Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (10): 1785-1792.doi: 10.3969/j.issn.2095-4344.2013.10.013
Previous Articles Next Articles
Identification of marker molecules for negative screening of pancreatic stem cells
Feng Rui-cheng, Lang Yan-he, Liang Yang, Wu Ying, Teng Chun-bo
- College of Life Science, Northeast Forestry University, Harbin 150040, Heilongjiang Province, China
-
Received:2012-11-12Revised:2012-12-11Online:2013-03-05Published:2013-03-05 -
Contact:Teng Chun-bo, M.D., Professor, College of Life Science, Northeast Forestry University, Harbin 150040, Heilongjiang Province, China chunboteng@yahoo.com.cn -
About author:Feng Rui-cheng★, Studying for master’s degree, College of Life Science, Northeast Forestry University, Harbin 150040, Heilongjiang Province, China
CLC Number:
Cite this article
Feng Rui-cheng, Lang Yan-he, Liang Yang, Wu Ying, Teng Chun-bo. Identification of marker molecules for negative screening of pancreatic stem cells[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(10): 1785-1792.
share this article
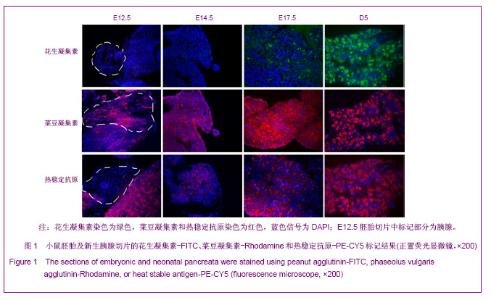
2.1 PNA、PHA和HSA对小鼠胚胎时期胰腺细胞的标记结果 胰腺干/祖细胞起源于胚胎期胰芽中。在小鼠E8.5到E9.5,前后肠交界处内胚层上皮向胰腺细胞命运特化,胰腺干细胞出现于从原始肠管外凸期的胰腺芽基中,这个时期称为第1次转换;在E13.5到E15.5,未分化的胰腺干/祖细胞开始向内外分泌方向快速分化,这个时期称为第2次转换,2次转换的胚胎胰腺中包含大量前体细胞;在E17.5到新生胰腺中,腺泡细胞大量增加,内分泌细胞聚集成胰岛,这段时期称为第3次转换[24-25]。 为了研究PNA、PHA和HSA对胚胎期胰腺干/祖细胞、前体细胞和分化细胞的标记情况,对小鼠E12.5、E14.5、E17.5及出生后5 d(D5)胰腺切片进行了染色。结果发现,在E12.5和E14.5,PNA不与任何胰腺细胞结合;在E17.5,PNA能结合到清晰可辨的腺泡细胞中;出生后5 d,PNA与腺泡细胞强烈结合,见图1。因而,PNA不能标记胰腺干/祖细胞及前体细胞,而强标记分化后的腺泡细胞。"
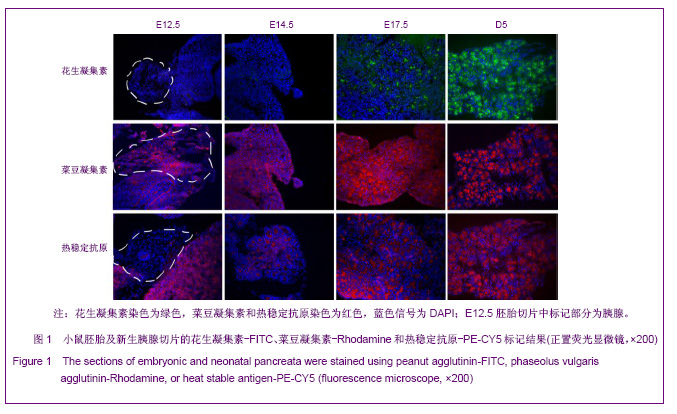
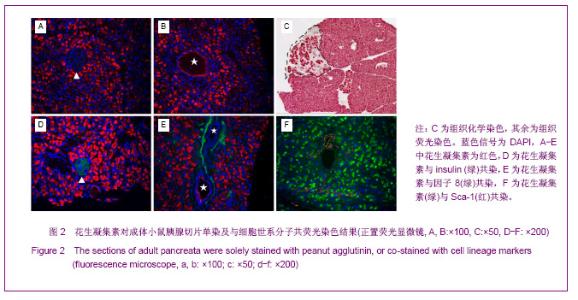
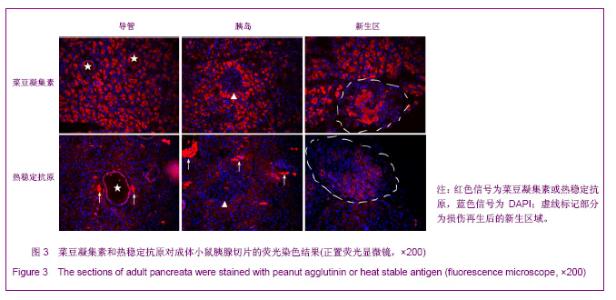
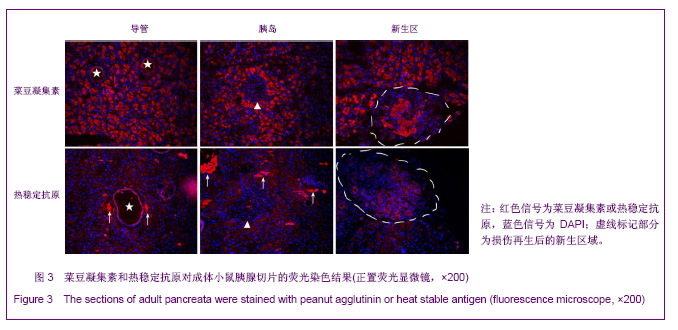
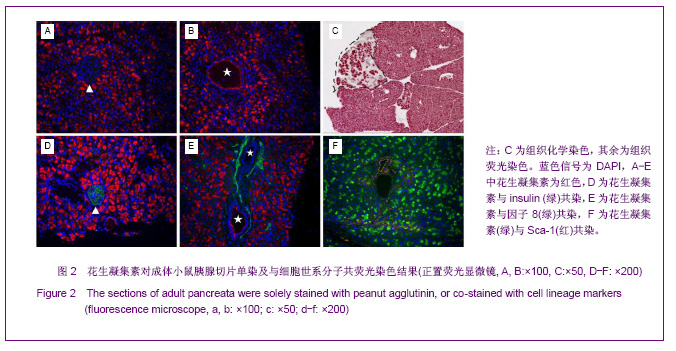
利用PHA对胚胎胰腺染色发现,PHA与E12.5大部分胰腺细胞弱结合;在E14.5,PHA结合的胰腺细胞数量增加;在E17.5,PHA与大多数胰腺细胞强结合;出生后5 d,PHA能与结构清晰可辨的腺泡细胞强烈结合,见图1。早期胰芽的广泛染色结果暗示,PHA不仅能够标记分化的腺泡细胞,也能标记胰腺祖及前体细胞。 HSA在E12.5小鼠胰腺仅能与胰芽部分上皮细胞结合;在E14.5,HSA与胰腺芽基初步分化产生的管状上皮细胞结合;在E17.5和出生后5 d,HSA能与大多数胰腺细胞结合,见图1。因而,HSA对正在分化的胰腺祖细胞、前体和终末分化的腺泡能很好地标记。 2.2 PNA、PHA和HSA对小鼠成体胰腺细胞的标记结果 见图2。 进一步利用荧光组织化学染色分析了PNA、PHA和HSA对成体小鼠胰腺细胞的标记情况。结果发现,PNA能与腺泡细胞强结合,与部分导管上皮内侧也有结合,但与胰腺组织中的某些区域不能结合,见图2A,B。 为了进一步分析PNA不能结合哪些细胞,作者利用细胞世系标志分子与PNA进行了共染色。因子8常被作为血管上皮的特异性标记分子,Sca-1是造血世系细胞的标志性分子,胰岛素能够特异性的标记胰岛细胞。共染结果发现,PNA不能与insulin共染,见图2D,不能与因子8共染,见图2E,也不能与Sca-1共定位,见图2F,表明PNA不能标记胰岛细胞、血管内皮细胞及造血世系干细胞。 已有研究发现,胰腺损伤后干/祖细胞被刺激增殖分化,参与胰腺再生[26]。胰腺大部切除后再生过程中能产生新生区,其中包括未分化的胰腺祖细胞[23,26]。为了进一步分析PNA对成体胰腺干/祖细胞的标记情况,作者制作了小鼠胰腺损伤再生模型。结果发现,在损伤再生胰腺中,PNA不但能与正常组织腺泡细胞结合,还能与再生组织中的腺泡细胞结合,见图2C,但不能与未分化细胞结合。上述结果表明,PNA可以作为负向筛选分子排除腺泡及部分导管细胞。 与PNA相类似,PHA能与正常及损伤后成体胰腺腺泡细胞结合,也能与部分导管上皮细胞所分泌的蛋白结合,但不能结合胰岛,见图3。HSA能弱标记成体腺泡细胞、导管细胞(尤其是小导管)和部分胰岛细胞,强标记血管及血细胞,见图3。在损伤再生胰腺中,HSA与再生的管状上皮细胞结合,见图3。结合胚胎期的染色结果,HSA在成体胰腺中可以标记大部分分化的胰腺细胞类型,正在分化的祖细胞和前体细胞,及血管和造血世系细胞。"
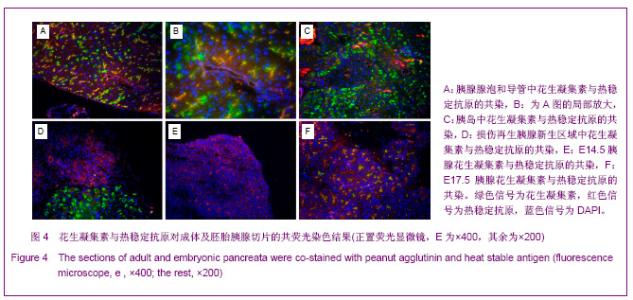
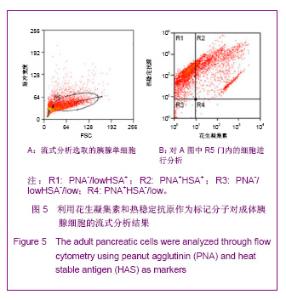
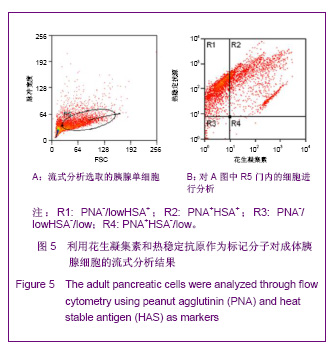
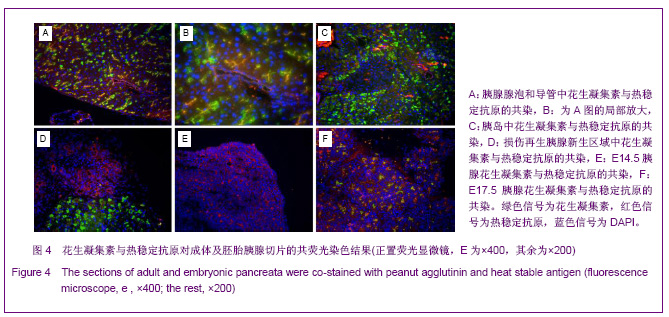
2.3 PNA与HSA在成体及胚胎小鼠胰腺中共染分析结果 见图4。 进一步检测PNA和HSA共同使用对成体及胚胎小鼠胰腺细胞的标记情况。PNA和HSA共染能结合腺泡细胞、导管上皮细胞、血管内皮细胞、血细胞,及部分胰岛细胞,见图4A-C。在损伤再生的小鼠胰腺中,PNA和HSA共染不但能与上述正常胰腺细胞结合,而且还能与再生组织中正在分化的前体细胞及刚分化的腺泡细胞结合,见图4C。PNA和HSA共同使用对胚胎胰腺染色结果相类似,能够标记正在分化的腺泡、导管及已经完全分化的腺泡和导管细胞,见图4E,F。以上结果表明,PNA和HSA可作为负向筛选分子从胚胎及成体胰腺中富集胰腺干细胞。 2.4 PNA与HSA作为负向筛选标志分子对成体胰腺细胞的流式分析结果 利用流式细胞术分析了PNA和HSA阳性及阴性细胞所占的比例。结果显示,PNA和HSA可将细胞分为4群:PNA+HSA+、PNA+HSA-/low、PNA-/lowHSA+和PNA-/lowHSA-/low,在成体中所占比例分别为69.73%,0.06%,28.61%和1.60%,见图5。PNA+HSA+和PNA-/lowHSA+细胞占胰腺细胞总量的98.34%,与前述免疫染色结果相吻合,而PNA-/ lowHSA-/low细胞仅占细胞总量的1.6%,见图5B,符合干/祖细胞含量较低的特点。"
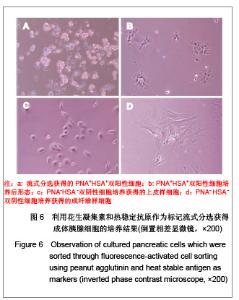
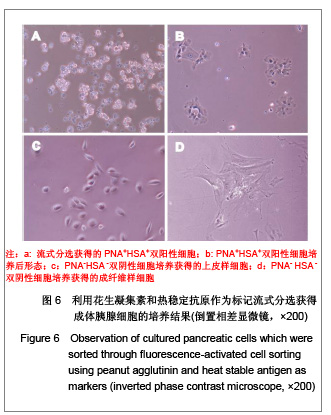
2.5 流式分选细胞的体外培养观察结果 对PNA和HSA作为标记进行流式分选的细胞原代培养,相差显微镜下观察发现:PNA和HSA荧光最强(PNA+HSA+)的细胞体积大,分选后在显微镜下观察可见细胞内有很多酶原颗粒,培养时呈现腺泡细胞培养特征,见图6A,B,传代后细胞均死亡。HSA+细胞表现为小而亮,在培养时不贴壁也不生长。而PNA-/lowHSA-/low细胞培养后形态上包括两群:一群具有早期胰腺发育期细胞的形态学特征,即体积较小、为不规则三角形或多角形的上皮细胞特征,可进行连续传代培养,见图6C;另一群细胞在培养5-7 d后呈现成纤维细胞形态,传代培养时抑制胰腺来源的上皮细胞生长,见图6D。"
| [1] Jiang FX, Morahan G. Pancreatic Stem Cells: From Possible to Probable. Stem Cell Rev. 2012;8(3):647-657. [2] Wagner RT, Lewis J, Cooney A, et al. Stem cell approaches for the treatment of type 1 diabetes mellitus. Transl Res. 2010; 156(3):169-179. [3] Stanekzai J, Isenovic ER, Mousa SA. Treatment options for diabetes: Potential role of stem cells. Diabetes Res Clin Pract. 2012. pii: S0168-8227(12)00314-2.[4] Sims E, Evans-Molina C. Stem cells as a tool to improve outcomes of islet transplantation. J Transplant. 2012; 2012: 736491.[5] Godfrey KJ, Mathew B, Bulman JC, et al. Stem cell-based treatments for Type 1 diabetes mellitus: bone marrow, embryonic, hepatic, pancreatic and induced pluripotent stem cells. Diabet Med. 2012; 29(1):14-23.[6] Zhang LX, Teng CB, An TZ. Shengwu Gongcheng Xuebao. 2008;24(2): 177-182. 张丽新,滕春波,安铁洙.利用成体干细胞治疗糖尿病[J].生物工程学报,2008,24(2):177-182.[7] Ramiya VK, Maraist M, Arfors KE, et al. Reversal of insulin-dependent diabetes using islets generated in vitro from pancreatic stem cells. Nat Med. 2000; 6:278-282.[8] Hao E, Tyrberg B, Itkin-Ansari P, et al. Beta-cell differentiation from nonendocrine epithelial cells of the adult human pancreas. Nat Med. 2006; 12:310-316.[9] Noguchi H, Oishi K, Ueda M, et al. Establishment of mouse pancreatic stem cell line. Cell Transplant. 2009; 18:563-571.[10] Smukler SR, Arntfield ME, Razavi R, et al. The adult mouse and human pancreas contain rare multipotent stem cells that express insulin. Cell Stem Cell. 2011; 8:281-93.[11] Ioannou M, Serafimidis I, Arnes L, et al. ALDH1B1 is a potential stem/progenitor marker for multiple pancreas progenitor pools. Dev Biol.2012. pii: S0012-1606(12)00601-X.[12] Wang H, Wang S, Hu J, et al. Oct4 is expressed in Nestin-positive cells as a marker for pancreatic endocrine progenitor. Histochem Cell Biol. 2009;131(5):553-563. [13] Inada A, Nienaber C, Katsuta H, et al. Carbonic anhydrase II-positive pancreatic cells are progenitors for both endocrine and exocrine pancreas after birth. Proc Natl Acad Sci U S A. 2008;105(50):19915-19919.[14] Zulewski H, Abraham EJ, Gerlach MJ, et al. Multipotential nestin-positive stem cells isolated from adult pancreatic islets differentiate ex vivo into pancreatic endocrine, exocrine, and hepatic phenotypes. Diabetes. 2001; 50:521-533.[15] Suzuki A, Nakauchi H, Taniguchi H. Prospective isolation of multipotent pancreatic progenitors using flow-cytometric cell sorting. Diabetes. 2004; 53: 2143-2152.[16] Oshima Y, Suzuki A, Kawashimo K, et al. Isolation of mouse pancreatic ductal progenitor cells expressing CD133 and c-Met by flow cytometric cell sorting. Gastroenterology. 2007; 132(2):720-732.[17] Rovira M, Scott SG, Liss AS, et al. Isolation and characterization of centroacinar/terminal ductal progenitor cells in adult mouse pancreas. Proc Natl Acad Sci USA. 2010; 107: 75-80.[18] Zhou Q, Law AC, Rajagopal J, et al. A Multipotent Progenitor Domain Guides Pancreatic Organogenesis. Dev Cell. 2007; 13(1):103-114.[19] Wagers AJ, Allsopp RC, Weissman IL. Changes in integrin expression are associated with altered homing properties of Lin(-/lo)Thy1.1(lo)Sca-1(+)c-kit(+) hematopoietic stem cells following mobilization by cyclophosphamide/granulocyte colony-stimulating factor. Exp Hematol. 2002; 30:176-185.[20] Toyoda M, Yamazaki-Inoue M, Itakura Y, et al. Lectin microarray analysis of pluripotent and multipotent stem cells. Genes Cells. 2011; 16(1): 1-11.[21] Kitada M, Kuroda Y, Dezawa M. Lectins as a tool for detecting neural stem/progenitor cells in the adult mouse brain. Anat Rec (Hoboken). 2011; 294(2): 305-321.[22] Rietze RL, Valcanis H, Brooker GF, et al. Purification of a pluripotent neural stem cell from the adult mouse brain. Nature. 2001; 412(6848): 736-739.[23] Zhang LX, Ju XF, Wang F, et al. Shengwu Gongcheng Xuebao. 2008; 24(4) :604-609. 张丽新,鞠晓芳,王法,等.利用假型反转录病毒对大部分胰腺切除后再生细胞的世系追踪[J].生物工程学报,2008,24(4):604-609.[24] Slack JM. Developmental Biology of the Pancreas. Development.1995;121(6):1569-1580.[25] Jensen J. Gene regulatory factors in pancreatic development. Dev Dyn. 2004; 229:176-200.[26] Bonner-weir S, Baxter LA, Schuppin GT, et al. A second pathway for regegneration of adult exocrine and endocrine pancreas: A possible recapitulation of embryonic development. Diabetes. 1993; 42: 1715-1720.[27] Kaligin MS, Gumerova AA, Titova MA, et al. C-kit is a marker of human pancreatic endocrinocyte stem cells. Morfologiia. 2011; 140(4):32-37. [28] Lin HT, Chiou SH, Kao CL, et al. Characterization of pancreatic stem cells derived from adult human pancreas ducts by fluorescence activated cell sorting. World J Gastroenterol. 2006; 12(28):4529-4535.[29] Newman RA, Klein PJ, Rudland PS. Binding of peanut lectin to breast epithelium, human carcinomas, and a cultured rat mammary stem cell: use of the lectin as a marker of mammary differentiation. J Natl Cancer Inst. 1979; 63: 1339-1346.[30] Schulte BA, Spicer SS. Histochemical methods for characterizing secretory and cell surface sialoglycoconjugates. J Histochem Cytochem. 1985;33:427-438.[31] Barresi G, Vitarelli E, Grosso M, et al. Peanut lectin binding sites in human foetal and neonatal pancreas. Eur J Histochem. 1993; 37(4):329-334.[32] Venable A, Mitalipova M, Lyons I, et al. Lectin binding profiles of SSEA-4 enriched, pluripotent human embryonic stem cell surfaces. BMC Dev Biol. 2005; 5:15.[33] Allen LW, Svenson RH, Yachnin S. Purification of mitogenic proteins derived from Phaseolus vulgaris: isolation of potent and weak phytohemagglutinins possessing mitogenic activity. Proc Natl Acad Sci U S A. 1969; 63(2):334-341.[34] Wang X, Hu J, Zhao D, et al. Nestinneg CD24low/- population from fetal Nestin-EGFP transgenic mice enriches the pancreatic endocrine progenitor cells. Pancreas. 2005; 31(4): 385-391.[35] Li D, Feng RC, Zhang LX, et al. Xiandai Yixue Jinzhan. 2011; 11(12):2242-2246. 李丹,冯锐成,张丽新,等.通过G418处理离体纯化小鼠胰腺上皮细胞[J]. 现代医学进展, 2011; 11(12):2242-2246.[36] Miyazaki S, Yamato E, Miyazaki J. Regulated expression of pdx-1 promotes in vitro differentiation of insulin-producing cells from embryonic stem cells. Diabetes. 2004; 53(4): 1030-1037.[37] Zhou L, Pelengaris S, Abouna S, et al. Re-expression of IGF-II is important for beta cell regeneration in adult mice. PLoS One. 2012; 7(9):e43623. [38] Domínguez-Bendala J, Lanzoni G, Inverardi L, et al. Concise review: mesenchymal stem cells for diabetes. Stem Cells Transl Med.2012; 1:59-63. [39] Kadam S, Muthyala S, Nair P, et al. Human placenta-derived mesenchymal stem cells and islet-like cell clusters generated from these cells as a novel source for stem cell therapy in diabetes. Rev Diabet Stud. 2010;7(2):168-182.[40] Xu X, D'Hoker J, Stangé G, et al. Beta cells can be generated from endogenous progenitors in injured adult mouse pancreas. Cell. 2008; 132(2):197-207. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [3] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [4] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [5] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [6] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [7] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [8] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [9] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [10] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [11] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [12] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [13] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [14] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [15] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||