Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (24): 6297-6305.doi: 10.12307/2026.181
Previous Articles Next Articles
The interaction and balance between cellular senescence and tissue repair
Kan Weiyi1, Wang Linrong1, 2, 3, Cheng Leping1, 2, 3
- 1Collaborative Innovation Centre of Regenerative Medicine and Medical BioResource Development and Application Co-constructed by the Province and Ministry, Guangxi Key Laboratory of Regenerative Medicine, Nanning 530021, Guangxi Zhuang Autonomous Region, China; 2Institute of Neuroscience, Basic Medical College, Guangxi Medical University; Guangxi Key Laboratory of Brain Sciences, Key Laboratory of Basic Research on Brain Function and Brain Diseases (Guangxi Medical University), Health Commission of Guangxi Zhuang Autonomous Region, Nanning 530021, Guangxi Zhuang Autonomous Region, China; 3Translational Medicine Research Center, Guangxi Medical University; Key Laboratory of Longevity and Aging-related Diseases, Ministry of Education, Nanning 530021, Guangxi Zhuang Autonomous Region, China
-
Received:2025-06-06Revised:2025-07-28Online:2026-08-28Published:2026-02-03 -
Contact:Cheng Leping, PhD, Professor, Master’s and Doctoral supervisor, Collaborative Innovation Centre of Regenerative Medicine and Medical BioResource Development and Application Co-constructed by the Province and Ministry, Guangxi Key Laboratory of Regenerative Medicine, Nanning 530021, Guangxi Zhuang Autonomous Region, China; Institute of Neuroscience, Basic Medical College, Guangxi Medical University; Guangxi Key Laboratory of Brain Sciences, Key Laboratory of Basic Research on Brain Function and Brain Diseases (Guangxi Medical University), Health Commission of Guangxi Zhuang Autonomous Region, Nanning 530021, Guangxi Zhuang Autonomous Region, China; Translational Medicine Research Center, Guangxi Medical University; Key Laboratory of Longevity and Aging-related Diseases, Ministry of Education, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
About author:Kan Weiyi, MS candidate, Collaborative Innovation Centre of Regenerative Medicine and Medical BioResource Development and Application Co-constructed by the Province and Ministry, Guangxi Key Laboratory of Regenerative Medicine, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
Supported by:National Natural Science Foundation of China, No. 32070976 (to CLP); Guangxi Science and Technology Base and Talent Special Project, No. AD21075052 (to CLP)
CLC Number:
Cite this article
Kan Weiyi, Wang Linrong, Cheng Leping. The interaction and balance between cellular senescence and tissue repair[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6297-6305.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
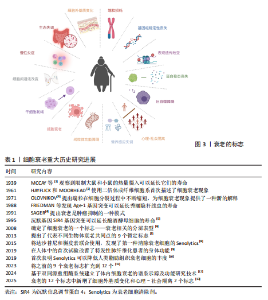
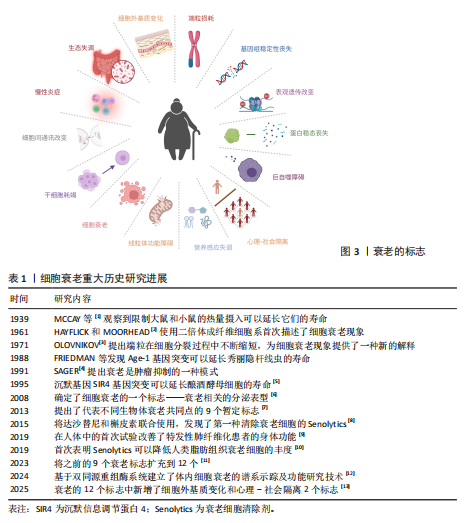
2.1 细胞衰老 衰老被认为是一种应激反应,由许多不同的内在和外在损伤因素引起,其主要特征性标志包括基因组稳定性丧失、端粒消耗、表观遗传改变、蛋白质稳态丧失、巨自噬障碍、营养感应失调、线粒体功能障碍、细胞衰老、干细胞耗竭、细胞间通讯改变、慢性炎症、生态失调、细胞外基质变化和心理-社会隔离,见图3。其中,细胞衰老被认为是衰老和衰老相关疾病的关键驱动因素。细胞衰老研究的重大历史进展见表1[1-13]。衰老细胞的特征是细胞永久性退出细胞周期,失去增殖能力,即使在有丝分裂刺激的情况下也无法增殖[14]。1961年,HAYFLICK和MOORHEAD使用二倍体成纤维细胞系首次描述了细胞衰老现象,该细胞系在停滞生长之前可以达到最大细胞分裂次数[2]。这一发现提出了一个假说,即组织衰老是由细胞逐渐丧失增殖能力并转化为衰老细胞所驱动。衰老细胞持续积累进一步加剧了组织功能的衰退,从而促进机体衰老及相关功能障碍[15]。 细胞衰老是一种应激反应,由多种应激源诱导,包括癌基因激活、氧化和基因毒性应激、线粒体功能障碍、DNA损伤及端粒缩短与损伤等,与肿瘤抑制、组织修复、胚胎发育和机体衰老等过程密切相关[16]。核DNA损伤通常为衰老的潜在原因,主要以DNA双链断裂的形式存在,这些损伤会激活DNA损伤反应通路。DNA损伤反应因子在DNA损伤位点积累,形成焦点以标记单个DNA损伤位点,并在损伤修复前通过检查点停滞细胞周期。如果DNA损伤持续存在,会导致DNA损伤反应信号延长, DNA损伤反应信号激酶进一步激活肿瘤抑制蛋白p53,并刺激细胞周期依赖性激酶抑制剂p21的表达,最终以细胞衰老的形式引发持续的增殖停滞。端粒酶的缺失或端粒间的重组会引起端粒缩短,此时形成的DNA双链断裂是"
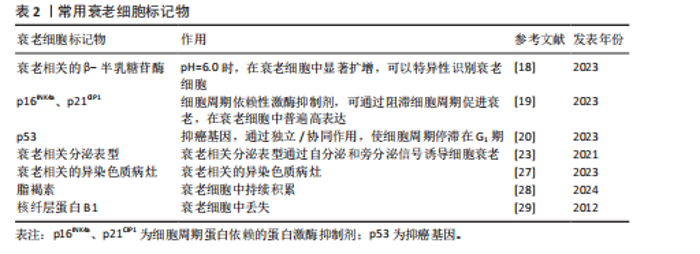
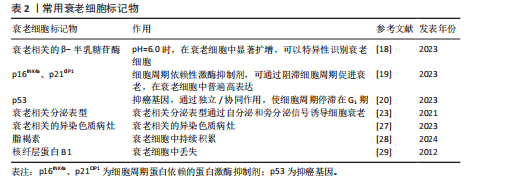
“单端”断裂(仅1条染色体末端断裂),这种断裂无法通过常规修复机制(如同源重组)有效修复从而触发DNA损伤反应信号,诱导细胞衰老。肿瘤细胞中活性氧积累,激活癌基因并触发一个初始的过度增殖阶段,引起DNA复制应激,在易碎位点处发生DNA损伤,通过激活DNA损伤反应通路导致衰老。线粒体去乙酰化酶(sirtuins)(一组在不同物种中调控衰老的进化保守蛋白)的耗竭以及线粒体功能的选择性化学抑制会触发衰老[17]。 衰老相关的β-半乳糖苷酶活性是最早被描述的衰老标志物之一,用于证明多种哺乳动物中与衰老相关的患病部位及老年组织中存在衰老细胞积累。衰老细胞的一个显著特征是细胞周期依赖性激酶抑制剂的蛋白表达增加。其中,p16INK4A可持久维持细胞增殖停滞状态,是衰老细胞积累过程中最重要的抑制剂。在pH值为6.0时,溶酶体含量增加,在衰老细胞中显著扩增,此时对β-半乳糖苷酶活性进行组织化学检测,可以特异性识别衰老细胞[18]。细胞衰老主要激活p16INK4a/Rb和p53/p21CIP1肿瘤抑制通路[19],其中p16INK4a和p21CIP1是细胞周期依赖性激酶抑制剂,可通过阻滞细胞周期促进衰老,在衰老细胞中普遍高表达。p53是抑癌基因,它们通过独立/协同作用,使细胞周期停滞在G1期[20]。如果触发应激是短暂的,p53诱导细胞周期停滞(静止状态)并激活DNA修复过程。待应激解决后,细胞周期可以恢复循环[21],而持续应激或其他信号可激活p16INK4a(细胞周期依赖性激酶4和细胞周期依赖性激酶6的抑制剂),导致细胞周期长期停滞[22]。在衰老细胞中,DNA损伤反应和p38丝裂原活化蛋白激酶信号通路持续激活,转录因子GATA结合蛋白4通过诱导白细胞介素1α表达,激活核因子κB依赖性炎症基因,两者协同转录激活衰老相关分泌表型因子的表达,其中包括多种生物活性因子,例如炎性细胞因子、趋化因子、生长因子、基质金属蛋白酶、脂质、核苷酸、细胞外囊泡和可溶性因子等[23]。组蛋白乙酰化阅读器溴结构域蛋白4 (bromodomain-containing protein 4,BRD4)被募集到与衰老相关分泌表型基因相邻的新激活的超级增强子,环磷酸鸟苷-磷酸腺苷酸合成酶-干扰素基因刺激物通路的异常激活、炎症小体也参与衰老相关分泌表型基因的激活,从而引起慢性炎症[24]。衰老相关分泌表型通过自分泌诱导细胞内功能障碍,还可以通过旁分泌信号诱导非恶性增殖的邻近细胞经历衰老[25]。衰老细胞具有显著的形态学特征,如细胞呈现较大、扁平形态,使它与正常细胞区别开来[26],并表现出衰老相关的异染色质病灶(senescence-associated heterochromatin foci,SAHF)形成[27]、脂褐素积累[28]、DNA 损伤病灶[29]、核纤层蛋白B1丢失[30]、衰老相关的卫星膨胀(senescence-associated distension of satellites,SADSs)[31]、人类分化型胚胎软骨细胞表达基因1 (differentiated embryonic chondrocyte gene1,DEC1)和诱骗受体2(decoy receptor 2,DCR2)的表达[32]。综上所述,衰老特征、诱因、形成机制、形态学等多方面都能够定义并应用成为衰老细胞的标志物[33]。目前,衰老细胞的鉴定不能单独使用一个标记物,而是依赖于多个标记的组合[34],具体信息见表2。 2.2 细胞衰老在组织损伤修复方面的作用 组织损伤修复是一个比较复杂的生理过程,它包含细胞损伤、炎症反应、修复以及再生等众多环节。一般而言,可以把它分成4个阶段:急性损伤阶段、炎症反应阶段、修复阶段以及重塑阶段,随着这些阶段依次推进,就能够有效地对组织损伤加以修复。各个阶段中细胞的各种活动及其相互之间的联系,对于最后的修复效果均具有非常关键的意义。 2.2.1 细胞衰老在组织损伤修复方面的积极作用 (1)组织再生与伤口愈合:有研究指出在急性伤口愈合过程中,短暂的细胞衰老可以促进组织重塑并帮助恢复正常的皮肤结构和功能[35]。 衰老细胞释放的衰老相关分泌表型中的细胞因子和趋化因子如白细胞介素6、白细胞介素8来招募免疫细胞如巨噬细胞,衰老的成纤维细胞通过分泌血小板衍生生长因子A(platelet-derived growth factor,PDGF-A)促进细胞外基质重塑来促进急性伤口愈合[36]。 (2)胚胎发育与组织形成:细胞衰老作为一种防御机制清除胚胎发育过程中的异常受损细胞后,引导组织进行重建过程,促进组织恢复正常功能和再生[37]。有研究表明小鼠胚胎干细胞中p53程序的激活能够通过促进分化相关基因的转录并且抑制多能性基因的表达来调控哺乳动物的胚胎发育和组织形成过程[15]。衰老细胞分泌的衰老相关分泌表型可以通过自分泌或旁分泌等形式促进邻近细胞的增殖、迁移和分化,从而推动损伤组织的修复和再生[38]。 (3)肿瘤抑制:在组织损伤发生时,细胞可能因受到损伤刺激而出现DNA损伤等异常情况。此时细胞衰老作为一种自我保护机制,抑制受损细胞进一步增殖,从而限制异常细胞的积累以及避免突变积累导致的癌变,预防肿瘤的发生,进而为受损组织的修复提供一个相对安全的细胞环境[39]。 (4)免疫调控与炎症:衰老细胞通过分泌衰老相关分泌表型,募集单核细胞和巨噬细胞,形成促炎微环境[40]。巨噬细胞通过清除凋亡细胞触发自身向抗炎表型(M2)转化,M2型巨噬细胞在炎症消退阶段负责吞噬细胞碎片[41],并分泌促修复因子(如促血管生成因子)促进血管生成和细胞外基质重塑[42],进而促进组织修复。研究发现,消除p16INK4a阳性的内皮细胞会加重肝损伤,表明衰老的内皮细胞可能在促进血管生成、调节免疫反应以及促进组织修复方面起到重要作用[12]。 (5)细胞重编程:在小鼠骨骼肌损伤模型中,短暂诱导的衰老细胞通过分泌衰老相关分泌表型因子和表皮生长因子受体(epidermal growth factor receptor,EGFR)的配体双调蛋白(Amphiregulin,AREG)加速邻近干细胞的增殖和间充质-上皮转化,显著增强体内重编程效率,并减少供者年龄对体外重编程的负面影响[43]。 研究表明,在水螅中,截肢损伤诱导少量局部细胞转变为衰老细胞,分泌衰老相关分泌表型破坏相邻体细胞的差异状态,将它们重编程为能够驱动全身组织再生的干细胞[44]。同样,在蝾螈中,衰老细胞衍生的分泌因子激活成纤维细胞生长因子-细胞外信号调节激酶信号轴,促进蝾螈肌管细胞再次进入细胞周期并且逆转肌肉身份,促进肌肉组织的去分化和产生肌肉再生祖细胞,增强肢体的组织再生能力[45]。 (6)干细胞功能调节:研究表明,肺上皮干细胞附近的基底膜中的p16INK4a阳性成纤维细胞具有监测屏障完整性的能力,可以感知损伤过程中浸润免疫细胞的存在,并通过快速激活核因子κB信号通路,增加分泌衰老相关分泌表型来激活干细胞修复,促进上皮再生[46]。有研究提出衰老细胞能够通过增强小鼠体内邻近干细胞的活性以刺激组织再生,从而促进毛发生长[47]。 2.2.2 细胞衰老在组织损伤修复方面的不利作用 (1)组织损伤后难以自我修复:研究表明,皮肤组织在受到紫外线辐射或化学物质刺激时,会导致DNA损伤,进而激活DNA损伤反应信号通路(p53信号通路),导致p21CIP1表达增加,最终使细胞周期停滞在G1期,进入衰老状态[48-49]。衰老细胞失去增殖能力后无法提供足够的新生细胞支持组织损伤后的修复和再生,导致组织的修复和再生能力下降[50-51]。研究人员利用年轻和老年小鼠分别构建了8种组织损伤模型,经过检测发现衰老组织中的再生相关免疫细胞(如巨噬细胞)被抑制,导致损伤组织的再生修复能力显著下降[52]。研究表明,免疫细胞衰老可能导致免疫系统功能受损,从而导致衰老细胞的清除受损[53]。 随着衰老细胞在组织中的积累,这种负面影响会逐渐加剧,使得组织损伤后难以自我修复,导致患慢性疾病的风险增加[54],包括糖尿病、癌症、骨关节炎和阿尔茨海默病等[55-57]。 (2)形成炎性微环境:衰老细胞分泌大量衰老相关分泌表型改变组织微环境,抑制组织损伤的正常修复过程[50,58],进而引发慢性炎症[59]。这种慢性炎症会破坏组织的稳态平衡,加剧周围健康细胞的二次衰老过程[60]。在高脂饮食诱导的非酒精性脂肪性肝病小鼠模型中,肝脏内皮细胞分泌的衰老相关分泌表型显著增加,通过分泌趋化因子配体10 (C-X-C motif chemokine ligand 10,CXCL10)等因子,促进M1型巨噬细胞极化增加,分泌大量促炎因子和趋化因子,同时诱导自身及邻近巨噬细胞的衰老[61]。 (3)驱使组织纤维化与器官功能障碍:研究表明,慢性伤口中衰老细胞的积累可能会导致伤口愈合延迟,进而导致慢性炎症和组织纤维化等不良后果[62]。在肺组织损伤后,衰老细胞会分泌衰老相关分泌表型,如转化生长因子β(transforming growth factor-β,TGF-β),"
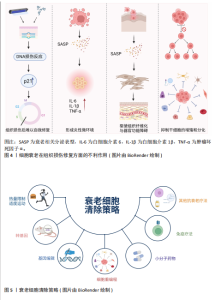
这些因子会促进成纤维细胞的活化和增殖,使细胞外基质过度沉积,导致肺纤维化[63]。类似的机制也见于肾脏等器官的慢性损伤修复过程中[16]。还有研究表明,脊髓损伤后反应性星形胶质细胞增生,随后成为星形胶质细胞,最终导致病变部位周围形成神经胶质细胞瘢痕,限制轴突再生和运动/感觉功能恢复[64]。 (4)抑制干细胞的增殖和分化:随着年龄的增长,干细胞数量耗竭和功能障碍也会随之增加,从而导致多个组织和系统的正常功能受到阻碍[65]。同时,衰老细胞可能抑制干细胞的增殖和分化能力,减少干细胞的数量和活性,进而限制干细胞在组织损伤修复中的关键作用,降低组织的修复效率和再生能力[66-67]。 综上所述,衰老细胞在组织修复中呈现双向调控作用,在急性损伤早期,其通过分泌衰老相关分泌表型因子募集免疫细胞并促进细胞外基质重塑,促进损伤修复;而在慢性或持续性损伤中,衰老细胞的长期滞留导致促纤维化表型及慢性炎症,最终加剧组织功能障碍,见图4。这一动态平衡受微环境信号及衰老时相的精确调控,因此靶向清除衰老细胞是令人信服的延长健康寿命的方法[68]。 2.3 衰老细胞清除策略 随着年龄的增长,机体的免疫功能逐渐失衡,衰老细胞不断积累。少量的衰老细胞会引起慢性无菌性炎症,打破组织的稳态,降低机体应对压力的恢复能力,最终发展成为炎性衰老,因此减少衰老细胞负担,对于延长健康寿命、减少年龄相关疾病的发生具有治疗作用。目前,针对衰老的干预方法主要分为非靶向和靶向两类,见图5。 2.3.1 非靶向干预 非靶向干预方式与人的生活密切相关,热量限制饮食和适度运动等饮食习惯和锻炼方式对人体代谢具有一定的调节作用[69-71],可以在一定程度上延缓衰老。同时,研究发现将年轻小鼠和老年小鼠交换血液或“异体共生”[72-73],可以恢复老年小鼠肌肉组织和肝脏的再生能力,并且可以将老年小鼠的血浆稀释[74],从而延缓老年小鼠肌肉组织的衰减和减轻老年小鼠肝脏的纤维化程度。尽管热量限制和其他抗衰老方法取得了重大进展,但它们的临床应用仍面临许多挑战。 2.3.2 靶向干预 (1)转基因技术介导的选择性清除:INK-ATTAC(Inducible Knockout-AP20187-Mediated Targeted Apoptosis through Caspase activation)是一种用于研究衰老和年龄相关疾病的转基因小鼠模型。INK-ATTAC转基因小鼠模型通过特异性清除衰老细胞来研究这些细胞在衰老和疾病中的作用。当给予小鼠特定的药物(AP20187)时,药物会与FK506结合蛋白(FK506 binding protein,FKBP)结合,诱导天冬氨酸特异性的半胱氨酸蛋白水解酶8(cysteinyl aspartate specific proteinas-8,caspase-8)激活,从而导致细胞发生凋亡,进而选择性地清除表达p16INK4a的细胞,而不影响其他正常细胞。因此,INK-ATTAC转基因小鼠模型通过特异性清除p16INK4a阳性衰老细胞为研究细胞衰老在多种疾病中的作用提供了强大的实验基础。研究表明,在脂肪组织、骨骼肌和眼睛等组织中,特异性清除表达p16INK4a的细胞可延迟衰老表型的发生。p16-3MR小鼠模型也是靶向清除p16高表达细胞以研究衰老常用的转基因模型[55]。此外,有研究人员采用p21-Cre 转基因小鼠模型与条件性细胞消融工具鼠杂交,每月少量清除p21CIP1高表达的衰老细胞,显著延长小鼠寿命,改善健康状况,并且增强身体功能[75]。尽管当前的研究还处于临床前阶段,利用转基因技术靶向清除衰老细胞的策略为抗衰老治疗提供了新的思路,并可能在未来成为一种有效的治疗方法。 (2)基因编辑技术:CRISPR (Clustered Regularly Interspaced Short Palindromic Repeats)-Cas9(CRISPR-associated protein 9)系统可以通过向导RNA (guide RNA,gRNA)精确地识别并定位到目标基因的特定突变位点,此时Cas9就可以在该位点进行切割,之后可以引入突变,从而达到基因表达抑制或激活的目的[76]。有研究显示CRISPR-Cas技术激活了部分与软骨细胞增殖和分化有关的SOX5基因,从而促进了软骨细胞的再生,提升了软骨的修复能力,减轻了由衰老和炎症引起的软骨损伤[77]。但是CRISPR-Cas技术在骨关节炎的治疗上还有许多困难,例如CRISPR-Cas技术的安全性,以及如何避免其带来的不良反应和伦理问题。 (3)细胞重编程技术:早在2006年,日本学者发现Oct4、Sox2、Klf4和c-Myc(OSKM)可以进行体外细胞重编程,这些转录因子会完全去除终末分化体细胞在发育过程中形成的所有表观遗传状态,使其变成多能干细胞[78]。 近年来,科学家们用转录因子进行衰老细胞重编程,使用OSKM转录因子短暂地部分重编程后,能够提高小鼠运动能力、认知功能和皮肤厚度等衰老指标,显著减缓衰老进程[79-80]。后续研究显示基因疗法促使的短期部分重编程可"
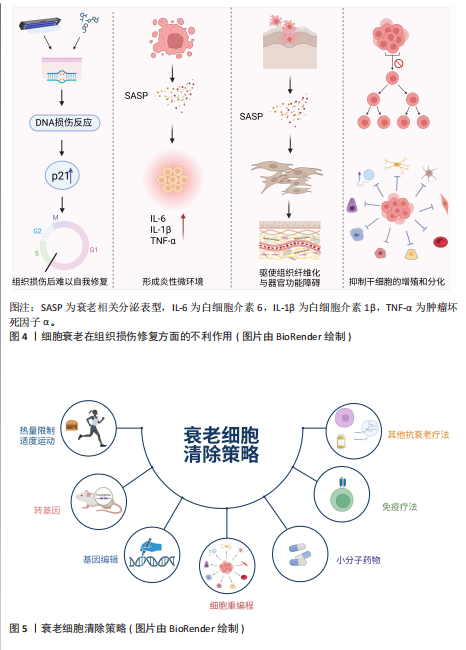
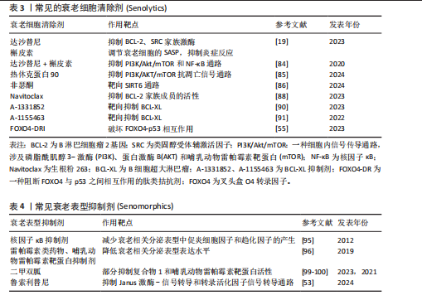
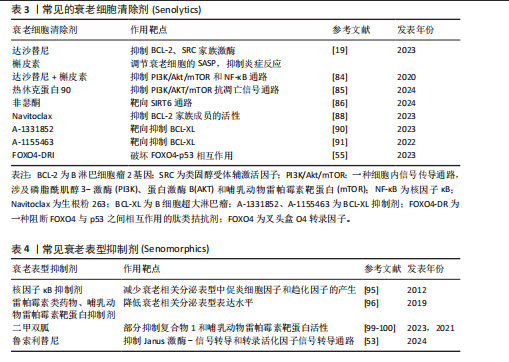
以逆转衰老的关键生理改变,从而延长小鼠的健康寿命,而且不会引发细胞过度增生或者肿瘤[81],这也许会变成以后衰老干预以及抗衰老治疗的新途径。不过应当注意,尽管这种方法在小鼠身上表现出积极的效果,但它是否适用于人类仍然要更多地去研究和证实。 (4)衰老细胞靶向清除策略:由于细胞衰老对组织损伤的影响,针对选择性清除衰老细胞的“衰老细胞清除剂”(Senolytics)的研究正在变得越来越普遍[82]。自2015年首次报道以来,许多衰老细胞清除剂已实现临床试验的转化。衰老细胞清除剂是一类通过清除衰老细胞来减缓或逆转与衰老相关的疾病、改善健康状况和延长寿命的药物。研究显示,通过遗传方法或衰老细胞清除剂去除小鼠体内的衰老细胞可减轻衰老和年龄相关疾病的多种病理表现[83]。达沙替尼(Dasatinib,D)通过抑制B淋巴细胞瘤2家族蛋白质(B-cell lymphoma-2,BCL-2)、SRC家族激酶等,促进衰老细胞的凋亡。槲皮素(Quercetin,Q)则通过调节衰老细胞的衰老相关分泌表型,抑制炎症反应并诱导衰老细胞死亡[19]。达沙替尼和槲皮素联合使用是首个体内验证的衰老细胞清除剂方案,比单一使用效果更好[55],可以通过抑制磷脂酰肌醇3激酶/蛋白激酶B/哺乳动物雷帕霉素靶蛋白和核因子κB通路,有效清除子宫组织中的衰老细胞,减缓衰老过程,同时抑制子宫纤维化的发生,并改善子宫的结构和功能[84]。研究表明,热休克蛋白90抑制剂通过靶向抑制磷脂酰肌醇3激酶/蛋白激酶B/哺乳动物雷帕霉素靶蛋白抗凋亡信号通路的活化,选择性地诱导衰老细胞凋亡[85]。非瑟酮(Fisetin)通过靶向沉默信息调节因子6抑制软骨细胞衰老,减缓骨关节炎的进展[86],同时有研究显示其通过调节肠道微生物群、抑制衰老和炎症反应,缓解了葡聚糖硫酸钠诱导的结肠炎进展[87]。衰老细胞通常会上调凋亡的负调控因子,包括BCL-2家族成员(如BCL-2、BCL-W和BCL-XL)。衰老细胞清除剂ABT-737和ABT-263(也称为navitoclax)能够抑制BCL-2家族成员的活性,从而促进衰老细胞启动凋亡过程[88]。一项临床试验发现鲁索利替尼(ruxolitinib)和ABT-263 (navitoclax)的联合使用可以改善原发性或者继发性骨髓纤维化患者的血红蛋白以及骨髓纤维化反应[89]。此外,A-1331852和A-1155463通过靶向抑制BCL-XL,发挥清除衰老细胞的活性[90-91]。据报道,FOXO4-DRI是一种细胞渗透性肽拮抗剂,可阻断叉头盒O4转录因子和p53的相互作用,导致复合物解离,释放p53并引发其核排斥反应,即p53从核内转移至胞质[55],有效地靶向衰老细胞凋亡,能够增强非小细胞肺癌的放射敏感性,还可以显著减少放射治疗引起的肺纤维化[92]。研究者通过给肺动脉高压小鼠口服生物制剂ABT-263 (navitoclax)清除衰老细胞可以改善肺动脉高压的症状,减轻肺血管重塑,并恢复右心功能。值得注意的是,过度清除衰老细胞可能反而加速肺动脉高压的进展,因此在治疗中需要维持衰老细胞的平衡[93]。见表3。 衰老表型抑制剂(Senomorphics)是一种通过减弱衰老细胞的衰老相关分泌表型而不消除衰老细胞的治疗策略,可用于缓解细胞衰老相关表型或疾病,其作用机制是通过抑制核因子κB、Janus激酶-信号转导和转录活化因子、磷脂酰肌醇3激酶/蛋白激酶B/哺乳动物雷帕霉素靶蛋白信号转导通路、线粒体复合物1和4相关靶标或其他途径来直接或间接减弱衰老细胞的衰老相关分泌表型表达幅度[94]。核因子κB抑制剂通过减少衰老相关分泌表型中促炎细胞因子和趋化因子的产生来延缓衰老进程[95]。Rapalogs(雷帕霉素类药物)和哺乳动物雷帕霉素靶蛋白抑制剂减少衰老相关分泌表型表达水平以延缓衰老进程[96]。在皮肤有衰老迹象的受试者(年龄≥40 岁)手上局部涂抹含有雷帕霉素(10 μmol/L)的乳膏,持续8个月后研究者们观察到受试者皮肤组织中的衰老相关基因产物p16INK4a和p21Cip1/Waf1减少,Ⅶ胶原蛋白表达增加,以及受试者皮肤外观的恢复[97]。16 周低剂量的哺乳动物雷帕霉素靶蛋白抑制剂治疗增加了老年人的抗病毒免疫力,并且降低了受试者病毒性呼吸道感染的发生率[98]。抗糖尿病药物二甲双胍通过部分抑制复合物显著提高衰老小鼠对缺血性损伤的耐受力[99],通过抑制哺乳动物雷帕霉素靶蛋白活性来减轻辐射小鼠支气管上皮组织的炎症和功能障碍[100],从而减轻实验动物和人的许多衰老相关疾病,包括代谢功能障碍、胰岛素抵抗、 2型糖尿病、心血管疾病、认知功能障碍和癌症的发生与转移等[101]。研究者们在9项随机对照试验的荟萃分析中观察到二甲双胍可以改善2型糖尿病患者的心肌耗氧量,同时降低了与心力衰竭发生有关的重要标志物(如低密度脂蛋白)的表达水平[102]。Janus激酶抑制剂通过抑制Janus激酶-信号转导和转录活化因子信号通路减少炎症因子的分泌,减轻衰老相关分泌表型引发的慢性低度炎症。Janus激酶抑制剂鲁索利替尼(ruxolitinib)抑制Janus激酶-信号转导和转录活化因子信号转导通路,抑制衰老相关分泌表型中促炎性细胞因子(白细胞介素6、白细胞介素1、肿瘤坏死因子)的产生,减少虚弱、老年小鼠的全身炎症,改善身体功能[53]。鲁索利替尼能够部分改善老年患者与衰弱相关的临床症状,如体质量减轻、肌力下降和食欲减退等。衰老表型抑制剂在动物模型研究中已经证明能够显著延缓与衰老相关的病理进程,尤其在间歇性给药模式下效果更加显著[101]。衰老相关分泌表型的抑制效果通常需要持续给药才能维持,然而连续使用衰老相关分泌表型抑制剂可能会产生“脱靶”效应。雷帕霉素(rapamycin)通过抑制哺乳动物雷帕霉素靶蛋白复合体2途径导致胰岛素抗性[103],还可能会导致白内障等与抑制哺乳动物雷帕霉素靶蛋白复合体2无关的不良反应[15]。见表4。 (5)免疫疗法:传统的衰老细胞清除方法虽然有效,但可能存在不良反应和非靶向效应[104]。为了克服这些问题,研究者Amor带领团队鉴定出尿激酶纤溶酶原激活剂受体(urokinase-type plasminogen activator receptor,uPAR)是在衰老细胞中广泛表达且特异性上调的细胞表面蛋白[105],尿激酶纤溶酶原激活剂受体在与配体结合后部分被蛋白水解切割,生成可溶性尿激酶纤溶酶原激活剂受体。可溶性尿激酶纤溶酶原激活剂受体是由衰老细胞分泌的衰老相关分泌表型的一部分,可作为肾脏疾病和糖尿病的血清生物标志物。靶向尿激酶纤溶酶原激活剂受体的嵌合抗原受体T(chimeric antigen receptor T,CAR-T)细胞能够在体外和体内高效清除衰老细胞。嵌合抗原受体T是通过基因工程改造的T细胞,可以特异性识别并攻击表达特定抗原的细胞[106]。注射靶向尿激酶纤溶酶原激活剂受体的嵌合抗原受体T细胞后,经化学或饮食诱导的肝纤维化小鼠的肝脏纤维化程度下降,肝功能指标得到改善;延长了经衰老诱导药物组合治疗的肺腺癌小鼠的生存期[56]。研究团队以小鼠和非人灵长类动物(猕猴)为衰老模型,构建的靶向自然杀伤细胞受体2D的嵌合抗原受体T细胞可有效清除衰老细胞,并改善衰老模型动物的健康状况[107]。另一个研究结果提供了一些靶向自然杀伤细胞受体2D的嵌合抗原受体T细胞改善衰老小鼠胚胎成纤维细胞和星形胶质细胞的初步证据,提出了管理衰老相关疾病的潜在新方法[108]。 自然杀伤细胞是先天免疫系统的淋巴细胞,其激活自然杀伤细胞受体2D和DNAM-1 (一种在人类调节性T细胞上表达的免疫受体)能够迅速应对衰老细胞中细胞周期停滞引起的应激反应,促使自然杀伤细胞释放细胞毒性颗粒(如穿孔素、颗粒酶)杀伤衰老细胞,从而调控组织稳态。研究表明,体外扩增和激活的自体自然杀伤细胞能够有效减少血液中的衰老标志物,并分泌细胞因子,激活巨噬细胞,进而增强免疫反应[109]。自然杀伤细胞在决定衰老细胞的存活与清除中起关键作用,能够通过免疫识别和杀伤机制清除衰老细胞。随着衰老进程,自然杀伤细胞功能下降,导致衰老细胞积累,从而加速组织老化和疾病发生。增强自然杀伤细胞活性或工程化改造(如嵌合抗原受体自然杀伤细胞),有望成为抗衰老和治疗衰老相关疾病的新策略[110]。有研究团队针对年龄较大"

个体进行自体自然杀伤细胞输注后发现可减少衰老相关慢性炎症,提高免疫稳态[111]。这些研究为自然杀伤细胞的抗衰老临床应用前景提供了有力支持。相比于嵌合抗原受体自然杀伤细胞疗法,由于嵌合抗原受体T细胞持久性差、免疫抑制性肿瘤微环境或癌细胞耐药等原因,大量患者的缓解是短暂的[106,112]。此外,嵌合抗原受体T细胞疗法的治疗效益常伴随剂量限制性毒性,其核心机制涉及T细胞过度活化引发的细胞因子风暴及神经血管稳态失衡,造成细胞因子释放综合征和免疫效应细胞相关神经毒性综合征等病症[113]。相较于嵌合抗原受体T细胞,嵌合抗原受体自然杀伤细胞具有安全性、低成本及可及性的优点,正成为细胞治疗领域中更具优势的策略[114]。目前嵌合抗原受体自然杀伤细胞疗法尚未在临床环境中得到广泛应用,未来研究仍然面临多项挑战。 (6)其他抗衰疗法:近年来,干细胞、外泌体和富血小板血浆在靶向清除衰老细胞方面已取得突破性进展,这些生物材料通过不同机制与衰老细胞发生相互作用,有望为衰老治疗提供新的策略。 干细胞具有自我更新和分化的生物学特性,已经成为目前抗衰领域中很有前景的解决方案。除内在衰老外,皮肤常暴露于各种环境压力因素,包括紫外线辐射、创伤、化学物质、吸烟和活性氧。随着年龄的增长,这些因素会引发黑色素细胞、成纤维细胞和胶原合成下降以及弹性纤维破坏,造成皮肤老化,干细胞通过旁分泌途径分泌生长因子和细胞因子来发挥抗炎和抗凋亡的作用,修复受损组织并促进组织重塑[115]。间充质干细胞在清除衰老细胞方面具有巨大的潜力[116]。有研究表明羊水间充质干细胞来源条件培养基通过提供促进愈合以及刺激胶原蛋白和细胞外基质重要成分生成的生长因子,改善了传统皮肤治疗方法的效果,减少了细纹和皱纹[117-118]。有研究者把临床级人脐带来源间充质干细胞用于SAMP8小鼠和D-半乳糖诱发的老化小鼠,发现间充质干细胞通过改善细胞外基质相关抗肌萎缩蛋白和层粘连蛋白的表达量,推动卫星细胞的活化,加强自噬作用并阻止细胞老化等手段,明显改善了这两种小鼠的老化骨骼肌力量和性能[119]。值得注意的是,尽管间充质干细胞在动物实验中表现出明显的疗效,但在临床使用上仍然碰上不少难题,主要涉及干细胞的获取是否规范、体外扩增进程的安全性、移植之后的长远疗效以及可能出现的不良反应等[120-121]。 衰老的成纤维细胞会产生更多的活性氧和基质金属蛋白酶,最终导致皮肤老化。然而,干细胞衍生的外泌体可以激活丝裂原活化蛋白激酶、蛋白激酶B、信号转导和转录活化因子3和细胞外调节蛋白激酶1/2信号通路,以增强成纤维细胞的增殖和迁移能力,从而逆转紫外线B诱导的衰老[122]。研究表明,外泌体通过调节转化生长因子β/Smad信号通路改善卵巢早衰疾病的卵巢功能[123]。年轻间充质干细胞分泌的外泌体可显著促进老年大鼠在骨拉伸成骨过程中的新骨形成[124]。尽管这一领域已取得显著进展,但外泌体疗法在临床应用中仍面临诸多挑战,包括生产与运输的高成本、在创伤部位的快速清除或滞留效率低,以及角质层吸收受限等技术瓶颈[125-127]。 富血小板血浆通过浓缩血小板、血小板衍生生长因子、转化生长因子β、表皮生长因子等生长因子,促使皮肤细胞的修复和再生,在皮肤修复、再生医学、美容等领域得到广泛应用[128]。富血小板血浆治疗男性脱发在临床上有较好的疗效,当血小板较多、某些生长因子含量较高时,疗效更好[129]。多项临床研究证实富血小板血浆借助生长因子促进皮肤细胞增殖、分化与迁移,增强皮肤的自我恢复能力[130]。 经过研究发现,增强型富血小板血浆促使成纤维细胞沉默信息调节因子1转录活性以延缓细胞老化,从而极大地加快了创伤愈合速度[131]。此外,富血小板血浆与微针、射频等医美结合使用时,皮肤质地显著改善,皱纹也明显减少[132]。"

| [1] MCCAY CM, MAYNARD LA, SPERLING G, et al. The Journal of Nutrition. Volume 18 July--December, 1939. Pages 1--13. Retarded growth, life span, ultimate body size and age changes in the albino rat after feeding diets restricted in calories. Nutr Rev. 1975;33(8):241-243. [2] HAYFLICK L, MOORHEAD PS. The serial cultivation of human diploid cell strains. Exp Cell Res. 1961; 25:585-621. [3] OLOVNIKOV AM. Principle of marginotomy in template synthesis of polynucleotides. Dokl Akad Nauk SSSR. 1971;201(6):1496-1499. [4] SAGER R. Senescence as a mode of tumor suppression. Environ Health Perspect. 1991;93: 59-62. [5] KENNEDY BK, AUSTRIACO NR JR, ZHANG J, et al. Mutation in the silencing gene SIR4 can delay aging in S. cerevisiae. Cell. 1995;80(3):485-496. [6] COPPÉ JP, PATIL CK, RODIER F, et al. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 2008;6(12): 2853-2868. [7] LÓPEZ-OTÍN C, BLASCO MA, PARTRIDGE L, et al. The hallmarks of aging. Cell. 2013;153(6):1194-1217. [8] ZHU Y, TCHKONIA T, PIRTSKHALAVA T, et al. The Achilles’ heel of senescent cells: from transcriptome to senolytic drugs. Aging Cell. 2015;14(4):644-658. [9] JUSTICE JN, NAMBIAR AM, TCHKONIA T, et al. Senolytics in idiopathic pulmonary fibrosis: Results from a first-in-human, open-label, pilot study. EBioMedicine. 2019;40:554-563. [10] HICKSON LJ, LANGHI PRATA LGP, BOBART SA, et al. Senolytics decrease senescent cells in humans: Preliminary report from a clinical trial of Dasatinib plus Quercetin in individuals with diabetic kidney disease. EBioMedicine. 2019;47:446-456. [11] LÓPEZ-OTÍN C, BLASCO MA, PARTRIDGE L, et al. Hallmarks of aging: An expanding universe. Cell. 2023;186(2):243-278. [12] ZHAO H, LIU Z, CHEN H, et al. Identifying specific functional roles for senescence across cell types. Cell. 2024;187(25):7314-7334. [13] KROEMER G, MAIER AB, CUERVO AM, et al. From geroscience to precision geromedicine: Understanding and managing aging. Cell. 2025; 188(8):2043-2062. [14] IZQUIERDO JM. Mitochondria-cGAS-STING axis is a potential therapeutic target for senescence-dependent inflammaging-associated neurodegeneration. Neural Regen Res. 2025;20(3): 805-807. [15] MA K, LI M, SONG Y, et al. Bazi Bushen alleviates reproductive aging in aged male mice. Chin J Nat Med. 2024;22(5):416-425. [16] ZHANG XR, ZHANG TS, ZHANG YN, et al. Aurora A Kinase Plays a Key Role in Mitosis Skip during Senescence Induced by Ionizing Radiation. Biomed Environ Sci. 2023;36(10):903-916. [17] YOUSEFZADEH M, HENPITA C, VYAS R, et al. DNA damage-how and why we age? Elife. 2021;10: e62852. [18] WU F, LIU J, TAO M, et al. β-Galactosidase-Activatable Fluorescent and Photoacoustic Imaging of Tumor Senescence. Anal Chem. 2023; 95(28):10481-10485. [19] ZHOU Y, NISHIURA A, MORIKUNI H, et al. RANKL(+) senescent cells under mechanical stress: a therapeutic target for orthodontic root resorption using senolytics. Int J Oral Sci. 2023;15(1):20. [20] LUCAS V, CAVADAS C, AVELEIRA CA. Cellular Senescence: From Mechanisms to Current Biomarkers and Senotherapies. Pharmacol Rev. 2023;75(4):675-713. [21] LIU Y, SU Z, TAVANA O, et al. Understanding the complexity of p53 in a new era of tumor suppression. Cancer Cell. 2024;42(6):946-967. [22] SHARPLESS NE, DEPINHO RA. The mighty mouse: genetically engineered mouse models in cancer drug development. Nat Rev Drug Discov. 2006;5(9):741-754. [23] FITSIOU E, PULIDO T, CAMPISI J, et al. Cellular Senescence and the Senescence-Associated Secretory Phenotype as Drivers of Skin Photoaging. J Invest Dermatol. 2021;141(4S):1119-1126. [24] TASDEMIR N, BANITO A, ROE JS, et al. BRD4 Connects Enhancer Remodeling to Senescence Immune Surveillance. Cancer Discov. 2016;6(6):612-629. [25] LI X, FENG L, ZHANG C, et al. Insulin-like growth factor binding proteins 7 prevents dental pulp-derived mesenchymal stem cell senescence via metabolic downregulation of p21. Sci China Life Sci. 2022;65(11):2218-2232. [26] LIANG WZ, LIU YX, XU DD, et al. Decoding the Molecular Mechanisms of BRAF V600E-Induced Nevi Formation. Biomed Environ Sci. 2024;37(7):774-784. [27] WANG Y, JIN S, LUO D, et al. Prim-O-glucosylcimifugin ameliorates aging-impaired endogenous tendon regeneration by rejuvenating senescent tendon stem/progenitor cells. Bone Res. 2023;11(1):54. [28] DAVAN-WETTON CSA, MONTERO-MELENDEZ T. An optimised protocol for the detection of lipofuscin, a versatile and quantifiable marker of cellular senescence. PLoS One. 2024;19(7):e0306275. [29] HEWITT G, JURK D, MARQUES FD, et al. Telomeres are favoured targets of a persistent DNA damage response in ageing and stress-induced senescence. Nat Commun. 2012;3:708. [30] MATIAS I, DINIZ LP, DAMICO IV, et al. Loss of lamin-B1 and defective nuclear morphology are hallmarks of astrocyte senescence in vitro and in the aging human hippocampus. Aging Cell. 2022;21(1):e13521. [31] SWANSON EC, MANNING B, ZHANG H, et al. Higher-order unfolding of satellite heterochromatin is a consistent and early event in cell senescence. J Cell Biol. 2013;203(6):929-942. [32] COLLADO M, GIL J, EFEYAN A, et al. Tumour biology: senescence in premalignant tumours. Nature. 2005;436(7051):642. [33] LI Q, LI Y, QIAO Q, et al. Oral administration of Bifidobacterium breve improves anti-angiogenic drugs-derived oral mucosal wound healing impairment via upregulation of interleukin-10. Int J Oral Sci. 2023;15(1):56. [34] NI JY, WANG X, XIE HY, et al. Deubiquitinating enzyme USP11 promotes renal tubular cell senescence and fibrosis via inhibiting the ubiquitin degradation of TGF-β receptor II. Acta Pharmacol Sin. 2023;44(3):584-595. [35] O’REILLY S, MARKIEWICZ E, IDOWU OC. Aging, senescence, and cutaneous wound healing-a complex relationship. Front Immunol. 2024;15:1429716. [36] DEMARIA M, OHTANI N, YOUSSEF SA, et al. An essential role for senescent cells in optimal wound healing through secretion of PDGF-AA. Dev Cell. 2014;31(6):722-733. [37] RING N AR, VALDIVIESO K, GRILLARI J, et al. The role of senescence in cellular plasticity: Lessons from regeneration and development and implications for age-related diseases. Dev Cell. 2022;57(9):1083-1101. [38] OGUMA Y, ALESSIO N, APRILE D, et al. Meta-analysis of senescent cell secretomes to identify common and specific features of the different senescent phenotypes: a tool for developing new senotherapeutics. Cell Commun Signal. 2023; 21(1):262. [39] DONG Z, LUO Y, YUAN Z, et al. Cellular senescence and SASP in tumor progression and therapeutic opportunities. Mol Cancer. 2024;23(1):181. [40] FANG YP, ZHAO Y, HUANG JY, et al. The functional role of cellular senescence during vascular calcification in chronic kidney disease. Front Endocrinol (Lausanne). 2024;15:1330942. [41] CHAINTREUIL P, KERRENEUR E, BOURGOIN M, et al. The generation, activation, and polarization of monocyte-derived macrophages in human malignancies. Front Immunol. 2023; 14:1178337. [42] COOKE JP, LAI L. Transflammation in tissue regeneration and response to injury: How cell-autonomous inflammatory signaling mediates cell plasticity. Adv Drug Deliv Rev. 2023;203:115118. [43] VON JOEST M, CHEN C, DOUCHÉ T, et al. Amphiregulin mediates non-cell-autonomous effect of senescence on reprogramming. Cell Rep. 2022;40(2):111074. [44] SALINAS-SAAVEDRA M, FEBRIMARSA, KRASOVEC G, et al. Senescence-induced cellular reprogramming drives cnidarian whole-body regeneration. Cell Rep. 2023;42(7):112687. [45] WALTERS HE, TROYANOVSKIY KE, GRAF AM, et al. Senescent cells enhance newt limb regeneration by promoting muscle dedifferentiation. Aging Cell. 2023;22(6):e13826. [46] REYES NS, KRASILNIKOV M, ALLEN NC, et al. Sentinel p16INK4a+ cells in the basement membrane form a reparative niche in the lung. Science. 2022;378(6616):192-201. [47] WANG X, RAMOS R, PHAN AQ, et al. Signalling by senescent melanocytes hyperactivates hair growth. Nature. 2023;618(7966):808-817. [48] TAKAYA K, ASOU T, KISHI K. Cathepsin F is a potential marker for senescent human skin fibroblasts and keratinocytes associated with skin aging. Geroscience. 2023;45(1):427-437. [49] MA X, QI D, SUN X, et al. TRPM7 controls skin keratinocyte senescence by targeting intracellular calcium signaling. FEBS J. 2024;291(21):4680-4695. [50] CHIN T, LEE XE, NG PY, et al. The role of cellular senescence in skin aging and age-related skin pathologies. Front Physiol. 2023;14:1297637. [51] BATEMAN G, GUO-PARKE H, RODGERS AM, et al. Airway Epithelium Senescence as a Driving Mechanism in COPD Pathogenesis. Biomedicines. 2023;11(7):2702. [52] CAI Y, XIONG M, XIN Z, et al. Decoding aging-dependent regenerative decline across tissues at single-cell resolution. Cell Stem Cell. 2023; 30(12):1674-1691. [53] MI B, XIONG Y, KNOEDLER S, et al. Ageing-related bone and immunity changes: insights into the complex interplay between the skeleton and the immune system. Bone Res. 2024;12(1):42. [54] ZHENG Z, LI J, LIU T, et al. DNA methylation clocks for estimating biological age in Chinese cohorts. Protein Cell. 2024;15(8):575-593. [55] AGING BIOMARKER CONSORTIUM, BAO H, CAO J, et al. Biomarkers of aging. Sci China Life Sci. 2023; 66(5):893-1066. [56] DAI K, GENG Z, ZHANG W, et al. Biomaterial design for regenerating aged bone: materiobiological advances and paradigmatic shifts. Natl Sci Rev. 2024;11(5):nwae076. [57] YANG T, WAN R, TU W, et al. Aryl hydrocarbon receptor: Linking environment to aging process in elderly patients with asthma. Chin Med J (Engl). 2024;137(4):382-393. [58] BEVILACQUA A, HO PC, FRANCO F. Metabolic reprogramming in inflammaging and aging in T cells. Life Metab. 2023;2(5):load028. [59] KITA A, YAMAMOTO S, SAITO Y, et al. Cellular senescence and wound healing in aged and diabetic skin. Front Physiol. 2024;15:1344116. [60] ZHANG L, PITCHER LE, YOUSEFZADEH MJ, et al. Cellular senescence: a key therapeutic target in aging and diseases. J Clin Invest. 2022; 132(15):e158450. [61] ZHOU C, SHEN Z, SHEN B, et al. FABP4 in LSECs promotes CXCL10-mediated macrophage recruitment and M1 polarization during NAFLD progression. Biochim Biophys Acta Mol Basis Dis. 2023;1869(7):166810. [62] YANG H, ZHANG X, XUE B. New insights into the role of cellular senescence and chronic wounds. Front Endocrinol (Lausanne). 2024;15:1400462. [63] YAMADA Z, NISHIO J, MOTOMURA K, et al. Senescence of alveolar epithelial cells impacts initiation and chronic phases of murine fibrosing interstitial lung disease. Front Immunol. 2022; 13:935114. [64] TAMARU T, KOBAYAKAWA K, SAIWAI H, et al. Glial scar survives until the chronic phase by recruiting scar-forming astrocytes after spinal cord injury. Exp Neurol. 2023;359:114264. [65] CAI Y, WANG S, QU J, et al. Rejuvenation of Tissue Stem Cells by Intrinsic and Extrinsic Factors. Stem Cells Transl Med. 2022;11(3):231-238. [66] NANIC L, CEDILAK A, VIDACEK NS, et al. In Vivo Skin Regeneration and Wound Healing Using Cell Micro-Transplantation. Pharmaceutics. 2022; 14(9):1955. [67] HUANG Y, HE W, ZHANG Y, et al. Targeting SIRT2 in Aging-Associated Fibrosis Pathophysiology. Aging Dis. 2024;16(4):2036-2053. [68] CHENG H, ZHAO Z, LIU D, et al. Early senescence of pancreatic β cells induced by unfolded protein response deficiency prevents type 1 diabetes. J Zhejiang Univ Sci B. 2024;25(9):796-799. [69] LEE MB, HILL CM, BITTO A, et al. Antiaging diets: Separating fact from fiction. Science. 2021; 374(6570):eabe7365. [70] DIWAN B, SHARMA R. Nutritional components as mitigators of cellular senescence in organismal aging: a comprehensive review. Food Sci Biotechnol. 2022;31(9):1089-1109. [71] XIROUCHAKI CE, JIA Y, MCGRATH MJ, et al. Skeletal muscle NOX4 is required for adaptive responses that prevent insulin resistance. Sci Adv. 2021;7(51):eabl4988. [72] REBO J, MEHDIPOUR M, GATHWALA R, et al. A single heterochronic blood exchange reveals rapid inhibition of multiple tissues by old blood. Nat Commun. 2016;7:13363. [73] YANG C, LIU ZL, WANG J, et al. Parabiosis modeling: protocol, application and perspectives. Zool Res. 2021;42(3):253-261. [74] MEHDIPOUR M, SKINNER C, WONG N, et al. Rejuvenation of three germ layers tissues by exchanging old blood plasma with saline-albumin. Aging (Albany NY). 2020;12(10):8790-8819. [75] WANG B, WANG L, GASEK NS, et al. Intermittent clearance of p21-highly-expressing cells extends lifespan and confers sustained benefits to health and physical function. Cell Metab. 2024;36(8):1795-1805. [76] YUAN Z. From Origin to the Present: Establishment, Mechanism, Evolutions and Biomedical Applications of the CRISPR/Cas-Based Macromolecular System in Brief. Molecules. 2025;30(4):947. [77] JING Y, JIANG X, JI Q, et al. Genome-wide CRISPR activation screening in senescent cells reveals SOX5 as a driver and therapeutic target of rejuvenation. Cell Stem Cell. 2023;30(11):1452-1471. [78] TAKAHASHI K, YAMANAKA S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126(4):663-676. [79] ALLE Q, LE BORGNE E, BENSADOUN P, et al. A single short reprogramming early in life initiates and propagates an epigenetically related mechanism improving fitness and promoting an increased healthy lifespan. Aging Cell. 2022; 21(11):e13714. [80] BROWDER KC, REDDY P, YAMAMOTO M, et al. In vivo partial reprogramming alters age-associated molecular changes during physiological aging in mice. Nat Aging. 2022;2(3):243-253. [81] MACIP CC, HASAN R, HOZNEK V, et al. Gene Therapy-Mediated Partial Reprogramming Extends Lifespan and Reverses Age-Related Changes in Aged Mice. Cell Reprogram. 2024; 26(1):24-32. [82] POWER H, VALTCHEV P, DEHGHANI F, et al. Strategies for senolytic drug discovery. Aging Cell. 2023;22(10):e13948. [83] ROBBINS PD, JURK D, KHOSLA S, et al. Senolytic Drugs: Reducing Senescent Cell Viability to Extend Health Span. Annu Rev Pharmacol Toxicol. 2021;61:779-803. [84] CAVALCANTE MB, SACCON TD, NUNES A DC, et al. Dasatinib plus quercetin prevents uterine age-related dysfunction and fibrosis in mice. Aging (Albany NY). 2020;12(3):2711-2722. [85] GUAN Q, ZHANG Y, WANG ZK, et al. Skeletal phenotypes and molecular mechanisms in aging mice. Zool Res. 2024;45(4):724-746. [86] WANG X, LI X, ZHOU J, et al. Fisetin suppresses chondrocyte senescence and attenuates osteoarthritis progression by targeting sirtuin 6. Chem Biol Interact. 2024;390:110890. [87] ASHIQUEALI SA, CHAUDHARI D, ZHU X, et al. Fisetin modulates the gut microbiota alongside biomarkers of senescence and inflammation in a DSS-induced murine model of colitis. Geroscience. 2024;46(3):3085-3103. [88] NIYOGI U, JARA CP, CARLSON MA. Treatment of aged wound healing models with FGF2 and ABT-737 reduces the senescent cell population and increases wound closure rate. Wound Repair Regen. 2023;31(5):613-626. [89] HARRISON CN, GARCIA JS, SOMERVAILLE TCP, et al. Addition of Navitoclax to Ongoing Ruxolitinib Therapy for Patients With Myelofibrosis With Progression or Suboptimal Response: Phase II Safety and Efficacy. J Clin Oncol. 2022;40(15): 1671-1680. [90] YANG JH, PETTY CA, DIXON-MCDOUGALL T, et al. Chemically induced reprogramming to reverse cellular aging. Aging (Albany NY). 2023;15(13):5966-5989. [91] TSUJI K, KIDA Y, KOSHIKAWA N, et al. Suppression of non-small-cell lung cancer A549 tumor growth by an mtDNA mutation-targeting pyrrole-imidazole polyamide-triphenylphosphonium and a senolytic drug. Cancer Sci. 2022;113(4):1321-1337. [92] WU M, LIAO Y, TANG L. Non-small cell lung cancer organoids: Advances and challenges in current applications. Chin J Cancer Res. 2024;36(5):455-473. [93] BORN E, LIPSKAIA L, BREAU M, et al. Eliminating Senescent Cells Can Promote Pulmonary Hypertension Development and Progression. Circulation. 2023;147(8):650-666. [94] ZHANG L, PITCHER LE, PRAHALAD V, et al. Targeting cellular senescence with senotherapeutics: senolytics and senomorphics. FEBS J. 2023;290(5):1362-1383. [95] TILSTRA JS, ROBINSON AR, WANG J, et al. NF-κB inhibition delays DNA damage-induced senescence and aging in mice J Clin Invest. 2012;122(7):2601-2612. [96] TANG H, GENG A, ZHANG T, et al. Single senescent cell sequencing reveals heterogeneity in senescent cells induced by telomere erosion. Protein Cell. 2019; 10(5):370-375. [97] CHUNG CL, LAWRENCE I, HOFFMAN M, et al. Topical rapamycin reduces markers of senescence and aging in human skin: an exploratory, prospective, randomized trial. Geroscience. 2019;41(6):861-869. [98] MANNICK JB, TEO G, BERNARDO P, et al. Targeting the biology of ageing with mTOR inhibitors to improve immune function in older adults: phase 2b and phase 3 randomised trials. Lancet Healthy Longev. 2021;2(5):e250-e262. [99] LI Z, WANG H, ZOUNGRANA LI, et al. Administration of metformin rescues age-related vulnerability to ischemic insults through mitochondrial energy metabolism. Biochem Biophys Res Commun. 2023;659: 46-53. [100] HANSEL C, BARR S, SCHEMANN AV, et al. Metformin Protects against Radiation-Induced Acute Effects by Limiting Senescence of Bronchial-Epithelial Cells. Int J Mol Sci. 2021;22(13):7064. [101] HUFFMAN DM, JUSTICE JN, STOUT MB, et al. Evaluating Health Span in Preclinical Models of Aging and Disease: Guidelines, Challenges, and Opportunities for Geroscience. J Gerontol A Biol Sci Med Sci. 2016;71(11):1395-1406. [102] DLUDLA PV, NYAMBUYA TM, JOHNSON R, et al. Metformin and heart failure-related outcomes in patients with or without diabetes: a systematic review of randomized controlled trials. Heart Fail Rev. 2021;26(6):1437-1445. [103] FAN GH, ZHANG CZ, GAO FQ, et al. A mixed blessing for liver transplantation patients - Rapamycin. Hepatobiliary Pancreat Dis Int. 2023; 22(1):14-21. [104] YANG S, LIU C, JIANG M, et al. A single-nucleus transcriptomic atlas of primate liver aging uncovers the pro-senescence role of SREBP2 in hepatocytes. Protein Cell. 2024;15(2):98-120. [105] SHOU LH, CAO D, DONG XH, et al. Bone Marrow Urokinase Plasminogen Activator Receptor Levels are Associated with the Progress of Multiple Myeloma. Chin Med Sci J. 2016;31(3):155-160. [106] TANG L, HUANG ZP, MEI H, et al. Insights gained from single-cell analysis of chimeric antigen receptor T-cell immunotherapy in cancer. Mil Med Res. 2023;10(1):52. [107] YANG D, SUN B, LI S, et al. NKG2D-CAR T cells eliminate senescent cells in aged mice and nonhuman primates. Sci Transl Med. 2023; 15(709):eadd1951. [108] DENG Y, KUMAR A, XIE K, et al. Targeting senescent cells with NKG2D-CAR T cells. Cell Death Discov. 2024;10(1):217. [109] CHELYAPOV N, NGUYEN TT, GONZALEZ R. Autologous NK cells propagated and activated ex vivo decrease senescence markers in human PBMCs. Biochem Biophys Rep. 2022;32: 101380. [110] ANTONANGELI F, ZINGONI A, SORIANI A, et al. Senescent cells: Living or dying is a matter of NK cells. J Leukoc Biol. 2019;105(6):1275-1283. [111] TANG X, DENG B, ZANG A, et al. Characterization of age-related immune features after autologous NK cell infusion: Protocol for an open-label and randomized controlled trial. Front Immunol. 2022;13:940577. [112] ZHANG Y, XU Y, DANG X, et al. Challenges and optimal strategies of CAR T therapy for hematological malignancies. Chin Med J (Engl). 2023;136(3):269-279. [113] DIORIO C, SHRAIM R, MYERS R, et al. Comprehensive Serum Proteome Profiling of Cytokine Release Syndrome and Immune Effector Cell-Associated Neurotoxicity Syndrome Patients with B-Cell ALL Receiving CAR T19. Clin Cancer Res. 2022;28(17):3804-3813. [114] LI W, WANG X, ZHANG X, et al. CAR-NK Cell Therapy: A Transformative Approach to Overcoming Oncological Challenges. Biomolecules. 2024;14(8):1035. [115] WANG JV, SCHOENBERG E, ZAYA R, et al. The rise of stem cells in skin rejuvenation: A new frontier. Clin Dermatol. 2020; 38(4):494-496. [116] POURANG A, ROCKWELL H, KARIMI K. New Frontiers in Skin Rejuvenation, Including Stem Cells and Autologous Therapies. Facial Plast Surg Clin North Am. 2020;28(1):101-117. [117] EL-DOMYATI M, MOFTAH NH, NASIF GA, et al. Facial rejuvenation using stem cell conditioned media combined with skin needling: A split-face comparative study.J Cosmet Dermatol. 2020;19(9):2404-2410. [118] EL-DOMYATI M, MOFTAH NH, NASIF GA, et al. Amniotic fluid-derived mesenchymal stem cell products combined with microneedling for acne scars: A split-face clinical, histological, and histometric study. J Cosmet Dermatol. 2019; 18(5):1300-1306. [119] WANG C, ZHAO B, ZHAI J, et al. Clinical-grade human umbilical cord-derived mesenchymal stem cells improved skeletal muscle dysfunction in age-associated sarcopenia mice. Cell Death Dis. 2023;14(5):321. [120] FANI N, MORADI M, ZAVARI R, et al. Current Advances in Wound Healing and Regenerative Medicine. Curr Stem Cell Res Ther. 2024;19(3): 277-291. [121] DING JY, CHEN MJ, WU LF, et al. Mesenchymal stem cell-derived extracellular vesicles in skin wound healing: roles, opportunities and challenges. Mil Med Res. 2023;10(1):36. [122] WU JY, WU SN, ZHANG LP, et al. Stem Cell-Derived Exosomes: A New Method for Reversing Skin Aging. Tissue Eng Regen Med. 2022;19(5):961-968. [123] HUANG B, LU J, DING C, et al. Exosomes derived from human adipose mesenchymal stem cells improve ovary function of premature ovarian insufficiency by targeting SMAD. Stem Cell Res Ther. 2018;9(1):216. [124] JIA Y, QIU S, XU J, et al. Exosomes Secreted by Young Mesenchymal Stem Cells Promote New Bone Formation During Distraction Osteogenesis in Older Rats. Calcif Tissue Int. 2020;106(5):509-517. [125] LOPES D, LOPES J, PEREIRA-SILVA M, et al. Bioengineered exosomal-membrane-camouflaged abiotic nanocarriers: neurodegenerative diseases, tissue engineering and regenerative medicine. Mil Med Res. 2023;10(1):19. [126] VYAS KS, KAUFMAN J, MUNAVALLI GS, et al. Exosomes: the latest in regenerative aesthetics. Regen Med. 2023;18(2):181-194. [127] RIAU AK, ONG HS, YAM GHF, et al. Sustained Delivery System for Stem Cell-Derived Exosomes. Front Pharmacol. 2019;10:1368. [128] HAUSAUER AK, HUMPHREY S. The Physician’s Guide to Platelet-Rich Plasma in Dermatologic Surgery Part II: Clinical Evidence. Dermatol Surg. 2020;46(4):447-456. [129] RODRIGUES BL, MONTALVAO SAL, CANCELA RBB, et al. Treatment of male pattern alopecia with platelet-rich plasma: A double-blind controlled study with analysis of platelet number and growth factor levels. J Am Acad Dermatol. 2019;80(3): 694-700. [130] MAISEL-CAMPBELL AL, ISMAIL A, REYNOLDS KA, et al. A systematic review of the safety and effectiveness of platelet-rich plasma (PRP) for skin aging. Arch Dermatol Res. 2020;312(5):301-315. [131] WENG HP, CHENG YY, LEE HL, et al. Enhanced Platelet-Rich Plasma (ePRP) Stimulates Wound Healing through Effects on Metabolic Reprogramming in Fibroblasts. Int J Mol Sci. 2021;22(23): 12623. [132] LI KC, LIN ZZ, ZHANG ZD, et al. Comparing the Effectiveness of Platelet-Rich Plasma Alone Versus Combined With Microneedles or Radiofrequency for Neck Wrinkle Treatment. J Cosmet Dermatol. 2025;24(1):e16651. [133] GASEK NS, KUCHEL GA, KIRKLAND JL, et al. Strategies for Targeting Senescent Cells in Human Disease. Nat Aging. 2021;1(10): 870-879. [134] LI Y, TIAN X, LUO J, et al. Molecular mechanisms of aging and anti-aging strategies. Cell Commun Signal. 2024; 22(1):285. [135] ZHENG L, HE S, WANG H, et al. Targeting Cellular Senescence in Aging and Age-Related Diseases: Challenges, Considerations, and the Emerging Role of Senolytic and Senomorphic Therapies. Aging Dis. 2024;15(6):2554-2594. [136] HUANG W, HICKSON LJ, EIRIN A, et al. Cellular senescence: the good, the bad and the unknown. Nat Rev Nephrol. 2022;18(10):611-627. [137] DI MICCO R, KRIZHANOVSKY V, BAKER D, et al. Cellular senescence in ageing: from mechanisms to therapeutic opportunities. Nat Rev Mol Cell Biol. 2021;22(2):75-95. |
| [1] | Cao Yong, Teng Hongliang, Tai Pengfei, Li Junda, Zhu Tengqi, Li Zhaojin. Interactions between cytokines and satellite cells in muscle regeneration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1808-1817. |
| [2] | Cai Ziming, Yu Qinghe, Ma Pengfei, Zhang Xin, Zhou Longqian, Zhang Chongyang, Lin Wenping. Heme oxygenase-1 alleviates lipopolysaccharide-induced inflammatory response in nucleus pulposus mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1624-1631. |
| [3] | He Jiale, Huang Xi, Dong Hongfei, Chen Lang, Zhong Fangyu, Li Xianhui. Acellular dermal matrix combined with adipose-derived stem cell exosomes promotes burn wound healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1699-1710. |
| [4] | Xia Linfeng, Wang Lu, Long Qianfa, Tang Rongwu, Luo Haodong, Tang Yi, Zhong Jun, Liu Yang. Human umbilical cord mesenchymal stem cell-derived exosomes alleviate blood-brain barrier damage in mice with septic encephalopathy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1711-1719. |
| [5] | Cui Lianxu, Li Haomin, Xu Junrong, Tan Baodong, Lu Dahong, Peng Siwei, Wang Jinhui. Effect of umbilical cord mesenchymal stem cell conditioned medium on tissue repair after traumatic craniocerebral injury in miniature pigs [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1730-1735. |
| [6] | Zhang Haiwen, Zhang Xian, Xu Taichuan, Li Chao. Bibliometric and visual analysis of the research status and trends of senescence in osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1580-1591. |
| [7] | Hou Chaowen, Li Zhaojin, Kong Jianda, Zhang Shuli. Main physiological changes in skeletal muscle aging and the multimechanism regulatory role of exercise [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1464-1475. |
| [8] | You Huijuan, Wu Shuzhen, Rong Rong, Chen Liyuan, Zhao Yuqing, Wang Qinglu, Ou Xiaowei, Yang Fengying. Macrophage autophagy in lung diseases: two-sided effects [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1516-1526. |
| [9] | Yin Yongcheng, Zhao Xiangrui, Yang Zhijie, Li Zheng, Li Fang, Ning Bin. Effect and mechanism of peroxiredoxin 1 in microglial inflammation after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1106-1113. |
| [10] | Zhang Di, Zhao Jun, Ma Guangyue, Sun Hui, Jiang Rong. Mechanism of depression-like behavior in chronic social defeat stress mice based on high-throughput sequencing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1139-1146. |
| [11] | Li Haojing, Wang Xin, Song Chenglin, Zhang Shengnan, Chen Yunxin. Therapeutic efficacy of extracorporeal shock wave therapy in the upper trapezius muscle area combined with exercise control training in patients with chronic non-specific neck pain [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1162-1170. |
| [12] | Liu Yu, Lei Senlin, Zhou Jintao, Liu Hui, Li Xianhui. Mechanisms by which aerobic and resistance exercises improve obesity-related cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1171-1183. |
| [13] | Yu Huifen, Mo Licun, Cheng Leping. The position and role of 5-hydroxytryptamine in the repair of tissue injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1196-1206. |
| [14] | Wen Xiaolong, Weng Xiquan, Feng Yao, Cao Wenyan, Liu Yuqian, Wang Haitao. Effects of inflammation on serum hepcidin and iron metabolism related parameters in patients with type 2 diabetes mellitus: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1294-1301. |
| [15] | Chen Yixian, Chen Chen, Lu Liheng, Tang Jinpeng, Yu Xiaowei. Triptolide in the treatment of osteoarthritis: network pharmacology analysis and animal model validation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 805-815. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||