Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (16): 2562-2568.doi: 10.3969/j.issn.2095-4344.3131
Previous Articles Next Articles
Enzyme-responsive nanoparticles in tumor therapy: superiority of nanoparticles in accumulation and drug release
Gan Zhoujie, Pei Xibo
- Repair Department I, West China Hospital of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China
-
Received:2020-06-02Revised:2020-06-05Accepted:2020-07-06Online:2021-06-08Published:2021-01-07 -
Contact:Pei Xibo, Associate professor, Repair Department I, West China Hospital of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China -
About author:Gan Zhoujie, Master candidate, Repair Department I, West China Hospital of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China -
Supported by:the National Youth Science Fund Project, No. 81601613 (to PXB); the Youth Clinical Research Fund of Chinese Stomatological Association, No. CSA-B2018-09 (to PXB)
CLC Number:
Cite this article
Gan Zhoujie, Pei Xibo. Enzyme-responsive nanoparticles in tumor therapy: superiority of nanoparticles in accumulation and drug release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2562-2568.
share this article
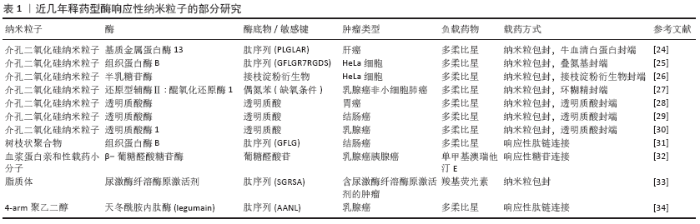
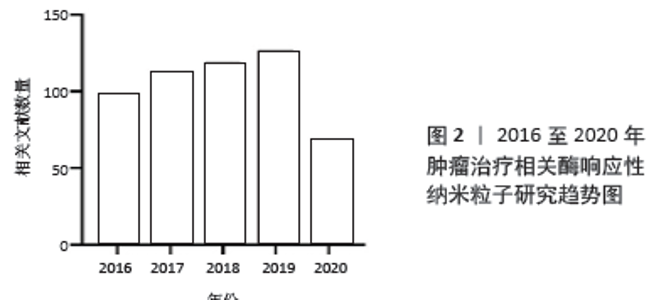
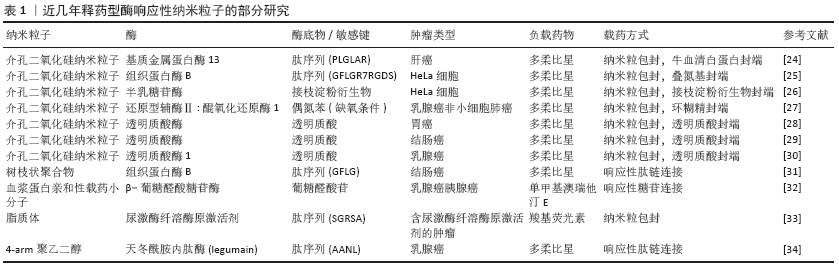
2.1 释药型酶响应纳米粒子抗肿瘤药物设计 释药型酶响应纳米粒子抗肿瘤药物是一类将酶响应与药物释放相关联的响应性纳米粒子,这类纳米粒子的设计特点在于通过肿瘤特异性酶来触发负载药物的释放,其主要优点在于:①通过提高药物在纳米粒子上的包封率,间接提高在靶向位点的递送效率[11-12];②降低非特异性释放,降低对非靶向器官的毒性[13-14];③提高药物在靶向位点的释放率,直接提高药物递送效率[15]。可简单归类为以下3种类型:连接型、包封型、前药激活型。 2.1.1 连接型载药体系 连接型载药体系将药物通过中间连接物与纳米粒子相连接,是一种重要的释药型酶响应纳米粒子的设计方式。ZHANG等[16]设计了一种树状大分子纳米粒子,通过组织蛋白酶D响应性的四肽接头将多柔比星缀合到纳米粒子表面,利用增强的渗漏与滞留效应及表面电中性,纳米粒子在肿瘤细胞中获得较高积累,并在溶酶体内响应组织蛋白酶D来释放多柔比星,诱导乳腺癌细胞发生凋亡。动物实验中在给药量相同的情况下,该纳米粒子药物的肿瘤生长抑制效果达到了61.9%,明显高于游离多柔比星的45.4%。此外,BEN-NUN等[17]设计了一种负载多柔比星的自组装纳米纤维短肽,这种短肽纤维利用增强的渗漏与滞留效应到达肿瘤组织,被肿瘤细胞摄取后响应溶酶体内的组织蛋白酶B,短肽降解并释放多柔比星,这种纳米短肽实现了较高的降解率,同时明显提高了多柔比星的释放率。因此,根据上述研究可以总结,这一类纳米粒子缀合药物的基本设计通常需要:①药物与载体上存在可连接的活性位点;②合成靶目标酶的响应性底物并连接药物与载体。 2.1.2 包封型载药体系 包封型载药体系把药物包封在纳米粒子内部,在分子门处设计酶响应性的开关,是另一类常见的释药型酶响应纳米粒子的设计方式。DE LA TORRE等[18]将介孔二氧化硅纳米粒子作为纳米粒子、以短肽(Abz-GIVRAK-OH)为封端装载番红素和多柔比星,这种纳米粒子被HeLa细胞等表达组织蛋白酶B的肿瘤细胞摄取后进入溶酶体,短肽响应组织蛋白酶B后分解,二氧化硅封端被打开释放多柔比星杀伤肿瘤细胞。而在不含该酶的MEFs CatB肿瘤细胞中,结果显示出无明显的包封物释放或杀伤肿瘤细胞的能力。VAN RIJT等[19]同样以介孔二氧化硅纳米粒子为纳米粒子负载顺铂,通过用与生物素基团连接的七肽包被介孔二氧化硅纳米粒子的外表面,响应基质金属蛋白酶9。纳米粒子进入肿瘤微环境后响应基质金属蛋白酶9,七肽被分解后纳米粒子释放顺铂杀伤肿瘤细胞,而在正常组织中则不释放顺铂。在体外动物及人组织培养模型中,纳米粒子表现出对肿瘤组织模型明显的杀伤作用,而对正常组织模型则无毒性作用。SHI及其团队[20]利用聚乙二醇与肽链合成聚乙二醇-肽二嵌段共聚物,该共聚物在结直肠癌微环境内特异性响应基质金属蛋白酶2,肽段被分解,胶束结构逐渐变得不稳定,从而触发纳米粒子在肿瘤中释放抗血管生成药与抗肿瘤药物,研究结果显示2种药物联合载药相对传统用药方式或单独载药效果大幅提高。这一类的纳米粒子基本设计要求为:①制备可包封药物的中空纳米粒子;②合成靶目标酶的响应性底物并连接在纳米粒子的封端起到开关作用。 2.1.3 前药激活型载药体系 前药激活型载药体系使药物在到达靶位点前维持前药的形态,避免药物对周围正常组织产生毒性,在到达靶位点后响应肿瘤内特异性高表达的酶,前药被转化为抗肿瘤药物并释放,从而实现精准靶向治疗。ZHOU及其团队[21]合成了一种负载人工基因proMel的纳米粒子AP30NP,该粒子表面通过表面缀合的AMD3100与脑转移乳腺癌肿瘤微环境中的CXCR4反应蓄积在肿瘤内,并利用proMel转染肿瘤细胞,使肿瘤细胞合成分泌褪黑素。褪黑素响应肿瘤微环境中高水平的基质金属蛋白酶2并被激活,释放游离的蜂毒肽,从而溶解邻近肿瘤细胞,实验结果显示这种纳米粒子的肿瘤抑制效率达到了60%,而对照组则不到10%。JALLOUK等[22]将具有非特异性高毒性的抗肿瘤药物蜂毒肽制备成了低毒性基质金属蛋白酶9激活的蜂毒肽衍生物,利用全氟化碳纳米粒子载药进一步降低其毒性,纳米粒子进入体内后该蜂毒肽衍生物只响应肿瘤微环境中特异性分布的基质金属蛋白酶9,特异性杀伤周围肿瘤细胞,用于抗黑色素瘤治疗;实验结果中相比对照组,用载有前药的纳米颗粒进行治疗可使肿瘤第14天大小缩小约54%,同时治疗后的白细胞计数无显著下降。YE等[23]在一项研究中设计了一种载多柔比星前药的级联放大释药纳米粒子用于治疗乳腺癌,该纳米粒子装载的β-拉帕酮可催化还原型辅酶Ⅱ: 醌氧化还原酶1,刺激肿瘤细胞内活性氧的水平上升,进而诱导多柔比星前药转化为多柔比星的级联扩增释放,这种纳米粒子的肿瘤生长抑制率达到了92.8%,远高于对照组的50.3%与64.4%。 和以上3种类似的释药型纳米粒子已有相当数量的研究,下表将罗列部分最近的研究进展,见表1[24-34]。 "
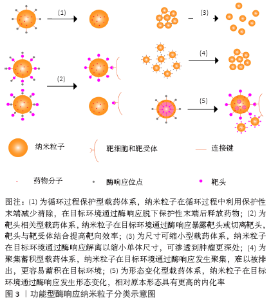
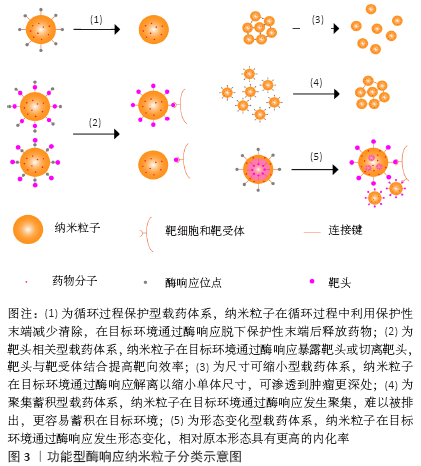
2.1.4 仅修饰了酶响应释药功能纳米粒子的局限性 上述研究中所设计纳米粒子均表现出了特异性响应肿瘤中高表达的酶,从而释放抗肿瘤相关分子的特点,但部分研究的响应环境与体内环境具有较大差异。例如DE LA Torre等[18]仅研究了多种肿瘤细胞在体外环境下的细胞摄取与药物释放情况;VAn RIJT等[19]则通过建立体外组织模型研究纳米粒子的响应情况,该2项研究均缺少体内过程,包括静脉注射后的体循环和实体肿瘤分布,这可能导致纳米粒子在动物实验中不能达到预期的效果,因为只修饰了酶响应释药功能的纳米粒子只在药物释放上具有特异性,但循环保护及分布渗透功能并未进行改善。而ZHOU等[21]通过在纳米粒子上负载AMD3100用于提高蓄积并在动物实验中表现出良好的肿瘤内分布。因此,在设计释药型酶响应性纳米粒子时,除了考虑释药的触发条件外还应注意添加提高纳米粒子靶向性的设计,以增强纳米粒子的抗肿瘤效率。 2.2 功能型酶响应纳米粒子抗肿瘤药物设计 功能型酶响应纳米粒子抗肿瘤药物是一类将酶响应性与纳米粒形态、大小、表面电荷等相关联的响应性纳米粒子,通过酶响应使纳米粒子的上述性质发生改变,从而使纳米粒子在肿瘤中渗透、富集与留存,提高纳米粒靶向性,最终改善药物的抗肿瘤效果。这类设计从递药机制上改善纳米粒在靶位点的递药效率:①既利用酶响应的高特异性优势又提高纳米粒在过程中的传递效率[35];②使纳米粒在不同的位点具有不同的功能,实现纳米粒的智能传递[36];③减少纳米粒在传递过程环节中的损耗,减小抗肿瘤药物的毒性[37]。该综述将这类酶响应性纳米粒子分为5类,分类示意图见图3。 2.2.1 循环过程保护型载药体系 许多纳米粒子由于其形态、表面电位和分子极性等因素在血液循环过程中易被免疫系统清除,为延长纳米粒子的体循环时间并减少非特异性分布,通过在纳米粒子表面包裹如透明质酸或聚乙二醇这类生物相容性较好的片段,可以有效提高纳米粒子在肿瘤的递送效率并降低毒性[38]。FENG等[36]提出一种用透明质酸覆盖近红外共振材料-空心中孔硫化铜表面用于递送多柔比星的纳米粒子,透明质酸在表面充当保护层的同时密封纳米粒子避免药物提前释放,到达肿瘤后纳米粒子表面的透明质酸既介导CD44受体内吞,又在肿瘤细胞溶酶体内被过度表达的透明质酸酶降解,使药物释放到肿瘤细胞中。该研究证明了被透明质酸包裹的纳米粒子肿瘤内蓄积明显提高。HAN等[39]利用基质金属蛋白酶2敏感肽接头将透明质酸连接到聚酰胺-胺型树枝状高分子表面,掩盖了纳米粒子表面的正电荷,用于递送多柔比星,这种纳米粒子到达肿瘤间质后特异性响应基质金属蛋白酶2,纳米粒子发生解离释放多柔比星。同时他们的研究结果还证明,这种基质金属蛋白酶2敏感的纳米粒子相对于对照组具有更好的肿瘤渗透效果。 2.2.2 靶头相关型载药体系 这类纳米粒子利用酶响应性的小分子片段连接在功能性靶头的内侧或外侧,以实现纳米粒子传递过程中的多级靶向。大部分纳米粒子药物通过增强的渗漏与滞留效应靶向肿瘤区域,然而肿瘤区域内的进一步渗透受到肿瘤微环境的影响,其中肿瘤间质中增高的组织液压力与纤维胶原蛋白抑制了纳米粒子的进一步渗透[40]。在表面连接功能性靶头或用酶响应性片段隔离靶头,使靶头只在靶向过程的特定步骤发挥作用以提高肿瘤靶向性,是这类纳米粒子的基本特点。 保护靶头型载药体系:这种类型的纳米粒子通过将响应性片段连接在靶头的外侧,只在靶向区域(通常是肿瘤间质)暴露靶头,提高纳米粒子的靶向性,增加纳米粒子的肿瘤细胞摄取从而改善治疗效果。LI等[41]利用乳腺癌内基质金属蛋白酶与叶酸受体均有过表达的特点制备了一种双靶向的聚合物胶束用于递送siRNA,聚乙二醇被基质金属蛋白酶7响应的肽链连接到叶酸嵌段外,实现了在血液循环中屏蔽对叶酸靶头的非特异性作用,并在肿瘤微环境下响应基质金属蛋白酶7与纳米粒子解离,暴露叶酸靶头进行第二阶段靶向,实验结果表明该纳米粒子显著降低了模型基因在乳腺癌细胞内的表达,并且未检测到对正常细胞的毒性。GUO等[42]开发了基于对基质金属蛋白酶敏感的功能化共聚物(mPEG-Peptide-PCL)酶反应性纳米粒子,以聚己内酯部分负载姜黄素,用于治疗非小细胞肺癌。这种纳米粒子的响应性肽段(GPLGLAGQr9)可响应基质金属蛋白酶2暴露细胞穿透肽(r9),用于增强肿瘤细胞对纳米粒子的吸收,研究结果显示相比不含响应性肽段的对照组,实验组在肿瘤组织中表现出更高的荧光亮度,表明实验组具有更高的肿瘤细胞穿透性。 切离靶头型载药体系:与保护靶头型的纳米粒子相比,切离靶头型纳米粒子可以在传递过程最后一步利用切离的靶头提高肿瘤细胞摄取率。PENG等[43]开发了一种甲基丙烯酰胺共聚物纳米粒子,将肿瘤穿透相关肽(iRGD)用基质金属蛋白酶2响应性的肽段连接到甲基丙烯酰胺表面,并搭载组织蛋白酶B响应释放的多柔比星,在肿瘤间质中肿瘤穿透相关肽靶头响应性解离并与细胞表面相关蛋白反应,提高纳米粒子穿透肿瘤细胞的能力,多柔比星在肿瘤溶酶体内响应组织蛋白酶B特异性释放。这种设计被证明有效提高了纳米粒子在肿瘤细胞中的积累与肿瘤实体的渗透,同时表现出比对照组更高的肿瘤细胞毒性。 2.2.3 尺寸可缩小型载药体系 一类尺寸可缩小的酶响应性纳米粒子在近年来受到了广泛的关注。纳米粒子在血液中的循环半衰期在一定范围内与纳米粒子的尺寸呈正相关,因为相对尺寸较大的纳米粒子渗透性较差,更不容易分布到正常组织中,而较小尺寸的纳米粒子则相反[44]。利用增强的渗漏与滞留效应,较大尺寸的纳米粒子能保持较长循环时间,并更多地渗透到肿瘤间质中[37];而较小尺寸的纳米粒子穿透性更强,更容易进入肿瘤组织深处,从而更好地发挥抗肿瘤作用[40]。基于该原理近年来研究者设计出了一系列的尺寸可缩小的酶响应性纳米粒子。ZHANG等[45]利用聚乙二醇、聚己内酯和肽段合成了一种负载多柔比星和选择性腺苷受体激动剂的中性粒细胞弹性蛋白酶响应纳米共聚物,用于治疗脑转移乳腺癌。这种纳米粒子进入肿瘤微环境后释放选择性腺苷受体激动剂,暂时提高血脑屏障通透性,诱导更多纳米共聚物穿透血脑屏障,同时响应中性粒细胞弹性蛋白酶收缩尺寸以提高递送效率。此外,该纳米共聚物还通过表面缀合的AMD3100与脑转移乳腺癌中CXCR4的反应提高肿瘤内蓄积率。动物实验结果显示,可缩小的纳米共聚物显著延长了脑转移乳腺癌小鼠的存活率,并且小鼠显示出较低的体质量减轻。SECRET等[46]设计了一种基质金属蛋白酶响应的聚乙二醇二丙烯酸酯水凝胶微粒,用于肺部肿瘤的递送,在肿瘤部位该微粒响应基质金属蛋白酶降解至2.8-4 μm之间的小颗粒,同时能发生溶胀避免肺部巨噬细胞清除,提高了递送效率。最近一项研究将基质金属蛋白酶2/9可裂解的寡肽(GK8)插入α-生育酚琥珀酸酯和被N-羟基琥珀酰亚胺活化的甲氧基-聚乙二醇中设计出了一种胶束纳米粒子,在肿瘤中响应基质金属蛋白酶2/9发生裂解,提高多西他赛在肿瘤中的蓄积[47]。与不敏感的胶束相比,负载多西他赛的胶束在HT1080细胞中的细胞摄取高出1倍,同时保持了最低的全身毒性。RUAN等[48]用基质金属蛋白酶2响应的明胶封装多柔比星负载的金纳米粒团,通过增强的渗漏与滞留效应穿过肿瘤血管壁后,在间质中被基质金属蛋白酶2裂解明胶释放出更小的金纳米粒,从而进一步穿透到肿瘤组织深处。体外实验证明,该纳米粒子在基质金属蛋白酶2降解下大小从186.5 nm缩小到59.3 nm,同时动物实验中该纳米粒子在肿瘤中的分布是所有组中程度最大的。 2.2.4 聚集蓄积型载药体系 利用酶响应触发纳米粒子的聚集以提高在肿瘤部位的积累,是一种提高靶向效率的重要方法。较小的纳米粒子进入肿瘤间质后易渗透到肿瘤深处,但因体积较小其增强的渗漏与滞留效应中的滞留效应较差,且在被肿瘤细胞摄取后容易被排出,导致蓄积率较低[44]。RUAN等[49]设计了2种天冬酰胺内肽酶响应的金纳米粒子用于递送多柔比星治疗C6脑胶质瘤。其中一种金纳米粒子以肽链(Ala-Ala-Asn-Cys-Lys)包封末端,该肽链在肿瘤细胞溶酶体内被天冬酰胺内肽酶水解暴露1,2-巯基氨基末端,与另一种金纳米粒子末端的连续氰基末端发生点击环加成反应,2种金纳米粒子发生聚集,体积增大并沉降在肿瘤细胞内,阻断排出过程并提高蓄积率,实现了多柔比星的靶向递送。动物实验结果显示与盐水组相比,该纳米粒子的中位生存时间增加到288%。PROETTO 等[50]开发了基质金属蛋白酶响应的负载Pt(Ⅱ)的胶束纳米粒子,表面携带响应性肽段(GPL GL AGGERDG),在富含基质金属蛋白酶的肿瘤微环境下,响应性肽段被酶切暴露出亲水性肽段(L AGGERDG),纳米粒子在该亲水性肽段的作用下蓄积在肿瘤微环境中,该研究证明了相对于非响应性的对照组,这种纳米粒子表现出增加的肿瘤内蓄积并明显抑制肿瘤生长。 2.2.5 形态变化型载药体系 设计形态可变化的纳米粒子,使其在循环与分布过程的形态中具有较低的内化率,而在靶向位点发生形态变化,使新的形态具有较高的细胞摄取效率。LI等[51]把细胞穿透肽片段(PGPMA)用基质金属蛋白酶2响应的肽段与聚乙二醇包裹,形成负载紫杉醇的聚乙二醇-GPLGVRG-b-聚己内酯-b-聚(3-胍基丙基甲基丙烯酰胺)(PEG-GPLGVRG-PCL-PGPMA)不对称双层聚合物囊泡,用于靶向的细胞穿透肽片段主要分布于内层。使用基质金属蛋白酶2切割聚乙二醇后,聚合物形态与结构发生重构,转化为融合的多腔囊泡和纳米微粒并使大量细胞穿透肽片段暴露于外层表面,相对未切割的聚合物,响应基质金属蛋白酶2后该纳米粒子的内化效率提高了近10倍,并对HT1080细胞的毒性显著增强。 2.2.6 功能型酶响应纳米粒子的局限性 功能型酶响应纳米粒子借助功能性的酶响应修饰可以获得更好的肿瘤内分布和更高的传递效率,但这种修饰在某些特殊环境下仍然有一定的局限性。例如设计一种在正常血液循环中能够穿过血脑屏障到达脑组织的纳米粒子,用以治疗脑胶质瘤或脑转移肿瘤时,由于血脑屏障的阻隔,肿瘤中特异性表达的酶不能为纳米粒子穿透血脑屏障提供帮助,这时往往需要结合利用其他的靶向或者响应方式,如在纳米粒子表面接枝糖基末端以诱导血管内皮细胞的转胞吞作用,从而实现针对肿瘤的靶向给药[52];还可以通过封装增强血脑屏障通透性的小分子,在肿瘤间质中响应基质金属蛋白酶特异性释放,诱导更多纳米粒子穿透血脑屏障形成正反馈,提高递送效率[45]。所以在设计功能型酶响应性纳米粒子时,应详细分析纳米粒子靶向肿瘤细胞的整个过程中遇到的障碍,添加合适的设计使每个靶向环节都具有较高的传递效率,而非仅仅响应肿瘤内的酶,以提高纳米粒子在肿瘤组织内的靶向性。"
| [1] WILD CP, WEIDERPASS E, STEWART BW. World cancer report: cancer research for cancer prevention. Lyon: International Agency for Research on Cancer, 2020. [2] 姚梦欢,王铁梅,林梓桐.纳米靶向给药系统在口腔鳞状细胞癌诊断与治疗的研究进展[J].口腔医学研究,2018,34(12):1277-1280. [3] 魏常博.纳米材料和纳米技术在肿瘤放疗增敏中的研究进展[J].口腔疾病防治,2017,25(11):744-748. [4] PRABHAKAR U, MAEDA H, JAIN K, et al. Challenges and key considerations of the enhanced permeability and retention effect for nanomedicine drug delivery in oncology. Cancer Res. 2013;73(8):2412-2417. [5] 刘艳红,周建平,霍美蓉.肿瘤微环境响应型智能纳米药物载体的研究进展[J].中国药科大学学报,2016,47(2):4-12. [6] 贺瑞,甘杨子,钟克焱,等.纳米抗肿瘤药物及其研究进展[J].现代养生(下半月版),2019(2):109-110. [7] AMREDDY N, BABU A, MURALIDHARAN R, et al. Recent Advances in Nanoparticle-Based Cancer Drug and Gene Delivery. Adv Cancer Res. 2018; 137:115-170. [8] BEHZADI S, SERPOOSHAN V, TAO W, et al. Cellular uptake of nanoparticles: journey inside the cell. Chem Soc Rev. 2017;46(14):4218-4244. [9] Chen B, Dai W, He B, et al. Current Multistage Drug Delivery Systems Based on the Tumor Microenvironment. Theranostics, 2017;7(3):538-558. [10] ZHANG H, LIU XL, ZHANG YF, et al. Magnetic nanoparticles based cancer therapy: current status and applications. Sci China Life Sci. 2018;61(4): 400-414. [11] 刘耀阳,王荣梅.酶响应纳米粒子共输送地西他滨阿霉素逆转乳腺癌多药耐药[J].药物生物技术,2018,25(4):288-293. [12] KUMAR B, KULANTHAIVEL S, MONDAL A, et al. Mesoporous silica nanoparticle based enzyme responsive system for colon specific drug delivery through guar gum capping. Colloids Surf B Biointerfaces. 2017;150: 352-361. [13] 刘新,马铭怿.基质金属蛋白酶响应的紫杉醇-二氢卟吩e6纳米粒子诱导结肠癌细胞凋亡[J].药物生物技术,2019,26(3):211-216. [14] JIANG H, SHI X, YU X, et al. Hyaluronidase Enzyme-responsive Targeted Nanoparticles for Effective Delivery of 5-Fluorouracil in Colon Cancer. Pharm Res. 2018;35(4):73. [15] 李辉盛,苏子桓,王冠海.酶刺激响应纳米载体的制备与负载姜黄素体外释放研究[J].山东化工,2018,47(20):18-19. [16] ZHANG C, PAN D, LUO K, et al. Peptide Dendrimer–doxorubicin Conjugate-Based Nanoparticles as an Enzyme-Responsive Drug Delivery System for Cancer Therapy. Adv Healthc Mater. 2014;3(8):1299-1308. [17] BEN-NUN Y, FICHMAN G, ADLER-ABRAMOVICH L, et al. Cathepsin nanofiber substrates as potential agents for targeted drug delivery. J Control Release. 2017;257:60-67. [18] DE LA TORRE C, MONDRAGÓN L, COLL C, et al. Cathepsin-B induced controlled release from peptide-capped mesoporous silica nanoparticles. Chemistry. 2014;20(47):15309-15314. [19] VAN RIJT SH, BÖLÜKBAS DA, ARGYO C, et al. Protease-Mediated Release of Chemotherapeutics from Mesoporous Silica Nanoparticles to ex Vivo Human and Mouse Lung Tumors. ACS Nano. 2015;9(3):2377-2389. [20] SHI L, HU Y, LIN A, et al. Matrix Metalloproteinase Responsive Nanoparticles for Synergistic Treatment of Colorectal Cancer via Simultaneous Anti-Angiogenesis and Chemotherapy. Bioconjug Chem. 2016;27(12):2943-2953. [21] ZHOU Y, ZHANG S, CHEN Z, et al. Targeted Delivery of Secretory Promelittin via Novel Poly(lactone-co-β-amino ester) Nanoparticles for Treatment of Breast Cancer Brain Metastases. Adv Sci (Weinh). 2020;7(5):1901866. [22] JALLOUK AP, PALEKAR RU, MARSH JN, et al. Delivery of a Protease-Activated Cytolytic Peptide Prodrug by Perfluorocarbon Nanoparticles. Bioconjug Chem. 2015;26(8):1640-1650. [23] YE M, HAN Y, TANG J, et al. A Tumor-Specific Cascade Amplification Drug Release Nanoparticle for Overcoming Multidrug Resistance in Cancers. Adv Mater. 2017;29(38):1702342. [24] LIU Y, DING X, LI J, et al. Enzyme responsive drug delivery system based on mesoporous silica nanoparticles for tumor therapyin vivo. Nanotechnology. 2015;26(14):145102. [25] CHENG YJ, LUO GF, ZHU JY, et al. Enzyme-induced and tumor-targeted drug delivery system based on multifunctional mesoporous silica nanoparticles. ACS Appl Mater Interfaces. 2015;7(17):9078-9087. [26] BERNARDOS A, MONDRAGON L, AZNAR E, et al. Enzyme-responsive intracellular controlled release using nanometric silica mesoporous supports capped with “saccharides”. ACS Nano. 2010;4(11):6353-6368. [27] LEE J, OH ET, YOON H, et al. Mesoporous nanocarriers with a stimulus-responsive cyclodextrin gatekeeper for targeting tumor hypoxia. Nanoscale. 2017;9(20):6901-6909. [28] NAZ S, WANG M, HAN Y, et al. Enzyme-responsive mesoporous silica nanoparticles for tumor cells and mitochondria multistage-targeted drug delivery. Int J Nanomedicine. 2019;14:2533-2542. [29] ZHANG M, XU C, WEN L, et al. A Hyaluronidase-Responsive Nanoparticle-Based Drug Delivery System for Targeting Colon Cancer Cells. Cancer Res. 2016;76(24):7208-7218. [30] CHEN Z, LI Z, LIN Y, et al. Bioresponsive hyaluronic acid-capped mesoporous silica nanoparticles for targeted drug delivery. Chemistry. 2013;19(5): 1778-1783. [31] LEE SJ, JEONG YI, PARK HK, et al. Enzyme-responsive doxorubicin release from dendrimer nanoparticles for anticancer drug delivery. Int J Nanomedicine. 2015;10:5489-5503. [32] RENOUX B, RAES F, LEGIGAN T, et al. Targeting the tumour microenvironment with an enzyme-responsive drug delivery system for the efficient therapy of breast and pancreatic cancers. Chem Sci. 2017;8(5):3427-3433. [33] BASEL MT, SHRESTHA TB, TROYER DL, et al. Protease-sensitive, polymer-caged liposomes: a method for making highly targeted liposomes using triggered release. ACS Nano. 2011;5(3):2162-2175. [34] 王丁丁,孙中洋,南军,等.胞内酶敏感肿瘤靶向递药体系的合成及应用[J].西北药学杂志,2019,34(2):203-208. [35] CALLMANN CE, BARBACK CV, THOMPSON MP, et al. Therapeutic Enzyme-Responsive Nanoparticles for Targeted Delivery and Accumulation in Tumors. Adv Mater. 2015;27(31):4611-4615. [36] FENG Q, ZHANG Y, ZHANG W, et al. Tumor-targeted and multi-stimuli responsive drug delivery system for near-infrared light induced chemo-phototherapy and photoacoustic tomography. Acta Biomater. 2016;38: 129-142. [37] CABRAL H, MATSUMOTO Y, MIZUNO K, et al. Accumulation of sub-100 nm polymeric micelles in poorly permeable tumours depends on size. Nat Nanotechnol. 2011;6(12):815-823. [38] CHOI KY, SARAVANAKUMAR G, PARK JH, et al. Hyaluronic acid-based nanocarriers for intracellular targeting: interfacial interactions with proteins in cancer. Colloids Surf B Biointerfaces. 2012;99:82-94. [39] HAN M, HUANG-FU MY, GUO WW, et al. MMP-2-Sensitive HA End-Conjugated Poly(amidoamine) Dendrimers via Click Reaction To Enhance Drug Penetration into Solid Tumor. ACS Appl Mater Interfaces. 2017;9(49):42459-42470. [40] FERNANDES C, SUARES D, YERGERI MC. Tumor Microenvironment Targeted Nanotherapy. Front Pharmacol. 2018;9:1230. [41] LI H, MITEVA M, KIRKBRIDE KC, et al. Dual MMP7-proximity-activated and folate receptor-targeted nanoparticles for siRNA delivery. Biomacromolecules. 2015;16(1):192-201. [42] GUO F, WU J, WU W, et al. PEGylated self-assembled enzyme-responsive nanoparticles for effective targeted therapy against lung tumors. J Nanobiotechnology. 2018;16(1):57. [43] PENG ZH, KOPEČEK J. Enhancing Accumulation and Penetration of HPMA Copolymer-Doxorubicin Conjugates in 2D and 3D Prostate Cancer Cells via iRGD Conjugation with an MMP-2 Cleavable Spacer. J Am Chem Soc. 2015;137(21):6726-6729. [44] POPOVIĆ Z, LIU W, CHAUHAN VP, et al. A nanoparticle size series for in vivo fluorescence imaging. Angew Chem Int Ed Engl. 2010;49(46):8649-8652. [45] ZHANG S, DENG G, LIU F, et al. Autocatalytic Delivery of Brain Tumor-targeting, Size-shrinkable Nanoparticles for Treatment of Breast Cancer Brain Metastases. Adv Funct Mater. 2020;30(14):1910651. [46] SECRET E, KELLY SJ, CRANNELL KE, et al. Enzyme-responsive hydrogel microparticles for pulmonary drug delivery. ACS Appl Mater Interfaces. 2014;6(13):10313-10321. [47] ZHANG X, WANG X, ZHONG W, et al. Matrix metalloproteinases-2/9-sensitive peptide-conjugated polymer micelles for site-specific release of drugs and enhancing tumor accumulation: preparation and in vitro and in vivo evaluation. Int J Nanomedicine. 2016;11:1643-1661. [48] RUAN S, CAO X, CUN X, et al. Matrix metalloproteinase-sensitive size-shrinkable nanoparticles for deep tumor penetration and pH triggered doxorubicin release. Biomaterials. 2015;60:100-110. [49] RUAN S, HU C, TANG X, et al. Increased Gold Nanoparticle Retention in Brain Tumors by in Situ Enzyme-Induced Aggregation. ACS Nano. 2016;10(11):10086-10098. [50] PROETTO MT, CALLMANN CE, CLIFF J, et al. Tumor Retention of Enzyme-Responsive Pt(II) Drug-Loaded Nanoparticles Imaged by Nanoscale Secondary Ion Mass Spectrometry and Fluorescence Microscopy. ACS Cent Sci. 2018;4(11):1477-1484. [51] LI J, XIAO S, XU Y, et al. Smart Asymmetric Vesicles with Triggered Availability of Inner Cell-Penetrating Shells for Specific Intracellular Drug Delivery. ACS Appl Mater Interfaces. 2017;9(21):17727-17735. [52] RUAN S, QIN L, XIAO W, et al. Acid-Responsive Transferrin Dissociation and GLUT Mediated Exocytosis for Increased Blood–Brain Barrier Transcytosis and Programmed Glioma Targeting Delivery. Adv Funct Mater, 2018;28(30):1802227. [53] ANSELMO AC, MITRAGOTRI S. Nanoparticles in the clinic. Bioeng Transl Med. 2016;1(1):10-29. [54] CHATTERJEE N, BIVONA TG. Polytherapy and Targeted Cancer Drug Resistance. Trends Cancer. 2019;5(3):170-182. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Jiang Yong, Luo Yi, Ding Yongli, Zhou Yong, Min Li, Tang Fan, Zhang Wenli, Duan Hong, Tu Chongqi. Von Mises stress on the influence of pelvic stability by precise sacral resection and clinical validation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1318-1323. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [5] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [6] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [7] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [8] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [9] | Chai Le, Lü Jianlan, Hu Jintao, Hu Huahui, Xu Qingjun, Yu Jinwei, Quan Renfu. Signal pathway variation after induction of inflammatory response in rats with acute spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1218-1223. |
| [10] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [11] | Gu Xia, Zhao Min, Wang Pingyi, Li Yimei, Li Wenhua. Relationship between hypoxia inducible factor 1 alpha and hypoxia signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1284-1289. |
| [12] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [13] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [14] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| [15] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||