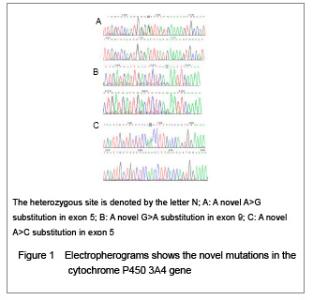
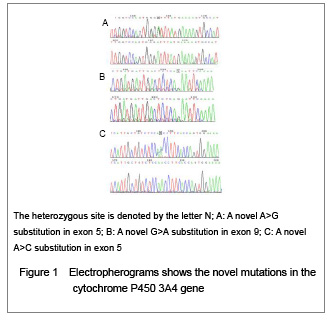
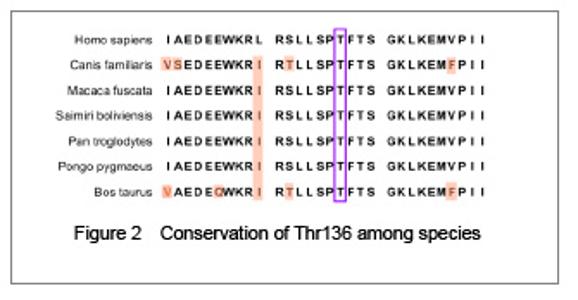
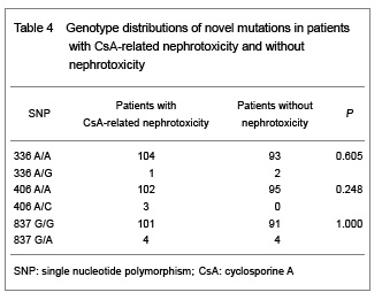
| [1] Fletcher JT, Nankivell BJ, Alexander SI. Chronic allograft nephropathy. Pediatr Nephrol. 2009;24(8):1465-1471. [2] Morales JM, Wramner L, Kreis H, et al. Sirolimus does not exhibit nephrotoxicity compared to cyclosporine in renal transplant recipients. Am J Transplant. 2002;2(5): 436-442. [3] Roy MK, Takenaka M, Kobori M, et al. Apoptosis, necrosis and cell proliferation-inhibition by cyclosporine A in U937 cells (a human monocytic cell line). Pharmacol Res. 2006; 53(3):293-302. [4] Olyaei AJ, de Mattos AM, Bennett WM. Nephrotoxicity of immunosuppressive drugs: new insight and preventive strategies. Curr Opin Crit Care. 2001;7(6):384-389. [5] Du J, Xing Q, Xu L, et al. Systematic screening for polymorphisms in the CYP3A4 gene in the Chinese population. Pharmacogenomics. 2006;7(6):831-841. [6] Ellouk-Achard S, Martin C, Pham-Huy C, et al. Implication of CYP 3A in the toxicity of cyclosporin G (CsG), cyclosporin A (Cyclosporine A) and FK506 on rat hepatocytes in primary culture. Arch Toxicol. 1997;71(7):437-442. [7] Sarah C Sim. Home Page of the Human Cytochrome P450 (CYP) Allele Nomenclature Committee. [8] Sata F, Sapone A, Elizondo G, et al. CYP3A4 allelic variants with amino acid substitutions in exons 7 and 12: evidence for an allelic variant with altered catalytic activity. Clin Pharmacol Ther. 2000;67(1):48-56. [9] Hsieh KP, Lin YY, Cheng CL, et al. Novel mutations of CYP3A4 in Chinese. Drug Metab Dispos. 2001;29(3): 268-273. [10] Lamba JK, Lin YS, Thummel K, et al. Common allelic variants of cytochrome P4503A4 and their prevalence in different populations. Pharmacogenetics. 2002;12(2): 121-132. [11] Dai D, Tang J, Rose R, et al. Identification of variants of CYP3A4 and characterization of their abilities to metabolize testosterone and chlorpyrifos. J Pharmacol Exp Ther. 2001; 299(3):825-831. [12] Hauser IA, Schaeffeler E, Gauer S, et al. ABCB1 genotype of the donor but not of the recipient is a major risk factor for cyclosporine-related nephrotoxicity after renal transplantation. J Am Soc Nephrol. 2005;16(5):1501-1511. [13] Kelly P, Kahan BD. Review: Metabolism of immunosuppressant drugs. Curr Drug Metab. 2002;3(3): 275-287.[14] Min DI, Ellingrod VL. Association of the CYP3A4*1B 5’-flanking region polymorphism with cyclosporine pharmacokinetics in healthy subjects. Ther Drug Monit. 2003;25(3):305-309. [15] Qiu XY, Jiao Z, Zhang M, et al. Association of MDR1, CYP3A4*18B, and CYP3A5*3 polymorphisms with cyclosporine pharmacokinetics in Chinese renal transplant recipients. Eur J Clin Pharmacol. 2008;64(11):1069-1084. [16] Hu YF, Qiu W, Liu ZQ, et al. Effects of genetic polymorphisms of CYP3A4, CYP3A5 and MDR1 on cyclosporine pharmacokinetics after renal transplantation. Clin Exp Pharmacol P. 2006;33(11):1093-1098. [17] Gallon L, Akalin E, Lynch P, et al. ACE gene D/D genotype as a risk factor for chronic nephrotoxicity from calcineurin inhibitors in liver transplant recipients. Transplantation. 2006;81(3):463-468. [18] Smith HE, Jones JP 3rd, Kalhorn TF, et al. Role of cytochrome P450 2C8 and 2J2 genotypes in calcineurin inhibitor-induced chronic kidney disease. Pharmacogenet Genom. 2008;18(11):943-953. [19] Hawwa AF, McKiernan PJ, Shields M, et al Influence of ABCB1 polymorphisms and haplotypes on tacrolimus nephrotoxicity and dosage requirements in children with liver transplant. Br J Clin Pharmacol. 2009;68(3):413-421. [20] Kuypers DR, de Jonge H, Naesens M, et al CYP3A5 and CYP3A4 but not MDR1 single-nucleotide polymorphisms determine long-term tacrolimus disposition and drug-related nephrotoxicity in renal recipients. Clin Pharmacol Ther. 2007; 82(6):711-725. [21] Shi L, Bao Z, Zhu Y, et al. Correlation between CYP3A5 genetic polymorphism and calcineurin inhibitor-induced chronic nephrotoxicity following renal transplantation. Zhongguo Zuzhi Gongcheng Yanjiu yu Linchuang Kangfu. 2011;15(5):806-809.[22] Zhou Q, Yu X, Shu C, et al. Analysis of CYP3A4 genetic polymorphisms in Han Chinese. J Hum Genet. 2011;56(6): 415-422. [23] Eiselt R, Domanski TL, Zibat A, et al. Identification and functional characterization of eight CYP3A4 protein variants. Pharmacogenetics. 2001;11(5):447-458. [24] State Council of the People's Republic of China. Administrative Regulations on Medical Institution. 1994-09-01. |