Chinese Journal of Tissue Engineering Research ›› 2017, Vol. 21 ›› Issue (18): 2839-2845.doi: 10.3969/j.issn.2095-4344.2017.18.009
Previous Articles Next Articles
Biological evaluation of a novel porous tantalum scaffold
Xie Hui1, 2, Ma Zhi-jie2, Wang Jian-chuan2, Wang Ben-jie2, Wang Wei2, Wei Xiao-wei2, Zhao De-wei1, 2
- 1Department of Biomedical Engineering, Faculty of Electronic Information and Electrical Engineering, Dalian University of Technology, Dalian 116023, Liaoning Province, China; 2Department of Orthopedics, Zhongshan Hospital of Dalian University, Dalian 116001, Liaoning Province, China
-
Received:2017-04-01Online:2017-06-28Published:2017-07-07 -
Contact:Zhao De-wei, Chief physician, Professor, Department of Biomedical Engineering, Faculty of Electronic Information and Electrical Engineering, Dalian University of Technology, Dalian 116023, Liaoning Province, China; Department of Orthopedics, Zhongshan Hospital of Dalian University, Dalian 116001, Liaoning Province, China -
About author:Xie Hui, Studying for doctorate, Attending physician, Department of Biomedical Engineering, Faculty of Electronic Information and Electrical Engineering, Dalian University of Technology, Dalian 116023, Liaoning Province, China; Department of Orthopedics, Zhongshan Hospital of Dalian University, Dalian 116001, Liaoning Province, China -
Supported by:the Key Projects in the National Science & Technology Pillar Program during the Twelfth Five-Year Plan Period, No. 2012BAI17B02; the National Major Research and Development Plan during the Thirteenth Five-Year Plan Period, No. 2016YFC1102000
CLC Number:
Cite this article
Xie Hui, Ma Zhi-jie, Wang Jian-chuan, Wang Ben-jie, Wang Wei, Wei Xiao-wei, Zhao De-wei.
share this article
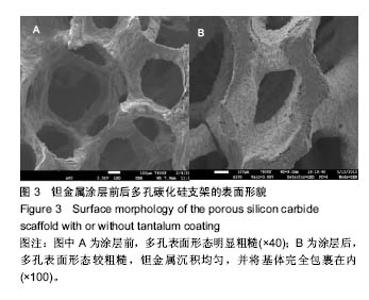
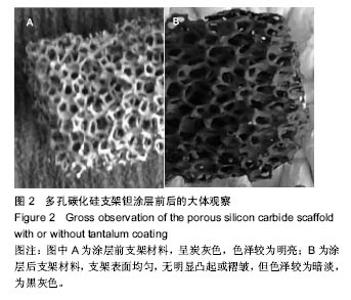
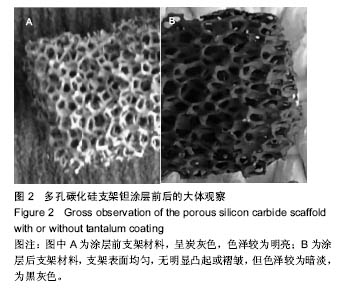
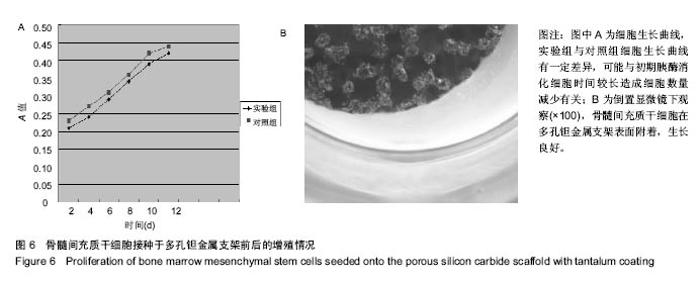
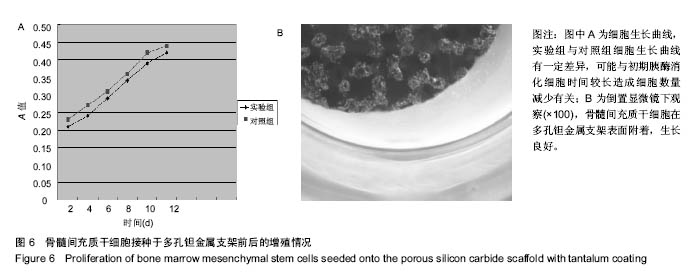
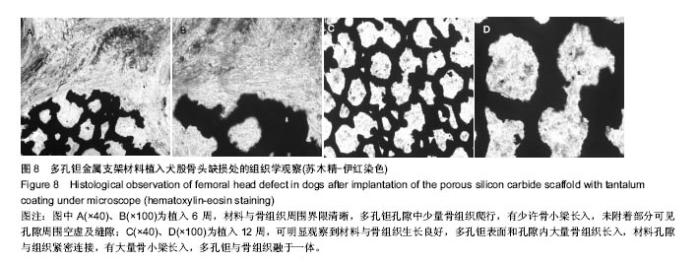
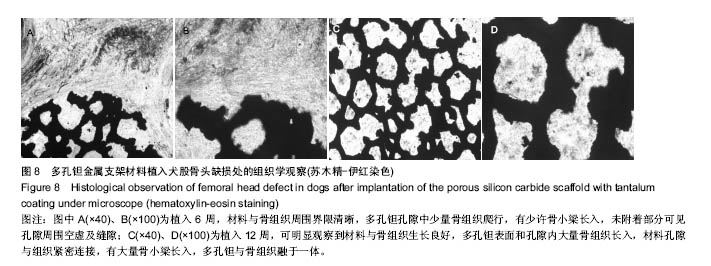
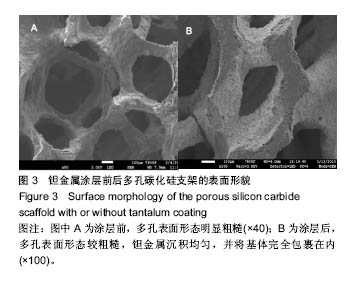
2.2 钽金属涂层前后多孔碳化硅支架的表面形貌 利用超景深三维数字显显微系统观察多孔碳化硅支架喷涂前后表面特征变化,结果见图3所示,涂层前其多孔表面形态明显粗糙,造成这种微观表面结构的原因是采用快速成形技术的特点决定的,从图中可看出涂层前后孔隙表面形貌发生了明显变化,涂层后的形貌比较粗糙,这种表面的微观结构改变是由快速成形及烧结技术特点决定的。因为快速成形及烧结技术是分层累积烧结成形技术,在分层制造过程中必然会形成上述表面特征;另外,可见涂层后的多孔表面钽金属沉积均匀,并将基体完全包裹在内。目前已有研究证实,这些凹凸不平的粗糙表面不仅有利于细胞的粘附和组织的嵌入,而且还有利于组织的爬行替代,形成完整的支架,增加与骨组织的连接强度。 由于涂层前多孔碳化硅支架的色泽比较明亮,在数字显微图像中显示了较强的反光性,不利于细微结构清晰显示,但涂层后的多孔碳化硅支架表面的反光性明显减弱,可以清晰的观察多孔支架的细微结构和孔的形态特征,展现了良好的三维连通性。 "
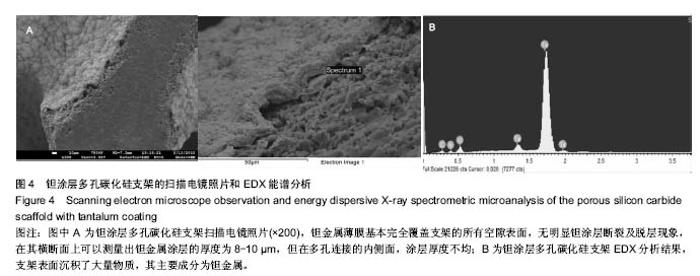
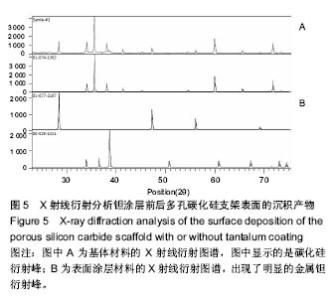
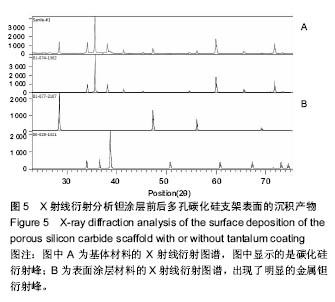
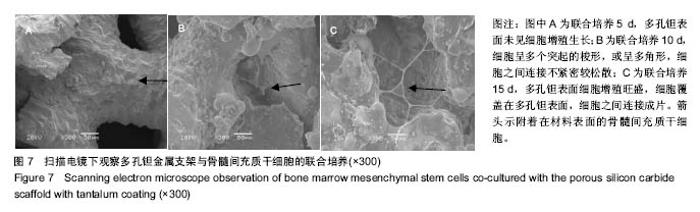
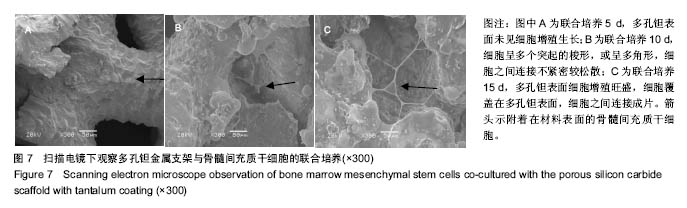
2.3 钽涂层多孔碳化硅支架的扫描电镜照片和EDX能谱分析 图4为钽涂层多孔碳化硅支架的扫描电镜照片和EDX能谱分析结果。钽金属薄膜基本完全覆盖支架的所有空隙表面,无明显钽涂层断裂及脱层现象,在其横断面上可以测量出钽金属涂层的厚度为8-10 μm,但在多孔连接的内侧面,涂层厚度有所不均(图4A),因此,在植入体内后可能会影响支架的生物学性能。因此,需要对化学气相沉积涂层工艺进一步优化,以提高钽金属涂层厚度的均匀性及力学强度,避免植入后对细胞和组织长入产生不利影响。EDX结果分析显示,支架表面沉积了大量物质,其主要成分为钽金属(图4B);因此可以证明采用化学气相沉积技术是完全可将五氯化钽均匀沉积在多孔碳化硅支架表面,进而得到新的多孔钽金属材料。 "

| [1]Reach JS Jr,Dickey ID,Zobitz ME.Direct tendon attachment and healing to porous tantalum: An experimental animal study.Bone Joint Surg Am.2007;89(5):1000-1009.[2]Jonitz A,Lochner K,Lindner T,et al.Oxygen consumption, acidification and migration capacity of human primary osteoblasts within a three-dimensional tantalum scaffold.J Mater Sci Mater Med.2011;22(9):2089-2095.[3]Liu G,Wang J,Yang S,et al.Effect of a porous tantalum rod on early and intermediate stages of necrosis of the femoral head. Biomed Mater.2010;5(6):65-68.[4]于晓明,谭丽丽,杨柯.医用金属表面的钽涂层制备及其临床应用趋势[J].中国骨科临床与基础研究杂志,2013,5(2):115-119.[5]Wu L,Xu X,Wang L,et al.Study on cytocompatability and animal implantation test of foam SiC.J Inorg Mater.2010;25: 211-215.[6]吴琳,许兴祥,李波,等.泡沫碳化硅的生物相容性[J].材料研究学报,2008,22(1):58-62.[7]Stivastava S,Stephen DG.Screening of in cyto toxicity by the adhesive test.Biomaterials.1990;11(3):133-136.[8]Ersev H,Schmalz G,Bayirli G,et al.Cytotoxic and mutagenic potencies of various root canal filling materials in eukaryotic and prokaryotic cells in vitro.J Endod.1999;25(5):359-363.[9]Ingham E,Green TR,Stone MH,et al.Production of TNF and bone resorbing activity bymacrophages in response todifferent types of bone cement particles.Biomaterials. 2000;21(10):1005-1013.[10]Balla VK,Bodhak S,Bose S,et al.Porous tantalum structures for bone implants: fabrication, mechanical and in vitro biological properties.Acta Biomaterialia.2010;6(8): 3349-3359.[11]Mrosek EH,Schagemann JC,Chung HW,et al.Porous tantalum and poly-epsilom-caprolactone biocomposites for osteochondral defect repair:preliminary studies in rabbits.J Orthop Res.2010;28 2:141-148.[12]Findlay DM,Welldon NK,Atkins GJ,et al.The proliferation and ph- enotypic expression of human osteoblasts on tantalum metal.Biomaterials.2004;25(12):2215-2227.[13]Tsao AK,Roberson JR,Christie MJ,et al.Biomechanical and clinical evaluations of a porous tantalum implant for the treatment of Early-stage osteonecrosis.J Bone Joint Surg Am.2005;8 Suppl 2:22-27.[14]Bobyn JD,Hacking SA,Chan SP,et al.Characterization of a new poro us tantalum biomaterial for reconstructive orthopaedics.Presented as a scientific exhibit at the Annual Meeting of the American A cademy of Orthopaedic Surgeons, Anaheim,1999:4-8.[15]庞海涛,甘洪全,王茜,等.转化生长因子β1对多孔钽/MG63成骨样细胞复合物细胞增殖及分泌功能的影响[J]. 中国组织工程研究,2016,20(25):3680-3686.[16]任原.缺氧对多孔钽-人成骨细胞复合物细胞增殖、分泌功能及成骨基因表达的影响[D].河北联合大学, 2015.[17]张辉,王茜,赵阳,等.碱性成纤维细胞生长因子对国产多孔钽-软骨细胞复合物软骨细胞表型维持及去分化的影响[J].第二军医大学学报,2015,36(3):248-255.[18]Wei X,Zhao D,Wang B,et al.Tantalum coating of porous carbon scaffold supplemented with autologous bone marrow stromal stem cells for bone regeneration in vitro and in vivo. Exp Biol Med.2016; 241(6):592.[19]Tang Z,Xie Y,Yang F,et al.Porous tantalum coatings prepared by vacuum plasma spraying enhance bmscs osteogenic differentiation and bone regeneration in vitro and in vivo.PloS One.2013;8(6):e66263.[20]Cai F,Wu XT,Xie XH,et al.Evaluation of intervertebral disc regeneration with implantation of bone marrow mesenchymal stem cells (BMSCs) using quantitative T2 mapping: a study in rabbits.IntOrthop.2015;39(1):149-159.[21]Li Q,Wan W,Gao H,et al.BMSCs laden injectable amino-diethoxypropane modified alginate-chitosan hydrogel for hyaline cartilage reconstruction.J Mater ChemB.2015;3(9): 1990-2005.[22]Yazdimamaghani M,Razavi M,Vashaee D,et al.Porous magnesium-based scaffolds for tissue engineering.Mater Sci Eng C Mater Biol Appl.2017;71:1253-1266. [23]Li G,Cao H,Zhang W,et al.Enhanced Osseointegration of Hierarchical Micro/Nanotopographic Titanium Fabricated by Microarc Oxidation and Electrochemical Treatment.ACS Appl Mater Interfaces. 2016;8(6):3840-3852.[24]Wang H,Jiang Z,Zhang J,et al.Enhanced osteogenic differentiation of rat bone marrow mesenchymal stem cells on titanium substrates by inhibiting Notch3.Arch Oral Biol.2017; 80:34-40.[25]Chen X,Li M,Yan J,et al.Alcohol Induces Cellular Senescence and Impairs Osteogenic Potential in Bone Marrow-Derived Mesenchymal Stem Cells.Alcohol Alcohol.2017:1-9.[26]Li W,Xu R,Huang J,et al.Treatment of rabbit growth plate injuries with oriented ECM scaffold and autologous BMSCs. Sci Rep.2017;7:44140.[27]Abdallah BM.Marrow adipocytes inhibit the differentiation of mesenchymal stem cells into osteoblasts via suppressing BMP-signaling.J Biomed Sci.2017;24(1):11.[28]Gong X,Yu W,Zhao H,et al.Skeletal Site-specific Effects of Zoledronate on in vivo Bone Remodeling and in vitro BMSCs Osteogenic Activity.Sci Rep.2017;7:36129.[29]Du X,Huang F,Zhang S,et al.Carboxymethylcellulose with phenolic hydroxyl microcapsules enclosinggene-modified BMSCs for controlled BMP-2 release in vitro.Artif Cells Nanomed Biotechnol. 2017:1-14.[30]Qiu G,Shi Z,Xu HH,et al.Bone regeneration in minipigs via calcium phosphate cement scaffold delivering autologous BMSCs and platelet-rich plasma.J Tissue Eng Regen Med.2017.doi: 10.1002/term.2416. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [3] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [4] | Wen Dandan, Li Qiang, Shen Caiqi, Ji Zhe, Jin Peisheng. Nocardia rubra cell wall skeleton for extemal use improves the viability of adipogenic mesenchymal stem cells and promotes diabetes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1038-1044. |
| [5] | Zhu Bingbing, Deng Jianghua, Chen Jingjing, Mu Xiaoling. Interleukin-8 receptor enhances the migration and adhesion of umbilical cord mesenchymal stem cells to injured endothelium [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1045-1050. |
| [6] | Luo Xiaoling, Zhang Li, Yang Maohua, Xu Jie, Xu Xiaomei. Effect of naringenin on osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1051-1056. |
| [7] | Wang Xinmin, Liu Fei, Xu Jie, Bai Yuxi, Lü Jian. Core decompression combined with dental pulp stem cells in the treatment of steroid-associated femoral head necrosis in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1074-1079. |
| [8] | Fang Xiaolei, Leng Jun, Zhang Chen, Liu Huimin, Guo Wen. Systematic evaluation of different therapeutic effects of mesenchymal stem cell transplantation in the treatment of ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1085-1092. |
| [9] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| [10] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [11] | Huang Chenwei, Fei Yankang, Zhu Mengmei, Li Penghao, Yu Bing. Important role of glutathione in stemness and regulation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1119-1124. |
| [12] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [13] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 985-991. |
| [14] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1005-1011. |
| [15] | Zhou Ying, Zhang Huan, Liao Song, Hu Fanqi, Yi Jing, Liu Yubin, Jin Jide. Immunomodulatory effects of deferoxamine and interferon gamma on human dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1012-1019. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||