Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (7): 1074-1079.doi: 10.12307/2022.147
Previous Articles Next Articles
Core decompression combined with dental pulp stem cells in the treatment of steroid-associated femoral head necrosis in rabbits
Wang Xinmin, Liu Fei, Xu Jie, Bai Yuxi, Lü Jian
- Ward Two, Department of Orthopedics, First Hospital of Qinhuangdao, Qinhuangdao 066000, Hebei Province, China
-
Received:2021-03-16Revised:2021-03-17Accepted:2021-06-01Online:2022-03-08Published:2021-10-29 -
Contact:Liu Fei, Professor, Master’s supervisor, Ward Two, Department of Orthopedics, First Hospital of Qinhuangdao, Qinhuangdao 066000, Hebei Province, China -
About author:Wang Xinmin, MD, Associate chief physician, Ward Two, Department of Orthopedics, First Hospital of Qinhuangdao, Qinhuangdao 066000, Hebei Province, China -
Supported by:Special Fund Project of Central Government Guiding Local Science and Technology Development in 2019, No. 20190214175848679 (to LF)
CLC Number:
Cite this article
Wang Xinmin, Liu Fei, Xu Jie, Bai Yuxi, Lü Jian. Core decompression combined with dental pulp stem cells in the treatment of steroid-associated femoral head necrosis in rabbits[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1074-1079.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
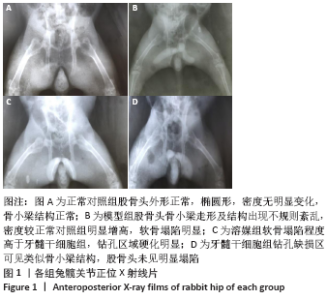
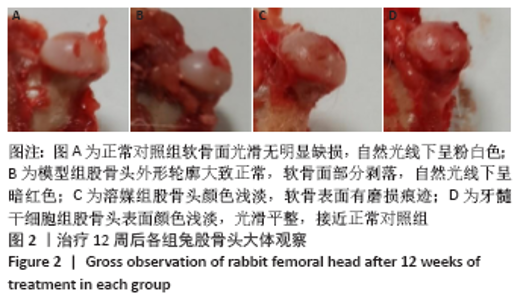
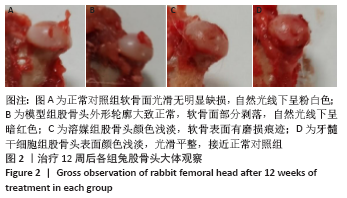
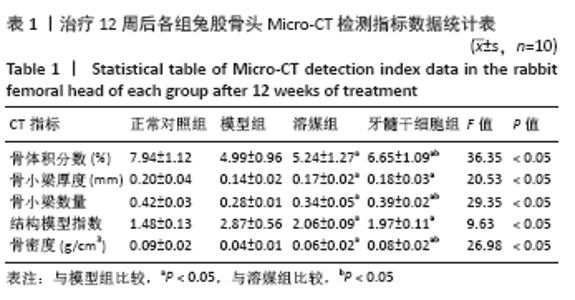
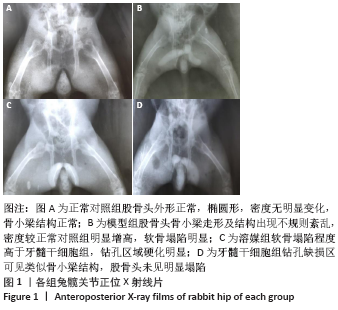
2.1 一般情况 模型组注射醋酸泼尼松龙三四周时能明显观察到右侧股骨变细,摄食量减小,反应迟钝,体态偏瘦;髓芯减压术后1周内,溶媒组和干细胞组实验动物活动及摄食量减少,动物精神萎靡,术后2周活动量及摄食量增加,3周时活动增多,摄食量及活动逐渐恢复正常。所有52只实验动物生存状态良好,无死亡,模型组有5只、溶媒组有3只、干细胞组有1只实验动物轻度跛行。 2.2 X射线片观察结果 各组标本在钻孔缺损区以外变化大致相似,其中正常对照组股骨头外形正常,椭圆形,密度无明显变化,骨小梁结构正常,见图1;模型组股骨头骨小梁走形及结构出现不规则紊乱,密度较正常对照组明显增高,软骨塌陷明显,出现软骨下囊性病变;模型组、溶媒组股骨头软骨比牙髓干细胞组塌陷更明显,溶媒组、牙髓干细胞组钻孔缺损内有着不同的变化,牙髓干细胞组钻孔缺损区可见类似骨小梁结构,钻孔缺损区中心密度低于溶媒组,两组钻孔区外边缘密度有所增高,未见明显差别,见图1。"
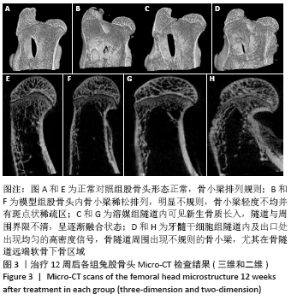
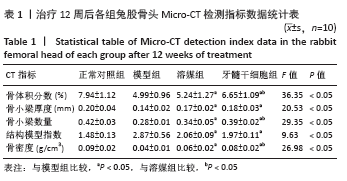
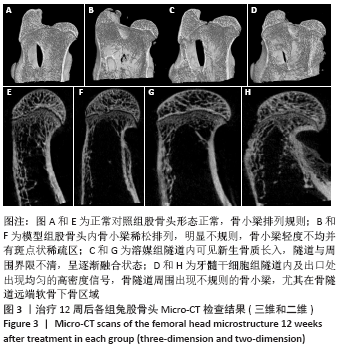
2.4 Micro-CT检测结果 牙髓干细胞组隧道内及出口处出现均匀的高密度信号,骨隧道周围出现不规则的骨小梁,尤其在骨隧道远端,软骨下骨区域;溶媒组、牙髓干细胞组隧道内均可见新生骨质长入,隧道与周围界限不清,呈逐渐融合状态,其中牙髓干细胞组更明显;模型组股骨头内骨小梁稀松排列,明显不规则,骨小梁轻度不均并斑点状稀疏区,4组股骨头外形大致正常,均未见到明显塌陷,见图3。采用Micro-CT对兔股骨头进行扫描并三维重建,使用CT-AN软件进行三维分析,并进行感兴趣区域骨密度测定,结果显示:溶媒组和牙髓干细胞组均较模型组有显著改善,差异有显著性意义(P < 0.05);与溶媒组比较,牙髓干细胞组股骨头骨体积分数、骨小梁数量、骨密度改善更明显,差异有显著性意义(P < 0.05),见表1。"
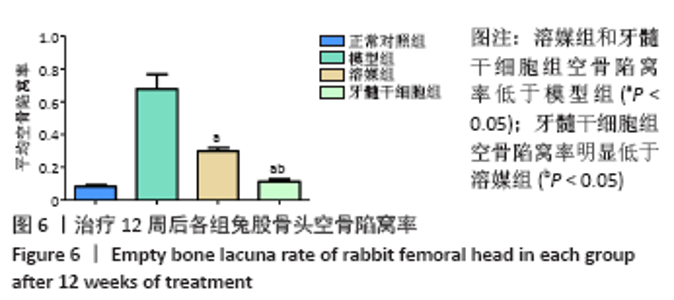
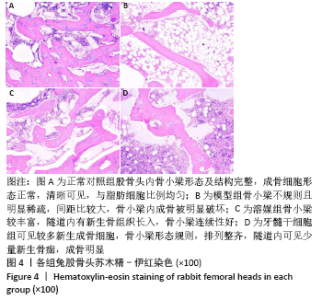
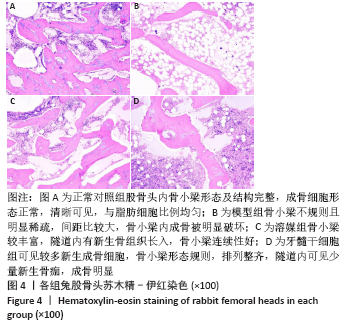
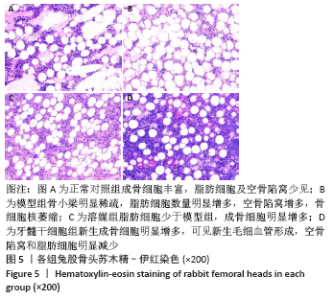
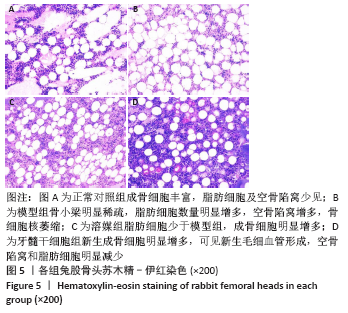
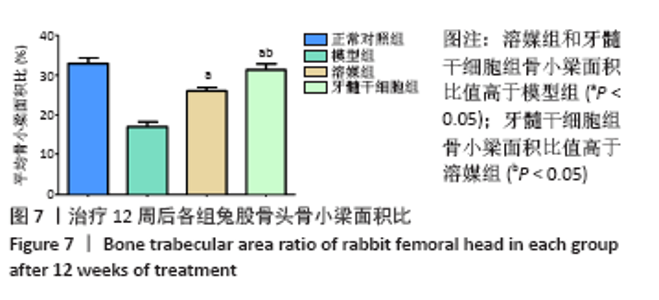
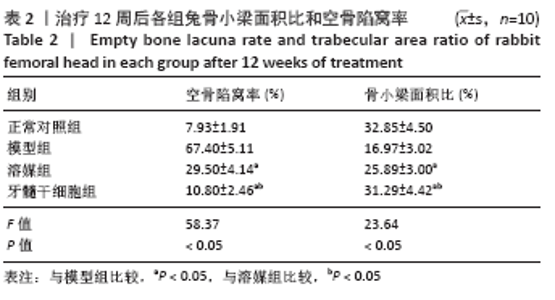
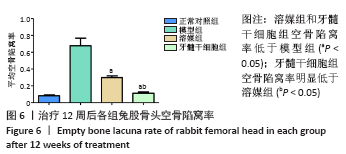
2.5.2 Masson染色结果 模型组可见大多数骨组织呈绿色,骨小梁混乱且不规则,骨小梁中骨细胞空骨陷窝明显多于溶媒组和牙髓干细胞组;溶媒组骨组织中呈现红绿相间分布,红染区域较模型组明显增多,隧道内出现片状或条状被红染的成熟骨组织;牙髓干细胞组红色成熟骨组织多于模型组和溶媒组,其中红染区域比例高于模型组和溶媒组,骨髓内脂肪细胞较模型组和溶媒组减少,但绿染区域较正常对照组明显增多。 2.6 空骨陷窝率及骨小梁面积比 牙髓干细胞组空骨陷窝率和骨小梁面积比与正常对照组更为接近,两组间比较差异无显著性意义,溶媒组空骨陷窝率高于正常对照组,骨小梁面积比低于正常对照组,两组间比较差异有显著性意义,见图6和图7;溶媒组、牙髓干细胞组空骨陷窝率明显低于模型组,骨小梁面积比明显高于模型组;与溶媒组相比,牙髓干细胞组空骨陷窝率及骨小梁面积比更接近于正常对照组,差异有显著性意义,见表2。 2.7 生物相容性 治疗12周后溶媒组、牙髓干细胞组髓芯减压隧道内组织相容性好,并未发现明显的细胞排斥反应及炎性细胞浸润。 "
| [1] LI B, YANG M, YU L. Vascular bundle transplantation combined with porous bone substituted scaffold for the treatment of early-stage avascular necrosis of femoral head. Med Hypotheses. 2019;132:109374. [2] FU Q, TANG NN, ZHANG Q, et al. Preclinical Study of Cell Therapy for Osteonecrosis of the Femoral Head with Allogenic Peripheral Blood-Derived Mesenchymal Stem Cells. Yonsei Med J. 2016;57(4):1006-1015. [3] YUAN HF, ZHANG J, GUO CA, et al. Clinical outcomes of osteonecrosis of the femoral head after autologous bone marrow stem cell implantation: a meta-analysis of seven case-control studies. Clinics (Sao Paulo). 2016;71(2):110-113. [4] FANG SH, LI YF, JIANG JR, et al. Relationship of α2-Macroglobulin with Steroid-Induced Femoral Head Necrosis: A Chinese Population-Based Association Study in Southeast China. Orthop Surg. 2019;11(3):481-486. [5] GÓMEZ-MONT LANDERRECHE JG, GIL-ORBEZO F, MORALES-DOMINGUEZ H, et al. Nontraumatic causes of bilateral avascular necrosis of the femoral head: link between hepatitis C and pegylated interferon. Acta Ortop Mex. 2015;29(3): 172-175. [6] CHAO PC, CUI MY, LI XA, et al. Correlation between miR-1207-5p expression with steroid-induced necrosis of femoral head and VEGF expression. Eur Rev Med Pharmacol Sci. 2019;23(7):2710-2718. [7] WANG Z, SUN QM, ZHANG FQ, et al. Core decompression combined with autologous bone marrow stem cells versus core decompression alone for patients with osteonecrosis of the femoral head: A meta-analysis. Int J Surg. 2019;69:23-31. [8] ANDRONIC O, SHOMAN H, WEISS O, et al. What are the outcomes of core decompression in patients with avascular necrosis? Protocol for a systematic review. F1000Res. 2020;9:71. [9] WANG SL, HU YB, CHEN H, et al. Efficacy of bone marrow stem cells combined with core decompression in the treatment of osteonecrosis of the femoral head: A PRISMA-compliant meta-analysis. Medicine (Baltimore). 2020;99(25):e20509. [10] HERNIGOU P, DUBORY A, HOMMA Y, et al. Cell therapy versus simultaneous contralateral decompression in symptomatic corticosteroid osteonecrosis: a thirty year follow-up prospective randomized study of one hundred and twenty five adult patients. Int Orthop. 2018;42(7):1639-1649. [11] ZHANG XL, SHI KQ, JIA PT, et al. Effects of platelet-rich plasma on angiogenesis and osteogenesis-associated factors in rabbits with avascular necrosis of the femoral head. Eur Rev Med Pharmacol Sci. 2018;22(7):2143-2152. [12] 骆瑜,李若涵,彭友俭.牙髓干细胞来源外泌体修复兔颞下颌关节软骨损伤后软骨组织修复的实验研究[J]. 临床口腔医学杂志,2020,36(4):202-205. [13] ARAÚJO AB, FURLAN JM, SALTON GD, et al. Isolation of human mesenchymal stem cells from amnion, chorion, placental decidua and umbilical cord: comparison of four enzymatic protocols. Biotechnol Lett. 2018;40(6):989-998. [14] PHAM H, TONAI R, WU M, et al. CD73, CD90, CD105 and Cadherin-11 RT-PCR Screening for Mesenchymal Stem Cells from Cryopreserved Human Cord Tissue. Int J Stem Cells. 2018;11(1):26-38. [15] KOBOLAK J, DINNYES A, MEMIC A, et al. Mesenchymal stem cells: Identification, phenotypic characterization, biological properties and potential for regenerative medicine through biomaterial micro-engineering of their niche. Methods. 2016; 99:62-68. [16] XIAO D, YE M, LI X, et al. Biomechanics analysis for the treatment of ischemic necrosis of the femoral head by using an interior supporting system. Int J Clin Exp Med. 2015;8(3):4551-4556. [17] HUA KC, YANG XG, FENG JT, et al. The efficacy and safety of core decompression for the treatment of femoral head necrosis: a systematic review and meta-analysis. J Orthop Surg Res. 2019;14(1):306. [18] THEOPOLD J, ARMONIES S, PIEROH P, et al. Nontraumatic avascular necrosis of the femoral head : Arthroscopic and navigation-supported core decompression. Oper Orthop Traumatol. 2020;32(2):107-115. [19] XU S, ZHANG L, JIN H, et al. Autologous Stem Cells Combined Core Decompression for Treatment of Avascular Necrosis of the Femoral Head: A Systematic Meta-Analysis. Biomed Res Int. 2017;2017:6136205. [20] PENG K, WANG Y, ZHU J, et al. Repair of non-traumatic femoral head necrosis by marrow core decompression with bone grafting and porous tantalum rod implantation. Pak J Med Sci. 2020;36(6):1392-1396. [21] KOSUKEGAWA I, OKAZAKI S, Yamamoto M, et al. The proton pump inhibitor, lansoprazole, prevents the development of non-traumatic osteonecrosis of the femoral head: an experimental and prospective clinical trial. Eur J Orthop Surg Traumatol. 2020;30(4):713-721. [22] FAN L, ZHANG C, YU Z, et al. Transplantation of hypoxia preconditioned bone marrow mesenchymal stem cells enhances angiogenesis and osteogenesis in rabbit femoral head osteonecrosis. Bone. 2015;81:544-553. [23] HERNIGOU P, FLOUZAT-LACHANIETTE CH, DELAMBRE J, et al. Osteonecrosis repair with bone marrow cell therapies: state of the clinical art. Bone. 2015;70:102-109. [24] JUNIOR AL, PINHEIRO CCG, TANIKAWA DYS, et al. Mesenchymal Stem Cells from Human Exfoliated Deciduous Teeth and the Orbicularis Oris Muscle: How Do They Behave When Exposed to a Proinflammatory Stimulus? Stem Cells Int. 2020; 2020:3670412. [25] AYOUB S, BERBÉRI A, FAYYAD-KAZAN M. An update on human periapical cyst-mesenchymal stem cells and their potential applications in regenerative medicine. Mol Biol Rep. 2020;47(3):2381-2389. [26] HENG BC, ZHU S, XU J, et al. Effects of decellularized matrices derived from periodontal ligament stem cells and SHED on the adhesion, proliferation and osteogenic differentiation of human dental pulp stem cells in vitro. Tissue Cell. 2016;48(2):133-143. [27] CHEN Y, ZHANG F, FU Q, et al. In vitro proliferation and osteogenic differentiation of human dental pulp stem cells in injectable thermo-sensitive chitosan/β-glycerophosphate/hydroxyapatite hydrogel. J Biomater Appl. 2016;31(3):317-327. [28] PINHEIRO CCG, LEYENDECKER JUNIOR A, TANIKAWA DYS, et al. Is There a Noninvasive Source of MSCs Isolated with GMP Methods with Better Osteogenic Potential? Stem Cells Int. 2019;2019:7951696. [29] CHAMIEH F, COLLIGNON AM, COYAC BR, et al. Accelerated craniofacial bone regeneration through dense collagen gel scaffolds seeded with dental pulp stem cells. Sci Rep. 2016;6:38814. [30] KERMANI S, MEGAT ABDUL WAHAB R, ZARINA ZAINOL ABIDIN I, et al. Differentiation capacity of mouse dental pulp stem cells into osteoblasts and osteoclasts. Cell J. 2014;16(1):31-42. [31] WOLOSZYK A, HOLSTEN DIRCKSEN S, BOSTANCI N, et al. Influence of the mechanical environment on the engineering of mineralised tissues using human dental pulp stem cells and silk fibroin scaffolds. PLoS One. 2014;9(10):e111010. [32] 毛丽霞,刘加强,赵晶蕾,等.人牙髓干细胞体外成骨向分化的实验研究[J].口腔颌面修复学杂志,2014,15(4):193-198. [33] WANG C, WANG Y, MENG HY, et al. Application of bone marrow mesenchymal stem cells to the treatment of osteonecrosis of the femoral head. Int J Clin Exp Med. 2015;8(3):3127-3135. [34] HERNIGOU P, BEAUJEAN F. Abnormalities in the bone marrow of the iliac crest in patients who have osteonecrosis secondary to corticosteroid therapy or alcohol abuse. J Bone Joint Surg Am. 1997;79(7):1047-1053. |
| [1] | Tan Xinfang, Guo Yanxing, Qin Xiaofei, Zhang Binqing, Zhao Dongliang, Pan Kunkun, Li Yuzhuo, Chen Haoyu. Effect of uniaxial fatigue exercise on patellofemoral cartilage injury in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(在线): 1-6. |
| [2] | Wang Baojuan, Zheng Shuguang, Zhang Qi, Li Tianyang. Miao medicine fumigation can delay extracellular matrix destruction in a rabbit model of knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1236-1242. |
| [3] | Lü Yiyan, Li Hanbing, Ma Xiaoqing, Zhang Han, Zhang Yuhang, Li Genlin. Establishment and characteristic analysis of interior heat and diabetes mouse model using compound factors [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1243-1249. |
| [4] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1316-1322. |
| [5] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1323-1329. |
| [6] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1330-1335. |
| [7] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [8] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [9] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1005-1011. |
| [10] | Zhou Ying, Zhang Huan, Liao Song, Hu Fanqi, Yi Jing, Liu Yubin, Jin Jide. Immunomodulatory effects of deferoxamine and interferon gamma on human dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1012-1019. |
| [11] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 985-991. |
| [12] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1020-1026. |
| [13] | Wang Jifang, Bao Zhen, Qiao Yahong. miR-206 regulates EVI1 gene expression and cell biological behavior in stem cells of small cell lung cancer [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1027-1031. |
| [14] | Liu Feng, Peng Yuhuan, Luo Liangping, Wu Benqing. Plant-derived basic fibroblast growth factor maintains the growth and differentiation of human embryonic stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1032-1037. |
| [15] | Wen Dandan, Li Qiang, Shen Caiqi, Ji Zhe, Jin Peisheng. Nocardia rubra cell wall skeleton for extemal use improves the viability of adipogenic mesenchymal stem cells and promotes diabetes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1038-1044. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||