Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (35): 5603-5609.doi: 10.12307/2023.890
Previous Articles Next Articles
Mechanism by which high-intensity intermittent exercise improves skeletal muscle injury and enhances exercise capacity in rats
Zhang Liumei1, Liu Jingjing1, Lin Xiaoye2, Liu Lin1, Lu Jiao1, 3
- 1School of Sports and Health, Nanjing Sport Institute, Nanjing 210014, Jiangsu Province, China; 2School of Sports and Health, Shanghai University of Sport, Shanghai 200000, China; 3Jiangsu Collaborative Innovation Center for Sport and Health, Nanjing 210014, Jiangsu Province, China
-
Received:2022-10-13Accepted:2022-12-14Online:2023-12-18Published:2023-06-01 -
Contact:Lu Jiao, PhD, Lecturer, School of Sports and Health, Nanjing Sport Institute, Nanjing 210014, Jiangsu Province, China; Jiangsu Collaborative Innovation Center for Sport and Health, Nanjing 210014, Jiangsu Province, China -
About author:Zhang Liumei, Master candidate, School of Sports and Health, Nanjing Sport Institute, Nanjing 210014, Jiangsu Province, China -
Supported by:National Natural Science Foundation of China, No. 32000829 (to LL); Natural Science Research of Jiangsu Higher Education Institutions of China, No. 19KJB180019 (to LJ); Jiangsu Collaborative Innovation Center for Sport and Health, No. JSCIC-YP21003 (to LJ); National Key R&D Program of China, No. 2020YFC2007002 (to LJ)
CLC Number:
Cite this article
Zhang Liumei, Liu Jingjing, Lin Xiaoye, Liu Lin, Lu Jiao. Mechanism by which high-intensity intermittent exercise improves skeletal muscle injury and enhances exercise capacity in rats[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(35): 5603-5609.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
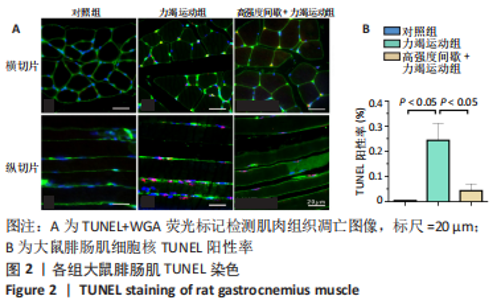
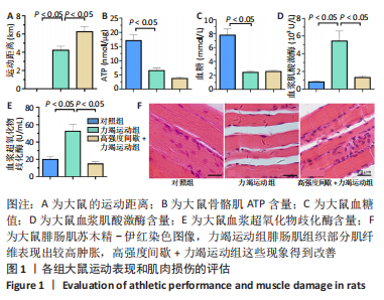
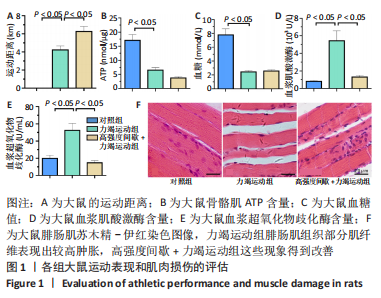
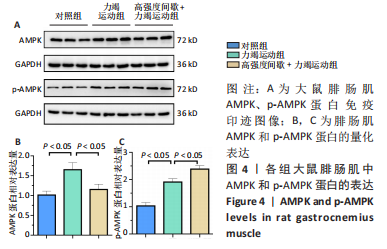
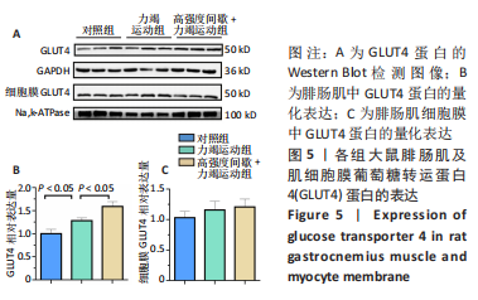
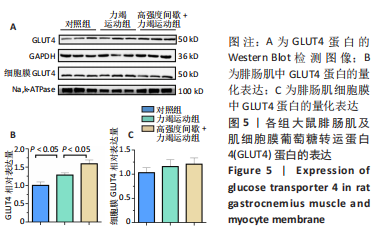
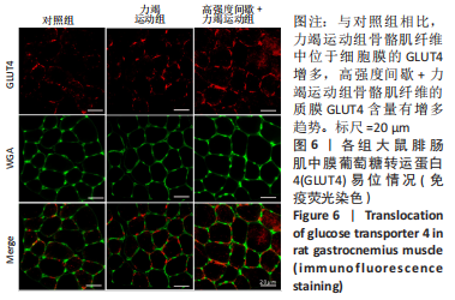
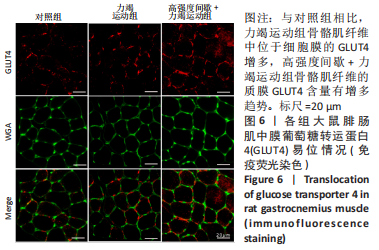
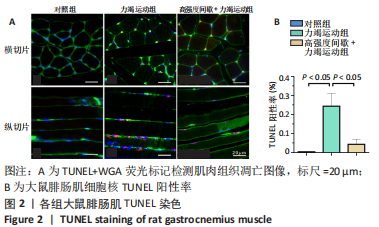
高强度间歇+力竭运动组大鼠的运动距离明显远于力竭运动组(P < 0.05),提示高强度间歇运动干预后,大鼠的运动能力提高,见图1A。与对照组比较,高强度间歇+力竭运动组和力竭运动组大鼠腓肠肌ATP浓度下降,高强度间歇+力竭运动组和力竭运动组间ATP浓度比较差异无显著性意义(P > 0.05),见图1B。血糖水平结果与ATP浓度变化趋势一致,与对照组比较,高强度间歇+力竭运动组和力竭运动组大鼠血糖水平明显下调(P < 0.05),见图1C。与对照组比较,力竭运动组大鼠血浆肌酸激酶和超氧化物歧化酶水平显著升高(P < 0.05);与力竭运动组比较,高强度间歇+力竭运动组大鼠血浆酸激酶和超氧化物歧化酶水平显著降低(P < 0.05),见图1D,E。苏木精-伊红染色显示,力竭运动组腓肠肌组织部分肌纤维表现出较高肿胀,高强度间歇+力竭运动组这些现象得到改善,见图1F。 2.3 高强度间歇运动对大鼠腓肠肌纤维凋亡情况的影响 如图2所示,在腓肠肌横切切片中,力竭运动组存在更多的梅红色细胞核;在腓肠肌骨骼肌纵切片中,一条肌纤维中存在多个核,部分核呈现梅红色荧光信号。与力竭运动组相比,高强度间歇+力竭运动组存在较少的梅红色细胞核。力竭运动组与高强度间歇+力竭运动组细胞核大部分位于细胞间质,少数位于肌细胞内。"
| [1] LI J, LI Y, ATAKAN MM, et al. The Molecular Adaptive Responses of Skeletal Muscle to High-Intensity Exercise/Training and Hypoxia. Antioxidants. 2020;9(8):656. [2] ADDIS P, SHECTERLE LM, ST CYR JA. Cellular Protection During Oxidative Stress: A Potential Role for D-ribose and Antioxidants. J Diet Suppl. 2012;9(3):178-182. [3] WILBER RL, STRAY-GUNDERSEN J, LEVINE BD. Effect of Hypoxic “Dose” on Physiological Responses and Sea-Level Performance. Med Sci Sports Exerc. 2007;39(9):1590-1599. [4] FEBBRAIO MA, HISCOCK N, SACCHETTI M, et al. Interleukin-6 Is a Novel Factor Mediating Glucose Homeostasis During Skeletal Muscle Contraction. Diabetes. 2004;53(7):1643-1648. [5] BAATI N, MOUGENOT N, LEMAITRE M, et al. Alteration of skeletal and cardiac muscles function inDBA/2J mdx mice background: a focus on high intensity interval training. Intractable Rare Dis Res. 2021;10(4):269-275. [6] RAI S, CHOWDHURY A, RENIERS R, et al. A pilot study to assess the effect of acute exercise on brain glutathione. Free Radic Res. 2018; 52(1):57-69. [7] 刘敬祺,苏浩,芦建东,等.业余冰球运动员一次性HIIT训练后混合饮料补充对唾液SIgA、LZM、AMS的影响[J].辽宁体育科技, 2022,44(3):69-74. [8] THOMSON JS, ALI A, ROWLANDS DS. Leucine-protein supplemented recovery feeding enhances subsequent cycling performance in well-trained men. Appl Physiol Nutr Metab. 2011;36(2):242-253. [9] RAHIMI M, SHEKARFOROUSH S, ASGARI AR, et al. The effect of high intensity interval training on cardioprotection against ischemia-reperfusion injury in wistar rats. EXCLI J. 2015;14:237-246. [10] 石海燕,杨珂,文安.6周HIIT对男大学生心肺耐力与身体成分影响的研究[J].福建体育科技,2020,39(2):33-35. [11] 纪天一,邱俊强.高强度间歇训练对静坐少动女大学生心肺耐力的影响[J].中国学校卫生,2020,41(8):1220-1223. [12] GORZI A, RAHMANI A, MOHAMMADI Z, et al. Effects of different lengths of high-intensity interval training microcycles on the systemic and hippocampal inflammatory state and antioxidant balance of immature rats. Mol Biol Rep. 2021;48(6):5003-5011. [13] OLÁH A, NÉMETH BT, MÁTYÁS C, et al. Cardiac effects of acute exhaustive exercise in a rat model. Int J Cardiol. 2015;182:258-266. [14] LUNDSGAARD A, FRITZEN AM, KIENS B. The Importance of Fatty Acids as Nutrients during Post-Exercise Recovery. Nutrients. 2020;12(2):280. [15] BEYFUSS K, HOOD DA. A systematic review of p53 regulation of oxidative stress in skeletal muscle. Redox Rep. 2018;23(1):100-117. [16] 曹师承,赵刚,常波,等.运动对2型糖尿病大鼠骨骼肌胰岛素信号通路PI3K/PKB磷酸化与表达的影响[J].南方医科大学学报,2010, 30(6):1217-1221. [17] 刘霞,金其贯,罗强.运动和膳食控制对2型糖尿病大鼠脂联素- AMPK-GLUT4通路的影响[J].北京体育大学学报,2013,36(1):55-58. [18] MARTINEZ-HUENCHULLAN SF, FOX SL, TAM CS, et al. Constant-moderate versus high-intensity interval training on heart adiponectin levels in high-fat fed mice: a preventive and treatment approach. Arch Physiol Biochem. 2020:1-5.doi: 10.1080/13813455.2020.1797098. [19] HOLLOSZY JO. Regulation of mitochondrial biogenesis and GLUT4 expression by exercise. Compr Physiol. 2011;1(2):921. [20] 王世强,吕万刚,常芸,等.马拉松运动对心脏的影响:生理适应和潜在风险[J].中国体育科技,2020,56(6):9-21. [21] HUNG B, SUN C, CHANG N, et al. Effects of Different Kinesio-Taping Applications for Delayed Onset Muscle Soreness after High-Intensity Interval Training Exercise: A Randomized Controlled Trial. Evid Based Complement Alternat Med. 2021;2021:1-10. [22] MARLEGA J, MICKIEWICZ A, FIJALKOWSKA J, et al. Exertional heat stroke in an amateur runner - Challenges in diagnostics and the role of unhealthy competition. J Sports Sci. 2020;38(22):2597-2602. [23] 赵亭,郭黎.冲刺间歇训练研究进展[J].中国体育科技,2022,58(2): 47-54. [24] GRANATA C, OLIVEIRA RSF, LITTLE JP, et al. Training intensity modulates changes in PGC‐1α and p53 protein content and mitochondrial respiration, but not markers of mitochondrial content in human skeletal muscle. FASEB J. 2015;30(2):959-970. [25] ABE T, KITAOKA Y, KIKUCHI DM, et al. High-intensity interval training-induced metabolic adaptation coupled with an increase in Hif-1α and glycolytic protein expression. J Appl Physiol (1985). 2015;119(11): 1297-1302. [26] LLOYD JM, MORRIS MG, JAKEMAN JR. Effect of Work: Rest Ratio on Cycling Performance Following Sprint Interval Training: A Randomized Control Trial. J Strength Cond Res. 2019;33(12):3263-3268. [27] KRUSNAUSKAS R, EIMANTAS N, BARANAUSKIENE N, et al. Response to Three Weeks of Sprint Interval Training Cannot Be Explained by the Exertional Level. Medicina (Kaunas). 2020;56(8):395. [28] 张星,李嘉,高峰.运动裨益心血管健康:从分子机制到临床应用[J].中国科学:生命科学,2022,52(2):174-189. [29] ESER P, TRACHSEL LD, MARCIN T, et al. Short- and Long-Term Effects of High-Intensity Interval Training vs. Moderate-Intensity Continuous Training on Left Ventricular Remodeling in Patients Early After ST-Segment Elevation Myocardial Infarction-The HIIT-EARLY Randomized Controlled Trial. Front Cardiovasc Med. 2022;9:869501. [30] ROTHSCHILD JA, ISLAM H, BISHOP DJ, et al. Factors Influencing AMPK Activation During Cycling Exercise: A Pooled Analysis and Meta-Regression. Sports Med. 2022;52(6):1273-1294. [31] 王林佳,苏浩,梁春瑜,等.不同时长HIIT与中等强度运动后大鼠VO2max及血液心血管风险指标的变化[J].中国体育科技,2016, 52(6):81-85. [32] KWON I, SONG W, JANG Y, et al. Elevation of hepatic autophagy and antioxidative capacity by endurance exercise is associated with suppression of apoptosis in mice. Ann Hepatol. 2020;19(1):69-78. [33] MERLE A, JOLLET M, BRITTO FA, et al. Endurance exercise decreases protein synthesis and ER-mitochondria contacts in mice skeletal. J Appl Physiol (1985). 2019;127(5):1297-1306. [34] SYLOW L, NIELSEN IL, KLEINERT M, et al. Rac1 governs exercise-stimulated glucose uptake in skeletal muscle through regulation of GLUT4 translocation in mice. J Physiol. 2016;594(17):4997-5008. [35] 李雪梅,王正珍.运动与骨骼肌葡萄糖转运通路研究进展[J].北京体育大学学报,2010,33(12):72-76. [36] CHEN S, LI A, WU J, et al. Dexmedetomidine reduces myocardial ischemia-reperfusion injury in young mice through MIF/AMPK/GLUT4 axis. BMC Anesthesiol. 2022;22(1):289. [37] 王瑞瑶,霍梦露,李超,等.小檗碱对高糖诱导心肌细胞AMPK-AS160-GLUT4通路的调节及保护作用[J]. 中国临床保健杂志,2020, 23(2):241-246. [38] KOH J, HANCOCK CR, HAN D, et al. AMPK and PPARβ positive feedback loop regulates endurance exercise training-mediated GLUT4 expression in skeletal muscle. Am J Physiol Endocrinol Metab. 2019; 316(5):E931-E939. [39] WANG T, NIU Y, LIU S, et al. Exercise improves glucose uptake in murine myotubes through the AMPKα2-mediated induction of Sestrins. Biochim Biophys Acta Mol Basis Dis. 2018;1864(10):3368-3377. [40] DEHGHAN F, HAJIAGHAALIPOUR F, YUSOF A, et al. Saffron with resistance exercise improves diabetic parameters through the GLUT4/AMPK pathway in-vitro and in-vivo. Sci Rep. 2016;6(1):189. [41] KIDO K, ATO S, YOKOKAWA T, et al. Acute resistance exercise‐induced IGF1 expression and subsequent GLUT4 translocation. Physiol Rep. 2016;4(16):e12907. |
| [1] | Yang Yifeng, Ye Nan, Wang Lin, Guo Shuaicheng, Huang Jian. Signaling pathway of dexmedetomidine against ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1464-1469. |
| [2] | Yue Yun, Wang Peipei, Yuan Zhaohe, He Shengcun, Jia Xusheng, Liu Qian, Li Zhantao, Fu Huiling, Song Fei, Jia Menghui. Effects of croton cream on JNK/p38 MAPK signaling pathway and neuronal apoptosis in cerebral ischemia-reperfusion injury rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1186-1192. |
| [3] | Shen Feiyan, Yao Jixiang, Su Shanshan, Zhao Zhongmin, Tang Weidong. Knockdown of circRNA WD repeat containing protein 1 inhibits proliferation and induces apoptosis of chondrocytes in knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 499-504. |
| [4] | Chen Zepeng, Hou Yonghui, Chen Shudong, Hou Yu, Lin Dingkun. Tauroursodeoxycholic acid treats spinal cord injury by reducing apoptosis of spinal cord neurons under glucose and oxygen deprivation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 528-534. |
| [5] | Duan Yanzhe, Hua Jianlin, Ding Zhibin, Jiang Nan, Song Lijuan, Yan Yuqing, Ma Cungen. Visual analysis of the effect of apoptosis on ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(26): 4145-4150. |
| [6] | Xie Ting, Liu Tingting, Zeng Xuehui, Li Yamin, Zhou Panghu, Yi Nianhua. Fucoxanthin alleviates glucocorticoid-induced osteoblast apoptosis by activating nuclear factor erythroid-2-related factor 2 [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(23): 3609-3614. |
| [7] | Ma Wanli, Yang Hongsheng, Qu Bo, Zhang Zhengdong, Gong Kai, Lin Yanshui. Mechanisms by which baicalein protects against steriod-induced osteonecrosis of the femoral head in rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(23): 3661-3668. |
| [8] | Zhu Yongzhao, Fang Chao, Zhao Fang, Zhang Qing, Zhao Dan. Mechanism by which lycium barbarum polysaccharides inhibit keratinocyte apoptosis in burn wounds via autophagy [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(23): 3686-3691. |
| [9] | Wang Xin, Wubulikasimu·Mijiti, Huang Jinyong, Xie Zengru. Regulation of bone tissue cells by tumor necrosis factor-alpha [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(21): 3400-3406. |
| [10] | Ning Taoli, Xie Yan, Wang Na, Wang Qingfeng, Ji Jian, Zhang Dongna. Grape seed extract inhibits apoptosis in growth plate chondrocytes and promotes tibial growth in rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3216-3222. |
| [11] | Ran Lei, Han Haihui, Xu Bo, Wang Jianye, Shen Jun, Xiao Lianbo, Shi Qi. Molecular docking analysis of the anti-inflammatory mechanism of Cibotium barometz and Epimedium for rheumatoid arthritis: animal experiment validation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 208-215. |
| [12] | Zuo Jun, Ma Shaolin. Mechanism of beta-sitosterol on hypertrophic scar fibroblasts: an analysis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 216-223. |
| [13] | Wang Qian, Lu Ziang, Li Lihe, Lyu Chaoliang, Wang Meng, Zhang Cunxin. Sinomenine effectively inhibits interleukin-1beta-induced apoptosis in nucleus pulposus cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 224-230. |
| [14] | Chen Simin, Hu Yingjun, Yan Wenrui, Ji Le, Shao Mengli, Sun Ze, Zheng Hongxing, Qi Shanshan. Establishment and evaluation of a streptozotocin-induced diabetic encephalopathy rat model [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 237-241. |
| [15] | Guo Qin, Wu Minmin, Tao Ying. Oxidized high-density lipoprotein promotes rat ovarian granulosa cell apoptosis through reactive oxygen species-initiated p38 signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(19): 3055-3060. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||