Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (23): 3661-3668.doi: 10.12307/2024.391
Previous Articles Next Articles
Mechanisms by which baicalein protects against steriod-induced osteonecrosis of the femoral head in rats
Ma Wanli, Yang Hongsheng, Qu Bo, Zhang Zhengdong, Gong Kai, Lin Yanshui
- Department of Orthopedics, First Affiliated Hospital of Chengdu Medical College, Chengdu 610500, Sichuan Province, China
-
Received:2023-03-21Accepted:2023-06-02Online:2024-08-18Published:2023-09-13 -
Contact:Lin Yanshui, Chief physician, Professor, Master’s supervisor, Department of Orthopedics, First Affiliated Hospital of Chengdu Medical College, Chengdu 610500, Sichuan Province, China -
About author:Ma Wanli, Master candidate, Physician, Department of Orthopedics, First Affiliated Hospital of Chengdu Medical College, Chengdu 610500, Sichuan Province, China -
Supported by:Chengdu Medical College - Chengdu Seventh People’s Hospital Joint Research Fund (Major Project), No. 2022LHJYZD-03 (to YHS)
CLC Number:
Cite this article
Ma Wanli, Yang Hongsheng, Qu Bo, Zhang Zhengdong, Gong Kai, Lin Yanshui. Mechanisms by which baicalein protects against steriod-induced osteonecrosis of the femoral head in rats[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(23): 3661-3668.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
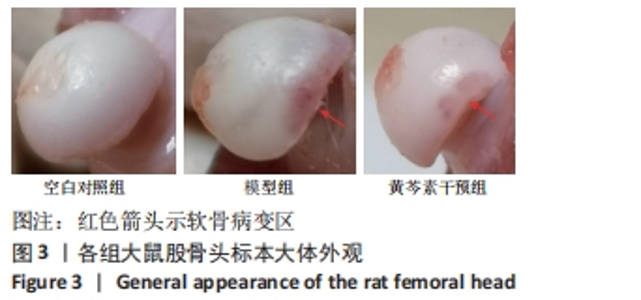
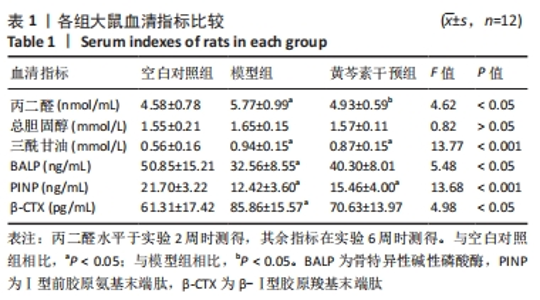
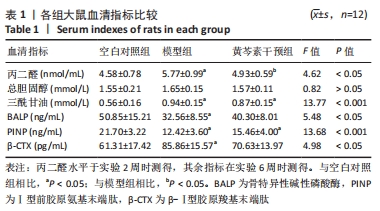
2.2.2 血脂水平 总胆固醇在第6周时组间无明显统计学差异(P > 0.05);三酰甘油的水平表现为模型组与黄芩素干预组均高于空白对照组,差异有显著性意义(P均< 0.05),但模型组与黄芩素干预组无明显组间差异(P > 0.05),提示黄芩素未能改善造模药物诱发的血脂异常。见表1。 2.2.3 骨代谢标志物水平 见表1。 BALP水平:模型组<空白对照组(P < 0.05),黄芩素干预组与另两组无明显组间差异(P > 0.05)。 PINP水平:模型组<空白对照组(P < 0.05),黄芩素干预组<空白对照组(P < 0.05),模型组与黄芩素干预组无明显组间差异(P > 0.05)。 β-CTX水平:模型组>空白对照组(P < 0.05),黄芩素干预组与另两组无明显组间差异(P > 0.05)。 由骨代谢标志物水平的组间差异可知,此次研究中黄芩素对于GIOFH大鼠模型的骨形成代谢促进作用尚不显著,骨吸收代谢的影响暂不明确。 2.3 股骨头大体外观情况 空白对照组股骨头外观结构完好,表面软骨光滑润泽呈现乳白色;模型组与空白对照组相比股骨头外形未见明显塌陷变扁,结构完整,但软骨区域较粗糙且色泽稍暗淡,部分标本在软骨周缘区域出现暗红色病变区;黄芩素干预组结构同样均完整无缺失,无明显塌陷变形,软骨质地、色泽较空白对照组无明显差异,部分标本在软骨周缘区域出现暗红色病变但与模型组相比范围较小、程度较轻,提示黄芩素的早期干预对股骨头软骨的病理改变有一定的益处。见图3。"
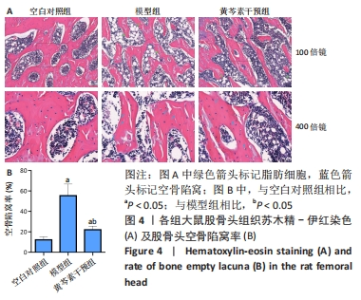
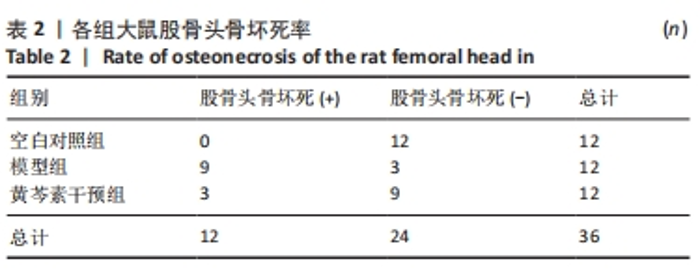
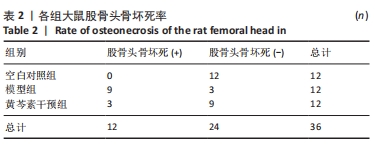
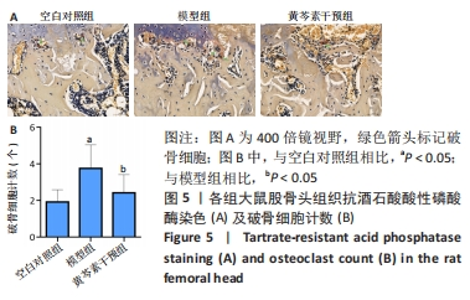
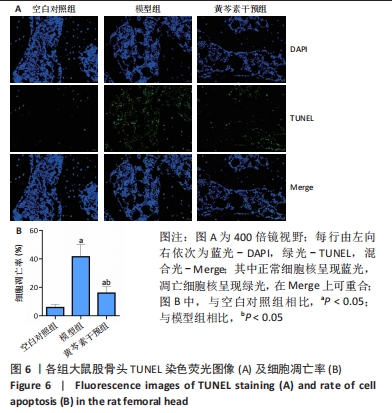
2.4 股骨头组织切片苏木精-伊红染色 2.4.1 切片观察分析 如图4A所示,空白对照组组织结构完整,可见粗壮的骨小梁及周围骨髓腔内少量脂肪细胞、大量造血细胞,骨小梁的结构连续性无明显异常,排列规整,其中偶尔可见空骨陷窝;模型组骨小梁较空白对照组明显地厚度变薄,排列稀疏,连续性出现较多中断,骨髓腔的脂肪细胞明显增多,骨小梁上的空骨陷窝明显增多;黄芩素干预组骨小梁厚度同模型组相似地比空白对照组变薄,排列较为稀疏,连续性时有中断,骨髓腔内的脂肪细胞也有增多,部分区域可见量空骨陷窝但较模型组少,提示黄芩素的早期干预对减少股骨头中空骨陷窝的形成以及骨小梁结构连续性的维持有一定改善作用。股骨头空骨陷窝率分析结果如图4B所示,模型组>黄芩素干预组>空白对照组,组间差异均有显著性意义(P < 0.05),提示黄芩素干预显著减少了空骨陷窝的形成。"

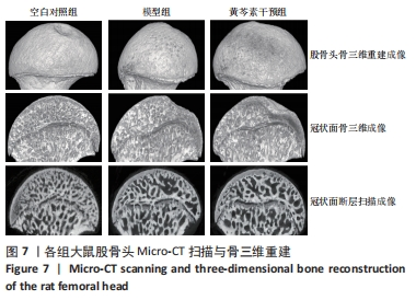
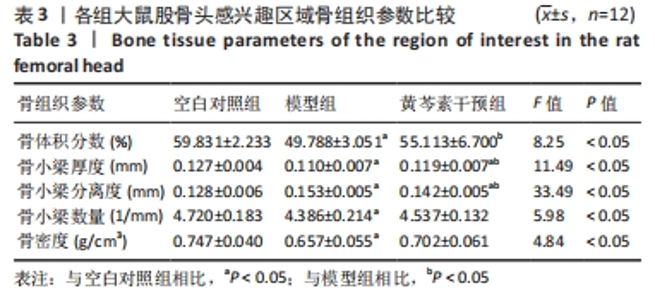
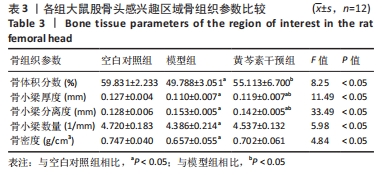
空白对照组vs.模型组:骨体积分数、骨小梁厚度、骨小梁数量、骨密度方面,空白对照组>模型组(P < 0.05);骨小梁分离度方面模型组>空白对照组(P < 0.05)。 空白对照组vs.黄芩素干预组:骨体积分数、骨小梁数量、骨密度都无明显组间差异;骨小梁厚度方面空白对照组>黄芩素干预组(P < 0.05),骨小梁分离度方面空白对照组<黄芩素干预组(P < 0.05)。 模型组vs. 黄芩素干预组:骨体积分数、骨小梁厚度方面黄芩素干预组>模型组(P < 0.05);骨小梁分离度方面黄芩素干预组<模型组(P < 0.05);骨小梁数量、骨密度方面黄芩素干预组与模型组无明显组间差异。 结果提示模型组的各项骨组织参数均下降,而黄芩素可一定程度地改善股骨头骨组织参数,主要体现在骨体积分数、骨小梁厚度、骨小梁分离度方面。 2.7.2 Micro-CT扫描与骨三维重建 3组大鼠股骨头外形均无明显变扁塌陷,但模型组与空白对照组相比可见骨小梁数量减少,部分区域骨小梁连续性断裂,骨小梁分离度增加,厚度变薄,整体上表现为明显的骨质量丢失改变;黄芩素干预组改变情况与此大致相似但程度较轻。扫描图像的组间差异与骨组织参数的组间改变基本一致,见图7。"

| [1] KURODA T, TANABE N, WAKAMATSU A, et al. High triglyceride is a risk factor for silent osteonecrosis of the femoral head in systemic lupus erythematosus. Clin Rheumatol. 2015;34(12):2071-2077. [2] HOUDEK MT, WYLES CC, PACKARD BD, et al. Decreased Osteogenic Activity of Mesenchymal Stem Cells in Patients With Corticosteroid-Induced Osteonecrosis of the Femoral Head. J Arthroplasty. 2016;31(4):893-898. [3] XIE XH, WANG XL, YANG HL, et al. Steroid-associated osteonecrosis: Epidemiology, pathophysiology, animal model, prevention, and potential treatments (an overview). J Orthop Translat. 2015;3(2):58-70. [4] KIM HJ. Glucocorticoids suppress bone formation via the osteoclast. J Clin Invest. 2006;116(8):2152-2160. [5] CHEN K, LIU Y, HE J, et al. Steroid-induced osteonecrosis of the femoral head reveals enhanced reactive oxygen species and hyperactive osteoclasts. Int J Biol Sci. 2020;16(11):1888-1900. [6] JIA D, O’BRIEN CA, STEWART SA, et al. Glucocorticoids Act Directly on Osteoclasts to Increase Their Life Span and Reduce Bone Density. Endocrinology. 2006;147(12):5592-5599. [7] ZHAN J, YAN Z, ZHAO M, et al. Allicin inhibits osteoblast apoptosis and steroid-induced necrosis of femoral head progression by activating the PI3K/AKT pathway. Food Funct. 2020;11(9):7830-7841. [8] JIA YB, JIANG DM, REN YZ, et al. Inhibitory effects of vitamin E on osteocyte apoptosis and DNA oxidative damage in bone marrow hemopoietic cells at early stage of steroid-induced femoral head necrosis. Mol Med Rep. 2017;15(4):1585-1592. [9] MONT MA, SALEM HS, PIUZZI NS, et al. Nontraumatic Osteonecrosis of the Femoral Head: Where Do We Stand Today? J Bone Joint Surg Am. 2020; 102(12):1084-1099. [10] LEE Y J, CUI Q, KOO KH. Is There a Role of Pharmacological Treatments in the Prevention or Treatment of Osteonecrosis of the Femoral Head?: A Systematic Review. J Bone Metab. 2019;26(1):13. [11] FU F, HUANG Z, YE H, et al. Mechanisms and Molecular Targets of the Tao-Hong-Si-Wu-Tang Formula for Treatment of Osteonecrosis of Femoral Head: A Network Pharmacology Study. Evid Based Complement Alternat Med. 2020;2020:1-13. [12] ZHANG P, XU H, WANG P, et al. Yougui pills exert osteoprotective effects on rabbit steroid-related osteonecrosis of the femoral head by activating β-catenin. Biomed Pharmacother. 2019;120:109520. [13] MEI R, CHEN D, ZHONG D, et al. Metabolic Profiling Analysis of the Effect and Mechanism of Gushiling Capsule in Rabbits With Glucocorticoid-Induced Osteonecrosis of the Femoral Head. Front Pharmacol. 2022;13:845856. [14] HUANG Z, FU F, YE H, et al. Chinese herbal Huo-Gu formula for the treatment of steroid-associated osteonecrosis of femoral head: A 14-year follow-up of convalescent SARS patients. J Orthop Translat. 2020;23:122-131. [15] LI H, JIANG Y, CHEN F. Separation methods used for Scutellaria baicalensis active components. J Chromatogr B Analyt Technol Biomed Life Sci. 2004; 812(1):277-290. [16] CHA MH, KIM IC, LEE BH, et al. Baicalein Inhibits Adipocyte Differentiation by Enhancing COX-2 Expression. J Med Food. 2006;9(2):145-153. [17] SEO MJ, CHOI HS, JEON HJ, et al. Baicalein inhibits lipid accumulation by regulating early adipogenesis and m-TOR signaling. Food Chem Toxicol. 2014;67:57-64. [18] RAHIMI VB, ASKARI VR, HOSSEINZADEH H. Promising influences of Scutellaria baicalensis and its two active constituents, baicalin, and baicalein, against metabolic syndrome: A review. Phytother Res. 2021;35(7):3558-3574. [19] CAI P, LU Y, YIN Z, et al. Baicalein ameliorates osteoporosis via AKT/FOXO1 signaling. Aging (Albany NY). 2021;13(13):17370-17379. [20] JIN D, LI S, TANG J, et al. Regulation of bone formation by baicalein via the mTORC1 pathway. Drug Des Devel Ther. 2015;9:5169-5183. [21] KIM JM, LEE SU, KIM YS, et al. Baicalein stimulates osteoblast differentiation via coordinating activation of MAP kinases and transcription factors. J Cell Biochem. 2008;104(5):1906-1917. [22] KIM MH, RYU SY, BAE MA, et al. Baicalein inhibits osteoclast differentiation and induces mature osteoclast apoptosis. Food Chem Toxicol. 2008;46(11): 3375-3382. [23] ZHANG X, ZHU Y, CHEN X, et al. Baicalein ameliorates inflammatory-related apoptotic and catabolic phenotypes in human chondrocytes. Int Immunopharmacol. 2014;21(2):301-308. [24] YI N, MI Y, XU X, et al. Baicalein Alleviates Osteoarthritis Progression in Mice by Protecting Subchondral Bone and Suppressing Chondrocyte Apoptosis Based on Network Pharmacology. Front Pharmacol. 2022;12:788392. [25] YUNE TY, LEE JY, CUI CM, et al. Neuroprotective effect of Scutellaria baicalensis on spinal cord injury in rats . J Neurochem. 2009;110(4):1276-1287. [26] DONG Q, CHU F, WU C, et al. Scutellaria baicalensis Georgi extract protects against alcohol-induced acute liver injury in mice and affects the mechanism of ER stress. Mol Med Rep. 2016;13(4):3052-3062. [27] DONG Y, LI Y, HUANG C, et al. Systemic application of teriparatide for steroid induced osteonecrosis in a rat model. BMC Musculoskelet Disord. 2015;16:163. [28] BAEK SH, KIM KH, LEE WK, et al. Abnormal Lipid Profiles in Nontraumatic Osteonecrosis of the Femoral Head: A Comparison with Osteoarthritis Using Propensity Score Matching. J Bone Joint Surg Am. 2022;104(Suppl 2):19-24. [29] WU X, GENG C, SUN W, et al. Incidence and Risk Factors of Osteonecrosis of Femoral Head in Multiple Myeloma Patients Undergoing Dexamethasone-Based Regimens. Biomed Res Int. 2020;2020:1-7. [30] SAAD B, GHAREEB B, KMAIL A. Metabolic and Epigenetics Action Mechanisms of Antiobesity Medicinal Plants and Phytochemicals. Evid Based Complement Alternat Med. 2021;2021:1-19. [31] WANG XH, GUO YW, TOLBA E, et al. Two-Armed Activation of Bone Mineral Deposition by the Flavones Baicalin and Baicalein, Encapsulated in Polyphosphate Microparticles. Am J Chin Med. 2017;45(3):533-555. [32] SAUL D, GLEITZ S, NGUYEN HH, et al. Effect of the lipoxygenase-inhibitors baicalein and zileuton on the vertebra in ovariectomized rats. Bone. 2017; 101:134-144. [33] 桂林源, 刘世宇, 邱新毓, 等. 黄芩小分子化合物对雌激素缺乏骨质疏松的预防和治疗作用[J]. 牙体牙髓牙周病学杂志,2016,26(5):294-300. [34] 付方胜, 丁佳昕, 邵思远, 等. 黄芩素抑制RANKL诱导的破骨细胞分化和功能[J]. 锦州医科大学学报,2019,40(2):18-20+118-119. [35] MAZUR CM, ANDRADE CDC, TOKAVANICH N, et al. Partial prevention of glucocorticoid-induced osteocyte deterioration in young male mice with osteocrin gene therapy. iScience. 2022;25(9):105019. [36] O’BRIEN CA, JIA D, PLOTKIN LI, et al. Glucocorticoids Act Directly on Osteoblasts and Osteocytes to Induce Their Apoptosis and Reduce Bone Formation and Strength. Endocrinology. 2004;145(4):1835-1841. [37] MOUTSATSOU P, KASSI E, PAPAVASSILIOU AG. Glucocorticoid receptor signaling in bone cells. Trends Mol Med. 2012;18(6):348-359. [38] FENG Z, ZHENG W, TANG Q, et al. Fludarabine inhibits STAT1-mediated up-regulation of caspase-3 expression in dexamethasone-induced osteoblasts apoptosis and slows the progression of steroid-induced avascular necrosis of the femoral head in rats. Apoptosis. 2017;22(8):1001-1012. [39] TAO SC, YUAN T, RUI BY, et al. Exosomes derived from human platelet-rich plasma prevent apoptosis induced by glucocorticoid-associated endoplasmic reticulum stress in rat osteonecrosis of the femoral head via the Akt/Bad/Bcl-2 signal pathway. Theranostics. 2017;7(3):733-750. [40] XUE X, FENG Z, LI Z, et al. Salidroside inhibits steroid-induced avascular necrosis of the femoral head via the PI3K/Akt signaling pathway: In vitro and in vivo studies. Mol Med Rep. 2017;17(3):3751-3757. [41] ZHANG S, YE J, DONG G. Neuroprotective effect of baicalein on hydrogen peroxide-mediated oxidative stress and mitochondrial dysfunction in PC12 cells. J Mol Neurosci. 2009;40(3):311-320. [42] JIN S, YANG L, MENG C, et al. Sequential Epiphyseal Cartilage Changes of Femoral Heads in C57BL/6 Female Mice Treated with Excessive Glucocorticoids. Cartilage. 2020;13(2_suppl):453S-464S. [43] LU C, QI H, XU H, et al. Global research trends of steroid-induced osteonecrosis of the femoral head: A 30-year bibliometric analysis. Front Endocrinol (Lausanne). 2022;13:1027603. [44] LU BB, LI KH. Lipoic acid prevents steroid-induced osteonecrosis in rabbits. Rheumatol Int. 2011;32(6):1679-1683. [45] IUCHI T, AKAIKE M, MITSUI T, et al. Glucocorticoid Excess Induces Superoxide Production in Vascular Endothelial Cells and Elicits Vascular Endothelial Dysfunction. Circ Res. 2003;92(1):81-87. [46] 周年, 刘波, 徐彭. 氧化应激与骨质疏松症的研究进展[J]. 中国骨质疏松杂志,2014,20(12):1485-1489. [47] 代重山, 汤树生, 张青杰, 等. 黄芩苷和黄芩素的生物学功能及其在动物生产中的应用[J]. 中国饲料,2015(8):11-14+18. [48] DOK-GO H, LEE KH, KIM HJ, et al. Neuroprotective effects of antioxidative flavonoids, quercetin, (+)-dihydroquercetin and quercetin 3-methyl ether, isolated from Opuntia ficus-indica var. saboten. Brain Res. 2003;965(1-2): 130-136. [49] GRIGALIUS I, PETRIKAITE V. Relationship between Antioxidant and Anticancer Activity of Trihydroxyflavones. Molecules. 2017;22(12):2169. [50] 李东, 宋启春, 赵研, 等. 白藜芦醇对兔激素诱导股骨头缺血性坏死抗氧化应激和抗凋亡作用的实验研究[J]. 中国骨与关节损伤杂志,2022, 37(12):1276-1280. [51] 刘靖丽, 崔莹超. 黄芩素和黄芩苷抗氧化活性与结构关系的密度泛函理论研究[J]. 云南民族大学学报(自然科学版),2017,26(6):461-464. [52] JIANG WB, ZHAO W, CHEN H, et al. Baicalin protects H9c2 cardiomyocytes against hypoxia/reoxygenation-induced apoptosis and oxidative stress through activation of mitochondrial aldehyde dehydrogenase 2. Clin Exp Pharmacol Physiol. 2017;45(3):303-311. |
| [1] | Yang Yufang, Yang Zhishan, Duan Mianmian, Liu Yiheng, Tang Zhenglong, Wang Yu. Application and prospects of erythropoietin in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1443-1449. |
| [2] | Yang Yifeng, Ye Nan, Wang Lin, Guo Shuaicheng, Huang Jian. Signaling pathway of dexmedetomidine against ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1464-1469. |
| [3] | Huang Haoran, Fan Yinuo, Wei-Yang Wenxiang, Jiang Mengyu, Fang Hanjun, Wang Haibin, Chen Zhenqiu, Liu Yuhao, Zhou Chi. Urolithin A mediates p38/MAPK pathway to inhibit osteoclast activity [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1149-1154. |
| [4] | Lou Guo, Zhang Yan, Fu Changxi. Role of endothelial nitric oxide synthase in exercise preconditioning-induced improvement of myocardial ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1283-1288. |
| [5] | Liu Jianhong, Liao Shijie, Li Boxiang, Tang Shengping, Wei Zhendi, Ding Xiaofei. Extracellular vesicles carrying non-coding RNA regulate the activation of osteoclasts [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1076-1082. |
| [6] | Pan Xiaolong, Fan Feiyan, Ying Chunmiao, Liu Feixiang, Zhang Yunke. Effect and mechanism of traditional Chinese medicine on inhibiting the aging of mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1091-1098. |
| [7] | Chen Zepeng, Hou Yonghui, Chen Shudong, Hou Yu, Lin Dingkun. Tauroursodeoxycholic acid treats spinal cord injury by reducing apoptosis of spinal cord neurons under glucose and oxygen deprivation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 528-534. |
| [8] | Wei Yuanxun, Chen Feng, Lin Zonghan, Zhang Chi, Pan Chengzhen, Wei Zongbo. The mechanism of Notch signaling pathway in osteoporosis and its prevention and treatment with traditional Chinese medicine [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 587-593. |
| [9] | Cao Sheng, Kong Lingwei, Xu Kun, Sun Zhijie. Effect of gelatin methacryloyl hydrogel loaded with salvianolic acid B on intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 380-386. |
| [10] | Zheng Jianbin, Lu Yuchun, Jiang Zixian, Li Xiufang, Wang Tao, Wang Wenjing. Yi medicine Sambucus adnata Wall methanol extract in a rat osteoarthritis model: therapeutic effect and target prediction [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(23): 3627-3635. |
| [11] | Ye Zhikui, Zhang Zhimin, Cui Linna, Jiang Xiaowen. Mechanism by which alendronate promotes rapid mandibular distraction osteogenesis in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(23): 3642-3648. |
| [12] | Han Jie, Peng Qinglin, Xu Zhiwei, Wu Yukun, Ren Guowu, Xie Xiaozhong, Jin Wanqing, Yang Ling. Active ingredients of Panax notoginseng regulate signaling pathways related to steroid-induced necrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(23): 3751-3758. |
| [13] | Zhang Jiahao, Liu Yuhao, Zhou Chi, Mo Liang, Fang Hanjun, Chen Zhenqiu. The pathological progression of steroid-induced osteonecrosis of the femoral head caused by oxidative stress-induced osteoblast ferroptosis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3202-3208. |
| [14] | Hai Zhen, Ning Zhongping. Protective effect of salidroside on angiotensin II-induced fibrosis in cardiac fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3137-3142. |
| [15] | Ji Weixiu, Bai Yi, Wang Shuo, Zhao Yungang. Protective effect and mechanism of 3-nitro-N-methyl salicylamide on the skeletal muscle of rats with limb ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3164-3169. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||