78. [132] XU C, WANG J, ZHU T, et al. Cross-Talking Between PPAR and WNT Signaling and its Regulation in Mesenchymal Stem Cell Differentiation. Curr Stem Cell Res Ther. 2016;11(3):247-254. [133] 李磊,高绚照,马连萍,等.积雪草苷通过激活PI3K/Akt信号通路缓解局灶性脑梗死大鼠脑组织损伤和诱导血管新生[J].中国免疫学杂志,2019,35(17):2068-2072,2078. [134] HUANG Y, MAO Y, LI H, et al. Knockdown of Nrf2 inhibits angiogenesis by downregulating VEGF expression through PI3K/Akt signaling pathway in cerebral microvascular endothelial cells under hypoxic conditions. Biochem Cell Biol. 2018;96(4):475-482. [135] SUN D, ZHANG F, QIAN J, et al. 4’-hydroxywogonin inhibits colorectal cancer angiogenesis by disrupting PI3K/AKT signaling. Chem Biol Interact. 2018;296:26-33. [136] CUI HJ, YANG AL, ZHOU HJ, et al. Buyang huanwu decoction promotes angiogenesis via vascular endothelial growth factor receptor-2 activation through the PI3K/Akt pathway in a mouse model of intracerebral hemorrhage. BMC Complement Altern Med. 2015;15:91. [137] 曾新生,姚微微.SDF-1/CXCR4生物轴与眼部新生血管的研究进展[J].华夏医学,2015,28(1):167-170. [138] 张晗祥,何斌,胡侦明.SDF1/CXCR4信号轴在退变髓核细胞诱导血管新生中的作用[J].基础医学与临床,2019,39(5):682-689. [139] XIANG Y, YAO X, WANG X, et al. Houshiheisan promotes angiogenesis via HIF-1α/VEGF and SDF-1/CXCR4 pathways: in vivo and in vitro. Biosci Rep. 2019;39(10):BSR20191006. [140] 胡惠琼,吴雄伟,杨玉燕,等.SDF-1/CXCR4轴及其对老年血管新生的影响机制进展[J].四川生理科学杂志,2015,37(3):137-139. [141] 吴晓光,李义学,尹瑞新,等.缺血再灌注大脑皮质区微血管、神经元的损伤及补阳还五汤干预作用[J].时珍国医国药,2011,22(4): 1013-1014. [142] 檀荣方,夏爱华,吴晓光,等.山楂叶总黄酮抑制慢性脑缺血模型大鼠脑组织神经元的凋亡(英文)[J].中国组织工程研究,2014, 18(49):7879-7883. [143] 王瑾.针刺内关对MCAO大鼠血管新生的影响SDF-1/CXCR4信号轴调节机制研究[D].济南:山东中医药大学,2015. [144] 殷沈华,唐德才.益气活血类中药通过SDF-1/CXCR4通路影响血管生成的研究[J].陕西中医,2013,34(5):638-639,641. [145] 戴儒丽.川芎嗪对骨髓间质干细胞增殖、SDF-1mRNA表达及向心肌样细胞分化的实验研究[D].广州:广州中医药大学,2010. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Yuan Jiawei, Zhang Haitao, Jie Ke, Cao Houran, Zeng Yirong. Underlying targets and mechanism of Taohong Siwu Decoction in prosthetic joint infection on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1428-1433. |
| [4] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [5] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [6] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [7] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [8] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [9] | Geng Qiudong, Ge Haiya, Wang Heming, Li Nan. Role and mechanism of Guilu Erxianjiao in treatment of osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1229-1236. |
| [10] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [11] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [12] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [13] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [14] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [15] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||