Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (1): 153-158.doi: 10.12307/2023.755
Previous Articles Next Articles
Traditional Chinese medicine monomer in the prevention and treatment of flap necrosis by regulating “autophagy”
Ma Suilu1, He Zhijun2, Liu Tao2, Li Yan2, He Yuanxu1, He Bo1, Wang Weiwei1, Wei Xiaotao1
- 1Clinical College of Traditional Chinese Medicine, Gansu University of Chinese Medicine, Lanzhou 730030, Gansu Province, China; 2Department of Foot and Ankle Orthopedics, Gansu Provincial Hospital of TCM, Lanzhou 730050, Gansu Province, China
-
Received:2022-10-24Accepted:2022-12-24Online:2024-01-08Published:2023-06-29 -
Contact:He Zhijun, Professor, Chief physician, Master’s supervisor, Department of Foot and Ankle Orthopedics, Gansu Provincial Hospital of TCM, Lanzhou 730050, Gansu Province, China -
About author:Ma Suilu, Master candidate, Clinical College of Traditional Chinese Medicine, Gansu University of Chinese Medicine, Lanzhou 730030, Gansu Province, China -
Supported by:National Natural Science Foundation of China, No. 81660802 (to HZJ); Key Project of Gansu Administration of Traditional Chinese Medicine, No. GZKZ-2020-2 (to HZJ); Gansu Provincial Key Research and Development Program, No. 21YF5FA021 (to HZJ); Gansu Youth Science and Technology Fund Program, No. 21JR11RA211 (to LT); Gansu Talent Innovation and Entrepreneurship Project, No. 2019-RC-63 (to HZJ)
CLC Number:
Cite this article
Ma Suilu, He Zhijun, Liu Tao, Li Yan, He Yuanxu, He Bo, Wang Weiwei, Wei Xiaotao. Traditional Chinese medicine monomer in the prevention and treatment of flap necrosis by regulating “autophagy”[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 153-158.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
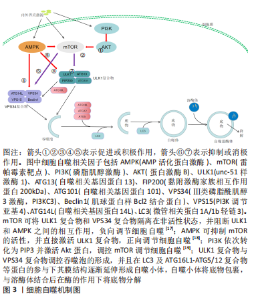
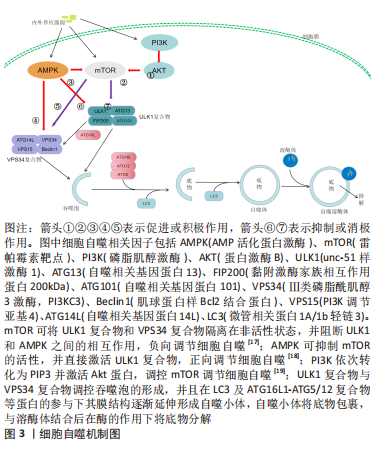
2.1 细胞自噬与皮瓣坏死概述 细胞自噬是维持细胞内环境稳态的关键,也是细胞内物质循环的参与者。在缺血再灌注损伤类疾病的研究中,激活细胞自噬可减少内皮细胞的凋亡,抑制氧化应激与炎性反应、促进微循环的建立,从而加快疾病的好转。皮瓣坏死是皮瓣移植后的严重并发症,与血管损伤、氧化应激及细胞凋亡等因素有关,促进血管再生,减少细胞凋亡与氧化损伤是防治皮瓣坏死的有利条件。因此,通过研究细胞自噬在防治皮瓣坏死中的作用,可为皮瓣坏死的防治提供一个新的思路。 2.1.1 细胞自噬概述及作用机制 细胞自噬起始于自噬小体的形成,细胞在内外因素的刺激下,上游信号分子被激活,并开始调控细胞自噬过程,而这一过程是由丝氨酸/苏氨酸蛋白激酶unc-51样激酶1 (ULK1)介导的。ULK1与自噬相关基因蛋白13(autophagy related gene,ATG13)、黏附激酶家族相互作用蛋白200 kD (FAK Family Interacting Protein of 200 kD,FIP200)以及ATG101形成ULK1特异性复合物[10];同时,ULK1复合物会易位到自噬起始位点,招募Ⅲ类磷脂酰肌醇3激酶(phosphatidylinositol 3-kinase catalytic subunit type 3,PI3KC3,也被称为VPS34)、肌球蛋白样Bcl2结合蛋白(myosin-like BCL2 interacting protein,Beclin1)、VPS15(PI3K调节亚基4,PI3KR4)和ATG14L形成VPS34复合物[11];VPS34复合物可作为一种信号分子,募集WIPI家族的PI3P结合蛋白参与吞噬体的形成并使其边缘延伸并融合,从而形成自噬小体[12]。此外,ATG家族的诸多蛋白参与细胞自噬过程。ATG9作为ATG家族中惟一已知的跨膜蛋白,通过形成的ATG9A参与自噬小体膜的形成[13]。ATG8蛋白被认为在底物识别、自噬小体的关闭和与溶酶体的融合中发挥着重要的作用,WIPI2招募ATG16L1及其ATG5-ATG12等蛋白,形成E3样ATG16L1-ATG5/12复合物,通过泛素样作用催化ATG8家族复合物与吞噬体膜中的脂质磷脂酰乙醇胺结合,控制自噬囊泡的扩张[14]。微管相关蛋白1A/1b轻链3(microtubule-associated protein 1A/1B-light chain 3,LC3)是一种可溶性蛋白质,通过与磷脂酰乙醇胺结合形成LC3-磷脂酰乙醇胺共轭物(LC3-Ⅱ),参与自噬溶酶体的形成,并反映细胞自噬的活性[15]。Beclin1不仅参与VPS34复合物的形成,还与抗凋亡蛋白Bcl2结合,形成Beclin 1-Bcl2复合物,减少细胞凋亡的同时确保细胞自噬的稳定[16]。既往研究发现,细胞自噬的调控与AMP活化蛋白激酶(AMP-activated protein kinase,AMPK)/雷帕霉素靶蛋白(mammalian target of rapamycin,mTOR)信号通路及磷脂肌醇激酶(phosphatidylinositol-3-kinase,PI3K)/蛋白激酶B(protein kinase B,AKT)信号通路密切相关[17-19],见图3。"

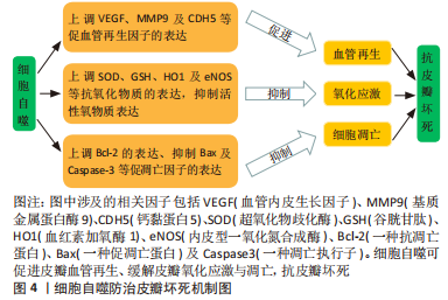
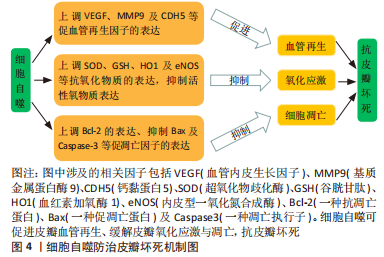
新生血管生成需要清除先前的细胞连接、促进内皮细胞有丝分裂、增殖、萌发、迁移以及成熟[20]。既往研究发现,激活细胞自噬可促进缺血皮瓣中血管内皮生长因子(vascular endothelial growth factor,VEGF)、基质金属蛋白酶9(matrix metalloproteinase 9,MMP9)以及钙黏蛋白5(cadherin5,CDH5)的表达,加快皮瓣微血管的再生与稳定[21]。并且可缓解皮瓣氧化应激与细胞凋亡,提高皮瓣存活率[22]。血管内皮生长因子是血管生成的关键因子,可通过多个途径参与新生血管萌发,促皮瓣存活[23]。MMP9可断开血管细胞间的细胞间桥,从而加强血管内皮细胞的迁移与血管的重构[24],参与新生血管的再生。CDH5又称“血管内皮钙黏蛋白(VE-cadherin)”,特异性表达于内皮细胞中的黏附连接中,可促进血管内皮细胞形成血管样结构[25]。氧化应激是体内氧化与抗氧化失衡的结果,由于细胞内超氧化物歧化酶(superoxide dismutase,SOD)、谷胱甘肽(glutathione,GSH)以及血红素加氧酶1(heme oxygenase,HO1)及内皮型一氧化氮合成酶(endothelial nitric oxide synthase,eNOS)等抗氧化物无法消除过多的活性氧类物质,最终引起皮瓣组织的氧化损伤[26-27]。细胞凋亡是由基因控制的细胞自主有序的死亡过程,主要依赖于Bcl-2家族与Caspase家族等凋亡基因的调控[28]。研究发现,过度的细胞凋亡会导致缺血缺氧皮瓣血管容量、血流速率以及皮瓣活力下降[29],引起皮瓣坏死。 2.2 调控细胞自噬防治皮瓣坏死的中药单体 临床上常用于防治皮瓣坏死的药物大多数易引起出血、颅脑水肿等不良反应。而中药具有补血活血、化瘀不伤正的特点,在防治皮瓣坏死方面具有显著的疗效。并且,随着医药学与组织工程学的不断发展,许多中药的有效成分及其防治皮瓣坏死的作用机制被逐渐发现,细胞自噬作为维持细胞内环境稳态的有利条件,在皮瓣坏死的研究中也较为深入。因此,文章总结调控细胞自噬防治皮瓣坏死的中药单体,为中药防治皮瓣坏死提供部分理论依据。 2.2.1 萜类化合物 萜类化合物是一类广泛存在于自然界的天然化合物的总称,同时也是中草药中比较重要的化合物。研究发现来源于中药的萜类化合物梓醇、三七三萜、穿心莲内酯和桦木酸可通过诱导细胞自噬防治皮瓣坏死。桦木酸又称白桦脂酸,是一种天然存在的五环三萜化合物,主要来源于中药桦木皮,具有抗炎、抗氧化和抗肿瘤的功效[30]。有研究表明桦木酸对于糖尿病创面愈合有显著的疗效[31]。LI等[32]研究桦木酸在小鼠随机皮瓣中的作用时,发现桦木酸干预的小鼠随机皮瓣组织中的LC3-Ⅱ、VPS34、Beclin1、CTSD,HO1、eNOS、SOD1、血管内皮生长因子、MMP9以及CDH5的表达显著升高,而Caspase-3、Bax、CYC以及p62等蛋白的表达显著下降,为了进一步研究桦木酸的作用机制,使用自噬抑制剂干预后,上述检测指标发生反转,这提示桦木酸可通过激活细胞自噬来抑制皮瓣组织氧化应激与凋亡,加快血管生长,发挥促皮瓣成活的作用。穿心莲内酯是中药穿心莲的生物活性成分之一,属于二萜内酯类化合物,具有抗氧化及抗凋亡等药理作用[33]。JIANG等[34]研究发现,穿心莲内酯通过调控PI3K/Akt信号通路能够显著升高小鼠皮瓣组织中的LC3II、VPS-34、Beclin1、CTSD、HO1、eNOS、SOD1、血管内皮生长因子、MMP9以及CDH5标志物的表达,降低 Bax、CYC、Caspase-3以及p62的表达,表明穿心莲内酯可促进随机皮瓣自噬与血管再生,缓解细胞凋亡与氧化应激反应,以防止皮瓣坏死,但当合并使用自噬抑制剂后,穿心莲内酯在随机皮瓣中抗氧化、抗凋亡及促血管再生的作用被抑制,提示穿心莲内酯促皮瓣成活是通过诱导细胞自噬而发挥作用的。 三七三萜是中药三七的提取物之一,具有抗炎、抗缺血的功效[35]。研究发现,三七三萜可促进体外血管生成及血管内皮生长因子蛋白表达,对缺血再灌注损伤类疾病具有积极意义[36]。HUANG等[37]通过建立小鼠随机皮瓣模型验证了三七三萜可以促进血管内皮细胞增殖和迁移,以及增加皮瓣组织血管再生与血液灌注,深入研究发现,三七三萜对随机皮瓣的保护作用是通过激活Beclin-1/VPS34/LC3信号通路,诱导细胞自噬并上调血管内皮生长因子、MMP9、CDH5、HO1、eNOS以及SOD1蛋白的表达,促进血管再生,抑制皮瓣氧化应激,从而提高随机皮瓣的成活率。梓醇是从中药地黄中提取出的一种环烯醚萜苷,具有抗炎、抗缺血及保护多巴胺神经能等功效[38]。有研究发现,梓醇不仅能上调血管内皮生长因子和血管内皮生长因子受体2表达,促进血管内皮细胞的增殖、迁移[39],还可抑制血管内皮细胞的凋亡[40],从而发挥血管保护的作用。JIANG等[41]研究发现,经梓醇干预的随机皮瓣组织中自噬相关蛋白Beclin1、LC3Ⅱ、CTSD、抗凋亡蛋白Bcl-2、抗氧化蛋白SOD1、eNOS、HO1、血管再生相关蛋白血管内皮生长因子、MMP9及CDH5的表达升高,而自噬底物蛋白p62以及促凋亡的Bax、Caspase-3蛋白表达降低,然而,抑制梓醇诱导的细胞自噬后上述蛋白的表达发生逆转,这提示梓醇可能通过激活细胞自噬以减轻随机皮瓣细胞凋亡与氧化应激,促进皮瓣血管再生,从而提高随机皮瓣的存活率;进一步研究发现,梓醇的这一保护作用可能与SIRT1-AMPK-mTOR通路的激活有关。上述研究具体汇总见表1。"


2.2.2 酚类化合物 酚类化合物是芳烃的含羟基衍生物的总称,广泛存在与自然界中,也是诸多中药内的主要成分。有研究发现,中药来源的酚类化合物白藜芦醇、姜黄素以及天麻素可通过诱导细胞自噬防治皮瓣坏死。白藜芦醇是一种非黄酮类多酚化合物,天然来源于中药虎杖,具有抗炎、抗感染、保护神经血管的功效[42]。以往的研究发现,白藜芦醇在缺血性疾病中具有良好的保护作用[43],此外,在创面的愈合方面也具有显著疗效[44]。LIN等[45]研究白藜芦醇对随机皮瓣存活的保护作用时发现,白藜芦醇能够使皮瓣组织中血管内皮生长因子、MMP9、CDH5、SOD1、eNOS及HO1过表达,而CYC、Bax、Caspase-3低表达,通过缓解皮瓣组织氧化应激与凋亡,增强皮瓣血管再生,促进随机型皮瓣的存活。此外,白藜芦醇还上调了VPS34、Beclin1及LC3Ⅱ等蛋白的表达,激活了细胞自噬,这可能是促进皮瓣存活的一个潜在机制。姜黄素是一种天然植物来源的多酚类化合物,主要来源于中药姜黄,具有抗炎、抗凝、抗肿瘤等药理作用[46]。研究发现,姜黄素发挥通过抗炎、抗氧化等功效,减轻皮瓣缺血再灌注损伤,防治皮瓣坏死[47]。TU等[48]在研究姜黄素促进缺血随机皮瓣存活时发现,经过姜黄素处理的小鼠背部皮瓣中细胞自噬被激活,Beclin1、LC3-Ⅱ、VPS34、CTSD蛋白表达显著上升,进而上调血管内皮生长因子、MMP9、CDH5、SOD1、eNOS及HO1等蛋白的表达,下调CYC、Bax、Caspase-3凋亡因子的表达,通过激活细胞自噬缓解皮瓣组织轻氧化应激与凋亡,促进皮瓣血管再生,减轻随机皮瓣的坏死,此外,该研究还证实了姜黄素通过激活细胞自噬防治皮瓣可能是通过PI3K/AKT/mTOR信号通路介导的。天麻素是一种来源于中药天麻的酚类糖苷,具有抗炎、抗凋亡以及扩张血管的药理作用,在缺血缺氧性疾病中具有显著疗效[49]。CHEN等[50]研究天麻素促进随机皮瓣存活时发现,天麻素可抑制大鼠缺血缺氧皮瓣组织中凋亡蛋白CYC、Bax、Caspase-3的表达,增加血管内皮生长因子、MMP9、CDH5、SOD1、eNOS及HO1的表达,通过促进血管再生,减轻细胞凋亡与氧化应激反应从而防治皮瓣坏死,进一步研究发现天麻素的治疗作用是通过上调自噬相关蛋白Vps34、Beclin1、LC3-Ⅱ以及CTSD的表达,激活细胞自噬而诱导产生的。上述研究具体汇总见表2。"


2.2.3 其他化合物 丹酚酸B是从中药丹参中提取出的一种水溶性的酚酸类化合物,具有抗氧化、抗凋亡及促进血管生长等药理作用[51]。有研究表明,丹酚酸B能上调缺血再灌注损伤皮瓣组织中血管内皮生长因子和SOD-1的表达,并抑制丙二醛的产生,提示丹酚酸B可通过促进血管新生、减轻氧化应激反应来保护皮瓣组织[52]。LIN等[53]研究发现丹酚酸B能够使缺血缺氧大鼠皮瓣组织中Beclin1、LC3-Ⅱ、VPS34以及CTSD蛋白的表达升高,p62以及Caspase3、Bax、CYC表达降低,同时研究发现通过激活细胞自噬可促进皮瓣组织中血管内皮生长因子、MMP9、CDH5以及SOD1、eNOS、HO1等细胞因子的表达,并降低丙二醛的含量,这表明丹酚酸B能够通过激活细胞自噬来促进血管新生,抑制细胞凋亡与氧化应激反应,从而保护随机皮瓣存活。拟人参皂甙F11是来源于中药西洋参的类固醇化合物,具有抗炎、抗氧化以及抗缺血的功效[54]。ZHOU等[55]研究发现伪人参皂甙F11不仅可以通过增强细胞自噬来促进新生血管的萌发、抑制氧化应激反应与细胞凋亡,从而减轻缺血再灌注皮瓣组织的水肿,促进其存活,深入研究后发现,拟人参皂甙可能是通过调控AMPK-MTOR信号通路促进皮瓣组织TFEB核易位,从而激活细胞自噬,提高随机皮瓣的成活率。"

| [1] FUSE Y, YAMAMOTO T, KAGEYAMA T, et al. Domino free flap transfer using a superficial circumflex iliac artery perforator flap for the toe flap donor site. Ann Plast Surg. 2022;88(3):293-297. [2] CHAMSEDDINE AH, DIB AA, WARDANI HM. The instep flap for anterior ankle coverage with bone and hardware exposure. Eur J Orthop Surg Traumatol. 2022;32(4):775-781. [3] LEE MP, GIORDANO CN, HIGGINS HW 2ND, et al. Trends in interpolated flap repairs on the nose, eyelids, ears, and lips after skin cancer excision in the United States from 2007 to 2016. Dermatol Surg. 2021;47(2): 285-286. [4] 何志军,刘涛,宋渊,等.消肿止痛合剂诱导缺氧内皮细胞再生效应观察及机制研究[J].中华中医药杂志,2020,35(6):3123-3127. [5] 章盖,耿乐乐,方勇.缺血再灌注对小鼠胸部皮瓣的影响及机制研究[J].中华损伤与修复杂志(电子版),2019,14(6):416-425. [6] 王海刚,何志军.皮瓣缺血再灌注损伤的相关机制研究进展[J].甘肃科技,2022,38(20):129-134. [7] CASSIDY LD, NARITA M. Autophagy at the intersection of aging, senescence, and cancer. Mol Oncol. 2022;16(18):3259-3275. [8] KLIONSKY DJ, PETRONI G, AMARAVADI RK, et al. Autophagy in major human diseases. EMBO J. 2021;40(19):e108863. [9] ZHU X, HU X, LOU J, et al. Liraglutide, a TFEB-mediated autophagy agonist, promotes the viability of random-pattern skin flaps. Oxid Med Cell Longev. 2021;2021:6610603. [10] ZACHARI M, GANLEY IG. The mammalian ULK1 complex and autophagy initiation. Essays Biochem. 2017;61(6):585-596. [11] STORK B, DENGJEL J. Study of ULK1 catalytic activity and its regulation. Methods Enzymol. 2017;587:391-404. [12] YANG G, SONG W, POSTOAK JL, et al. Autophagy-related protein PIK3C3/VPS34 controls T cell metabolism and function. Autophagy. 2021;17(5):1193-1204. [13] NODA NN. Atg2 and Atg9: intermembrane and interleaflet lipid transporters driving autophagy. Biochim Biophys Acta Mol Cell Biol Lipids. 2021;1866(8):158956. [14] BENTO CF, RENNA M, GHISLAT G, et al. Mammalian autophagy: how does it work? Annu Rev Biochem. 2016;85:685-713. [15] TANIDA I. Autophagosome formation and molecular mechanism of autophagy. Antioxid Redox Signal. 2011;14(11):2201-2214. [16] XU HD, QIN ZH. Beclin 1, Bcl-2 and autophagy. Adv Exp Med Biol. 2019; 1206:109-126. [17] DELEYTO-SELDAS N, EFEYAN A. The mTOR-autophagy axis and the control of metabolism. Front Cell Dev Biol. 2021;9:655731. [18] LI Y, CHEN Y. AMPK and Autophagy. Adv Exp Med Biol. 2019;1206:85-108. [19] LARSEN LJ, MØLLER LB. Crosstalk of Hedgehog and mTORC1 Pathways. Cells. 2020;9(10):2316. [20] NOWAK-SLIWINSKA P, ALITALO K, ALLEN E, et al. Consensus guidelines for the use and interpretation of angiogenesis assays. Angiogenesis. 2018; 21(3):425-532. [21] WU H, DING J, LI S, et al. Metformin promotes the survival of random-pattern skin flaps by inducing autophagy via the AMPK-mTOR-TFEB signaling pathway. Int J Biol Sci. 2019;15(2):325-340. [22] LOU J, ZHANG H, QI J, et al. Cyclic helix B peptide promotes random-pattern skin flap survival via TFE3-mediated enhancement of autophagy and reduction of ROS levels. Br J Pharmacol. 2022;179(2):301-321. [23] 姚兴璋,刘涛,何志军,等.消肿止痛合剂对大鼠缺血皮瓣血管再生及VEGF-Dll4/Notch信号通路的影响[J].暨南大学学报(自然科学与医学版),2021,42(2):164-171. [24] NISHIKAI-YAN SHEN T, KADO M, HAGIWARA H, et al. MMP9 secreted from mononuclear cell quality and quantity culture mediates STAT3 phosphorylation and fibroblast migration in wounds. Regen Ther. 2021; 18:464-471. [25] RHO SS, ANDO K, FUKUHARA S. Dynamic regulation of vascular permeability by vascular endothelial cadherin-mediated endothelial cell-cell junctions. J Nippon Med Sch. 2017;84(4):148-159. [26] 张畅. 脂肪来源干细胞及其条件培养基在防治血管内皮细胞氧化应激和皮瓣缺血再灌注中的研究[D].北京:北京协和医学院,2022. [27] 高子浚,张皓,李春霖,等.氧化应激影响移植后皮瓣愈合的研究进展[J].国际骨科学杂志,2021,42(1):35-39. [28] MAO X, CHENG R, ZHANG H, et al. Self-healing and injectable hydrogel for matching skin flap regeneration. Adv Sci (Weinh). 2018;6(3): 1801555. [29] BALI U, AYDEMIR I, KEÇECI Y, et al. Effects of oxidative stress and apoptosis on vascularity and viability of perforator flaps. Biotech Histochem. 2021;96(7):526-535. [30] AN T, ZHA W, ZI J. Biotechnological production of betulinic acid and derivatives and their applications. Appl Microbiol Biotechnol. 2020;104(8):3339-3348. [31] XIE W, HU W, HUANG Z, et al. Betulinic acid accelerates diabetic wound healing by modulating hyperglycemia-induced oxidative stress, inflammation and glucose intolerance. Burns Trauma. 2022;10:tkac007. [32] Li J, Bao G, ALyafeai E, et al. Betulinic acid enhances the viability of random-pattern skin flaps by activating autophagy. Front Pharmacol. 2019;10:1017. [33] 秦慧真,林思,邓玲玉,等.穿心莲内酯药理作用及机制研究进展[J].中国实验方剂学杂志,2022,28(6):272-282. [34] JIANG J, JIN J, LOU J, et al. Positive effect of andrographolide induced autophagy on random-pattern skin flaps survival. Front Pharmacol. 2021; 12:653035. [35] XIE W, ZHU T, DONG X, et al. HMGB1-triggered inflammation inhibition of notoginseng leaf triterpenes against cerebral ischemia and reperfusion injury via MAPK and NF-κB signaling pathways. Biomolecules. 2019;9(10):512. [36] SHI HB, CHEN JD, CHEN XH, et al. Effects of salvianolic acid and notoginseng triterpenes on angiogenesis in EA-hy926 cells in vitro. Chin J Nat Med. 2013;11(3):254-257. [37] HUANG Z, LUO X, ZHANG Y, et al. Notoginseng triterpenes inhibited autophagy in random flaps via the Beclin-1/VPS34/LC3 signaling pathway to improve tissue survival. Front Bioeng Biotechnol. 2021; 9:771066. [38] 董炤,陈长勋.梓醇药理作用的研究进展[J].中成药,2013,35(5):1047-1051. [39] 周霞,张文倩,刘炬,等.从血管内皮生长因子及其受体调控角度探讨地黄梓醇促血管新生作用及分子机制研究[J].实用心脑肺血管病杂志,2020,28(2):64-68. [40] 曹萍,程梦馨,沈丹,等.梓醇对衰老大鼠血管内皮细胞的保护作用研究[J].中西医结合心脑血管病杂志,2018,16(18):2630-2634. [41] JIANG RH, DONG CJ, CHEN ZL, et al. Catalpol enhances random-pattern skin flap survival by activating sirt1-mediated enhancement of autophagy. Oxid Med Cell Longev. 2022;2022:5668226. [42] 张琪,苏兰,何龙希,等.白藜芦醇的药理作用及其临床应用的研究进展[J].华西药学杂志,2022,37(2):214-217. [43] BREUSS JM, ATANASOV AG, UHRIN P. Resveratrol and its effects on the vascular system. Int J Mol Sci. 2019;20(7):1523. [44] HECKER A, SCHELLNEGGER M, HOFMANN E, et al. The impact of resveratrol on skin wound healing, scarring, and aging. Int Wound J. 2022;19(1):9-28. [45] LIN J, LIN R, LI S, et al. Protective effects of resveratrol on random-pattern skin flap survival: an experimental study. Am J Transl Res. 2019;11(1):379-392. [46] 陈美霓,郭巍,郝琴,等.姜黄素的药理作用、临床应用及机制研究进展[J].延安大学学报(医学科学版),2021,19(3):96-99. [47] 周全,胡建中,黄莹,等.姜黄素对岛状皮瓣缺血再灌注损伤的影响[J].湖南中医药大学学报,2019,39(1):23-27. [48] TU Z, JIANG X, LI Y, et al. Curcumin promotes the survival of ischemic random skin flaps via autophagy. Am J Transl Res. 2021;13(3):1337-1351. [49] 胡国云,姜月华,赵海霞.天麻素的含量测定、结构表征和药理活性研究进展[J].当代化工,2022,51(1):169-173. [50] CHEN H, CHEN B, LI B, et al. Gastrodin promotes the survival of random-pattern skin flaps via autophagy flux stimulation. Oxid Med Cell Longev. 2021;2021:6611668. [51] 魏西羽,杨婷,刘厚汝,等.丹酚酸B的药理作用研究进展[J].药学研究,2021,40(11):748-752. [52] 刘焕兴,季日旭,沈新升,等.丹酚酸B能促进大鼠腹部岛状皮瓣缺血再灌注损伤后的皮瓣成活[J].中华显微外科杂志,2019(6):557-561. [53] LIN J, LIN R, LI S, et al. Salvianolic acid b promotes the survival of random-pattern skin flaps in rats by inducing autophagy. Front Pharmacol. 2018;9:1178. [54] LIU YY, ZHANG TY, XUE X, et al. Pseudoginsenoside-F11 attenuates cerebral ischemic injury by alleviating autophagic/lysosomal defects. CNS Neurosci Ther. 2017;23(7):567-579. [55] ZHOU F, ZHANG X, JIANG L, et al. Pseudoginsenoside F11 enhances the viability of random-pattern skin flaps by promoting TFEB nuclear translocation through AMPK-mTOR signal pathway. Front Pharmacol. 2021;12:667524. |
| [1] | Deng Rui, Huang Keming, Luo Jian, Chen Gong, Feng Jian, Huang Weiyi, Wei Gang. Effect of heme oxygenase-1-mediated atorvastatin on macrophage polarization and cholesterol accumulation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 62-67. |
| [2] | Fang Xingyan, Tian Zhenli, Zhao Zheyi, Wen Ping, Xie Tingting. Effects of sodium arsenite on human umbilical vein endothelial cell injury and sphingosine kinases 1/sphingosine 1-phosphate signaling axis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [3] | Guo Shuhui, Yang Ye, Jiang Yangyang, Xu Jianwen. Screening and validation of neurogenic bladder miRNA-mRNA regulatory network [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-8. |
| [4] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [5] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [6] | Liang Jiaqi, Liu Hengxu, Yang Jinxin, Yang Yi, Deng Xuhui, Tan Mingjian, Luo Jiong. Health benefit relationship between exercise and intestinal bacteria [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1292-1299. |
| [7] | Ruan Ling, Wang Guanghua, Wu Rongping, Jin Zhan, Lyu Zhenqing, Zhang Nan, Li Shoubang. Correlation between exercise intensity and lipid metabolism disorder and oxidative stress in a high-diet rat model [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1149-1155. |
| [8] | Xu Yan, Li Ping, Lai Chunhua, Zhu Peijun, Yang Shuo, Xu Shulan. Piezoelectric materials for vascularized bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1126-1132. |
| [9] | Tian Qinyu, Tian Xinggui, Tian Zhuang, Sui Xiang, Liu Shuyun, Lu Xiaobo, Guo Quanyi. Protection of manganese oxide nanoparticles for bone marrow mesenchymal stem cell spreading against oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 821-826. |
| [10] | Li Long, Li Guangdi, Shi Hao, Deng Keqi. Circular RNA as a competing endogenous RNA is involved in the regulation of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 751-757. |
| [11] | Li Zhichao, Tan Guoqing, Su Hui, Xu Zhanwang, Xue Haipeng. Regulatory role of non-coding RNAs as potential therapeutic targets in spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 758-764. |
| [12] | Wu Yujie, Wan Xiaofang, Wei Mianxing, Peng Shiyuan, Xu Xiaomei. Correlation between autophagy and the Hippo-YAP protein pathway in periodental ligament cells on the pressure side of a mouse model of orthodontic tooth movement [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 683-689. |
| [13] | Xiong Zhilin, Sun Hong, Liu Miao, Zhuang Yong. Roles of ferroptosis in intervertebral disc degeneration and osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(36): 5884-5890. |
| [14] | Liang Xuezhen, Luo Di, Li Jiacheng, Wen Mingtao, Zhang Jian, Xu Bo, Li Gang. PTGS2 and STAT3 in steroid-induced osteonecrosis of the femoral head: ferroptosis-related potential diagnostic biomarkers [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(36): 5898-5904. |
| [15] | Qin Yixiong, Yuan Zijian, Xie Shanzhou, Zhu Yingwen, Chen Minghui, Han Biao, Guo Yong. Effect of tea polyphenols on proliferation and differentiation of osteoblasts stimulated by lipopolysaccharide [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(35): 5665-5669. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||