Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (28): 7447-7455.doi: 10.12307/2026.830
Previous Articles Next Articles
Screening biomarkers for premature ovarian insufficiency based on cellular senescence and endoplasmic reticulum stress with experimental validation
Yan Yuge1, Wang Yanxi1, Qi Xiang2, Cao Shan3, Zou Xiaoyan1, Liu Yujuan2
- 1School of Nursing, 2College of Traditional Chinese Medicine (Zhongjing College), 3School of Medicine, Henan University of Chinese Medicine, Zhengzhou 450046, Henan Province, China
-
Received:2025-10-15Revised:2025-12-27Online:2026-10-08Published:2026-02-26 -
Contact:Wang Yanxi, PhD, Lecturer, School of Nursing, Henan University of Chinese Medicine, Zhengzhou 450046, Henan Province, China -
About author:Yan Yuge, MS candidate, School of Nursing, Henan University of Chinese Medicine, Zhengzhou 450046, Henan Province, China -
Supported by:Henan Provincial Natural Science Foundation, No. 232300421311 (to WYX); Henan Provincial Science and Technology Research Project, No. 252102311251 (to ZXY); Cui Yingmin National Famous Traditional Chinese Medicine Experts Inheritance Studio Construction Project, No. [2022]75 (to CS); Henan Provincial Traditional Chinese Medicine Culture and Management Research Project, No. TCM2025041 (to CS); 2023 Postgraduate Scientific Research Innovation Project of Henan University of Chinese Medicine, No. 2023KYCX054 (to YYG)
CLC Number:
Cite this article
Yan Yuge, Wang Yanxi, Qi Xiang, Cao Shan, Zou Xiaoyan, Liu Yujuan. Screening biomarkers for premature ovarian insufficiency based on cellular senescence and endoplasmic reticulum stress with experimental validation[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7447-7455.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
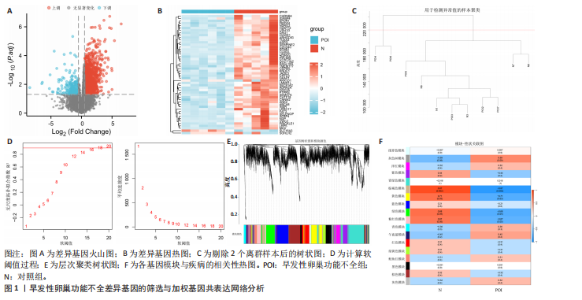
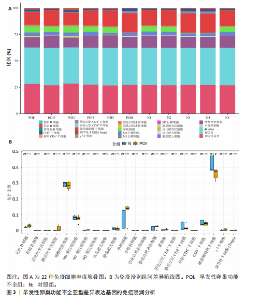
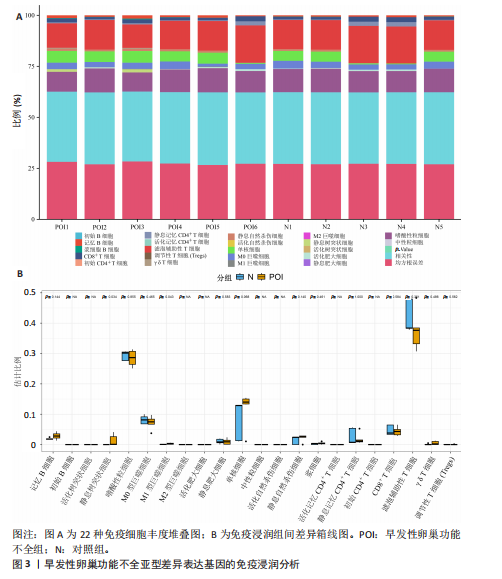
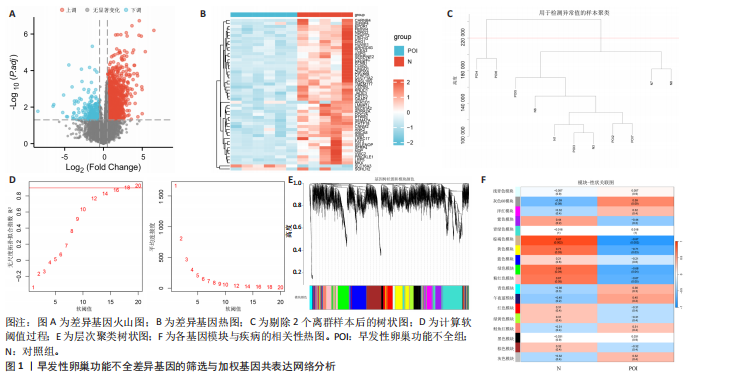
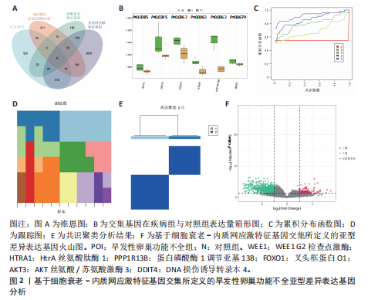
2.1 差异基因的筛选与加权基因共表达网络分析 使用“limma”包分析GSE201276数据集早发性卵巢功能不全疾病组与对照组的RNA-seq数据,共鉴定出1 766个差异基因,其中1 535个表达上调,231个表达下调(图1A,B)。采用加权基因共表达网络分析对GSE201276数据集基因与疾病的相关性开展分析,样本聚类后剔除2个离群样本(图1C)。为满足无尺度网络分布前提条件,将软阈值设置为6,最终鉴定出18个目标模块(图1D,E)。通过构建热图将各模块与早发性卵巢功能不全的相关性进行可视化,结果显示棕褐色模块(r=±0.87,P=0.000 2)和黄色模块(r=±0.71,P=0.03)共572个与疾病相关的关键基因(图1F)。 2.2 共识聚类分析 将模块基因与细胞衰老、内质网应激特征基因及差异基因取交集,共获得6个关键基因(图2A,B),分别为WEE1 G2检查点激酶(WEE1 G2 checkpoint kinase,WEE1)、 HtrA 丝氨酸肽酶1(HtrA serine peptidase 1,HTRA1)、蛋白磷酸酶1调节亚基13B(Protein phosphatase 1 regulatory subunit 13B,PPP1R13B)、叉头框蛋白O1(Forkhead box O1,FOXO1)、AKT丝氨酸/苏氨酸激酶3(AKT serine/threonine kinase 3,AKT3)和DNA损伤诱导转录本4(DNA damage inducible transcript 4,DDIT4)。 基于6个交集基因使用共识聚类算法进行无监督聚类分析,以识别与早发性卵巢功能不全发病相关的亚型差异基因。结合累积分布函数图确定聚类簇k的值为6(图2C),根据此次研究选取k=2作为聚类结果(图2D)。结合共识矩阵的热图发现,k=2时具有清晰明确的分类边界,基于此将数据集中6个疾病组基因分为A、B两个亚型(图2E)。进一步对A、B两个亚型进行差异分析,最终获取911个基于细胞衰老–内质网应激特征基因交集所定义的亚型差异表达基因(图2F)。 2.3 亚型差异表达基因的免疫浸润分析 免疫细胞丰度堆叠图中展示了22种免疫细胞的浸润比例,见图3A。免疫浸润组间差异箱线图结果显示,早发性卵巢功能不全组和对照组之"
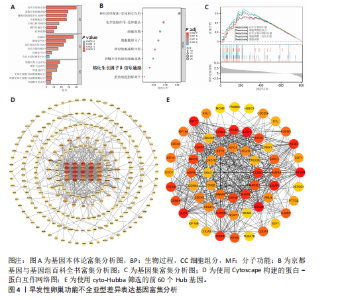
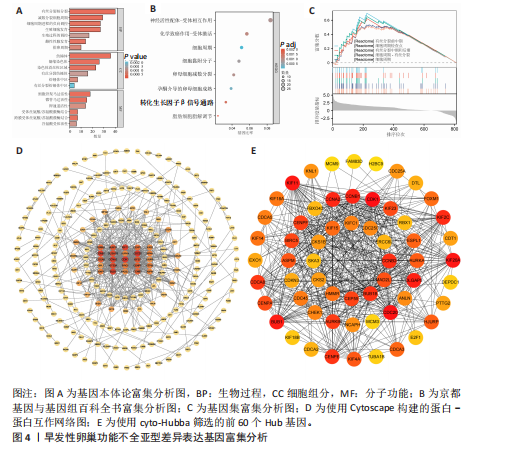
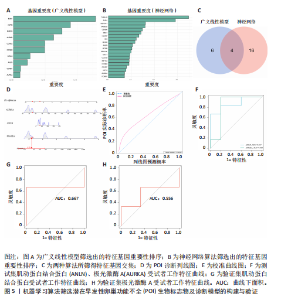
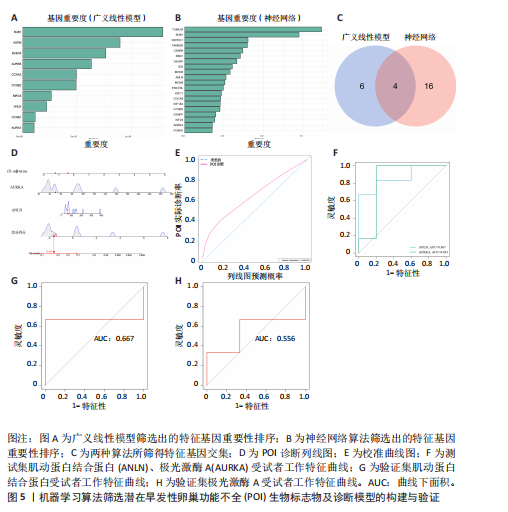
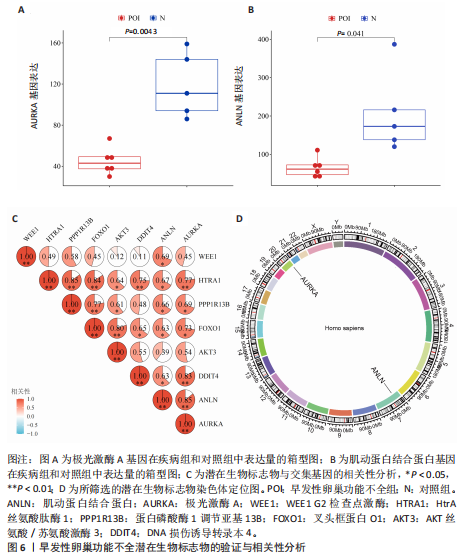
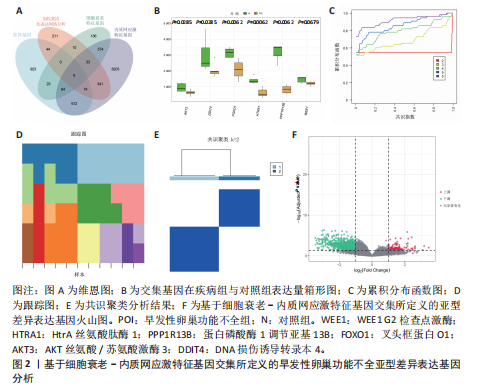
间有2种免疫细胞存在差异,分别为M1型巨噬细胞和静息树突状细胞。与对照组相比,这两种免疫细胞在疾病组浸润水平显著升高(P < 0.05),见图3B,提示这两种免疫细胞参与早发性卵巢功能不全的发病过程。 2.4 亚型差异表达基因富集分析 为分析疾病组与对照组之间的生物学功能差异,对亚型差异表达基因进行基因本体论、京都基因与基因组百科全书富集分析及基因集富集分析。结果显示,早发性卵巢功能不全发病涉及细胞周期负调控、减数分裂、雌性生殖腺发育等生物学过程,与卵母细胞减数分裂、孕激素介导卵母细胞成熟以及转化生长因子β等信号通路相关(图4A,B)。基因集富集分析结果显示,早发性卵巢功能不全发病与细胞有丝分裂、细胞周期相关通路的表达上调有关(图4C)。蛋白-蛋白互作网络分析结果共有879个节点,671条边,形成了一个由细胞衰老特征基因、内质网应激特征基因和早发性卵巢功能不全疾病靶基因构成的交互网络,细胞周期蛋白依赖性激酶1(Cyclin dependent kinase 1,CDK1)、细胞周期蛋白B1(Cyclin B1,CCNB1)和BUB1有丝分裂检查点丝氨酸/苏氨酸激酶(BUB1 mitotic checkpoint serine/threonine kinase,BUB1)等处于网络核心,提示基于细胞衰老-内质网应激可多靶点干预早发性卵巢功能不全(图4D)。应用cyto-hubba插件中最大团中心性算法筛选出得分最高的前60个基因(图4E)。 2.5 机器学习算法筛选潜在生物标志物及诊断模型的构建与验证 采用广义线性模型识别出10个特征基因,神经网络算法筛选出20个特征基因(图5A,B)。将两种机器学习算法的结果取交集得到4个关键基因,分别为细胞周期蛋白B2(Cyclin B2,CCNB2)、肌动蛋白结合蛋白(Anillin actin binding protein,ANLN)、极光激酶A(Aurora kinase A,AURKA)和BUB1有丝分裂检查点丝氨酸/苏氨酸激酶(图5C),以此构建列线图模型,计算早发性卵巢功能不全患者的患病风险概率。结果显示,极光激酶A和肌动蛋白结合蛋白两个基因具有良好的诊断性能(图5D)。校准曲线结果显示,实际患病风险与模型预测风险之间差异较小,说明该模型对早发性卵巢功能不全发病具有较好的预测能力(图5E)。测试集受试者工作特征曲线显示,极光激酶A和肌动蛋白结合蛋白的曲线下面积值均大于0.8(图5F)。验证集受试者工作特征曲线结果显示,肌动蛋白结合蛋白的曲线下面积值为0.667,极光激酶A的曲线下面积值为0.556,进一步证明极光激酶A和肌动蛋白结合蛋白对早发性卵巢功能不全具有较好的诊断价值(图5G,H)。 2.6 潜在生物标志物的验证与相关性分析 研究结果显示,早发性卵巢功能不全潜在生物标志物极光激酶A和肌动蛋白结合蛋白在早发性卵巢功能不全疾病组表达水平降低(P < 0.05)(图6A,B)。为进一步了解2个关键基因与细胞衰老-内质网应激特征基因之间表达的关联情况,此次研究进一步开展了相关性分析。结果显示,极光激酶A与HtrA丝氨酸肽酶1(r=0.77,P < 0.01)、蛋白磷酸酶1调节亚基13B(r=0.69,"
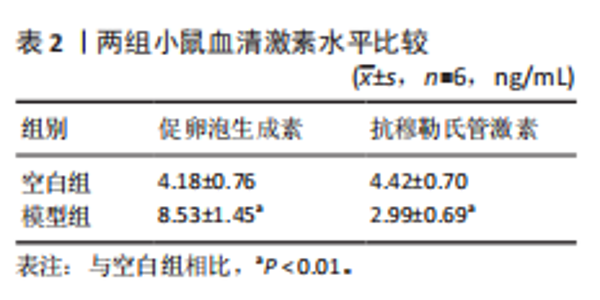
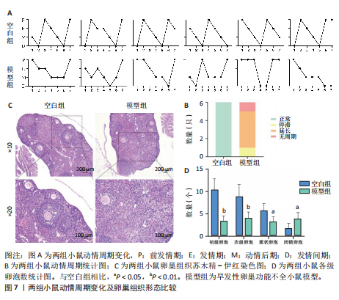
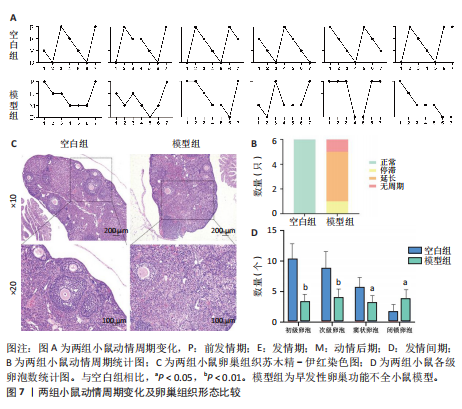
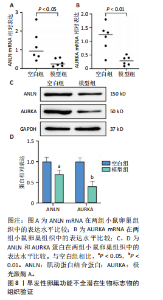
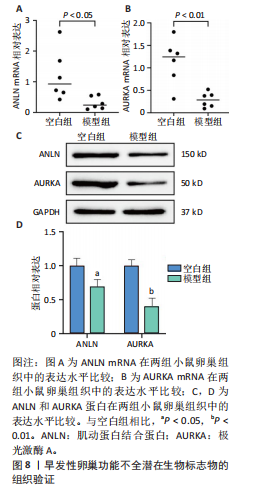
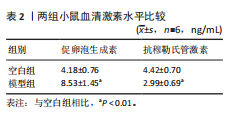
P=0.02)、叉头框蛋白O1(r=0.73,P=0.01)、DNA损伤诱导转录本4(r=0.83,P < 0.01)的表达呈正相关关系,肌动蛋白结合蛋白与WEE1 G2检查点激酶(r=0.69,P=0.02)、HtrA丝氨酸肽酶1(r=0.67,P=0.02)、蛋白磷酸酶1调节亚基13B(r=0.66,P=0.03)、叉头框蛋白O1(r=0.63,P=0.04)、DNA损伤诱导转录本4(r=0.63,P=0.04)的表达呈正相关关系,极光激酶A、肌动蛋白结合蛋白之间的表达水平也呈正相关关系(r=0.85,P < 0.01),见图6C。这些基因整体呈正相关性,表明它们之间可能存在正向相互作用。极光激酶A、肌动蛋白结合蛋白的染色体分布情况见图6D。 2.7 两组小鼠动情周期变化及卵巢组织形态比较 为进一步验证潜在生物标志物的表达情况,此次研究应用环磷酰胺构建早发性卵巢功能不全小鼠模型,用以检测所筛选潜在生物标志物的表达水平。动物实验研究期间,两组动物均无脱失。阴道脱落细胞涂片结果显示,空白组小鼠动情周期以四五天为1个周期,呈现出正常的规律性。而模型组小鼠在腹腔注射4次环磷酰胺后出现动情周期延长、停滞、无周期等紊乱现象,异常率为100%(图7A,B)。小鼠卵巢组织苏木精-伊红染色结果显示,空白组小鼠卵巢组织结构完整,各级卵泡均可见,窦卵泡数较多。与空白组相比,模型组小鼠卵巢萎缩明显,初级卵泡、次级卵泡及窦卵泡数量明显减少,闭锁卵泡数增加,差异有显著性意义(P < 0.05)(图7C,D)。 2.8 两组小鼠血清促卵泡生成素和抗穆勒氏管激素水平比较 应用 ELISA法检测两组小鼠血清性激素水平,模型组小鼠血清促卵泡生成素水平显著高于对照组,差异有显著性意义(P < 0.01);模型组小鼠血清抗穆勒氏管激素水平较空白组降低,差异有显著性意义(P < 0.01)。结合2.7部分实验结果,提示早发性卵巢功能不"
| [1] 卵巢储备功能减退临床诊治专家共识[J]. 生殖医学杂志,2022,31(4):425-434. [2] 程萌, 孔令伶俐, 许良智, 等. 卵巢储备功能减退临床诊治专家共识解读[J]. 实用妇产科杂志, 2022,38(10):743-745. [3] 冯晓玲, 李力, 曲凡, 等. 早发性卵巢功能不全中西医结合诊疗指南[J]. 中医杂志,2022, 63(12):1193-1198. [4] 王士萌, 赵小萱, 张杨, 等. 《早发性卵巢功能不全中西医结合诊疗指南》解读[J]. 中国临床医生杂志,2022,50(8):899-903. [5] 曾丽华, 张莹轩, 梁蕴仪, 等. 基于UPLC-Q-TOF-MS的左归丸干预早发性卵巢功能不全大鼠的血清代谢组学研究[J]. 中药新药与临床药理,2022,33(6):762-769. [6] 陈瑶, 唐丽, 田赛男, 等. 文献计量视域下中医药防治卵巢衰老研究现状与热点分析[J]. 中草药,2025,56(12):4369-4386. [7] 许庆龙, 姚顺发, 张君正, 等. 哺乳动物卵母细胞老化的相关机制研究进展[J]. 中国畜牧杂志, 2024, 60(6): 60-66. [8] ZHU J, ZHANG J, LI H, et al. Cumulus cells accelerate oocyte aging by releasing soluble Fas Ligand in mice. Sci Rep. 2015;5(1):1-8. [9] 张月婷, 李静林, 傅振燚, 等. 内质网应激促进铁死亡加重脑缺血再灌注损伤[J]. 中国组织工程研究,2026,30(11):2806-2813. [10] 尤佳, 孟冰瑶, 梁枭婷, 等. 黑色素瘤中SIRT7和自噬相关蛋白的表达相关性及其对放疗抵抗的影响[J]. 临床与实验病理学杂志,2022, 38(12):1462-1468. [11] 韩龙传, 李悦, 邹智慧, 等. 磷脂酰乙醇胺引起内质网应激促进巨噬细胞衰老及肝损伤[J]. 上海交通大学学报(医学版),2025,45(6):693-704. [12] UDDIN MS, TEWARI D, SHARMA G, et al. Molecular Mechanisms of ER Stress and UPR in the Pathogenesis of Alzheimer’s Disease. Mol Neurobiol. 2020;57(7):2902-2919. [13] 姜李乐. Sigma-1受体对卵巢储备低下患者颗粒细胞内质网应激凋亡通路的调控研究[D]. 郑州: 郑州大学,2019. [14] SONIGO C, BEAU I, GRYNBERG M, et al. AMH prevents primordial ovarian follicle loss and fertility alteration in cyclophosphamide‐treated mice. FASEB J. 2019;33(1):1278-1287. [15] 陆星星, 任豆豆, 徐华洲, 等.资癸益冲方对卵巢储备功能下降模型大鼠卵巢氧化损伤的影响及其作用机制[J].北京中医药大学学报,2020, 41(7):561-568. [16] 胡恒, 储继军, 李哲, 等. 基于TLR4/MyD88/NF-κB信号通路探讨益经汤改善卵巢储备功能减退大鼠卵巢炎症反应的作用机制[J]. 中国实验方剂学杂志,2025,31(11):20-30. [17] 阮祥燕. 医源性早发性卵巢功能不全临床治疗与管理指南(2025年版)[J]. 中国实用妇科与产科杂志,2025,41(1):76-87. [18] 谭容容, 吴洁. 早发性卵巢功能不全的遗传学病因[J]. 中国实用妇科与产科杂志,2023,39(9): 872-877. [19] 田赛男, 唐丽, 王圣羽, 等. 早发性卵巢功能不全在中医药的发展研究[J]. 中国中药杂志, 2025,50(11):2969-2981. [20] ROGER L, TOMAS F, GIRE V. Mechanisms and Regulation of Cellular Senescence. Int J Mol Sci. 2021;22(23):2969-2981. [21] 史薇, 刘敏, 王智超, 等. 新加苁蓉菟丝子汤经SIRT1/PGC-1α信号通路促进线粒体生物合成以修复卵巢颗粒细胞损伤的研究[J]. 世界科学技术-中医药现代化,2024,26(5):1269-1278. [22] YANG Y, TANG X, YAO T, et al. Metformin protects ovarian granulosa cells in chemotherapy-induced premature ovarian failure mice through AMPK/PPAR-γ/SIRT1 pathway. Sci Rep. 2024;14(1):1-16. [23] DENG J, TANG Y, LI L, et al. miR-143-3p Promotes Ovarian Granulosa Cell Senescence and Inhibits Estradiol Synthesis by Targeting UBE2E3 and LHCGR. Int J Mol Sci. 2023;24(16):1-17. [24] KOLOKO NGASSIE ML, BRANDSMA CA, GOSENS R, et al. The Stress of Lung Aging: Endoplasmic Reticulum and Senescence Tête-à-Tête. Physiology (Bethesda). 2021;36(3):150-159. [25] ZHANG CY, ZHONG WJ, LIU YB, et al. EETs alleviate alveolar epithelial cell senescence by inhibiting endoplasmic reticulum stress through the Trim25/Keap1/Nrf2 axis. Redox Biol. 2023;63:1-17. [26] 刘小虎, 赵志慧, 周玥, 等. PI3K/Akt/mTOR自噬通路在人参皂苷Rg1延缓D-gal诱导的卵巢早衰小鼠模型卵巢早衰中的作用[J]. 中国中药杂志,2020,45(24):6036-6042. [27] 苏娜, 蔡平平, 严如根, 等. 内质网应激—凋亡途径与卵巢衰老关系研究进展[J]. 山东医药, 2022,62(26):94-97. [28] ZHANG FL, FENG YQ, WANG JY, et al. Single cell epigenomic and transcriptomic analysis uncovers potential transcription factors regulating mitotic/meiotic switch. Cell Death Dis. 2023;14(2):1-12. [29] LIU W, CHEN C, GAO Y, et al. Transcriptome Dynamics and Cell Dialogs Between Oocytes and Granulosa Cells in Mouse Follicle Development. Genomics Proteomics Bioinformatics. 2024;22(2): 1-14. [30] ZHANG C, YU D, MEI Y, et al. Single-cell RNA sequencing of peripheral blood reveals immune cell dysfunction in premature ovarian insufficiency. Front Endocrinol (Lausanne). 2023;14(3):249-260. [31] 张天驰, 李沐哲, 牛园园, 等. 温肾通络止痛方抑制巨噬细胞衰老改善BMSC成骨分化和老年性骨质疏松模型小鼠骨丢失的研究[J]. 南京中医药大学学报,2024,40(3):249-260. [32] 魏守瑞, 张宁. 肠道菌群与生殖衰老的相关性[J]. 生命的化学,2025,45(4):698-707. [33] 胡嘉嘉, 黎业娟, 赵立强, 等. RGS1在不同反应性卵巢颗粒细胞中的表达及影响[J]. 系统医学,2024,9(14):15-18. [34] 孙梦迪, 孙志运, 王克婧, 等. 中药复方调控肺-肠轴治疗呼吸系统疾病的研究进展[J]. 药物评价研究,2025,48(9):2689-2702. [35] MANOURY B, MAISONNEUVE L, PODSYPANINA K. The role of endoplasmic reticulum stress in the MHC class I antigen presentation pathway of dendritic cells. Mol Immunol. 2022;144:44-48. [36] 王美娇. AuroraA/Bora,PLK1在小鼠受精卵有丝分裂中功能的研究[D]. 沈阳: 中国医科大学, 2018. [37] LIU Z, WU B, LIU X, et al. CD73/NT5E-mediated ubiquitination of AURKA regulates alcohol-related liver fibrosis via modulating hepatic stellate cell senescence. Int J Biol Sci. 2023;19(3):950-966. [38] ZHOU X, WU Y, QIN L, et al. Investigation of differentially expressed genes related to cellular senescence between high-risk and non-high-risk groups in neuroblastoma. Front Cell Dev Biol. 2024;12:1-9. [39] HUCK JJ, ZHANG M, MCDONALD A, et al. MLN8054, an inhibitor of Aurora A kinase, induces senescence in human tumor cells both in vitro and in vivo. Mol Cancer Res. 2010;8(3):373-384. [40] SHEN Z, YIN L, ZHOU H, et al. Combined inhibition of AURKA and HSF1 suppresses proliferation and promotes apoptosis in hepatocellular carcinoma by activating endoplasmic reticulum stress. Cell Oncol (Dordr). 2021;44(5):1035-1049. [41] 刘凤, 姚波, 莫晓龙, 等. Aurora激酶A调控卵母细胞减数分裂的分子机制[J]. 中国医学科学院学报,2022,44(1):142-148. [42] XU W, LIU X, HAN W, et al. Inhibiting HIF-1 signaling alleviates HTRA1-induced RPE senescence in retinal degeneration. Cell Commun Signal. 2023;21(1):1-17. [43] YOSHIDA K, KUSAMA K, AZUMI M, et al. Endoplasmic reticulum stress-regulated high temperature requirement A1 (HTRA1) modulates invasion and angiogenesis-related genes in human trophoblasts. J Pharmacol Sci. 2022;150(4):267-274. [44] YANG W, KIM DM, JIANG W, et al. Suppression of FOXO1 attenuates inflamm-aging and improves liver function during aging. Aging Cell. 2023; 22(10):1-21. [45] 王青青, 焦运, 吴欣妍, 等. FoxO1经ATF6调控Hcy诱导的肝细胞凋亡[J]. 中国药理学通报, 2021,37(2):203-209. [46] 刘东洋, 张楠, 李海玲, 等. ANLN在肾透明细胞癌中的表达及其与预后和免疫浸润相关性的生物信息学分析[J]. 中国免疫学杂志,2024, 40(8):1726-1734. [47] 欧阳晨雨, 代月黎, 方向军, 等. ANLN在肝内胆管细胞癌和肝细胞肝癌中的差异表达及其在二者诊断中的附加价值[J]. 临床放射学杂志, 2021,40(6):1145-1150. [48] MAGNUSSON K, GREMEL G, RYDÉN L, et al. ANLN is a prognostic biomarker independent of Ki-67 and essential for cell cycle progression in primary breast cancer. BMC Cancer. 2016;16(1):1-11. [49] ZENG S, YU X, MA C, et al. Transcriptome sequencing identifies ANLN as a promising prognostic biomarker in bladder urothelial carcinoma. Sci Rep. 2017;7(1):1-11. [50] LI H, LI G, GAO X, et al. Development of a reliable risk prognostic model for lung adenocarcinoma based on the genes related to endotheliocyte senescence. Sci Rep. 2025;15(1):1-16. [51] HALL G, LANE BM, KHAN K, et al. The Human FSGS-Causing ANLN R431C Mutation Induces Dysregulated PI3K/AKT/mTOR/Rac1 Signaling in Podocytes. J Am Soc Nephrol. 2018;29(8):2110-2122. |
| [1] | Lai Jiaming, , Song Yuling, Chen Zixi, Wei Jinghuan, Cai Hao, , Li Guoquan, . Screening of diagnostic markers for endothelial cell Senescence in mice with radiation-induced heart disease and analysis of immune infiltration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1450-1463. |
| [2] | Zhang Haiwen, Zhang Xian, Xu Taichuan, Li Chao. Bibliometric and visual analysis of the research status and trends of senescence in osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1580-1591. |
| [3] | Lyu Guoqing, Aizimaitijiang·Rouzi, Xiong Daohai. Irisin inhibits ferroptosis in human articular chondrocytes: roles and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1359-1367. |
| [4] | Liu Kexin, , Hao Kaimin, Zhuang Wenyue, , Li Zhengyi. Autophagy-related gene expression in pulmonary fibrosis models: bioinformatic analysis and experimental validation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1129-1138. |
| [5] | Wei Xinyi, Zheng Yan, Chen Qian, Ren Jiajia, Li Jian. Molecular dynamic characteristics of rat gastrocnemius muscle under acute and short-term exercise intervention during the subacute phase of spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6196-6206. |
| [6] | Kan Weiyi, Wang Linrong, Cheng Leping. The interaction and balance between cellular senescence and tissue repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6297-6305. |
| [7] | Zhou Wen, Yang Hongwei. Molecular mechanism and natural drug screening for ferroptosis-targeted therapy in rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6051-6061. |
| [8] | Liao Guibin, Wu Yixuan, Tang Jing, Huang Jinke, Wang Jun, Yan Ziqi, Liu Shujun, Zhang Haiyan. Shared genetic basis and causal relationship between nutrition, nutritional status and inflammatory bowel disease [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5876-5885. |
| [9] | Hu Xiaoyong, Song Qianhua, Yang Zhaoying, Tang Rui, Li Hongjian. Potential mechanism by which iroquois homeobox 3 regulates the browning of perivascular adipose tissue in vascular injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5671-5681. |
| [10] | Tang Cen, Hu Wanqin. Establishing a diagnostic model for recurrent spontaneous abortion based on the levels of autophagy-related genes in the endometrium [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5728-5738. |
| [11] | Wang Hengxin, Li Hongkun, Xu Nuo, Li Anping, Wang Xinjing, Zhang Tong. Quercetin promotes osteogenic differentiation of senescent jaw bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 4843-4852. |
| [12] | Li Wenhui, Fan Weijing, Liu Guobin. Impact of Zi-Zhu ointment on the miRNA expression profile in mouse models of diabetic ulcers: a high-throughput sequencing analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(17): 4337-4346. |
| [13] | Lu Liwei, Huang Keqi, Chen Yueping, Zhuo Yinghong, Zhu Naihui, Wei Peng. Bioinformatics-based analysis of shared genes and associations in immune mechanisms between rheumatoid arthritis and Crohn’s disease [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(16): 4253-4264. |
| [14] | Zhou Man, Long Meiting, Xin Guoyan, Huang Mengjun, Yao Zhenglian, Zhao Huajuan, Shen Linqiang, Wu Xijun, Yang Xiaoyan. Bioinformatics screening and experimental verification of core genes in chronic myeloid leukemia and imatinib resistance [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3331-3342. |
| [15] | Yang Yuxi, Xu Dan, Liu Zhongshan. Analysis of strategies to improve efficiency of in vitro culture of mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3359-3369. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||