Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (22): 3578-3583.doi: 10.3969/j.issn.2095-4344.3153
Previous Articles Next Articles
Hydrogel as drug scaffold in skin wound repair: challenges of clinical application possibilities
Gan Lili, Xiong Na, Liu Yanfei
- Key Laboratory of Cell Engineering in Guizhou Province, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China
-
Received:2020-06-10Revised:2020-06-15Accepted:2020-07-11Online:2021-08-08Published:2021-01-21 -
Contact:Liu Yanfei, PhD, Associate researcher, Master’s supervisor, Key Laboratory of Cell Engineering in Guizhou Province, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China -
About author:Gan Lili, Master candidate, Key Laboratory of Cell Engineering in Guizhou Province, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China -
Supported by:the Project of the Health and Family Planning Commission of Guizhou Province, No. Gzwjkj2017-1-036 (to LYF); the Fund for Less Developed Regions of the National Natural Science Foundation of China, No. 31860265 (to LYF)
CLC Number:
Cite this article
Gan Lili, Xiong Na, Liu Yanfei. Hydrogel as drug scaffold in skin wound repair: challenges of clinical application possibilities[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3578-3583.
share this article
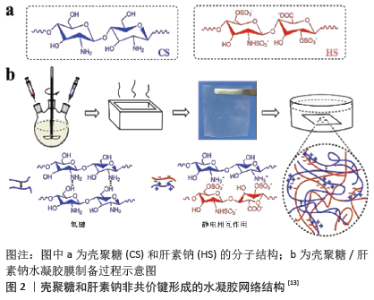
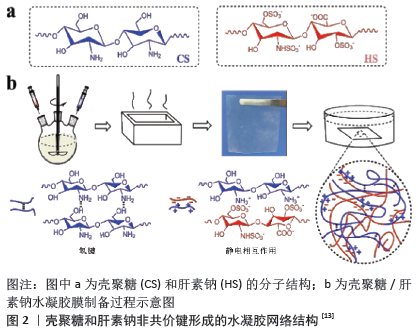
2.1 不同材料类型的水凝胶 水凝胶的分类按照结构可以分为α螺旋、β折叠、无规卷曲等;按照组成可分为短肽类、高分子类、多糖、DNA;还可以按照原料来源分为天然、合成及半合成,它既能单独使用,也能负载细胞、细胞因子和其他药物等。为处理单组分水凝胶机械强度低和功能单一的问题,两种或两种以上材料制备的复合水凝胶不失为一种良好策略[9]。在天然聚合物水凝胶材料中,临床研究较为深入的是壳聚糖、藻酸盐、胶原、纤维素和透明质酸等,这类高分子材料拥有良好的生物相容性、可降解性和细胞黏附性且代谢产物安全无毒,即使没有添加额外生物活性物质仍有明显的促愈合作用。YING等[10]研制一种由Ⅰ型胶原和透明质酸组成的可注射水凝胶,在全层创面模型实验中极大地促进了上皮化、血管新生和胶原纤维自发性形成。合成高分子基材料运用最多的是聚乙烯醇、聚乙烯吡咯烷酮、聚氨酯、聚乙二醇、聚丙烯酰胺等,虽然生物相容性不及天然材料,力学性能却更好,而且易于制备,如聚乙烯醇/聚甲基丙烯酸甲酯、聚乙烯醇/右旋乙醛、壳聚糖/肝素钠(图2)等复合水凝胶,拥有极好的溶胀、力学性能[11-13]。 "

总体来看在检索到的相关文献中,水凝胶作为缓释载体在急性创面的应用研究占比最大,约为43%。此外,负载细胞及细胞因子的研究报道在各类皮肤创面治疗选择的药物类型中均为最多,展现了许多研究者对水凝胶负载细胞或细胞因子极高的关注度。该文着重叙述水凝胶作为支架材料使负载的抗菌、清创和止血药物最大程度发挥其在各类型皮肤创面修复中的应用价值。 2.2 水凝胶在皮肤创面的应用 2.2.1 水凝胶在皮肤创面的药物递送与缓释 负载抗菌剂:如何避免创面感染是皮肤创面修复的临床难题。创面感染影响创面愈合速度,造成慢性创面,甚至还可能导致组织坏死、感染扩散,从而形成脓毒败血症。一些抗菌剂和抗生素往往存在刺激和毒副作用,研究者需要寻找更加广谱有效、安全持久、不易使细菌耐药的抗菌材料与方式。 部分天然材料来源水凝胶本身具备一定的抗感染作用,如壳聚糖、海藻酸水凝胶等[14],然而将抗菌剂分子固定在高分子载体上形成水不溶性聚合物抗菌剂会产生更加优良的抑菌效果。HOMANN等[15]制备聚乙烯吡咯烷酮碘脂质体水凝胶治疗43例深Ⅱ度烧伤患者,该水凝胶载体强化了脂质体的药物输送特性并提供了创面愈合所需的湿润环境,结果显示其抗感染效果优于洗必泰、脂肪纱布及磺胺嘧啶银软膏。OUYANG等[16]利用从罗非鱼中提取的海洋多肽制备壳聚糖-海洋多肽水凝胶敷料,该敷料具有抗菌活性,应用于烧伤动物模型可促进细胞增殖、迁移和皮肤再生。MOTAWEA等[17]设计水凝胶负载苯妥英钠脂质体治疗糖尿病足溃疡,未见不良反应且疗效显著。因此,在水凝胶中负载抗菌剂明显有助于预防创面感染,加快愈合速度。 值得一提的是水凝胶负载纳米级物质尤其是金属单质和抗菌肽,可成为创面愈合的纳米平台。MAHMOUD等[18]设计负载棒状和球状表面改性金纳米粒子的热敏水凝胶(泊洛沙姆407),具有较高的孔隙率和潜在的体外抑菌活性,延长了纳米粒子与创面的接触和释放时间,最大化促进创面皮肤细胞的快速构建和胶原的生长。MASOOD等[19]对纳米银浸渍的壳聚糖-聚乙二醇水凝胶进行研究发现,与水悬浮液中的银纳米颗粒相比,负载于水凝胶中的纳米银促愈合效果更好。LI等[20]将抗菌肽AP-57引入纳米颗粒并负载于热敏水凝胶,这种可注射原位成胶体系注射于创面后由溶胶转化为不流动的凝胶,具备低细胞毒性和持续的药物缓释特性。可引入金属氧化物和生物玻璃等抗菌材料,或与抗菌药物的聚合物微粒相结合作为水凝胶缓释体系应用于创面,以期增强水凝胶的抗感染能力和机械性能、促愈合效果[21-23]。 负载细胞及细胞因子:创面愈合是一个涉及多种细胞和细胞因子的复杂过程,这些活性因子通过不同的通路在创面修复中发挥重要作用,但如何在保持外源细胞及各种细胞因子活性的前提下大幅增加其在创面的维持时间一直是个难题,而水凝胶的可控性缓释是解决此问题的方法之一。 在三维环境下,脂肪源性干细胞内缺氧基因表达上调,增强旁分泌和自分泌效应促进血管新生和组织的再生[24-25]。XU等[26]构建功能性可注射温敏性壳聚糖水凝胶包裹人脐带间充质干细胞,结果显示可明显促进微循环、组织重塑、再上皮化和毛囊再生,抑制创面过度炎症,增加胶原沉积和角质形成标志物K1的表达,加速创面闭合。ALAPURE等[27]利用不饱和精氨酸聚酯酰胺和壳聚糖衍生物合成的杂化水凝胶负载骨髓间充质干细胞,应用于小鼠Ⅲ度烧伤创面实验,发现其能促进间充质干细胞的免疫调节功能,减轻晚期炎症反应。BURMEISTER等[28]将聚乙二醇纤维蛋白水凝胶中的同种异体脂肪干细胞作为网状自体移植物的附件,可以最大限度地减少供体部位,并通过防止收缩和增加新生血管来促进创面愈合。除了干细胞的递送,水凝胶将相关的皮肤组织细胞输送至创面也展现了良好的促愈合疗效。LOH等[29]用细菌纤维素/丙烯酸水凝胶输送人表皮角质形成细胞和真皮成纤维细胞治疗全层皮肤损伤,结果表明包埋在水凝胶中的细胞在4 h后就能转移到创面,对促使全层创面快速愈合有潜在的应用前景。SEMENIC等[30]以同种异体血小板凝胶形式激活富血小板血浆,用于治疗难愈性小腿溃疡的疗效明显优于Tegaderm敷料。综上,将干细胞或皮肤细胞在体外培养、扩增并移植到受损皮肤组织是促进修复的一个重要举施。水凝胶支架为加强细胞的活性及黏附、增殖和迁移能力提供了理想的仿生皮肤微环境,为促进皮肤创伤的快速愈合奠定了基础。 为提高传递效率,实现对生长因子的可控性输送,利用硫酸海藻酸盐、丝素蛋白等天然高分子基水凝胶作为潜在促愈合的生长因子载体能明显诱导血管再生,减轻炎症反应,上升多种活性因子的表达水平,加速皮肤组织的修复[31-32]。KIM等[33]通过明胶水凝胶负载基质衍生因子1和的鞘氨醇1磷酸激动剂诱导大量间充质干细胞和巨噬细胞的聚集,从而促进了实验小鼠皮肤缺损创面的修复。 针对创面瘢痕的形成,增强对胶原沉积、成纤维细胞的增生与凋亡的调节能力十分重要。HUANG等[34]在糖尿病小鼠皮肤创伤模型中利用酸性明胶水凝胶微球控释碱性成纤维细胞生长因子,结果表明能促进创面愈合,诱导肌成纤维细胞/成纤维细胞凋亡。LI等[35]合成人发角蛋白偶联胰岛素水凝胶,能明显抑制创面愈合后成纤维细胞的增生,预防瘢痕形成。ZHANG等[36]发现负载前列腺素E2的壳聚糖水凝胶延长了其与细胞接触的时间,维持细胞因子的结构稳定性和活性,使前列腺素E2有效参与皮肤组织修复与再生的生理过程,并抑制瘢痕增生。 负载动植物提取物:人们使用动植物来源药物治疗皮肤创伤的历史悠久,目前国内应用于各类皮肤创面的外用药物以膏剂形式的复方中药制剂多见,其中多种复杂的有效成分在创面发挥协调作用。这些具有活血化瘀、敛口止血、祛腐生肌、消肿止痛、杀菌抑菌等疗效的动植物成分,提高了创面修复的愈合效率。水凝胶作为良好的药物缓释载体和创面敷料,经其负载动植物提取物是未来创伤敷料的一大发展趋势。SH AHMED等[37]利用冻融技术制备负载积雪草苷的聚乙烯醇/聚乙二醇水凝胶,结果表明该水凝胶制剂促进创面愈合的速度比市售乳膏快15%,比未经处理的创面快40%以上,能促使成纤维细胞增殖和胶原合成,且生物相容性与安全性均可靠。DAVOODI-ROODBORDEII等[38]首次在活体模型中报道了由短梗烟堇提取物组成的水凝胶作为创面愈合促进剂的治疗潜力。 在临床应用上,强大的吸水保湿性使得水凝胶能承受一定压力而减少水分的丢失。王俊杰等[39]将白芨多糖制备的水凝胶作为天然高分子支架材料并负载当归提取物,结果显示对100例压疮患者的治疗有效率可达93%,其中四肢、外踝、足跟等部位的治疗有效率达100%。LAMMOGLIA-ORDIALES等[40]进行了一项随机双盲试验,发现载有细花含羞草树皮提取物水凝胶的治疗组有20/20的小腿静脉溃疡患者完全闭合,而对照组只有1/20的患者完全闭合。POH YUEN WEN等[41]研究发现,负载海参提取物的水凝胶在裂皮移植供体部位的治疗可与Duoderm?水凝胶相媲美。 经水凝胶缓释的提取物成分于烧伤、烫伤和糖尿病溃疡的应用广泛。例如唇形科植物种子中的百里酚成分有较强抗菌、抗炎功效,JIJI等[42]研制的百里酚细菌纤维素水凝胶可用作天然烧伤敷料,为受损组织的修复提供覆盖、保护和理想的修复条件。有研究报道,姜黄素纳米颗粒/水凝胶复合材料能解决姜黄素的低溶解度和低渗透性问题,有效提高了烧伤创面闭合率,促进肉芽组织形成、血管内皮生长因子和水通道蛋白3的表达,另有动物实验证实了这种姜黄素纳米颗粒复合凝胶能够加速糖尿病创面的修复[43-44]。AMIN等[45]发现由10%聚乙烯醇、0.6%壳聚糖和4%蜂毒组成的蜂毒水凝胶具有最佳的膨胀性、柔韧性和弹性,涂有该制剂的糖尿病大鼠创面组织显示出更高的羟脯氨酸和谷胱甘肽水平及更低的白细胞介素6水平。虽然动植物有效成分提取不易,有效剂量与毒性剂量不明确,但极大满足了医院临床对水凝胶敷料来源多样化的需求。因此,水凝胶负载动植物提取物促愈合组分在皮肤创面治疗方面有广阔的前景。水凝胶作为药物支架的缓释体系应用见表1。 2.2.2 水凝胶在皮肤创面的止血和清创作用 在创面修复的首要止血阶段,尤其是对急性皮损患者而言,迅速控制出血能避免机体遭受更大的损伤。粉剂、涂膜剂、泡沫喷剂及水凝胶等新型止血剂型较传统纱布绷带的效能更强,其中水凝胶便是这样一种具备高孔隙率和网络结构的速效止血剂,可促使创口快速凝血、闭合,还能增添抗炎和促愈合功效。比如生物相容性良好的自组装短肽有较高的黏附和弹性优势,能够不借助压力、烧灼和凝血交联剂等传统止血方法而实现快速止血效应,可被广泛应用于院前和术中止血[46]。ZHAO等[47]开发的可注射抗菌导电低温水凝胶,比明胶海绵和纱布具有更好的凝血效果、更强的血细胞和血小板黏附活化能力,可用于致命性不可压缩出血并促使创面闭合修复。此外有独特的成胶机制、良好的吸水和理化性能的可注射卡拉胶-纳米硅酸盐水凝胶,在促进创面组织再生的同时具有促凝血的巨大潜能[48-49]。水凝胶敷料对创面的强大促凝血能力有着不可忽视的潜力。 不仅如此,在对腐败溃烂创面的处理上,水凝胶清除坏死组织的疗效同样令人惊叹。已经发生坏死的皮肤组织如果不及时处理很可能导致感染的蔓延,甚至截肢和死亡。研究表明在糖尿病足溃疡坏死期使用水凝胶,其与组织接触时能连续吸收创面的渗液后形成凝胶,改换时无粘连,极大地避免了患者的疼痛[50]。通常创面清创所采用的蝇蛆生物敷料或外科削痂术的方法不尽人意,自溶性清创特性使水凝胶既能无痛清除腐烂组织和干燥创面的结痂,又因无色透明而便于观察愈合进展,是清除坏死组织的一种极佳的选择。BRITLAND等[51]设计一种负载冻干重组丝光绿蝇糜蛋白酶的流动水凝胶,经凝胶电泳定性证实糜蛋白酶/水凝胶制剂在体外能降解100 g/L的人慢性创面焦痂。自溶性清创依赖于创面组织的保持湿润,而水凝胶作为一种温和的清创剂能够促进非活性组织的再水化[52],为治疗各种急慢性皮肤创伤提供了有力保障。 "

| [1] MarketsandMarkets.com.Wound Care Market by Product (Foams, Hydrocolloids, Alginates, Antimicrobial Dressings, Assessment, NPWT Devices, Substitutes, Sutures, Staples, Tapes), Wound (Surgical, Trauma, Diabetic Ulcers, Burns), End-User, Region - Global Forecast to 2024. https://www.marketsandmarkets.com/Market-Reports/wound-care-market-371.html [2] BARROS SC, MARTINS JA, MARCOS JC, et al. Influence of secretory leukocyte protease inhibitor-based peptides on elastase activity and their incorporation in hyaluronic acid hydrogels for chronic wound therapy. Biopolymers. 2012;98(6):576-590. [3] VIG K, CHAUDHARI A, TRIPATHI S, et al. Advances in skin regeneration using tissue engineering. Int J Mol Sci. 2017;18(4):789. [4] WINTER GD. Formation of the scab and the rate of epithelisation of superficial wounds in the skin of the young domestic pig. J wound care. 1995;4(8):366-367. [5] XU Q, A S, GAO Y, et al. A hybrid injectable hydrogel from hyperbranched PEG macromer as a stem cell delivery and retention platform for diabetic wound healing. Acta Biomater. 2018;75:63-74. [6] WANG C, WANG M, XU T, et al. Engineering Bioactive Self-Healing Antibacterial Exosomes Hydrogel for Promoting Chronic Diabetic Wound Healing and Complete Skin Regeneration. Theranostics. 2019; 9(1):65-76. [7] LIU R, DAI L, SI C, et al. Antibacterial and hemostatic hydrogel via nanocomposite from cellulose nanofibers. Carbohydr Polym. 2018;195: 63-70. [8] ZHANG L, MA Y, PAN X, et al. A composite hydrogel of chitosan/heparin/poly (gamma-glutamic acid) loaded with superoxide dismutase for wound healing. Carbohydr Polym. 2018;180:168-174. [9] 韩颖,徐玉茵,田林奇,等.聚乙烯醇基水凝胶敷料的研究进展[J].中国医疗器械杂志,2018,42(6):437-439,443. [10] YING H, ZHOU J, WANG M, et al. In situ formed collagen-hyaluronic acid hydrogel as biomimetic dressing for promoting spontaneous wound healing. Mater Sci Eng C Mater Biol Appl. 2019;101:487-498. [11] HSIEH HT, CHANG HM, LIN WJ, et al. Poly-Methyl methacrylate/polyvinyl alcohol copolymer agents applied on diabetic wound dressing. Sci Rep. 2017;7(1):9531. [12] ZHENG C, LIU C, CHEN H, et al. Effective wound dressing based on poly (vinylalcohol)/dextran-aldehyde composite hydrogel. Int J Biol Macromol. 2019;132:1098-1105. [13] SHU M, LONG S, HUANG Y, et al. High strength and antibacterial polyelectrolyte complex CS/HS hydrogel films for wound healing. Soft Matter. 2019;15(38):7686-7694. [14] STRACCIA MC, D’AYALA GG, ROMANO I, et al. Alginate hydrogels coated with chitosan for wound dressing. Mar Drugs. 2015;13(5):2890-2908. [15] HOMANN HH, ROSBACH O, MOLL W, et al. A liposome hydrogel with polyvinyl-pyrrolidone iodine in the local treatment of partial-thickness burn wounds. Ann Plast Surg. 2007;59(4):423-427. [16] Ouyang QQ, Hu Z, Lin ZP, et al. Chitosan hydrogel in combination with marine peptides from tilapia for burns healing. Int J Biol Macromol. 2018;112:1191-1198. [17] MOTAWEA A, ABD EL-GAWAD AEH, BORG T, et al. The impact of topical phenytoin loaded nanostructured lipid carriers in diabetic foot ulceration. Foot (Edinb). 2019;40:14-21. [18] MAHMOUD NN, HIKMAT S, ABU GHITH D, et al. Gold nanoparticles loaded into polymeric hydrogel for wound healing in rats: Effect of nanoparticles’ shape and surface modification. Int J Pharm. 2019; 565:174-186. [19] MASOOD N, AHMED R, TARIQ M, et al. Silver nanoparticle impregnated chitosan-PEG hydrogel enhances wound healing in diabetes induced rabbits. Int J Pharm. 2019;559:23-36. [20] LI X, FAN R, TONG A, et al. In situ gel-forming AP-57 peptide delivery system for cutaneous wound healing. Int J Pharm. 2015;495(1): 560-571. [21] ZHAI M, XU Y, ZHOU B, et al. Keratin-chitosan/n-ZnO nanocomposite hydrogel for antimicrobial treatment of burn wound healing: Characterization and biomedical application. J Photochem Photobiol B. 2018;180:253-258. [22] KONG L, WU Z, ZHAO H, et al. Bioactive Injectable Hydrogels Containing Desferrioxamine and Bioglass for Diabetic Wound Healing. ACS Appl Mater Interfaces. 2018;10(36):30103-30114. [23] YASASVINI S, ANUSA RS, VEDHAHARI BN, et al. Topical hydrogel matrix loaded with Simvastatin microparticles for enhanced wound healing activity. Mater Sci Eng C Mater Biol Appl. 2017;72:160-167. [24] 凌建民,田爱玲,范鲁峰,等.脂肪间充质干细胞在功能化自组装纳米多肽水凝胶三维培养下旁分泌的研究[J].中华实验外科杂志, 2018,35(2):250-252. [25] KUO YR, WANG CT, CHENG JT, et al. Adipose-Derived Stem Cells Accelerate Diabetic Wound Healing through the Induction of Autocrine and Paracrine Effects. Cell Transplant. 2016;25(1):71-81. [26] XU H, HUANG S, WANG J, et al. Enhanced cutaneous wound healing by functional injectable thermo-sensitive chitosan-based hydrogel encapsulated human umbilical cord-mesenchymal stem cells. Int J Biol Macromol. 2019;137:433-441. [27] ALAPURE BV, LU Y, HE M, et al. Accelerate healing of severe burn wounds by mouse bone marrow mesenchymal stem cell-seeded biodegradable hydrogel scaffold synthesized from arginine-based poly(ester amide) and chitosan. Stem Cells Dev. 2018;27(23): 1605-1620. [28] BURMEISTER DM, STONE R, WRICE N, et al. Delivery of allogeneic adipose stem cells in polyethylene glycol-fibrin hydrogels as an adjunct to meshed autografts after sharp debridement of deep partial thickness burns. Stem Cells Transmed. 2018;7(4):360-372. [29] LOH EYX, MOHAMAD N, FAUZI MB, et al. Development of a bacterial cellulose-based hydrogel cell carrier containing keratinocytes and fibroblasts for fullthickness wound healing. Sci Rep. 2018;8(1):2875. [30] SEMENIC D, CIRMAN T, ROZMAN P, et al. Regeneration of chronic wounds with allogeneic platelet gel versus hydrogel treatment: A prospective study. Acta Clin Croat. 2018;57(3):434-442. [31] BABAVALIAN H, TEBYANIAN H, LATIFI AM, et al. The effect of synthetic alginate sulfate hydrogels with recombinant PDGF-BB on wound healing. BratislLek Listy. 2018;119(6):391-396. [32] Xu HL, Chen PP, ZhuGe DL, et al. Liposomes with silk fibroin hydrogel core to stabilize bFGF and promote the wound healing of mice with deep second-degree scald. Adv Healthc Mater. 2017;6(19). doi: 10.1002/adhm.201700344. [33] KIM YH, TABATA Y. Recruitment of mesenchymal stem cells and macrophages by dual release of stromal cell-derived factor-1 and a macrophage recruitment agent enhances wound closure. J Biomed Mater Res A. 2016;104(4):942-956. [34] HUANG C, ORBAY H, TOBITA M, et al. Proapoptotic effect of control-released basic fibroblast growth factor on skin wound healing in a diabetic mouse model. Wound Repair Regen. 2016;24(1):65-74. [35] LI W, GAO F, KAN J, et al. Synthesis and fabrication of a keratin-conjugated insulin hydrogel for the enhancement of wound healing. Colloids Surf B Biointerfaces. 2019;175:436-444. [36] ZHANG S, LIU Y, ZHANG X, et al. Prostaglandin E2 hydrogel improves cutaneous wound healing via M2 macrophages polarization. Theranostics. 2018;8(19):5348-5361. [37] SH AHMED A, TAHER M, MANDAL UK, et al. Pharmacological properties of Centella asiatica hydrogel in accelerating wound healing in rabbits. BMC Complement Alter Med. 2019;19(1):213. [38] DAVOODI-ROODBORDEII F, AFSHAR M, HAJI ABAS TABRIZI F, et al. Topical hydrogel containing Fumaria vaillantii Loisel. extract enhances wound healing in rats. BMC Complement Altern Med. 2019;19(1):254. [39] 王俊杰,王刚,益伟清,等.白及水凝胶敷料应用于Ⅰ Ⅱ期压疮的效果评价[J].中国药物与临床,2019,19(3):514-515. [40] LAMMOGLIA-ORDIALES L, VEGA-MEMIJE ME, HERRERA-ARELLANO A, et al. A randomised comparative trial on the use of a hydrogel with tepescohuite extract (Mimosa tenuiflora cortex extract-2G) in the treatment of venous leg ulcers. Int Wound J. 2012;9(4):412-418. [41] POH YUEN WEN A, HALIM AS, MAT SAAD AZ, et al. A prospective study evaluating wound healing with sea cucumber gel compared with hydrogel in treatment of skin graft donor sites. Complement Ther Med. 2018;41:261-266. [42] JIJI S, UDHAYAKUMAR S, ROSE C, et al. Thymol enriched bacterial cellulose hydrogel as effective material for third degree burn wound repair. Int J Biol Macromol. 2019;122:452-460. [43] DANG LH, NGUYEN TH, TRAN HLB, et al. Injectable nanocurcumin-formulated chitosan-g-pluronic hydrogel exhibiting a great potential for burn treatment. J Healthc Eng. 2018;2018:5754890. [44] KAMAR SS, ABDEL-KADER DH, RASHED LA. Beneficial effect of curcumin nanoparticls-hydrogel on excisional skin wound healing in type-I diabetic rat: Histological and immunohistochemical studies. Ann Anat. 2019;222:94-102. [45] AMIN MA, ABDEL-RAHEEM IT. Accelerated wound healing and anti-inflammatory effects of physically cross linked polyvinyl alcohol-chitosan hydrogel containing honey bee venom in diabetic rats. Arch Pharm Res. 2014;37(8):1016-1031. [46] 卫巍,刘燕飞,张玲,等.自组装短肽水凝胶:止血效应与机制研究进展[J].中国组织工程研究,2019,23(2):310-316. [47] ZHAO X, GUO B, WU H, et al. Injectable antibacterial conductive nanocomposite cryogels with rapid shape recovery for noncompressible hemorrhage and wound healing. Nat Commun. 2018;9(1):2784. [48] LOKHANDE G, CARROW JK, THAKUR T, et al. Nanoengineered injectable hydrogels for wound healing application. Acta Biomater. 2018;70:35-47. [49] YEGAPPAN R, SELVAPRITHIVIRAJ V, AMIRTHALINGAM S, et al. Carrageenan based hydrogels for drug delivery, tissue engineering and wound healing. Carbohydr Polym. 2018;198:385-400. [50] 聂海英,李昆,张全英.湿性敷料在老年糖尿病足溃疡换药中的应用[J].中国老年学杂志,2015,35(13):3582-3584. [51] BRITLAND S, SMITH A, FINTER W, et al. Recombinant Lucilia sericata chymotrypsin in a topical hydrogel formulation degrades human wound eschar ex vivo. Biotechnol Prog. 2011;27(3):870-874. [52] EDWARDS J. Hydrogels and their potential uses in burn wound management. Br J Nurs. 2010;19:12-16. [53] NALAMPANG K, PANJAKHA R, MOLLOY R, et al. Structural effects in photopolymerized sodium AMPS hydrogels crosslinked with poly(ethylene glycol) diacrylate for use as burn dressings. J Biomater Sci Polym Ed. 2013;24(11):1291-1304. [54] ZHU J, LI F, WANG X, et al. Hyaluronic Acid and Polyethylene Glycol Hybrid Hydrogel Encapsulating Nanogel with Hemostasis and Sustainable Antibacterial Property for Wound Healing. ACS Appl Mater Interfaces. 2018;10(16):13304-13316. [55] JIANG B, LARSON JC, DRAPALA PW, et al. Investigation of lysine acrylate containing poly(N-isopropylacrylamide) hydrogels as wound dressings in normal and infected wounds. J Biomed Mater Res B Appl Biomater. 2012;100(3):668-676. [56] HASEEB MT, HUSSAIN MA, ABBAS K, et al. Linseed hydrogel-mediated green synthesis of silver nanoparticles for antimicrobial and wound-dressing applications. Int J Nanomedicine. 2017;12:2845-2855. [57] JIN SG, KIM KS, KIM DW, et al. Development of a novel sodium fusidate-loaded triple polymer hydrogel wound dressing: Mechanical properties and effects on wound repair. Int J Pharm. 2016;497(1-2):114-122. [58] ZMEJKOSKI D, SPASOJEVIĆ D, ORLOVSKA I, et al. Bacterial cellulose-lignin composite hydrogel as a promising agent in chronic wound healing. Int J Biol Macromol. 2018;118(Pt A):494-503. [59] DIMATTEO R, DARLING NJ , SEGURA T. In situ forming injectable hydrogels for drug delivery and wound repair. Adv Drug Deliv Rev. 2018;127:167-184. [60] BURDICK JA, MAUCK RL, GERECHT S. To Serve and Protect: Hydrogels to Improve Stem Cell-Based Therapies. Cell Stem Cell. 2016;18(1):13-15. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [3] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [4] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [5] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [6] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [7] | Jiang Hongying, Zhu Liang, Yu Xi, Huang Jing, Xiang Xiaona, Lan Zhengyan, He Hongchen. Effect of platelet-rich plasma on pressure ulcers after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1149-1153. |
| [8] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [9] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [10] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [11] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| [12] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [13] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [14] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [15] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||