Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (20): 3137-3142.doi: 10.12307/2024.335
Previous Articles Next Articles
Protective effect of salidroside on angiotensin II-induced fibrosis in cardiac fibroblasts
Hai Zhen1, Ning Zhongping1, 2
- 1Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China; 2Department of Cardiology, Affiliated Zhoupu Hospital, Shanghai University of Medicine & Health Sciences, Shanghai 201318, China
-
Received:2023-04-20Accepted:2023-05-18Online:2024-07-18Published:2023-09-09 -
Contact:Ning Zhongping, Master, Chief physician, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China; Department of Cardiology, Affiliated Zhoupu Hospital, Shanghai University of Medicine & Health Sciences, Shanghai 201318, China -
About author:Hai Zhen, Master candidate, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China -
Supported by:Key Discipline Group Construction Project of Pudong New Area Health and Health Commission, No. PWZxq2022-11 (to NZP); Epidemiological Investigation of Atrial Fibrillation in Pudong New Area and Prospective Cohort Study on the Whole Process Management of Atrial Fibrillation under the Mode of Graded Diagnosis and Treatment, No. PKJ2021-Y33; Pudong New Area Health Committee Peak Discipline Construction, No. PWYgf2021-04 (to NZP)
CLC Number:
Cite this article
Hai Zhen, Ning Zhongping. Protective effect of salidroside on angiotensin II-induced fibrosis in cardiac fibroblasts[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3137-3142.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

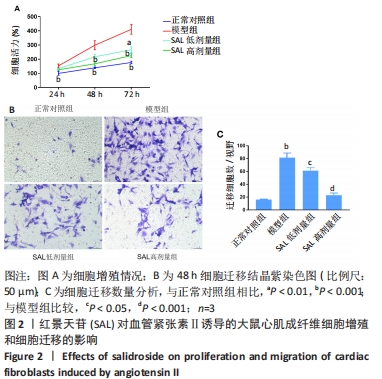
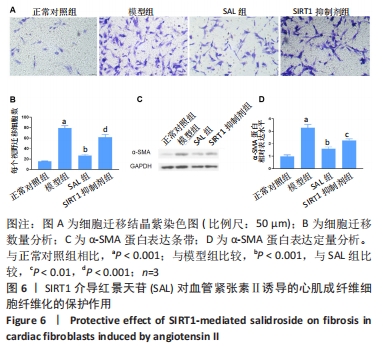
2.2.1 细胞增殖 CCK8法检测24,48 ,72 h 心脏成纤维细胞增殖情况,与正常对照组相比,模型组细胞均显著增殖,且72 h增殖率最高(P < 0.01);与模型组相比,红景天苷低剂量组于48,72 h时细胞增殖显著受到抑制(P < 0.05);红景天苷高剂量组在24,48,72 h均能明显抑制细胞的增殖(P < 0.05),且这种抑制呈剂量依赖性。 2.2.2 细胞迁移 Transwell迁移实验结果显示,与正常对照组相比,模型组细胞迁移率显著升高(P < 0.001);与模型组相比,红景天苷组细胞迁移率均显著降低,且呈剂量依赖性(P < 0.05)。 2.3 心肌成纤维细胞内活性氧生成和氧化应激结果 见图3。"
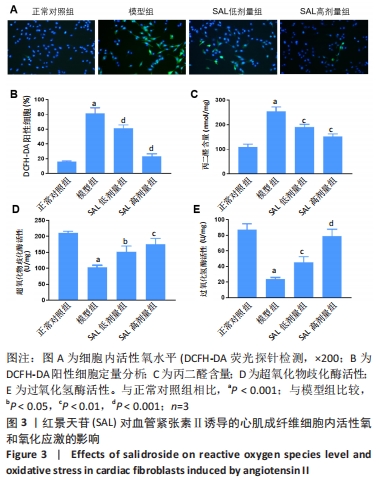

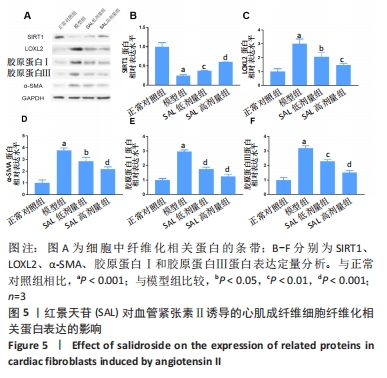
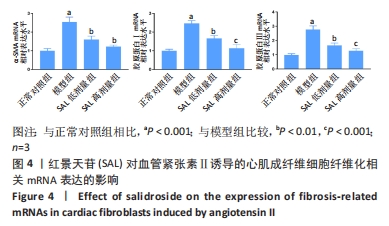
2.3.1 活性氧生成 DCFH-DA荧光探针检测心肌成纤维细胞内活性氧含量,与正常对照组相比,模型组细胞内活性氧含量显著增加(P < 0.001);与模型组相比,红景天苷组细胞内活性氧含量呈剂量依赖性减少(P < 0.001)。 2.3.2 氧化应激 采用试剂盒检测丙二醛含量、超氧化物歧化酶和过氧化氢酶活性,与正常对照组相比,模型组细胞中丙二醛含量显著增加(P < 0.001),超氧化物歧化酶和过氧化氢酶活性明显降低(P < 0.001);与模型组相比,红景天苷组细胞的丙二醛含量明显减少(P < 0.01),但无剂量依赖性,超氧化物歧化酶和过氧化氢酶活性呈剂量依赖性增加(P < 0.05)。 2.4 心肌成纤维细胞纤维化相关mRNA的表达 RT-qPCR检测结果显示,与正常对照组相比,模型组细胞中α-SMA、胶原蛋白Ⅰ、胶原蛋白Ⅲ的mRNA表达水平显著升高(P < 0.001);与模型组相比,红景天苷组细胞中α-SMA mRNA表达显著降低(P < 0.01),但无剂量依赖性,胶原蛋白Ⅰ、胶原蛋白Ⅲ的mRNA表达水平呈剂量依赖性降低(P < 0.01),见图4。"
| [1] ROTH GA, MENSAH GA, JOHNSON CO, et al. Global Burden of Cardiovascular Diseases and Risk Factors, 1990-2019: Update From the GBD 2019 Study. J Am Coll Cardiol. 2020; 76(25):2982-3021. [2] ROTH GA, MENSAH GA, FUSTER V. The Global Burden of Cardiovascular Diseases and Risks: A Compass for Global Action. J Am Coll Cardiol. 2020;76(25):2980-2981. [3] GONZALEZ A, SCHELBERT EB, DIEZ J, et al. Myocardial Interstitial Fibrosis in Heart Failure: Biological and Translational Perspectives. J Am Coll Cardiol. 2018;71(15):1696-1706. [4] QIN W, CAO L, MASSEY I Y. Role of PI3K/Akt signaling pathway in cardiac fibrosis. Mol Cell Biochem. 2021;476(11):4045-4059. [5] GARBERN JC, LEE RT. Heart regeneration: 20 years of progress and renewed optimism. Dev Cell. 2022;57(4):424-439. [6] REN C, ZHAO X, LIU K, et al. Research progress of natural medicine Astragalus mongholicus Bunge in treatment of myocardial fibrosis. J Ethnopharmacol. 2023;305: 116128. [7] LIU ZH, ZHANG Y, WANG X, et al. SIRT1 activation attenuates cardiac fibrosis by endothelial-to-mesenchymal transition. Biomed Pharmacother. 2019;118:109227. [8] ERASMUS M, SAMODIEN E, LECOUR S, et al. Linking LOXL2 to Cardiac Interstitial Fibrosis. Int J Mol Sci. 2020;21(16):5913. [9] AMES MK, ATKINS CE, PITT B. The renin-angiotensin-aldosterone system and its suppression. J Vet Intern Med. 2019;33(2):363-382. [10] MA ZG, YUAN YP, WU HM, et al. Cardiac fibrosis: new insights into the pathogenesis. Int J Biol Sci. 2018;14(12):1645-1657. [11] CONTESSOTTO P, PANDIT A. Therapies to prevent post-infarction remodelling: From repair to regeneration. Biomaterials. 2021;275:120906. [12] NI J, LI Y, XU Y, et al. Salidroside protects against cardiomyocyte apoptosis and ventricular remodeling by AKT/HO-1 signaling pathways in a diabetic cardiomyopathy mouse model. Phytomedicine. 2021;82:153406. [13] GAO H, LIU X, TIAN K, et al. Insight into the Protective Effect of Salidroside against H(2)O(2)-Induced Injury in H9C2 Cells. Oxid Med Cell Longev. 2021;2021:1060271. [14] LIEBEN LOUIS X, MEIKLE Z, CHAN L, et al. Divergent Effects of Resveratrol on Rat Cardiac Fibroblasts and Cardiomyocytes. Molecules. 2019;24(14):2604. [15] TARBIT E, SINGH I, PEART JN, et al. Increased release of serotonin from rat primary isolated adult cardiac myofibroblasts. Sci Rep.2021;11(1): 20376. [16] KUROSE H. Cardiac Fibrosis and Fibroblasts. Cells. 2021;10(7):1716. [17] MUHL L, GENOVE G, LEPTIDIS S, et al. Single-cell analysis uncovers fibroblast heterogeneity and criteria for fibroblast and mural cell identification and discrimination. Nat Commun. 2020;11(1):3953. [18] TARBIT E, SINGH I, PEART JN, et al. Biomarkers for the identification of cardiac fibroblast and myofibroblast cells. Heart Fail Rev. 2019;24(1):1-15. [19] CUI S, LIU Z, TAO B, et al. miR-145 attenuates cardiac fibrosis through the AKT/GSK-3beta/beta-catenin signaling pathway by directly targeting SOX9 in fibroblasts. J Cell Biochem. 2021;122(2):209-221. [20] HUANG C, WANG R, LU J, et al. MicroRNA-338-3p as a therapeutic target in cardiac fibrosis through FGFR2 suppression. J Clin Lab Anal. 2022;36(8):e24584. [21] KIM H, XUE X. Detection of Total Reactive Oxygen Species in Adherent Cells by 2’,7’-Dichlorodihydrofluorescein Diacetate Staining. J Vis Exp. 2020;(160): 10.3791/60682. [22] WEYDERT CJ, CULLEN JJ. Measurement of superoxide dismutase, catalase and glutathione peroxidase in cultured cells and tissue. Nat Protoc. 2010;5(1):51-66. [23] VINCENT A, THAUVIN M, QUEVRAIN E, et al. Evaluation of the compounds commonly known as superoxide dismutase and catalase mimics in cellular models. J Inorg Biochem. 2021;219:111431. [24] MIA MM, CIBI DM, GHANI S, et al. Loss of Yap/Taz in cardiac fibroblasts attenuates adverse remodelling and improves cardiac function. Cardiovasc Res. 2022;118(7):1785-1804. [25] KONG P, CHRISTIA P, FRANGOGIANNIS NG. The pathogenesis of cardiac fibrosis. Cell Mol Life Sci. 2014;71(4):549-574. [26] PAUKLIN P, ZILMER M, EHA J, et al. Markers of Inflammation, Oxidative Stress, and Fibrosis in Patients with Atrial Fibrillation. Oxid Med Cell Longev. 2022;2022:4556671. [27] REN C, LIU K, ZHAO X, et al. Research Progress of Traditional Chinese Medicine in Treatment of Myocardial fibrosis. Front Pharmacol. 2022;13:853289. [28] MAGANI SKJ, MUPPARTHI SD, GOLLAPALLI BP, et al. Salidroside - Can it be a Multifunctional Drug? . Curr Drug Metab.2020;21(7):512-524. [29] YANG S, PEI T, WANG L, et al. Salidroside Alleviates Renal Fibrosis in SAMP8 Mice by Inhibiting Ferroptosis. Molecules. 2022;27(22):8039. [30] QU B, LIU X, LIANG Y, et al. Salidroside in the Treatment of NAFLD/NASH. Chem Biodivers. 2022;19(12):e202200401. [31] MORLEY M E, RICHES K, PEERS C, et al. Hypoxic inhibition of human cardiac fibroblast invasion and MMP-2 activation may impair adaptive myocardial remodelling. Biochem Soc Trans. 2007;35(Pt 5):905-907. [32] FRANGOGIANNIS NG. Cardiac fibrosis. Cardiovasc Res. 2021;117(6):1450-1488. [33] LI X, MO N, LI Z. Ginsenosides: potential therapeutic source for fibrosis-associated human diseases. J Ginseng Res. 2020;44(3):386-398. [34] STONE RC, CHEN V, BURGESS J, et al. Genomics of Human Fibrotic Diseases: Disordered Wound Healing Response. Int J Mol Sci. 2020;21(22):8590. [35] CROSAS-MOLIST E, FABREGAT I. Role of NADPH oxidases in the redox biology of liver fibrosis. Redox Biol. 2015;6:106-111. [36] ZOROV DB, JUHASZOVA M, SOLLOTT SJ. Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol Rev. 2014;94(3):909-950. [37] RAMACHANDRA CJA, CONG S, CHAN X, et al. Oxidative stress in cardiac hypertrophy: From molecular mechanisms to novel therapeutic targets. Free Radic Biol Med. 2021; 166:297-312. [38] HAO S, SUI X, WANG J, et al. Secretory products from epicardial adipose tissue induce adverse myocardial remodeling after myocardial infarction by promoting reactive oxygen species accumulation. Cell Death Dis. 2021;12(9):848. [39] WANG CY, SUN ZN, WANG MX, et al. SIRT1 mediates salidroside-elicited protective effects against MPP(+) -induced apoptosis and oxidative stress in SH-SY5Y cells: involvement in suppressing MAPK pathways. Cell Biol Int. 2018;42(1):84-94. [40] HAN X, DING C, SANG X, et al. Targeting Sirtuin1 to treat aging-related tissue fibrosis: From prevention to therapy. Pharmacol Ther. 2022;229:107983. [41] BUGYEI-TWUM A, FORD C, CIVITARESE R, et al. Sirtuin 1 activation attenuates cardiac fibrosis in a rodent pressure overload model by modifying Smad2/3 transactivation. Cardiovasc Res. 2018;114(12):1629-1641. [42] ZHAO D, SUN X, LV S, et al. Salidroside attenuates oxidized lowdensity lipoproteininduced endothelial cell injury via promotion of the AMPK/SIRT1 pathway. Int J Mol Med. 2019;43(6):2279-2290. [43] ZEISBERG EM, TARNAVSKI O, ZEISBERG M, et al. Endothelial-to-mesenchymal transition contributes to cardiac fibrosis. Nat Med. 2007;13(8):952-961. [44] PARDALI E, SANCHEZ-DUFFHUES G, GOMEZ-PUERTO MC, et al. TGF-beta-Induced Endothelial-Mesenchymal Transition in Fibrotic Diseases. Int J Mol Sci. 2017;18(10):2157. [45] LI N, ZHOU H, MA ZG, et al. Geniposide Alleviates Isoproterenol-Induced Cardiac Fibrosis Partially via SIRT1 Activation in vivo and in vitro. Front Pharmacol. 2018;9:854. [46] ZERR P, PALUMBO-ZERR K, HUANG J, et al. Sirt1 regulates canonical TGF-beta signalling to control fibroblast activation and tissue fibrosis. Ann Rheum Dis. 2016;75(1):226-233. [47] LI S, ZHU Z, XUE M, et al. Fibroblast growth factor 21 protects the heart from angiotensin II-induced cardiac hypertrophy and dysfunction via SIRT1. Biochim Biophys Acta Mol Basis Dis. 2019;1865(6):1241-1252. [48] YANG J, SAVVATIS K, KANG JS, et al. Targeting LOXL2 for cardiac interstitial fibrosis and heart failure treatment. Nat Commun. 2016;7:13710. |
| [1] | Yang Yifeng, Ye Nan, Wang Lin, Guo Shuaicheng, Huang Jian. Signaling pathway of dexmedetomidine against ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1464-1469. |
| [2] | Shen Jiangyong, He Xi, Tang Yuting, Wang Jianjun, Liu Jinyi, Chen Yuanyuan, Wang Xinyi, Liu Tong, Sun Haoyuan. RAS-selective lethal small molecule 3 inhibits the fibrosis of pathological scar fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1168-1173. |
| [3] | Lou Guo, Zhang Yan, Fu Changxi. Role of endothelial nitric oxide synthase in exercise preconditioning-induced improvement of myocardial ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1283-1288. |
| [4] | Wang Weiqing, Zhou Yue. Chronic inflammation regulates adipose tissue fibrosis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1307-1312. |
| [5] | Sun Teng, Han Yu, Wang Shuang, Li Jialei, Cao Jimin. miR-20a regulates pressure overload-induced cardiac hypertrophy [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1021-1028. |
| [6] | Pan Xiaolong, Fan Feiyan, Ying Chunmiao, Liu Feixiang, Zhang Yunke. Effect and mechanism of traditional Chinese medicine on inhibiting the aging of mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1091-1098. |
| [7] | Cao Sheng, Kong Lingwei, Xu Kun, Sun Zhijie. Effect of gelatin methacryloyl hydrogel loaded with salvianolic acid B on intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 380-386. |
| [8] | Zhang Jiahao, Liu Yuhao, Zhou Chi, Mo Liang, Fang Hanjun, Chen Zhenqiu. The pathological progression of steroid-induced osteonecrosis of the femoral head caused by oxidative stress-induced osteoblast ferroptosis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3202-3208. |
| [9] | Yin Gonghua, Xu Ruoyao, Zhang Lijuan, Zhang Yifan, Qi Jie, Zhang Jun. Regulation of N6-methyladenosine on non-coding RNAs in pathological cardiac remodeling [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3252-2358. |
| [10] | Ji Weixiu, Bai Yi, Wang Shuo, Zhao Yungang. Protective effect and mechanism of 3-nitro-N-methyl salicylamide on the skeletal muscle of rats with limb ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3164-3169. |
| [11] | Sun Yuan, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Zhang Yan. Effects of early and late aerobic exercise on right heart failure induced by monocrotaline in rats with pulmonary hypertension [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 177-185. |
| [12] | Chen Simin, Hu Yingjun, Yan Wenrui, Ji Le, Shao Mengli, Sun Ze, Zheng Hongxing, Qi Shanshan. Establishment and evaluation of a streptozotocin-induced diabetic encephalopathy rat model [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 237-241. |
| [13] | Zhang Libing, Xu Shang, Jin Qiguan, Hu Yulong. Action mechanism of resveratrol intervention on ventricular remodeling in exercise-induced fatigue rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(16): 2587-2592. |
| [14] | Jiang Jie, Liu Juan, Yu Yanyan, Yang Yue, Wang Qianhui. Mechanism by which moxibustion pretreatment attenuates oxidative stress injury in a rat model of cerebral ischemia-reperfusion [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(16): 2488-2493. |
| [15] | Xie Wenguan, Liu Yutao, Cui Wenbo, Kuang Mingye. Melatonin inhibits hydrogen peroxide-induced injury of human nucleus pulposus cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(14): 2180-2185. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||