Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (9): 1441-1447.doi: 10.3969/j.issn.2095-4344.1582
Previous Articles Next Articles
Mesenchymal stem cell-derived exosomes for the treatment of neurodegenerative diseases: existing problems and prospects in application
Chen Chen1, 2, 3, Huang Hui1, 2, 3, Hu Wenjia1, 2, 3, Li Xiangyong1, 2, 3
- 1Institute of Biochemistry and Molecular Biology, 2Collaborative Innovation Center for Antitumor Active Substance Development, Guangdong Medical University, Zhanjiang 524023, Guangdong Province, China; 3Guangdong Key Laboratory of Medical Active Molecules, Zhanjiang 524023, Guangdong Province, China
-
Revised:2018-11-16Online:2019-03-28Published:2019-03-28 -
Contact:Li Xiangyong, MD, Associate professor, 1Institute of Biochemistry and Molecular Biology, 2Collaborative Innovation Center for Antitumor Active Substance Development, Guangdong Medical University, Zhanjiang 524023, Guangdong Province, China; 3Guangdong Key Laboratory of Medical Active Molecules, Zhanjiang 524023, Guangdong Province, China -
About author:Chen Chen, Master candidate, 1Institute of Biochemistry and Molecular Biology, 2Collaborative Innovation Center for Antitumor Active Substance Development, Guangdong Medical University, Zhanjiang 524023, Guangdong Province, China; 3Guangdong Key Laboratory of Medical Active Molecules, Zhanjiang 524023, Guangdong Province, China -
Supported by:the Guangdong Provincial Special Fund for Science and Technology Innovation Strategy, No. 2018A030310155 (to LHQ); Guangdong Provincial “Sailing Plan” for High-Level Talents Training, No. 4YF17001G (to LXY); Zhanjiang Municipal Financial Competition Project, No. 2016A01024 (to LHQ)
CLC Number:
Cite this article
Chen Chen, Huang Hui, Hu Wenjia, Li Xiangyong. Mesenchymal stem cell-derived exosomes for the treatment of neurodegenerative diseases: existing problems and prospects in application[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(9): 1441-1447.
share this article

2.1 间充质干细胞源性外泌体的起源及生物学特性 外泌体是一类直径为30-150 nm的膜性囊泡样小体,不仅包含特征性蛋白标志物如TSG101,CD9,CD63和CD81等,而且可携带包括蛋白、脂质、微小RNA和长链非编码RNA在内的各种生物分子来传递功能性遗传信息[10]。目前发现间充质干细胞是分泌外泌体能力最强的细胞[11],其可通过特定的信号轴来调控外泌体的释放[12],同时细胞外环境的改变也能影响间充质干细胞分泌外泌体的能力和活性[13]。利用无外泌体的血清培养间充质干细胞最终分离得到的间充质干细胞源性外泌体不仅共性表达外泌体标志蛋白如CD63,CD81等,还表达间充质干细胞的相关分子如CD29,CD44,CD90和CD73等[14]。 近年来,不断有研究证实间充质干细胞源性外泌体能够借助所携带的蛋白、非编码RNA、微小RNA等生物活性物质在多种疾病的治疗中发挥作用。新近的研究发现由富含外泌体的间充质干细胞衍生的分级分泌蛋白在肝损伤中可赋予肝脏保护作用[15]。此外,Ferguson等[16]通过对间充质干细胞源性外泌体中携带的内容物进行网络分析,不仅发现外泌体miRNA是调节心肌损伤修复的主要活性分子,而且明确其作用的生物学过程和途径。因此,目前逐渐认识到间充质干细胞源性外泌体能借助不同方式促进细胞间的信息传递和抗原呈递[17-18]、调节机体免疫应答[19]、促进细胞增殖、预防细胞凋亡、诱导血管生成、修复受损组织。 2.2 外泌体的分离鉴定与保存方法 外泌体独特的结构和临床应用的良好前景,使其逐渐成为研究的热点,而从细胞液中有效地提取并获得完整的外泌体是开展外泌体研究的前提与关键。当前外泌体的分离方法通常包括超速离心法、聚合物沉淀法、体积排阻与超滤法、免疫亲和捕获法和微流体芯片技术等,见表1。"

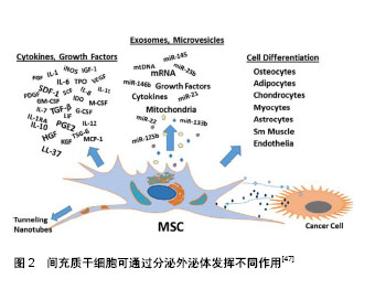
2.2.1 超速离心法 又称差速离心法是外泌体分离的标准方法,其一般步骤是先使用亚微米过滤器或低速离心法去除细胞碎片,微泡或凋亡小体;随后,以多次 100 000×g或更高的速度进行超速离心以消除大囊泡并使外泌体沉淀下来;最后去除上清液,在相对较少体积的缓冲液里重悬外泌体,从而得到用于研究的外泌体样品[20]。该方法虽然成本不高,但最终所得外泌体纯度低,且需要进行再提纯,回收率不稳定,故其是一个耗时耗力的过程[21]。最近也有实验将超速离心法与蔗糖密度梯度离心法组合起来用于分离低丰度外泌体[22]。 2.2.2 聚合沉淀法 目前常用的是System bioscience (SBI)研发的ExoQuickTM试剂盒,其原理是在4 ℃下将聚合物沉淀剂与样本共同孵育30 min,低速离心后获取沉淀并用PBS重悬后获取外泌体[23]。因为其操作简单且不需要额外的实验设备,在近年来饱受青睐[24]。由于这些试剂需要过夜处理且可能存在非外泌体材料的污染和其他蛋白与聚合物的共沉淀,所以会对后续的实验分析有一定的干扰性。 2.2.3 尺寸排阻色谱法与超滤法 这2种方法均以分子大小为基础进行分离。尺寸排阻色谱法可用于血浆样品及细胞培养上清液的外泌体分离,利用装填的多孔性材料色谱柱不仅可去除大多数的白蛋白和免疫球蛋白,而且洗脱分离的外泌体在形态上是完整的,非聚集的和有功能性的。最新研究证实使用尺寸排阻层析和密度垫超速离心结合的方法来分离血液中细胞外囊泡,能显著改善细胞外囊泡与脂蛋白的分离情况,从而允许研究者对血浆外泌体的蛋白质组进行详细分析,使得血液成为发现外泌体生物标记物的可行来源[25]。超滤法是利用截留不同相对分子质量的聚醚砜超滤膜对样品进行系统性分离从而获取外泌体[26]。 2.2.4 免疫亲和捕获法 此方法是基于外泌体特异性膜抗原与抗体特异性结合进行外泌体分离,包括酶联免疫吸附实验分离法和抗体包被的免疫磁珠法。基于免疫亲和原理,研究者在分离外泌体的实验中使用ExoELISA-ULTRA完整试剂盒,即通过CD63(一种常见的外泌体标志物)进行酶联免疫吸附分离得到了来自脐带血内皮祖细胞中的外泌体[27]。Zeming等[28]利用含微米尺寸柱状阵列的确定性横向位移装置来灵敏检测纳米尺寸生物颗粒,该方法依赖于测量捕获目标生物颗粒前后聚合物珠粒尺寸或静电荷的变化,进行外泌体的分离与定量检测。磁珠法虽具备特异性高、操作简单且实验中始终维持外泌体完整形态等优点,但所分离的外泌体容易被环境中的酸碱度干扰,限制了广泛推广。 2.2.5 微流体芯片技术 这是一项新兴的技术,具有低耗容、效率快和高灵敏度等优点,在外泌体分离和分析方面拥有着广泛前景。Marczak等[29]就在实验中提出了一种新的微流控技术,通过使用压力驱动流将样品输送至微流体芯片,利用横向电场迫使它们离开交叉流并进入琼脂糖凝胶,从而过滤掉不需要的细胞碎片,而离子选择性膜通过富集效应将外泌体浓缩。这项既可以分离又可以富集回收外泌体的技术,可能会在将来早期癌症的检测中发挥重要作用。 除此之外,随着生物技术的快速发展及人们对外泌体的深入了解,不断出现分离外泌体的新方法与技术,如利用亲和性膜离心柱分离方法可从外泌体中高效分离RNA,这种方法允许一次性处理较大体积的样本(最多4 mL)来有效探测低丰度RNA[30]。与传统超速离心法不同,这种技术能快速获得稳定的外泌体且可在敏感的下游得到应用。因此有理由相信越来越多分离外泌体的方法必将有效促进外泌体的深入研究和临床应用程度。 外泌体分离后进行有效鉴定是开展后续的研究或临床应用的前提与基础。目前对于外泌体的鉴定,主要方法包括流式细胞术、透射电子显微镜成像、生物免疫如SDS-PAGE/Western blot/抗体阵列技术和动态光散射技术。为避免单一技术的缺陷,研究者通常联合几种方法,同时结合外泌体的特性进行外泌体的鉴定[31-33]。 然而,需要指出的是外泌体的保存环境与保存时间对维持外泌体直径及完整性同样至关重要。Sokolova 等[34]研究指出6个月内存储在-20 ℃环境下的外泌体即使反复冻融多次也不会发生结构变异,而4 ℃和37 ℃环境下储存的外泌体却存在结构降解的风险。Cheng等[35]发现在24 h内,外泌体最佳的储存温度为4 ℃,此时较高的温度和冻融循环可能会影响外泌体膜结构并改变其特性,引起外泌体浓度的降低;而长期储存时,-80 ℃的低温环境更有利于维持外泌体的完整性;然而,该实验结果还表明酸性储存环境会降低外泌体的浓度,这与其他文献中报道的酸性pH可减少外泌体相关蛋白的降解结果相矛盾[36],故环境pH值对外泌体的影响值得进一步研究。 2.3 间充质干细胞源性外泌体在神经退行性疾病治疗中的应用进展 神经退行性疾病的发生是神经元或髓鞘的丧失导致,伴随着病程的进展而不断加重,最终引起神经功能障碍。其可分为急性神经退行性疾病和慢性神经退行性疾病,前者主要包括脑缺血、脑出血、脑损伤等;后者包括阿尔茨海默症、帕金森病、亨廷顿舞蹈症、肌萎缩侧索硬化、不同类型脊髓小脑共济失调、Pick病等[37-38]。近年来,随着神经退行性疾病的普遍性越来越高,对于科学界寻找神经退行性疾病的有效治疗途径是一项巨大的挑战。大量的研究发现间充质干细胞在组织再生中表现出明显的多分化、免疫调节和神经保护等功能,而且研究也证实了不同类型的间充质干细胞在神经退行性疾病的治疗中扮演着重要角色。因此,间充质干细胞可能成为治疗神经退行性疾病的重要手段之一[39]。Moayeri等[40]通过实验证实人牙髓间充质干细胞可被神经胶质诱导培养基诱导分化成少突胶质细胞,可能成为神经退行性疾病细胞疗法的重要来源。此外,过表达FOXQ1的脐带间充质干细胞移植到阿尔茨海默病动物模型后,伴随着动物脑中脐带间充质干细胞数量的增加,受试动物的认知功能和对抗病理性衰老的能力也不断改善[41]。Marei等[42]利用神经诱导培养基在体外诱导脂肪间充质干细胞分化成功能性神经元,促进和确保用于治疗神经退行性疾病性的脂肪间充质干细胞的重现性和标准化生产。同样的,骨髓间充质干细胞在神经方面的研究也在不断深入[43]。虽然以干细胞为基础的治疗提供了很大希望和承诺,但干细胞移植依然存在着如移植细胞的定向迁移、植入与存活能力较差等缺点[44]。而外泌体因为具有低免疫原性和长循环半衰期以及可携带小分子物质如微小RNA和蛋白质等穿过血脑屏障等特点,所以能引发并调节神经炎症、修饰神经原性小生境和神经的发生[45-46],如今不断有研究证实间充质干细胞源性外泌体有助于改善受损的神经系统功能和增强血管神经发生,从而可能为干细胞移植治疗神经退行性疾病提供了一种有效的替代方法[47],见图2。"
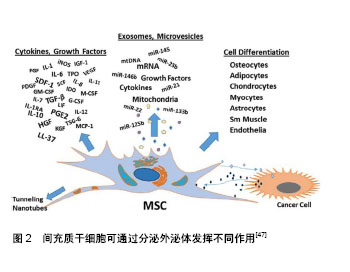
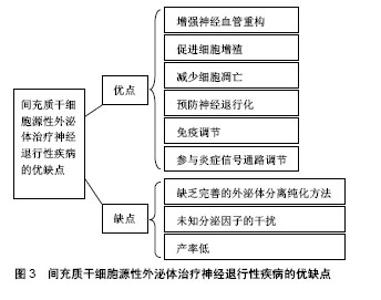
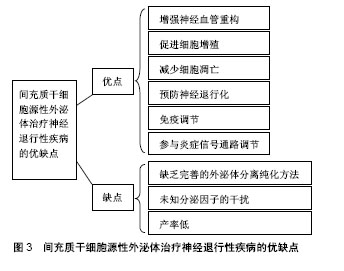
2.3.1 间充质干细胞源性外泌体与急性神经退行性疾病的研究 目前认为创伤性脑损伤会引起许多长期不利的神经学后果。Darkazalli等[48]使用京都基因和基因组百科全书(Kyoto Encyclopedia of Genes and Genomes,KEGG)数据库途径分析出创伤性脑损伤调控大量与神经元可塑性和神经退行性疾病有关的基因,导致神经退行性疾病状态的发生。同时,实验研究发现人类牙髓间充质干细胞分泌的外泌体可以促进啮齿动物创伤性脑损伤模型的小胶质细胞(中枢神经系统的主要活性细胞之一)发生M2极化,M2型小胶质细胞分泌的抗炎细胞因子可抑制神经炎症的发生,从而促进脑功能的恢复并减少神经元的退化[49]。而且间充质干细胞源性外泌体还能改善大鼠早产型脑损伤模型中的炎症诱导的细胞损伤、预防神经元死亡、恢复白质微观结构、减少胶质增生和促进脑功能的长期恢复,成为治疗脑损伤的一种新的治疗选择[50]。 脑出血是指非创伤性脑实质内血管破裂引起的出血,其发作大多与脑血管的病变有关,由于其高死亡率和高发病率,脑出血通常预后不佳。实验发现将miR-133b转染后的间充质干细胞源性外泌体经尾注射至脑出血大鼠模型后,其脑组织中的凋亡现象和退行性神经元明显减少,从而说明转染后的间充质干细胞源性外泌体在脑出血的治疗方面起到了神经保护作用[51]。 近年来,利用间充质干细胞源性外泌体治疗脑缺血后卒中的方法也引起了广泛的关注。研究发现间充质干细胞源性外泌体促进了啮齿动物卒中模型中缺血性脑组织的神经血管重构和脑内皮细胞的增殖,从而改善了脑功能[52]。同时间充质干细胞源性外泌体不仅可以促进已发生局灶性脑缺血小鼠的脑血管再生,还能拮抗缺血后发生的免疫抑制反应,为成功的脑重构提供适当的外部环境[53]。此外,骨髓来源的间充质干细胞分泌的外泌体在治疗早产儿大脑的缺氧缺血性损伤也存在希望[54]。 2.3.2 间充质干细胞源性外泌体与慢性神经退行性疾病的研究 阿尔茨海默症是最常见的痴呆类型,由慢性进行性中枢神经系统变性导致,并伴随记忆和认知功能减退。Lee等[55]发现脂肪间充质干细胞源性外泌体在减少阿尔茨海默症模型细胞凋亡的同时促进神经元干细胞的神经元生长从而改善受试小鼠的认知功能并修复受损的神经病理学机制。间充质干细胞源性外泌体可能是在神经退行性疾病如阿尔茨海默症中赋予神经保护作用的潜在治疗剂[56]。 帕金森病是阿尔茨海默症后第二大常见的神经退行性疾病以及最常见的运动障碍之一,帕金森病中的运动障碍很大程度上是黑质致密部中脑多巴胺神经元的丢失造成的。研究发现脱落乳牙间充质干细胞来源的外泌体能够抑制6-羟基多巴胺诱导的多巴胺能神经元凋亡,提示以间充质干细胞源性外泌体为基础的治疗方式可能成为帕金森病治疗的新途径[57]。 亨廷顿病是常染色体显性遗传的神经退行性疾病,疾病伴随亨廷顿蛋白的产生,最终引起纹状体细胞死亡。研究显示,人类脂肪间充质干细胞源性外泌体可通过减少亨廷顿蛋白的聚集、调节凋亡相关蛋白、保护线粒体进而促进细胞的存活,表明其在治疗亨廷顿病方面具有一定的潜力[58]。 肌萎缩侧索硬化是一种不可治愈的致命性进行性退行性疾病,涉及运动神经元细胞如NSC-34细胞的死亡,目前还未有较好的治疗方案。在不断探寻治疗神经退行性疾病新方法的同时,人们通过实验发现脂肪源性间充质干细胞所分泌的外泌体不仅能够保护运动神经元样细胞株NSC-34使其避免氧化损伤,增加细胞活力[59],还能减少超氧化物歧化酶1的聚集、阻止线粒体功能障碍的发生、调节肌萎缩侧索硬化的细胞类型[60],从而进一步突出了间充质干细胞源性外泌体在肌萎缩侧索硬化潜在治疗应用中的作用,见图3。"
| [1] McClarren B, Olabisi R. Strain and Vibration in Mesenchymal Stem Cells. Int J Biomater. 2018;2018:8686794. [2] Tsai YA, Liu RS, Lirng JF, et al. Treatment of Spinocerebellar Ataxia With Mesenchymal Stem Cells: A Phase I/IIa Clinical Study. Cell Transplant. 2017;26(3):503-512. [3] Hatzistergos KE, Saur D, Seidler B, et al. Stimulatory Effects of Mesenchymal Stem Cells on cKit+ Cardiac Stem Cells Are Mediated by SDF1/CXCR4 and SCF/cKit Signaling Pathways. Circ Res. 2016;119(8):921-930. [4] Iso Y, Spees JL, Serrano C, et al. Multipotent human stromal cells improve cardiac function after myocardial infarction in mice without long-term engraftment. Biochem Biophys Res Commun. 2007;354(3):700-706. [5] de Windt TS, Vonk LA, Slaper-Cortenbach IC, et al. Allogeneic Mesenchymal Stem Cells Stimulate Cartilage Regeneration and Are Safe for Single-Stage Cartilage Repair in Humans upon Mixture with Recycled Autologous Chondrons. Stem Cells. 2017; 35(1):256-264. [6] Lai RC, Arslan F, Lee MM, et al. Exosome secreted by MSC reduces myocardial ischemia/reperfusion injury. Stem Cell Res. 2010;4(3):214-222. [7] Lasso JM, Pérez Cano R, Castro Y, et al. Xenotransplantation of human adipose-derived stem cells in the regeneration of a rabbit peripheral nerve. J Plast Reconstr Aesthet Surg. 2015;68(12): e189-197. [8] Zhang Y, Chopp M, Liu XS, et al. Exosomes Derived from Mesenchymal Stromal Cells Promote Axonal Growth of Cortical Neurons. Mol Neurobiol. 2017;54(4):2659-2673. [9] Zhang Y, Chopp M, Meng Y, et al. Effect of exosomes derived from multipluripotent mesenchymal stromal cells on functional recovery and neurovascular plasticity in rats after traumatic brain injury. J Neurosurg. 2015;122(4):856-867. [10] Zhao H, Shang Q, Pan Z, et al. Exosomes From Adipose-Derived Stem Cells Attenuate Adipose Inflammation and Obesity Through Polarizing M2 Macrophages and Beiging in White Adipose Tissue. Diabetes. 2018;67(2):235-247. [11] Yeo RW, Lai RC, Zhang B, et al. Mesenchymal stem cell: an efficient mass producer of exosomes for drug delivery. Adv Drug Deliv Rev. 2013;65(3):336-341. [12] Kou X, Xu X, Chen C, et al. The Fas/Fap-1/Cav-1 complex regulates IL-1RA secretion in mesenchymal stem cells to accelerate wound healing. Sci Transl Med. 2018;10(432):eaai8524. [13] Cavallini C, Zannini C, Olivi E, et al. Restoring In Vivo-Like Membrane Lipidomics Promotes Exosome Trophic Behavior from Human Placental Mesenchymal Stromal/Stem Cells. Cell Transplant. 2018;27(1):55-69. [14] 张娟,刘峰,张薇,等.人脐血间充质干细胞来源的外泌体:分离鉴定及生物学特性[J].中国组织工程研究, 2014,18(37):5955-5960.[15] Damania A, Jaiman D, Teotia AK, et al. Mesenchymal stromal cell-derived exosome-rich fractionated secretome confers a hepatoprotective effect in liver injury. Stem Cell Res Ther. 2018; 9(1):31. [16] Ferguson SW, Wang J, Lee CJ, et al. The microRNA regulatory landscape of MSC-derived exosomes: a systems view. Sci Rep. 2018;8(1):1419. [17] Shen T, Zheng QQ, Shen J, et al. Effects of Adipose-derived Mesenchymal Stem Cell Exosomes on Corneal Stromal Fibroblast Viability and Extracellular Matrix Synthesis. Chin Med J (Engl). 2018;131(6):704-712. [18] Zhang B, Yeo RWY, Lai RC, et al. Mesenchymal stromal cell exosome-enhanced regulatory T-cell production through an antigen-presenting cell-mediated pathway. Cytotherapy. 2018; 20(5):687-696. [19] Tamura R, Uemoto S, Tabata Y. Immunosuppressive effect of mesenchymal stem cell-derived exosomes on a concanavalin A-induced liver injury model. Inflamm Regen. 2016;36:26. [20] Li X, Xie X, Lian W, et al. Exosomes from adipose-derived stem cells overexpressing Nrf2 accelerate cutaneous wound healing by promoting vascularization in a diabetic foot ulcer rat model. Exp Mol Med. 2018;50(4):29. [21] Bai Y, Han YD, Yan XL, et al. Adipose mesenchymal stem cell-derived exosomes stimulated by hydrogen peroxide enhanced skin flap recovery in ischemia-reperfusion injury. Biochem Biophys Res Commun. 2018;500(2):310-317. [22] Ren G, Wang Y, Yuan S, et al. Dendritic cells loaded with HeLa-derived exosomes simulate an antitumor immune response. Oncol Lett. 2018;15(5):6636-6640. [23] Liao TL, Hsieh SL, Chen YM, et al. Rituximab May Cause Increased Hepatitis C Virus Viremia in Rheumatoid Arthritis Patients Through Declining Exosomal MicroRNA-155. Arthritis Rheumatol. 2018; 70(8):1209-1219. [24] Brown M, Johnson LA, Leone DA, et al. Lymphatic exosomes promote dendritic cell migration along guidance cues. J Cell Biol. 2018;217(6):2205-2221. [25] Karimi N, Cvjetkovic A, Jang SC, et al. Detailed analysis of the plasma extracellular vesicle proteome after separation from lipoproteins. Cell Mol Life Sci. 2018;75(15):2873-2886. [26] 胡国文,李青,牛鑫,等.旋转超滤:一种提取细胞外泌体的新方法[J].第二军医大学学报,2014,35(6):598-602.[27] Zhou Y, Li P, Goodwin AJ, et al. Exosomes from Endothelial Progenitor Cells Improve the Outcome of a Murine Model of Sepsis. Mol Ther. 2018;26(5):1375-1384. [28] Zeming KK, Salafi T, Shikha S, et al. Fluorescent label-free quantitative detection of nano-sized bioparticles using a pillar array. Nat Commun. 2018;9(1):1254. [29] Marczak S, Richards K, Ramshani Z, et al. Simultaneous isolation and preconcentration of exosomes by ion concentration polarization. Electrophoresis. 2018. doi:10.1002/elps. 201700491. [30] Qiao Y, Liang X, Yan Y, et al. Identification of Exosomal miRNAs in Rats With Pulmonary Neutrophilic Inflammation Induced by Zinc Oxide Nanoparticles. Front Physiol. 2018;9:217. [31] Kahn S, Liao Y, Du X, et al. Exosomal MicroRNAs in Milk from Mothers Delivering Preterm Infants Survive in Vitro Digestion and Are Taken Up by Human Intestinal Cells. Mol Nutr Food Res. [32] Li Z, Jiang P, Li J, et al. Tumor-derived exosomal lnc-Sox2ot promotes EMT and stemness by acting as a ceRNA in pancreatic ductal adenocarcinoma. Oncogene. 2018;37(28):3822-3838. [33] Chen CY, Rao SS, Ren L, et al. Exosomal DMBT1 from human urine-derived stem cells facilitates diabetic wound repair by promoting angiogenesis. Theranostics. 2018;8(6):1607-1623. [34] Sokolova V, Ludwig AK, Hornung S, et al. Characterisation of exosomes derived from human cells by nanoparticle tracking analysis and scanning electron microscopy. Colloids Surf B Biointerfaces. 2011;87(1):146-150. [35] Cheng Y, Zeng Q, Han Q, et al. Effect of pH, temperature and freezing-thawing on quantity changes and cellular uptake of exosomes. Protein Cell. 2018. doi: 10. 1007/s13238-018-0529-4. [36] Zhao Y, Chen K, Li H, et al. Effect of pH on the isolation of urinary exosome. Int Urol Nephrol. 2017;49(1):165-169. [37] 薛小燕,郭小华,李敏,等.神经退行性疾病发病机制研究进展[J].中国老年学杂志,2015,35(11):3149-3152.[38] Chang YH, Wu KC, Harn HJ, et al. Exosomes and Stem Cells in Degenerative Disease Diagnosis and Therapy. Cell Transplant. 2018;27(3):349-363. [39] Lo Furno D, Mannino G, Giuffrida R. Functional role of mesenchymal stem cells in the treatment of chronic neurodegenerative diseases. J Cell Physiol. 2018;233(5): 3982-3999. [40] Moayeri A, Nazm Bojnordi M, Haratizadeh S, et al. Transdifferentiation of Human Dental Pulp Stem Cells Into Oligoprogenitor Cells. Basic Clin Neurosci. 2017;8(5):387-394. [41] Palomares T, Cordero M, Bruzos-Cidon C, et al. The Neuroprotective Effect of Conditioned Medium from Human Adipose-Derived Mesenchymal Stem Cells is Impaired by N-acetyl Cysteine Supplementation. Mol Neurobiol. 2018;55(1): 13-25. [42] Marei HES, El-Gamal A, Althani A, et al. Cholinergic and dopaminergic neuronal differentiation of human adipose tissue derived mesenchymal stem cells. J Cell Physiol. 2018;233(2): 936-945. [43] Mortazavi Y, Sheikhsaran F, Khamisipour GK, et al. The Evaluation of Nerve Growth Factor Over Expression on Neural Lineage Specific Genes in Human Mesenchymal Stem Cells. Cell J. 2016;18(2):189-196. [44] Zayed M, Adair S, Ursini T, et al. Concepts and challenges in the use of mesenchymal stem cells as a treatment for cartilage damage in the horse. Res Vet Sci. 2018;118:317-323. [45] Manek R, Moghieb A, Yang Z, et al. Protein Biomarkers and Neuroproteomics Characterization of Microvesicles/Exosomes from Human Cerebrospinal Fluid Following Traumatic Brain Injury. Mol Neurobiol. 2018;55(7):6112-6128. [46] Li JJ, Wang B, Kodali MC, et al. In vivo evidence for the contribution of peripheral circulating inflammatory exosomes to neuroinflammation. J Neuroinflammation. 2018;15(1):8. [47] Phinney DG, Pittenger MF. Concise Review: MSC-Derived Exosomes for Cell-Free Therapy. Stem Cells. 2017;35(4): 851-858. [48] Darkazalli A, Vied C, Badger CD, et al. Human Mesenchymal Stem Cell Treatment Normalizes Cortical Gene Expression after Traumatic Brain Injury. J Neurotrauma. 2017;34(1):204-212. [49] Li Y, Yang YY, Ren JL, et al. Exosomes secreted by stem cells from human exfoliated deciduous teeth contribute to functional recovery after traumatic brain injury by shifting microglia M1/M2 polarization in rats. Stem Cell Res Ther. 2017;8(1):198. [50] Drommelschmidt K, Serdar M, Bendix I, et al. Mesenchymal stem cell-derived extracellular vesicles ameliorate inflammation-induced preterm brain injury. Brain Behav Immun. 2017;60:220-232. [51] Shen H, Yao X, Li H, et al. Role of Exosomes Derived from miR-133b Modified MSCs in an Experimental Rat Model of Intracerebral Hemorrhage. J Mol Neurosci. 2018;64(3):421-430. [52] Xin H, Li Y, Cui Y, et al. Systemic administration of exosomes released from mesenchymal stromal cells promote functional recovery and neurovascular plasticity after stroke in rats. J Cereb Blood Flow Metab. 2013;33(11):1711-1715. [53] Doeppner TR, Herz J, Görgens A, et al. Extracellular Vesicles Improve Post-Stroke Neuroregeneration and Prevent Postischemic Immunosuppression. Stem Cells Transl Med. 2015;4(10):1131-1143. [54] Ophelders DR, Wolfs TG, Jellema RK, et al. Mesenchymal Stromal Cell-Derived Extracellular Vesicles Protect the Fetal Brain After Hypoxia-Ischemia. Stem Cells Transl Med. 2016;5(6): 754-763. [55] Lee M, Ban JJ, Yang S, et al. The exosome of adipose-derived stem cells reduces β-amyloid pathology and apoptosis of neuronal cells derived from the transgenic mouse model of Alzheimer's disease. Brain Res. 2018;1691:87-93. [56] Venugopal C, Shamir C, Senthilkumar S, et al. Dosage and Passage Dependent Neuroprotective Effects of Exosomes Derived from Rat Bone Marrow Mesenchymal Stem Cells: An In Vitro Analysis. Curr Gene Ther. 2017;17(5):379-390. [57] Jarmalavi?iūt? A, Tunaitis V, Pivorait? U, et al. Exosomes from dental pulp stem cells rescue human dopaminergic neurons from 6-hydroxy-dopamine-induced apoptosis. Cytotherapy. 2015;17(7): 932-939. [58] Lee M, Liu T, Im W, et al. Exosomes from adipose-derived stem cells ameliorate phenotype of Huntington's disease in vitro model. Eur J Neurosci. 2016;44(4):2114-2119. [59] Bonafede R, Scambi I, Peroni D, et al. Exosome derived from murine adipose-derived stromal cells: Neuroprotective effect on in vitro model of amyotrophic lateral sclerosis. Exp Cell Res. 2016; 340(1):150-158. [60] Lee M, Ban JJ, Kim KY, et al. Adipose-derived stem cell exosomes alleviate pathology of amyotrophic lateral sclerosis in vitro. Biochem Biophys Res Commun. 2016;479(3):434-439. [61] Lopez-Verrilli MA, Caviedes A, Cabrera A, et al. Mesenchymal stem cell-derived exosomes from different sources selectively promote neuritic outgrowth. Neuroscience. 2016;320:129-139. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [3] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| [4] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [5] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [6] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [7] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [8] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [9] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [10] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [11] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [12] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [13] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [14] | Chen Ziyang, Pu Rui, Deng Shuang, Yuan Lingyan. Regulatory effect of exosomes on exercise-mediated insulin resistance diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 4089-4094. |
| [15] | Gao Kun, Chen Dayu, Zhang Yong, Liu Weidong, Sun Shufen, Lai Wenqiang, Ma Dujun, Wu Yihong, Lin Zhanpeng, Jiang Yinglu, Yu Weiji. Achyranthes bidentata alcohol extract inhibits extracellular matrix degradation of the cartilage by regulating synovial fibroblast exosomes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3636-3640. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||