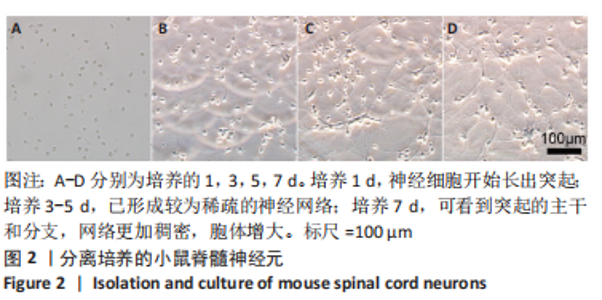
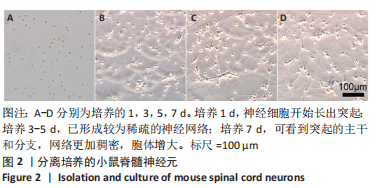
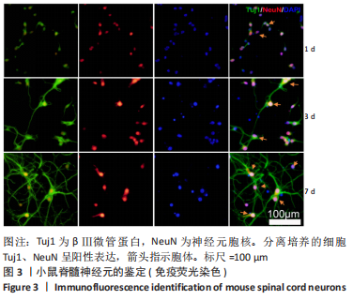
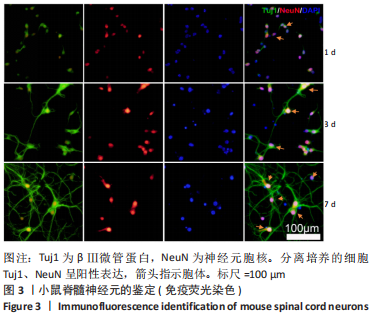
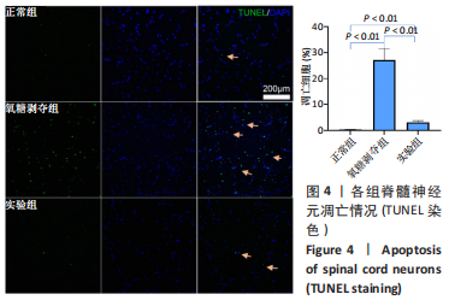
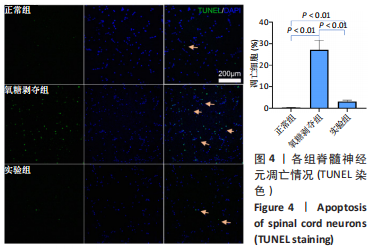
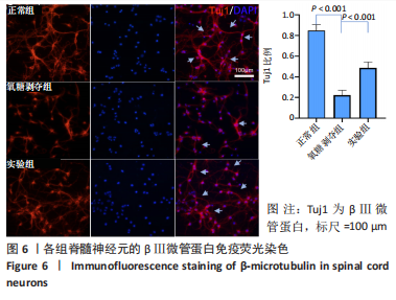
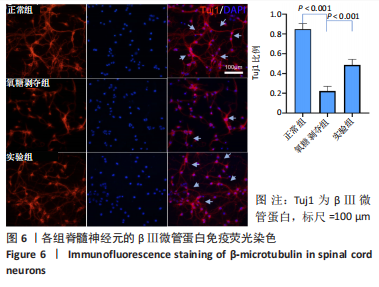
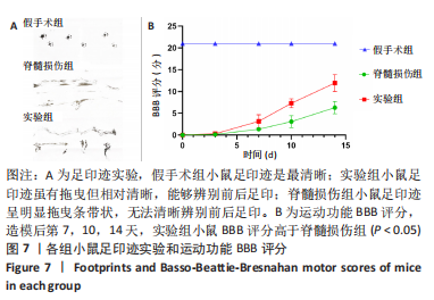
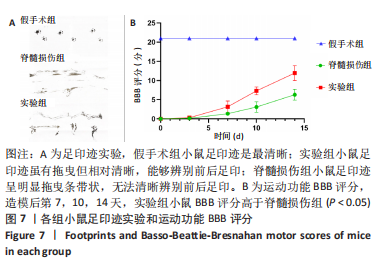
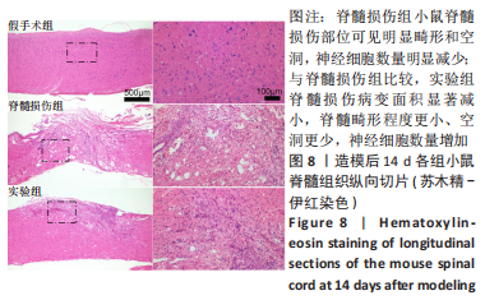
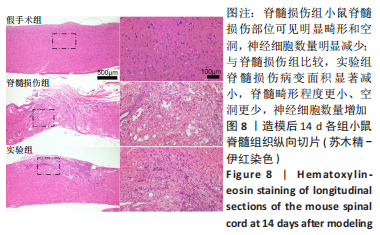
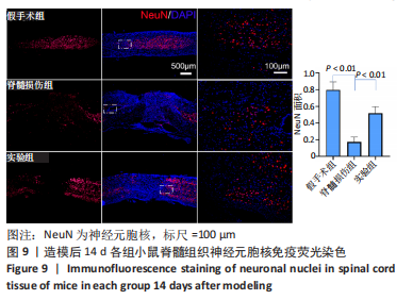
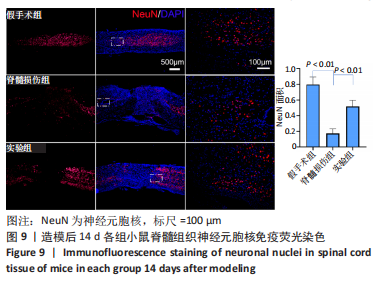
Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (4): 528-534.doi: 10.12307/2024.216
Previous Articles Next Articles
Tauroursodeoxycholic acid treats spinal cord injury by reducing apoptosis of spinal cord neurons under glucose and oxygen deprivation
Chen Zepeng1, 2, Hou Yonghui2, 3, Chen Shudong3, Hou Yu3, Lin Dingkun3
- 1The Second Clinical Medical College, Guangzhou University of Chinese Medicine, Guangzhou 510405, Guangdong Province, China; 2Lingnan Medical Research Center, Guangzhou University of Chinese Medicine, Guangzhou 510405, Guangdong Province, China; 3Department of Spinal Surgery, Guangdong Provincial Hospital of Chinese Medicine, Guangzhou 510120, Guangdong Province, China
-
Received:2023-01-17Accepted:2023-02-24Online:2024-02-08Published:2023-07-13 -
Contact:Lin Dingkun, Chief physician, Department of Spinal Surgery, Guangdong Provincial Hospital of Chinese Medicine, Guangzhou 510120, Guangdong Province, China -
About author:Chen Zepeng, MD, Attending physician, The Second Clinical Medical College, Guangzhou University of Chinese Medicine, Guangzhou 510405, Guangdong Province, China; Lingnan Medical Research Center, Guangzhou University of Chinese Medicine, Guangzhou 510405, Guangdong Province, China -
Supported by:National Natural Science Foundation of China, No. 82074451(to LDK); Natural Science Foundation of Guangdong Province, No. 2019A1515010323 (to LDK); Guangzhou Science and Technology Projects, Nos. 2023A03J0239 (to HYH) and 202102020542 (to CSD)
CLC Number:
Cite this article
Chen Zepeng, Hou Yonghui, Chen Shudong, Hou Yu, Lin Dingkun. Tauroursodeoxycholic acid treats spinal cord injury by reducing apoptosis of spinal cord neurons under glucose and oxygen deprivation[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 528-534.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
| [1] COUILLARD-DESPRES S, BIELER L, VOGL M. Pathophysiology of traumatic spinal cord injury. Neurological aspects of spinal cord injury. 2017:503-528. [2] TATOR CH, KOYANAGI I. Vascular mechanisms in the pathophysiology of human spinal cord injury. J Neurosurg. 1997;86(3):483-492. [3] KOYONAGI I, TATOR C, LEA P. Silicone rubber microangiography of acute spinal cord injury. Neurosurgery. 1993;32:260-268. [4] AGRAWAL SK, FEHLINGS MG. Mechanisms of secondary injury to spinal cord axons in vitro: role of Na+, Na (+)-K (+)-ATPase, the Na (+)-H+ exchanger, and the Na (+)-Ca2+ exchanger.J Neurosci. 1996;16(2):545. [5] DYCK S, KATARIA H, AKBARI-KELACHAYEH K, et al. LAR and PTPσ receptors are negative regulators of oligodendrogenesis and oligodendrocyte integrity in spinal cord injury. Glia. 2019;67(1):125-145. [6] CASHA S, YU W, FEHLINGS M. Oligodendroglial apoptosis occurs along degenerating axons and is associated with FAS and p75 expression following spinal cord injury in the rat. Neuroscience. 2001;103(1):203-218. [7] ALMAD A, SAHINKAYA FR, MCTIGUE DM. Oligodendrocyte fate after spinal cord injury. Neurotherapeutics. 2011;8:262-273. [8] BEATTIE MS, HERMANN GE, ROGERS RC, et al. Cell death in models of spinal cord injury. Prog Brain Res. 2002;137:37-47. [9] FUJIKAWA D, SHINMEI S, CAI B. Kainic acid-induced seizures produce necrotic, not apoptotic, neurons with internucleosomal DNA cleavage: implications for programmed cell death mechanisms. Neuroscience. 2000;98(1):41-53. [10] OYINBO CA. Secondary injury mechanisms in traumatic spinal cord injury: a nugget of this multiply cascade. Acta Neurobiol Exp (Wars). 2011;71(2):281-299. [11] VANG S, LONGLEY K, STEER CJ, et al. The unexpected uses of urso-and tauroursodeoxycholic acid in the treatment of non-liver diseases. Glob Adv Health Med. 2014;3(3):58-69. [12] CORTEZ L, SIM V. The therapeutic potential of chemical chaperones in protein folding diseases. Prion. 2014;8(2):197-202. [13] CORTEZ LM, CAMPEAU J, NORMAN G, et al. Bile acids reduce prion conversion, reduce neuronal loss, and prolong male survival in models of prion disease. J Virol. 2015;89(15):7660-7672. [14] LU X, YANG RR, ZHANG JL, et al. Tauroursodeoxycholic acid produces antidepressant‐like effects in a chronic unpredictable stress model of depression via attenuation of neuroinflammation, oxido‐nitrosative stress, and endoplasmic reticulum stress. Fundam Clin Pharmacol. 2018;32(4):363-377. [15] ZHAN J, LI X, LUO D, et al. Polydatin promotes the neuronal differentiation of bone marrow mesenchymal stem cells in vitro and in vivo: Involvement of Nrf2 signalling pathway. J Cell Mol Med. 2020;24(9):5317-5329. [16] BASSO DM, BEATTIE MS, BRESNAHAN JC. A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma. 1995;12(1):1-21. [17] RODRIGUES CM, STIEERS CL, KEENE CD, et al. Tauroursodeoxycholic acid partially prevents apoptosis induced by 3‐nitropropionic acid: evidence for a mitochondrial pathway independent of the permeability transition. J Neurochem. 2000;75(6):2368-2379. [18] CASTRO-CALDAS M, CARVALHO AN, RODRIGUES E, et al. Tauroursodeoxycholic acid prevents MPTP-induced dopaminergic cell death in a mouse model of Parkinson’s disease. Mol Neurobiol. 2012;46:475-486. [19] XIE Q, KHAOUSTOV VI, CHUNG CC, et al. Effect of tauroursodeoxycholic acid on endoplasmic reticulum stress–induced caspase‐12 activation. Hepatology. 2002;36(3):592-601. [20] VIANA R, NUNES A, CASTRO R, et al. Tauroursodeoxycholic acid prevents E22Q Alzheimer’s Aβ toxicity in human cerebral endothelial cells. Cell Mol Life Sci. 2009;66:1094-1104. [21] BREWER GJ, TORRICELLI JR. Isolation and culture of adult neurons and neurospheres. Nat Protoc. 2007;2(6):1490-1498. [22] CANNON JR, GREENAMYRE JT. NeuN is not a reliable marker of dopamine neurons in rat substantia nigra. Neurosci Lett. 2009;464(1): 14-17. [23] LEE MK, TUTTLE JB, REBHUN LI, et al. The expression and posttranslational modification of a neuron-specific β-tubulin isotype during chick embryogenesis. Cell Motil Cytoskeleton. 1990;17(2):118-132. [24] HACHEM LD, AHUJA CS, FEHLINGS MG. Assessment and management of acute spinal cord injury: From point of injury to rehabilitation. J Spinal Cord Med. 2017;40(6):665-675. [25] STEIN DM, KNIGHT WA. Emergency neurological life support: traumatic spine injury. Neurocrit Care. 2017;27:170-180. [26] KANG Y, REN X, YUAN X, et al. The effects of combined micron-scale surface and different nanoscale features on cell response. Adv Mater Sci Eng. 2018;2018:1-9. [27] CARLSON GD, GORDEN C. Current developments in spinal cord injury research. Spine J. 2002;2(2):116-128. [28] ALIZADEH A, DYCK SM, KARIMI-ABDOLREZAEE S. Traumatic spinal cord injury: an overview of pathophysiology, models and acute injury mechanisms. Front Neurol. 2019;10:282. [29] FAKHOURY M. Spinal cord injury: overview of experimental approaches used to restore locomotor activity. Rev Neurosci. 2015;26(4):397-405. [30] AHUJA CS, NORI S, TETREAULT L, et al. Traumatic spinal cord injury—repair and regeneration. Neurosurgery. 2017;80(3S):S9-S22. [31] KEENE CD, RODRIGUES CM, EICH T, et al. Tauroursodeoxycholic acid, a bile acid, is neuroprotective in a transgenic animal model of Huntington’s disease. Proc Natl Acad Sci U S A. 2002;99(16):10671-10676. [32] RODRIGUES CM, SOLÁ S, NAN Z, et al. Tauroursodeoxycholic acid reduces apoptosis and protects against neurological injury after acute hemorrhagic stroke in rats. Proc Natl Acad Sci U S A. 2003;100(10): 6087-6092. [33] NUNES AF, AMARAL JD, LO AC, et al. TUDCA, a bile acid, attenuates amyloid precursor protein processing and amyloid-β deposition in APP/PS1 mice. Mol Neurobiol. 2012;45(3):440-454. [34] GRONBECK KR, RODRIGUES CM, MAHMOUDI J, et al. Application of tauroursodeoxycholic acid for treatment of neurological and non-neurological diseases: is there a potential for treating traumatic brain injury? Neurocrit Care. 2016;25(1):153-166. [35] CASTRO-CALDAS M, CARVALHO AN, RODRIGUES E, et al. Tauroursodeoxycholic acid prevents MPTP-induced dopaminergic cell death in a mouse model of Parkinson’s disease. Mol Neurobiol. 2012;46(2):475-486. [36] JUG M, KEJŽAR N, VESEL M, et al. Neurological recovery after traumatic cervical spinal cord injury is superior if surgical decompression and instrumented fusion are performed within 8 hours versus 8 to 24 hours after injury: a single center experience. J Neurotrauma. 2015;32(18): 1385-1392. [37] FALASCA L, TISONE G, PALMIERI G, et al. PROTECTIVE ROLE OF TAUROURSODEOXYCHOLATE DURING HARVESTING AND COLD STORAGE OF HUMAN LIVER: A Pilot Study in Transplant Recipients: 1. Transplantation. 2001;71(9):1268-1276. [38] KIM SY, KWON YW, JUNG IL, et al. Tauroursodeoxycholate (TUDCA) inhibits neointimal hyperplasia by suppression of ERK via PKCα-mediated MKP-1 induction. Cardiovasc Res. 2011;92(2):307-316. [39] GUZY RD, HOYOS B, ROBIN E, et al. Mitochondrial complex III is required for hypoxia-induced ROS production and cellular oxygen sensing. Cell Metab. 2005;1(6):401-408. [40] SARNAT HB, NOCHLIN D, BORN DE. Neuronal nuclear antigen (NeuN): a marker of neuronal maturation in the early human fetal nervous system. Brain Dev. 1998;20(2):88-94. [41] HOU Y, LUAN J, HUANG T, et al. Tauroursodeoxycholic acid alleviates secondary injury in spinal cord injury mice by reducing oxidative stress, apoptosis, and inflammatory response. J Neuroinflammation. 2021;18: 1-13. [42] ZOROV DB, JUHASZOVA M, SOLLOTT SJ. Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol Rev. 2014;94(3): 909-950. [43] TRAN AP, WARREN PM, SILVER J. The biology of regeneration failure and success after spinal cord injury. Physiol Rev. 2018;98(2):881-917. [44] YANGUAS-CASáS N, BARREDA-MANSO MA, NIETO-SAMPEDRO M, et al. Tauroursodeoxycholic acid reduces glial cell activation in an animal model of acute neuroinflammation. J Neuroinflammation. 2014;11(1):50. [45] ACKERMAN HD, GERHARD GS. Bile acids in neurodegenerative disorders. Front Aging Neurosci. 2016;8:263. [46] XAVIER JM, MORGADO AL, RODRIGUES CM, et al. Tauroursodeoxycholic acid increases neural stem cell pool and neuronal conversion by regulating mitochondria-cell cycle retrograde signaling. Cell Cycle. 2014;13(22):3576-3589. [47] SOARES R, RIBEIRO FF, XAPELLI S, et al. Tauroursodeoxycholic acid enhances mitochondrial biogenesis, neural stem cell pool, and early neurogenesis in adult rats. Mol Neurobiol. 2018;55(5):3725-3738. |
| [1] | Chen Simin, Hu Yingjun, Yan Wenrui, Ji Le, Shao Mengli, Sun Ze, Zheng Hongxing, Qi Shanshan. Establishment and evaluation of a streptozotocin-induced diabetic encephalopathy rat model [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 237-241. |
| [2] | Long Qingxi, Zhang Pingshu, Liu Qing, Ou Ya, Zhang Lili, Yuan Xiaodong. Single-cell RNA sequencing reveals the heterogeneity of astrocytes [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 139-146. |
| [3] | Ma Suilu, He Zhijun, Liu Tao, Li Yan, He Yuanxu, He Bo, Wang Weiwei, Wei Xiaotao. Traditional Chinese medicine monomer in the prevention and treatment of flap necrosis by regulating “autophagy” [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 153-158. |
| [4] | Zheng Mingkui, Xue Chenhui, Guan Xiaoming, Ma Xun. Human umbilical cord mesenchymal stem cell-derived exosomes reduce the permeability of blood-spinal cord barrier after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 50-55. |
| [5] | Guo Shuhui, Yang Ye, Jiang Yangyang, Xu Jianwen. Screening and validation of neurogenic bladder miRNA-mRNA regulatory network [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-8. |
| [6] | He Wanyu, Cheng Leping. Strategies and advance on stem cell transplantation for repair of spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [7] | Yang Jiujie, Li Zhi, Wang Shujie, Tian Ye, Zhao Wei. Intraoperative neurophysiological monitoring of functional changes following durotomy with decompression for acute spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1232-1236. |
| [8] | Hao Liufang, Duan Hongmei, Wang Zijue, Hao Fei, Hao Peng, Zhao Wen, Gao Yudan, Yang Zhaoyang, Li Xiaoguang. Spatiotemporal dynamic changes of ependymal cells after spinal cord injury in transgenic mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 883-889. |
| [9] | Li Xiaoyin, Yang Xiaoqing, Chen Shulian, Li Zhengchao, Wang Ziqi, Song Zhen, Zhu Daren, Chen Xuyi. Collagen/silk fibroin scaffold combined with neural stem cells in the treatment of traumatic spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 890-896. |
| [10] | Zhang Qijian, Xu Ximing. Acquisition and application of ectodermal mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 928-934. |
| [11] | Li Zhichao, Tan Guoqing, Su Hui, Xu Zhanwang, Xue Haipeng. Regulatory role of non-coding RNAs as potential therapeutic targets in spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 758-764. |
| [12] | Wang Xiaoge, Liu Jiwen, Yang Shuai, Bao Jinyu, Li Cui. Effects of exercise on depression-like behaviors in chronic unpredictable mild stress rodent models: a systematic review and Meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 813-820. |
| [13] | Tao Xin, Xu Yi, Song Zhiwen, Liu Jinbo. Hippo signaling pathway in the regulation of spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 619-625. |
| [14] | Ding Yiqun, Li Xigong, Pan Wenming, Zhang Qin. Role and advance of mitophagy in spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(35): 5727-5733. |
| [15] | Zhou Heshan, Tan Longwang, Liu Chuang, Zhang Chi. Pretreatment methods improve the role of exosomes in spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(33): 5394-5403. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||