Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (6): 1122-1128.doi: 10.3969/j.issn.2095-4344.2013.06.029
Previous Articles Next Articles
Relationships between mesenchymal stem cells and tumor
Dong Hui-yue, Wang Jin, Huang Liang-hu, Tan Jian-ming
- Fujian Key Laboratory of Transplantation Biology, Fuzhou General Hospital of Nanjing Military Command, Fuzhou 350025, Fujian Province, China
-
Received:2012-06-23Revised:2012-08-11Online:2013-02-05Published:2013-02-05 -
Contact:Tan Jian-ming, Doctoral supervisor, Fujian Key Laboratory of Transplantation Biology, Fuzhou General Hospital of Nanjing Military Command, Fuzhou 350025, Fujian Province, China tanjianming59@yahoo.com.cn -
About author:Dong Hui-yue★, Master, Fujian Key Laboratory of Transplantation Biology, Fuzhou General Hospital of Nanjing Military Command, Fuzhou 350025, Fujian Province, China lilylinglong@163.com
CLC Number:
Cite this article
Dong Hui-yue, Wang Jin, Huang Liang-hu, Tan Jian-ming. Relationships between mesenchymal stem cells and tumor[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(6): 1122-1128.
share this article

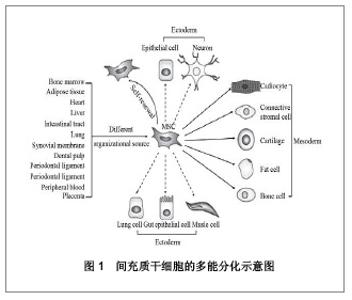
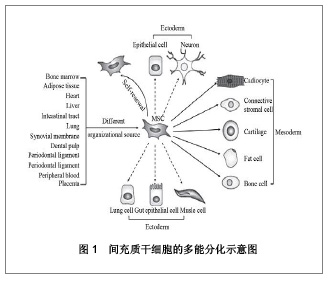
间充质干细胞不仅可以从骨髓中分离获得,还可以从其他组织中获得,例如:脐带、脂肪组织、羊膜液、外周血、胰腺组织等等。一些研究已显示间充质干细胞可以归巢到瘢痕和肿瘤组织的微环境,代替受损细胞的功能并活跃增殖。另外间充质干细胞具有低免疫原性的特点,到目前为止临床大量应用间充质干细胞治疗移植物抗宿主病和自身免疫性疾病。 2.2 间充质干细胞的肿瘤趋向性 间充质干细胞能特异性的归巢到各种肿瘤组织,第1次证实间充质干细胞的肿瘤趋向性的研究是大鼠同基因神经胶质瘤的间充质干细胞移植,随后大量研究证实间充质干细胞向肿瘤及转移肿瘤部位的趋向性。间充质干细胞向肿瘤部位迁移的特性在一些前临床体外迁移实验和肿瘤动物模型实验中得到证实,例如:恶性神经胶质瘤[2-4]、乳腺癌[5-7]、卡波西肉瘤、黑色素瘤、结肠癌[8]、卵巢癌和肺癌[9-10],以及在肿瘤过程中发生的转移[10]。间充质干细胞归巢至肿瘤组织的确切机制尚未明确,其最可能的机制是来自肿瘤的趋化因子和生长因子的释放,包括肿瘤细胞特异性受体和可溶性的肿瘤衍生因子,这些肿瘤细胞的特异性受体和因子可以和间充质干细胞表面受体结合。肿瘤细胞分泌的因子包括基质细胞衍生因子1、肿瘤细胞坏死因子α、白细胞介素类,其他一些经鉴定和未鉴定的炎症递质[11-12]。 CXCR4是牵涉到细胞迁移和归巢的最公认的趋化因子受体,研究发现CXCR4在间充质干细胞中高表达,并且主要在胞内表达。体外三维培养的间充质干细胞增强基质细胞衍生因子1α信号通路,此信号通路增强可以恢复基质细胞衍生因子1α的受体CXCR4的功能和归巢能力,对治疗应用发挥了决定性作用[13]。间充质干细胞归巢的机制也可能与白细胞从脉管系统外渗的机制相似,同样有黏附分子和整合素的参与。间充质干细胞沿血管壁内皮细胞表面滚动的过程中,P-选择素在间充质干细胞与内皮细胞结合上发挥了重要作用,极迟抗原4是黏附分子的一种整合素与其相对应的配体血管细胞黏附分子1构成极迟抗原4/血管细胞黏附分子1轴,在间充质干细胞黏附到内皮细胞上有重要作用,并且它们中的任一个经过抗体处理后均能减少间充质干细胞黏附到内皮细胞。可以推测,间充质干细胞与内皮细胞黏附、结合以及沿着内皮细胞血管壁滚动都涉及间充质干细胞归巢[14]。肿瘤的微环境被认为是慢性炎症部位,这种环境可以通过可溶性因子的释放介导间充质干细胞的迁移。最新研究发现肿瘤微环境中的炎症程度在间充质干细胞的募集能力中发挥了作用,在间充质干细胞-干扰素β介导的胰腺癌治疗研究中发现:抗炎治疗后间充质干细胞向肿瘤植入能力下降,逆转了对肿瘤的抑制作用[15]。 间充质干细胞到达肿瘤之后,表现出促肿瘤,发挥免疫抑制作用,抑制肿瘤凋亡[16],刺激上皮到间质转变,血管生成,增殖,外渗,迁移和转移[17]。 另外,在肉瘤、白血病研究中发现间充质干细胞具有抗肿瘤的作用,永生化的间充质干细胞还可以抑制原发肿瘤的生长和集落的形成[18]。 2.3 未修饰的间充质干细胞对体内外肿瘤的作用 2.3.1 促进肿瘤生长 许多研究发现间充质干细胞可以促进肿瘤生长。Karnoub等[5]将绿色荧光蛋白标记的人乳腺癌细胞系(MCF/Ras,MDA-MB-231, MDA-MB-435,HMLER)与间充质干细胞以1∶3的比例共同移植到免疫缺陷的小鼠体内时发现:间充质干细胞在MCF/Ras细胞系中加速肿瘤生长,而在其他3株细胞中未影响肿瘤的生长,但是间充质干细胞可以促进四株人乳腺癌细胞系的转移。成人和胚胎来源的间充质干细胞与结肠癌细胞(SW480和F6)共同移植到鼠的异体移植模型中发现:肿瘤的发生率明显提高,成人和胚胎来源的间充质干细胞具有相似的促进肿瘤增长的作用,但是成人来源的间充质干细胞比胚胎来源的间充质干细胞更能促进肿瘤的发生[19]。在没有与间充质干细胞共同移植的情况下,B16黑色素瘤细胞移植到同基因小鼠体内不会形成肿瘤,这也提示了间充质干细胞的免疫抑制的作用。间充质干细胞也对人滤泡型B淋巴细胞瘤的体外存活提供保护作用,并且经过肿瘤坏死因子α和淋巴霉素-α1β2处理的间充质干细胞保护作用增强[20]。 脂肪组织富含间充质干细胞,研究显示在同基因的小鼠模型中,脂肪来源的间充质干细胞可以促进肿瘤生长[21]。脂肪来源的间充质干细胞与乳腺癌细胞共同移植,与单独移植乳腺癌细胞相比,肿瘤增大,形成肿瘤速度加快[22]。另研究显示人脂肪来源的间充质干细胞可以促进裸鼠体内肿瘤细胞的生长,将人脂肪来源的间充质干细胞与肺癌细胞(H460)或神经胶质瘤细胞(U87MG)皮下注射到裸鼠体内,在肿瘤的大小和存活的肿瘤细胞数量上均明显增加[23]。 2.3.2 抑制肿瘤生长 许多研究报道间充质干细胞可以促进肿瘤生长,但是另外一些研究显示间充质干细胞也可以抑制肿瘤生长。间充质干细胞与结肠癌细胞按1∶1或10∶1的比例共同移植到大鼠里内,间充质干细胞可以抑制大鼠结肠癌的生长。在高炎症性和血管发生的卡波西肉瘤模型中,间充质干细胞可以抑制肿瘤生长[24]。体内外研究发现,胎儿皮肤来源的间充质干细胞可以抑制人肝癌细胞的生长,表现为降低细胞增殖、集落形成和癌基因的表达[25]。另体外研究发现胎儿皮肤来源的间充质干细胞可以抑制乳腺癌细胞MCF-7的生长[19]。间充质干细胞分泌Dickkopf相关蛋白1,干细胞转录因子NANOG调控Dickkopf相关蛋白1,而Dickkopf相关蛋白1介导间充质干细胞的肿瘤抑制作用,研究发现脂肪来源的间充质干细胞通过分泌Dickkopf相关蛋白1抑制白血病细胞的生长[26]。 间充质干细胞通过改变细胞周期进展来抑制胰腺肿瘤的生长,体外共培养研究发现,在胰腺肿瘤细胞中间充质干细胞增加G1期细胞比例。脂肪来源的间充质干细胞移植到胰腺肿瘤模型中抑制肿瘤的生长[27]。用升温处理的间充质干细胞的上清液培养卵巢癌细胞SKOV-3,SKOV-3细胞数量减少、活力降低。间充质干细胞分泌的因子在这一过程中发挥了作用,包括血管生成素、胰岛素样生长因子结合蛋白4、神经营养因子3和趋化因子配体18[28]。 2.4 修饰的间充质干细胞对体内外肿瘤的作用 未修饰的间充质干细胞既有促肿瘤的作用,也有抗肿瘤的作用。而对间充质干细胞修饰主要是为了抑制肿瘤,达到治疗肿瘤的目的。总体来说,间充质干细胞的肿瘤趋向性及逃避免疫清除的特性在肿瘤特异基因传递的应用上具有广阔研究空间[29-30]。对间充质干细胞进行修饰进行肿瘤基因治疗的方式主要是携带:自杀基因、凋亡基因、抗血管发生基因、免疫刺激基因或者溶瘤病毒载体。间充质干细胞携带这些基因或者载体能够克服直接应用间充质干细胞带来的不利因素:安全性;不完全的肿瘤靶向作用;无效的基因治疗导致的转基因免疫原性;病毒特异性受体对肿瘤细胞作用的局限性。目前主要是通过病毒载体将需携带的基因转染进间充质干细胞,病毒载体包括:腺病毒[31]、慢病毒和其他反转录病毒[32-33],或者用于治疗和追踪的腺病毒伴随病毒。 目前的抗肿瘤治疗主要是对间充质干细胞进行基因修饰,这些基因包括干扰素类、肿瘤坏死因子相关的凋亡诱导配体(TNF-related apoptosis- inducing ligand,TRAIL)、白细胞介素类、趋化因子CX3(CX3CL1)等。在小鼠黑色素瘤肺转移模型中,干扰素α基因修饰的间充质干细胞抑制肿瘤细胞增殖,减缓肺转移灶的生长[34]。体内外研究显示,腺病毒转染的间充质干细胞表达干扰素β可以有效的杀死神经胶质瘤细胞[35]。在黑色素瘤肺转移模型中,表达干扰素β的间充质干细胞抑制肿瘤生长并延长生存期,这种抗肿瘤作用是由于肿瘤部位干扰素β释放,提示间充质干细胞移植对肿瘤靶向治疗的重要性。前列腺癌肺转移模型中,表达干扰素β的间充质干细胞可以延长生存期和降低肿瘤负荷,其可能机制是干扰素β促进肿瘤细胞凋亡、抑制血管新生和增加自然杀伤细胞的活性[36]。在人胰腺癌肿瘤模型中,转染干扰素β的间充质干细胞抑制肿瘤细胞的生长[15]。腺病毒转染的间充质干细胞表达干扰素γ在体外可以抑制白血病细胞增殖并可以引起白血病细胞的凋亡[37]。在肺肿瘤模型中,TRAIL基因修饰的间充质干细胞可以抑制肿瘤生长[38]。在肺转移癌模型中,Loebinger等[32]研究发现携带TRAIL的间充质干细胞可以减少肿瘤生长和复发,并在多数小鼠中抑制肺转移灶的生长。利用间充质干细胞作为细胞工具将分泌型的TRAIL靶向运输到人神经胶质瘤处或在肿瘤部位分泌TRAIL,慢病毒转染人间充质干细胞,使间充质干细胞携带分泌型的TRAIL(S-TRAIL),体内研究发现间充质干细胞-S-TRAIL显著抑制肿瘤细胞生长并延长小鼠生存期[39]。最新研究显示,神经胶质瘤小鼠模型中,辐照后携带TRAIL的脐带间充质干细胞的肿瘤趋向性增强,间充质干细胞-TRAIL对肿瘤细胞的促凋亡作用增强[40]。在大鼠神经胶质瘤模型中,表达白细胞介素2的间充质干细胞具有明显的抗肿瘤作用[41]。在表达白细胞介素7的间充质干细胞的研究中发现了相似的效果。将白细胞介素12转染到间充质干细胞中,转染后的间充质干细胞具有抗肿瘤功能并且免疫监视的功能得到提高。有学者用转染白细胞介素12的间充质干细胞治疗肾细胞癌,对肿瘤生长起到明显抑制作用[42]。基因修饰的间充质干细胞表达白细胞介素18在大鼠神经胶质瘤移植模型中抑制肿瘤生长,减小肿瘤体积[43]。CX3CL1能激活T细胞和NK细胞,携带CX3CL1的间充质干细胞使结肠癌和黑素瘤静脉转移到肺转移灶的数目减少[44]。erbB2是肿瘤细胞常见的表面标志,Komarova等[9]将间充质干细胞修饰成带有erbB2的人工受体(MSCs-AR),MSCs-AR静脉注射到卵巢癌模型体内,聚集到肿瘤部位的间充质干细胞明显增多,提高了间充质干细胞的靶向作用,增强间充质干细胞为基础的肿瘤治疗作用。在乳腺癌动物模型中,表达碘化钠(NIS)的间充质干细胞通过静脉内或瘤内注射后,可以长久聚集在肿瘤部位并明显降低肿瘤生长[45]。细胞毒性药物代谢的集中可以通过表达胞嘧啶脱氨酶的间充质干细胞来实现。前体药物的集中转换,5-氟胞嘧啶到5-氟尿嘧啶,可以抑制结直肠癌和黑色素瘤肿瘤的生长[46-47]。将单纯疱疹病毒胸苷激酶(HSV-tk)利用反转录病毒转染到间充质干细胞,利用HSV-tk/gcv自杀基因治疗,体内外研究发现可以杀死神经胶质瘤细胞[48]。 间充质干细胞还可作为条件复制性腺病毒的载体,在体内减少肿瘤生长和转移。这些病毒通过复制产生新生病毒释放到肿瘤组织的周围,使肿瘤细胞发生溶解破坏。在鼠的原位肺癌和乳腺癌肿瘤模型中,利用这种治疗可以延长生存期。在肺转移癌模型中也取得了同样的效果[49]。腹膜内注射携带溶瘤腺病毒的间充质干细胞到卵巢癌模型中,发现宿主生存期延长,延缓肿瘤生长[50]。"

| [1] Dominici M, Le Blanc K, Mueller I, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006; 8: 315.[2] Sasportas LS, Kasmieh R, Wakimoto H, et al. Assessment of therapeutic efficacy and fate of engineered human mesenchymal stem cells for cancer therapy. Proc Natl Acad Sci USA.2009; 106: 4822-4827.[3] Sonabend AM, Ulasov IV, Tyler MA,et al. Mesenchymal stem cells effectively deliver an oncolytic adenovirus to intracranial glioma. Stem Cells.2008;26: 831-841.[4] Yang B, Wu X, Mao Y,et al. Dual-targeted antitumor effects against brainstem glioma by intravenous delivery of tumor necrosis factor-related, apoptosis-inducing, ligand-engineered human mesenchymal stem cells. Neurosurgery. 2009; 65: 610-624; discussion 624.[5] Karnoub AE, Dash AB, Vo AP,et al. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature.2007;449:557-563.[6] Liu S, Ginestier C, Ou SJ,et al. Breast cancer stem cells are regulated by mesenchymal stem cells through cytokine networks. Cancer Res.2011;71:614-624. [7] Goldstein RH, Reagan MR, Anderson K,et al. Human bone marrowderived MSCs can home to orthotopic breast cancer tumors and promote bone metastasis. Cancer Res 2010;70: 10044-10050.[8] Menon LG, Picinich S, Koneru R, et al. Differential gene expression associated with migration of mesenchymal stem cells to conditioned medium from tumor cells or bone marrow cells. Stem Cells 2007; 25: 520-528.[9] Komarova S, Roth J, Alvarez R,et al. Targeting of mesenchymal stem cells to ovarian tumors via an artificial receptor. J Ovarian Res.2010;3:12.[10] Loebinger MR, Kyrtatos PG, Turmaine M,et al. Magnetic resonance imaging of mesenchymal stem cells homing to pulmonary metastases using biocompatible magnetic nanoparticles. Cancer Res.2009; 69: 8862-8867.[11] Gao H, Priebe W, Glod J,et al. Activation of signal transducers and activators of transcription 3 and focal adhesion kinase by stromal cellderived factor 1 is required for migration of human mesenchymal stem cells in response to tumor cell-conditioned medium. Stem Cells.2009;27:857-865.[12] Klopp AH, Spaeth EL, Dembinski JL,et al. Tumor irradiation increases the recruitment of circulating mesenchymal stem cells into the tumor microenvironment. Cancer Res.2007;67: 11687-11695.[13] Potapova IA, Brink PR, Cohen IS,et al.Culturing of human mesenchymal stem cells as three-dimensional aggregates induces functional expression of CXCR4 that regulates adhesion to endothelial cells. J Biol Chem.2008;283: 13100-13107.[14] Rüster B,Göttig S,Ludwig RJ,et al. Mesenchymal stem cells display coordinated rolling and adhesion behavior on endothelial cells.Blood.2006;108:3938-3944. [15] Kidd S, Caldwell L, Dietrich M,et al.Mesenchymal stromal cells alone or expressing interferon-beta suppress pancreatic tumors in vivo, an eff ect countered by anti-infl ammatory treatment. Cytotherapy.2010;12:615-625.[16] Ramasamy R, Lam EW-F, Soeiro I,et al. Mesenchymal stem cells inhibit proliferation and apoptosis of tumor cells: Impact on in vivo tumor growth. Leukemia.2007;21:304-310.[17] Corcoran KE, Trzaska KA, Fernandes H,et al. Mesenchymal stemcells in early entry of breast cancer into bone marrow. PloS One.2008;3:e2563.[18] Qiao L, Xu ZL, Zhao TJ,et al. Dkk-1 secreted by mesenchymal stem cells inhibits growth of breast cancer cells via depression of Wnt signalling. Cancer Lett.2008;269:67-77.[19] Zhu W, Xu W, Jiang R,et al. Mesenchymal stem cells derived frombone marrow favor tumor cell growth in vivo. Exp Mol Pathol.2006;80:267-274.[20] Ame-Thomas P, Maby-El Hajjami H, Monvoisin C,et al. Human mesenchymal stem cells isolated from bone marrow and lymphoid organs support tumor B-cell growth: role of stromal cells in follicular lymphoma pathogenesis. Blood 2007;109:693-702.[21] Muehlberg F, Song YH, Krohn A,et al. Tissue resident stem cells promote breast cancer growth and metastasis. Carcinogenesis.2009;30:589-597. [22] Muehlberg FL, Song YH, Krohn A et al. Tissue-resident stem cells promote breast cancer growth and metastasis. Carcinogenesis 2009; 30:589-597.[23] Yu JM, Jun ES, Bae YC,et al. Mesenchymal stem cells derived from human adipose tissues favor tumor cell growth in vivo. Stem Cells Dev.2008;17:463-473.[24] Khakoo AY, Pati S, Anderson SA, et al. Human mesenchymal stem cells exert potent antitumorigenic effects in a model of Kaposi’s sarcoma. J Exp Med. 2006;203:1235-1247.[25] Qiao L, Xu Z, Zhao T,et al. Suppression of tumorigenesis by human mesenchymal stem cells in a hepatoma model. Cell Res.2008;18: 500-507.[26] Zhu Y, Sun Z, Han Q et al. Human mesenchymal stem cells inhibit cancer cell proliferation by secreting DKK-1. Leukemia. 2009;23: 925-933. [27] Cousin B, Ravet E, Poglio S et al. Adult stromal cells derived from human adipose tissue provoke pancreatic cancer cell death both in vitro and in vivo. Plos One.2009;4:e6278.[28] Cho JA, Park H, Kim HK,et al. Hyperthermia-treated mesenchymal stem cells exert antitumor effects on human carcinoma cell line. Cancer 2009;115:311-323.[29] Corsten MF, Shah K. Therapeutic stem-cells for cancer treatment: Hopes and hurdles in tactical warfare. Lancet Oncol.2008;9:376-384.[30] Hall B, Dembinski J, Sasser AK,et al. Mesenchymal stem cells in cancer: Tumor-associated fibroblasts and cell-based delivery vehicles. Int J Hematol. 2007;86:8-16.[31] Nakamizo A, Marini F, Amano T,et al. Human bone marrow-derived mesenchymal stem cells in the treatment of gliomas. Cancer Res.2005;65:3307-3318.[32] Loebinger MR, Eddaoudi A, Davies D,et al. Mesenchymal stem cell delivery of TRAIL can eliminate metastatic cancer. Cancer Res.2009; 69:4134-4142.[33] Wang N, Fallavollita L, Nguyen L,et al. Autologous bone marrow stromal cells genetically engineered to secrete an igf-I receptor decoy prevent the growth of liver metastases. Mol Ther.2009;17:1241-1249.[34] Ren C,Kumar S,Chanda D,et al. Therapeutic potential of mesenchymal stem cells producing interferon-alpha in a mouse melanoma lung metastasis model. Stem cells.2008; 26:2332-2338.[35] Nakamizo A, Marini F, Amano T et al. Human bone marrow-derived mesenchymal stem cells in the treatment of gliomas. Cancer Res.2005;65:3307-3318.[36] Ren C,Kumar S,Chanda D,et al. Cancer gene therapy using mesenchymal stem cells expressing interferon-beta in a mouse prostate cancer lung metastasis model.Gene Ther. 2008;15:1446-1453.[37] Li X,Lu Y,Huang W,et al.In vitro effect of adenovirus-mediated human gamma interferon gene transfer into human mesenchymal stem cells for chronic myelogenous leukemia. Hematol Oncol.2006;24:151-158.[38] Mohr A, Lyons M, Deedigan L,et al. Mesenchymal Stem Cells expressing TRAIL lead to tumour growth inhibition in an experimentallung cancer model. J Cell Mol Med.2008; 12: 2628-2643.[39] Lata GM,Kathleen K,Hong WY,et al. Human Bone Marrow-Derived Mesenchymal Stromal Cells Expressing S-TRAIL as a Cellular Delivery Vehicle for Human Glioma Therapy. Stem Cells.2009;27:2320-2330.[40] Seong MK,Ji HO,Soon AP,et al. Irradiation Enhances the Tumor Tropism and Therapeutic Potential of Tumor Necrosis Factor-Related Apoptosis-Inducing Ligand-Secreting Human Umbilical Cord Blood-Derived Mesenchymal Stem Cells in Glioma Therapy. Stem Cells.2010;28:2217-2228.[41] Nakamura K, Ito Y, Kawano Y,et al. Antitumor effect of genetically engineered mesenchymal stem cells in a rat glioma model. Gene Ther.2004;11:1155-1164.[42] Gao P,Ding Q,Wu Z,et al.Therapeutic potential of human mesenchymal stem cells producing IL-12 in a mouse xenograft model of renal cell carcinoma. Cancer Lett. 2010; 290:157-166.[43] Xu G, Jiang XD, Xu Y, et al. Adenoviral-mediated interleukin-18 expression in mesenchymal stem cells effectively suppresses the growth of glioma in rats. Cell Biol Int.2009;33(4):466-474.[44] Xin H, Kanehira M, Mizuguchi H,et al. Targeted delivery of CX3CL1 to multiple lung tumors by mesenchymal stem cells. StemCells.2007;25:1618-1626.[45] Dwyer RM, Ryan J, Havelin RJ, et al. Mesenchymal Stem Cell-Mediated Delivery of the Sodium Iodide Symporter Supports Radionuclide Imaging and Treatment of Breast Cancer. Stem Cells.2011;29:1149-1157.[46] Kucerova L, Altanerova V, Matuskova M,et al. Adipose tissuederived human mesenchymal stem cells mediated prodrug cancer gene therapy. Cancer Res. 2007;67: 6304-6313.[47] Kucerova L, Matuskova M, Pastorakova A,et al. Cytosine deaminase expressing human mesenchymal stem cells mediated tumour regression in melanoma bearing mice. J Gene Med.2008;10:1071-1082.[48] Uchibori R,Okada T,Ito T,et al.Retroviral vector–producing mesenchymal stem cells for targeted suicide cancer gene therapy. J Gene Med.2009;11:373-381.[49] Stoff-Khalili MA,Rivera AA,Mathis JM,et al.Mesenchymal stem cells as a vehicle for targeted delivery of CRAds to lung metastases of breast carcinoma.Breast Cancer Res Treat. 2007;105:157-167.[50] Komarova S, Kawakami Y, Stoff-Khalili MA,et al. Mesenchymal progenitor cells as cellular vehicles for delivery of oncolytic adenoviruses. Mol Cancer Ther. 2006;5:755-766. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Jiang Yong, Luo Yi, Ding Yongli, Zhou Yong, Min Li, Tang Fan, Zhang Wenli, Duan Hong, Tu Chongqi. Von Mises stress on the influence of pelvic stability by precise sacral resection and clinical validation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1318-1323. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [5] | Gu Xia, Zhao Min, Wang Pingyi, Li Yimei, Li Wenhua. Relationship between hypoxia inducible factor 1 alpha and hypoxia signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1284-1289. |
| [6] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [7] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [8] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [9] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [10] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| [11] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [12] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [13] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [14] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [15] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||