Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (1): 103-109.doi: 10.3969/j.issn.2095-4344.1530
Previous Articles Next Articles
Immunomodulatory properties of mesenchymal stem cells and their application in organ transplantation
Jiang Shanshan1, 2, Wang Feng3, Yu Limei1, 2, 4
- 1Key Laboratory of Cell Engineering in Guizhou Province, 2Biological Treatment Talent Base of Guizhou Province, 3Department of Cardiovascular Surgery, 4Zunyi Stem Cell and Regenerative Medicine Engineering Center, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China
-
Revised:2018-10-01Online:2019-01-08Published:2018-11-28 -
Contact:Yu Limei, MD, Professor, Master’s supervisor, 1Key Laboratory of Cell Engineering in Guizhou Province, 2Biological Treatment Talent Base of Guizhou Province, 4Zunyi Stem Cell and Regenerative Medicine Engineering Center, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China; Wang Feng, Doctorate candidate, Associate chief physician, Master’s supervisor, Department of Cardiovascular Surgery, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China -
About author:Jiang Shanshan, Master candidate, 1Key Laboratory of Cell Engineering in Guizhou Province, 2Biological Treatment Talent Base of Guizhou Province, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 81660074 (to WF); Guizhou Provincial Science and Technology Major Project, No. Qian-ke-he-zhong-da-zhuan-xiang-zi [2011]6002 (to YLM)
CLC Number:
Cite this article
Jiang Shanshan, Wang Feng, Yu Limei. Immunomodulatory properties of mesenchymal stem cells and their application in organ transplantation[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(1): 103-109.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
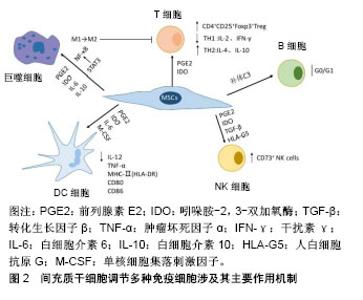
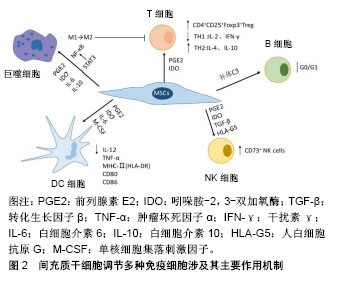
2.1 MSCs的基本特征 2006年国际细胞治疗协会(International Society for Cellular Therapy,ISCT)制定了MSCs的标准[2]:①在标准培养条件下,MSCs贴壁生长,具有类似成纤维细胞样形态;②大于95%的MSCs必须表达CD44、CD105、CD73和CD90,但不表达(≤2%)CD45、CD34、CD14(或CD11b)、CD79a(或CD19)和HLA-DR;③在体外特定条件下可诱导分化为成骨细胞、脂肪细胞和成软骨细胞。同时,MSCs大多数处于G0/G1期,表达Oct4、Nanog和SOX2等干性标志蛋白分子。 2.2 MSCs的免疫原性 来自于不同组织的MSCs均不表达或极低表达MHC-Ⅰ类分子,不表达MHC-Ⅱ类分子、Fas配体和共刺激分子B7-1、B7-2、CD40、CD40L;表达共抑制分子B7-H1和B7-H4[3-4]。而免疫应答需要细胞表达如MHC、黏附分子ICAM、共刺激分子等特异性分子及T细胞活化、功能发挥等多个环节与复杂机制的参与,一般认为效应T细胞活化依赖于MHC-Ⅱ类分子和共刺激分子,以上充分说明MSCs的低免疫原性。 应用单克隆抗体阻断B7-1的表达或沉默T细胞HLA-DRα表达,MSCs对T细胞增殖的抑制作用明显减弱[5],T细胞不能被很好地激活,从而诱导免疫耐受;移植的同种异体MSCs可以在体内存活很长一段时间后才被受体免疫细胞识别[6],也提示MSCs的低免疫原性,可能是MSCs在预防和治疗器官移植排斥反应中疗效维持的重要依据。 2.3 MSCs的免疫调节作用 2.3.1 对T淋巴细胞的影响 器官移植免疫排斥过程中的关键环节是T细胞对异种抗原的识别,其主要是两大信号途径:①直接识别,受者CD4+Th细胞或者CD8+CLT细胞可直接识别供体抗原提呈细胞(antigen presenting cells,APC)表面的MHCs分子和抗原肽-供者MHCs分子复合物;②间接识别,受者的APC如树突状细胞能够呈递供者抗原并通过自身的MHC分子-共刺激分子激活T细胞。在没有共刺激信号的情况下,受者的T细胞就处于无反应/克隆无能状态,因此通过阻断共刺激信号可以诱导免疫耐受。 调节性T细胞(regulatory T cell,Treg)是具有抑制免疫功能的CD4+T淋巴细胞,占外周CD4+T淋巴细胞的5%-10%。Treg是研究最多且临床上最为有效的Th细胞,尤其是CD4+CD25+Foxp3+Treg。主要涉及两种作用机制:①通过细胞表面标志物(CD28、CD2、LFA-1、CD8)与其他抗原提呈细胞的配体(LFA-3、ICAM-1、MHCⅠ、B7)接触来发挥抑制功能;②分泌一些免疫抑制性的细胞因子,间接发挥免疫抑制功能。最早是在小鼠皮肤移植模型中发现CD4+CD25+Treg可诱导移植免疫耐受[7],随后的研究均表明,CD4+CD25+Treg参与了多种机制诱导的移植免疫耐受。在气道炎症小鼠中,MSCs抑制过敏原特异性的Th2细胞反应就与其诱导Treg生成有关[8]。在小鼠自身免疫性脑脊髓炎模型和类风湿性关节炎患者T细胞体内外研究发现,MSCs均明显提高CD4+、CD25+Foxp3+或CD4+CD25+CD127(low/-)的Treg细胞比例[9-10]。在动物和人的器官移植研究中,MSCs能诱导同种异体移植物免疫耐受,作用机制也与Treg细胞比例上调密不可分,Treg细胞比例减少,则出现移植排斥反应[11]。MSCs还能显著抑制免疫性血小板减少症患者的Th1细胞分泌白细胞介素2和干扰素γ,同时促进Th2细胞分泌白细胞介素4和白细胞介素10,从而调节Th1/Th2的平衡;MSCs也可通过上调Treg的数量诱导ITP患者的免疫耐受[12]。脂肪MSCs还可通过减少活化的T细胞数量及促进Th2型免疫应答,而抑制鼻息肉患者的嗜酸性粒细胞性炎症反应[13],MSCs也可直接抑制CD4+T细胞增殖产生免疫抑制作用[14]。最近的研究报道,MSCs通过诱导前列腺素E2和吲哚胺-2,3-双加氧酶生成,抑制同种反应性T细胞激活,发挥免疫抑制作用[15-16]。 2.3.2 对B淋巴细胞的调节 B淋巴细胞在抗原和T细胞的双重作用下,可以大量增殖并进一步分化成浆细胞,从而分泌抗体参与免疫反应。关于MSCs对B淋巴细胞免疫调节作用的研究报道并不完全一致,但大多数的研究认为MSCs是抑制B细胞功能的。MSCs对B细胞的免疫调节作用是通过释放可溶性因子[17](干扰素γ、肿瘤坏死因子α、白细胞介素2、白细胞介素6、白细胞介素10、转化生长因子β)、细胞与细胞接触来抑制B淋巴细胞的增殖、抗体生成、分化和趋化。最近的研究还发现,在CpG刺激下,骨髓MSCs与CD3+T细胞的直接接触,明显提高MSCs对CD19+B细胞增殖的抑制效应[18]。MSCs可以将B细胞阻滞于G0/G1期,却不引起B细胞凋亡。当去除抑制因素后,MSCs对B淋巴细胞的抑制作用可被解除,B淋巴细胞增殖、转化形成IgM、IgG3的能力均可得以恢复[19],MSCs与B细胞非接触性共培养中发现,MSCs通过所分泌的物质介导B细胞终末分化主要调节因子Blimp-1表达的下调,PAX-5表达的上调来抑制B细胞的终末分化[20]。MSCs与B细胞接触可诱导MSCs产生更多的血管内皮生长因子,进而诱导AKT磷酸化增加,抑制Caspase-3介导的CD19+B细胞凋亡[21]。因而,MSCs可能是控制B细胞介导的移植物排斥和自身免疫性疾病的有效策略,然而MSCs如何与T细胞、树突状细胞或巨噬细胞相互作用,影响B细胞功能还了解甚少。 2.3.3 对NK细胞的影响 NK细胞是颗粒细胞毒性免疫细胞,除了有细胞杀伤功能外,还具有调节适应性免疫应答的作用。它们可以将自身和非自身细胞区别开来,通过裂解、激活靶细胞,产生炎症因子,引起炎症反应和组织器官损伤[22]。MSCs能够抑制活化的NK细胞的细胞毒作用以及NK细胞增殖和细胞因子分泌;也可通过降低活化标志物如NKp30、NKp44、NKG2D和CD132的表达改变NK细胞表型[23-24]。MSCs可以通过自分泌或旁分泌方式,经前列腺素E2、吲哚胺-2,3-双加氧酶、转化生长因子β和可溶性HLA-G5的调控介导对初始活化的及白细胞介素15或白细胞介素2诱导的NK细胞的增殖产生抑制效应[25-27],MSCs对NK细胞的影响仍需要细胞-细胞接触来产生[28]。另有研究认为,MSCs与NK细胞之间还具有双向调节作用[25],也就是说活化的NK细胞还可以通过受体与配体结合的溶胞途径清除非自体来源的MSCs。而MSCs表达MHC-Ⅰ分子,能躲避NK细胞的杀伤[27]。MSCs与NK细胞共培养后,CD73+在NK细胞中显著表达,但CD39+表达水平保持不变[29],表达CD73+的调节性NK细胞能够将5'-单磷酸腺苷转化为具有免疫调节作用的腺苷,从而控制炎症反应、维持免疫稳态,诱导免疫耐受[30]。 2.3.4 对树突状细胞的调节 树突状细胞作为唯一专职的抗原提呈细胞,主要是活化初始型T细胞,它在免疫应答的启动、调控和维持中起重要作用。树突状细胞不仅是T细胞启动的决定性因素,而且是维持体内自身耐受性的关键因素。未成熟的树突状细胞通常被认为是致免疫耐受性的,而成熟的树突状细胞则呈现免疫原性。在肿瘤坏死因子α刺激的条件下,不完全成熟的树突状细胞诱导无反应性T细胞或调节性T细胞(分泌白细胞介素10)增多而引起T细胞免疫耐受[31]。MSCs可影响树突状细胞的增殖、分化、抗原内吞、细胞因子释放及抗原递呈能力。当MSCs存在条件下培养树突状细胞刺激的外周血淋巴细胞时,T细胞的增殖可以被显著抑制,这种抑制效应是由MSCs所分泌的前列腺素E2所介导[32]。将同种异体胰岛和MSCs共同移植后,受体小鼠树突状细胞的成熟、内吞作用和促炎因子白细胞介素12分泌均被显著抑制[33];MSCs不仅能抑制单核细胞向树突状细胞的初始分化,还可通过MSCs衍生的前列腺素E2对早期树突状细胞的成熟和功能产生明显抑制作用[34-35];MSCs分泌的白细胞介素6和单核细胞集落刺激因子也可显著抑制树突状细胞的分化、内吞作用和白细胞介素12、肿瘤坏死因子α的分泌及诱导MHC Ⅱ类分子的低表达,而产生更高水平的白细胞介素1β和白细胞介素10[36]。当用MSCs处理成熟的树突状细胞时,抗原呈递分子HLA-DR、CD1a和共刺激分子CD80、CD86的表达降低,与下调的白细胞介素12一起,抑制成熟树突状细胞对同种异体T细胞的活化能力[34],树突状细胞转化或发育为诱导免疫耐受的免疫表型。MSCs对树突状细胞的影响是可逆的,这可能有益于更好地规避其他免疫抑制剂使用所致的长期免疫力低下等突出缺点。 2.3.5 对巨噬细胞的影响 巨噬细胞在加工呈递抗原和免疫调节方面发挥重要作用,既是一类APC,又是机体固有免疫的重要组成部分。根据巨噬细胞的激活和发挥免疫功能的方式,分为成M1型(致炎)和M2型(分泌白细胞介素10、抗炎)。在脂多糖刺激下,骨髓MSCs的Toll样受体4被激活,通过髓样分化因子88(MyD88)/NF-κB途径促进环氧合酶2(COX2)/前列腺素E2的分泌,作用于巨噬细胞的前列腺素受体EP2和EP4,诱导巨噬细胞向M2型分化,并促进抗炎因子白细胞介素10分泌[37]。随后进一步发现MSCs分泌的肿瘤坏死因子诱导蛋白6与巨噬细胞上的CD44相互作用,能减弱由酵母聚糖/TLR2/NF-κB通路诱导的炎症[38]。此外,MSCs的吲哚胺-2,3-双加氧酶激活对M2巨噬细胞的生成也有明显促进作用,而M2巨噬细胞还参与对T细胞的抑制效应,进一步增强MSCs免疫抑制作用[39]。MSCs在白细胞介素6的参与下通过前列腺素E2调节免疫抑制功能,同时前列腺素E2的分泌对M1巨噬细胞向M2巨噬细胞转化产生协同作用。在类风湿性关节炎小鼠中,使用白细胞介素6缺乏的MSCs,显著降低了在体内诱导Th2细胞的免疫应答作用,表明MSCs的免疫抑制功能主要依赖于白细胞介素6激活的信号通路,且前列腺素E2可能是其作用的下游信号分子[40];在诱导破骨细胞分化时,COX2/前列腺素E2能够刺激白细胞介素6产生,而白细胞介素6又反馈诱导破骨细胞中前列腺素E2、COX2的分泌增加[41]。前列腺素E2所介导的巨噬细胞分泌的白细胞介素10主要是p38-MAPK信号途径被激活所致,而白细胞介素6的增高与此信号途径无关。MSCs还可通过STAT3途径诱导M2型巨噬细胞的极化,从而抑制免疫炎症反应[42]。以上结果证实MSCs可影响巨噬细胞向M2型巨噬细胞的转化,增强了免疫抑制功能,减轻了免疫炎症反应。MSCs调节各免疫细胞涉及的相关机制,见图2。"
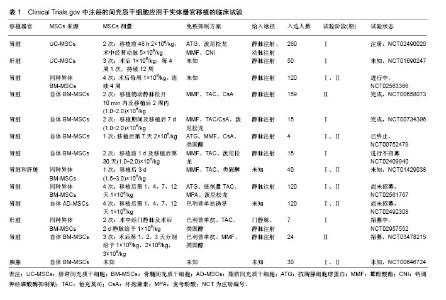
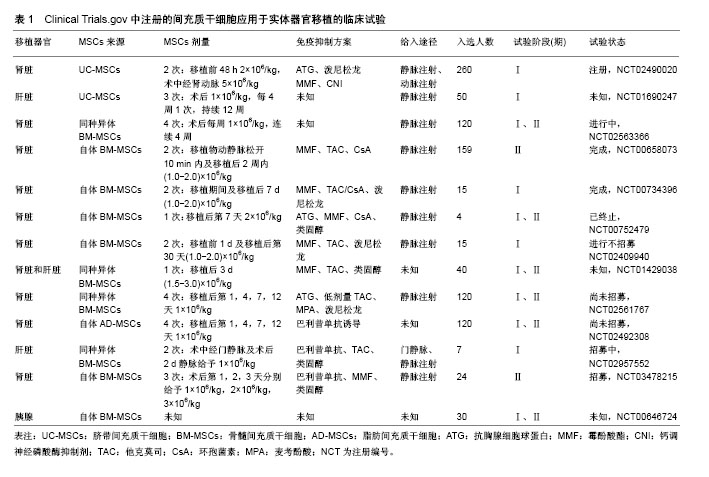
2.4 MSCs免疫调节特性在器官移植中的临床应用 MSCs对T细胞、B细胞、树突状细胞、NK细胞和巨噬细胞等发挥独特的免疫调节作用,显著降低排斥反应的发生,是其能够应用于器官移植领域的前提。大量基础研究及一些动物实验、临床研究已经证实,MSCs在调节免疫细胞功能和改善同种异体造血干细胞移植物抗宿主病及治疗自身免疫性疾病方面展现明显疗效,也为MSCs治疗器官移植排异反应奠定了较好的理论和实践依据。截止2018年3月,基于不同来源的MSCs(骨髓、脐带、脂肪等)在Clinical Trials.gov注册了734项临床试验[43],其中器官移植方面有13项,主要应用于肝、肾、胰腺移植领域,见表1。Vanikar等[44]首先使用供者脂肪MSCs进行了一项非随机临床试验,其主要目的是在100例终末期肾病患者中诱导肾移植的免疫低反应性,经过18个月随访,与100例对照组相比较,脂肪MSCs组移植物的存活率显著提高,肾小球滤过率高于对照组水平10.0 mL/min,肾功能明显改善,产生了持续的低水平的免疫抑制效应,此后的其他研究也得到了类似的结果。同期的一项为期1年的前瞻性、随机非盲临床试验中,比较了自体骨髓MSCs输注与抗CD25抗体巴利昔单抗诱导治疗结果的不同,在肾移植后1个月以及1年的随访期中观测到MSCs组肾小球滤过率明显高于对照组(平均差异9.1 mL/min),移植后肾功能得到了更明显的改善,同时,MSCs组的105例中仅有8例(7.5%)经活检证实存在急性排斥反应,低于对照组51例中有11例(21.6%)。MSCs组的急性排斥反应的发生率和排斥反应的严重程度均明显降低,机会性感染的发生率也较低[45]。脂肪或供者来源的MSCs结合低剂量环孢素A在肾脏移植的临床试验中也有相同的报道,即联合MSCs治疗器官移植排斥反应的效果更佳[46-47]。也有文献报道,短期内MSCs与低剂量免疫抑制药物联合使用可促进移植物免疫耐受,也存在潜在的损伤组织器官再生与功能恢复机制[48]。在第1例肝脏移植患者使用干细胞产品的临床试验中,为期1年的观察发现,最小化的使用免疫抑制剂药物不但更加安全,且移植物获得了更长期的存活,形成部分免疫耐受,提示器官移植后MSCs移植或MSCs配合免疫抑制剂治疗,可能是长期减少、减轻排异反应,减少免疫抑制药物用量和不良反应的有效手段,为提高器官移植的安全性和有效性提供了诱人的新技术应用前景。"
| [1] Baran DA.New directions in immunosuppression after heart transplantation.Nat Rev Cardiol. 2013;10(7):422-427. [2] Dominici M, Le Blanc K, Mueller I, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8(4):315-317. [3] Le Blanc K, Tammik C, Rosendahl K, et al. HLA expression and immunologic properties of differentiated and undifferentiated mesenchymal stem cells.ExpHematol. 2003;31(10):890-896. [4] Tse WT, Pendleton JD, Beyer WM, et al. Suppression of allogeneic T-cell proliferation by human marrow stromal cells: implications in transplantation.Transplantation. 2003;75(3): 389-397. [5] Jang IK, Yoon HH, Yang MS, et al. B7-H1 inhibits T cell proliferation through MHC class II in human mesenchymal stem cells.Transplant Proc. 2014;46(5):1638-1641. [6] Cornelissen AS, Maijenburg MW, Nolte MA, et al. Organ-specific migration of mesenchymal stromal cells: Who, when, where and why. Immunol Lett. 2015;168(2): 159-169. [7] Hall BM.CD4+CD25+ T Regulatory Cells in Transplantation Tolerance: 25 Years On.Transplantation. 2016;100(12):2533- 2547. [8] Kavanagh H, Mahon BP.Allogeneic mesenchymal stem cells prevent allergic airway inflammation by inducing murine regulatory T cells.Allergy. 2011;66(4):523-531. [9] Luz-Crawford P, Kurte M, Bravo-Alegría J, et al. Mesenchymal stem cells generate a CD4+CD25+Foxp3+ regulatory T cell population during the differentiation process of Th1 and Th17 cells.Stem Cell Res Ther. 2013;4(3):65. [10] 穆思捷.人羊膜间充质干细胞体外上调类风湿关节炎患者外周血Treg细胞比例[D].遵义:遵义医科大学,2015.[11] Kaundal U, Bagai U, Rakha A. et al. Immunomodulatory plasticity of mesenchymal stem cells: a potential key to successful solid organ transplantation.J Transl Med. 2018; 16(1):31. [12] Zhao X, Ding HF, Guo CS, et al. In vitro effects of mesenchymal stem cells on secreting function of T lymphocytes and CD4?CD25? T cells from patients with immune thrombocytopenia. ZhonghuaXue Ye XueZaZhi. 2013;34(12):1015-1019. [13] Cho KS, Kim YW, Kang MJ, et al. Immunomodulatory Effect of Mesenchymal Stem Cells on T Lymphocyte and Cytokine Expression in Nasal Polyps.Otolaryngol Head Neck Surg. 2014;150(6):1062-1070. [14] Tobin LM, Healy ME, English K, et al. Human mesenchymal stem cells suppress donor CD4(+) T cell proliferation and reduce pathology in a humanized mouse model of acute graft-versus-host disease.Clin ExpImmunol. 2013;172(2): 333-348. [15] Hsu WT, Lin CH, Chiang BL, et al. Prostaglandin E2 potentiates mesenchymal stem cell-induced IL-10+IFN-γ+ CD4+ regulatory T cells to control transplant arteriosclerosis.J Immunol. 2013;190(5):2372-2380. [16] Cagliani J, Grande D, Molmenti EP, et al. Immunomodulation by Mesenchymal Stromal Cells and Their Clinical Applications. J Stem Cell Regen Biol. 2017;3(2):1-26. [17] Franquesa M, Hoogduijn MJ, Bestard O, et al. Immunomodulatory effect of mesenchymal stem cells on B cells.Front Immunol. 2012;3:212. [18] Rosado MM, Bernardo ME, Scarsella M, et al. Inhibition of B-cell proliferation and antibody production by mesenchymal stromal cells is mediated by T cells.Stem Cells Dev. 2015; 24(1):93-103. [19] Tabera S, Pérez-Simón JA, Díez-Campelo M, et al. The effect of mesenchymal stem cells on the viability, proliferation and differentiation of B-lymphocytes.Haematologica. 2008;93(9): 1301-1309. [20] Asari S, Itakura S, Ferreri K, et al. Mesenchymal stem cells suppress B-cell terminal differentiation.ExpHematol. 2009; 37(5):604-615. [21] Healy ME, Bergin R, Mahon BP, et al. Mesenchymal stromal cells protect against caspase 3-mediated apoptosis of CD19(+) peripheral B cells through contact-dependent upregulation of VEGF.Stem Cells Dev. 2015; 24(20): 2391-2402. [22] Reinders ME, Hoogduijn MJ.NK Cells and MSCs: Possible Implications for MSC Therapy in Renal Transplantation.J Stem Cell Res Ther. 2014;4(2):1000166. [23] Spaggiari GM, Capobianco A, Becchetti S, et al. Mesenchymal stem cell-natural killer cell interactions: evidence that activated NK cells are capable of killing MSCs, whereas MSCs can inhibit IL-2-induced NK-cell proliferation. Blood. 2006;107(4):1484-1490. [24] Spaggiari GM, Capobianco A, Abdelrazik H, et al. Mesenchymal stem cells inhibit natural killer-cell proliferation, cytotoxicity, and cytokine production: role of indoleamine 2, 3-dioxygenase and prostaglandin E2.Blood. 2008;111(3): 1327-1333. [25] Sotiropoulou PA, Perez SA, Gritzapis AD, et al. Interactions between human mesenchymal stem cells and natural killer cells.Stem Cells. 2006;24(1):74-85. [26] Selmani Z, Naji A, Zidi I, et al. Human leukocyte antigen-G5 secretion by human mesenchymal stem cells is required to suppress T lymphocyte and natural killer function and to induce CD4+CD25highFOXP3+ regulatory T cells.Stem Cells. 2008;26(1):212-222. [27] Poggi A, Prevosto C, Massaro AM, et al. Interaction between human NK cells and bone marrow stromal cells induces NK cell triggering: role of NKp30 and NKG2D receptors.J Immunol. 2005;175(10):6352-6360. [28] Casado JG, Tarazona R, Sanchez-Margallo FM.NK and MSCs crosstalk: the sense of immunomodulation and their sensitivity.Stem Cell Rev. 2013;9(2):184-189. [29] Chatterjee D, Tufa DM, Baehre H, et al. Natural killer cells acquire CD73 expression upon exposure to mesenchymal stem cells.Blood. 2014;123(4):594-595. [30] Fu B, Tian Z, Wei H.Subsets of human natural killer cells and their regulatory effects.Immunology. 2014;141(4):483-489. [31] Hirata N, Yanagawa Y, Satoh M, et al. Dendritic cell-derived TNF-alpha is responsible for development of IL-10-producing CD4+ T cells.Cell Immunol. 2010;261(1):37-41.[32] van den Berk LC, Jansen BJ, Snowden S, et al. Cord blood mesenchymal stem cells suppress DC-T Cell proliferation via prostaglandin B2.Stem Cells Dev. 2014;23(14): 1582-1593. [33] Li FR, Wang XG, Deng CY, et al. Immune modulation of co-transplantation mesenchymal stem cells with islet on T and dendritic cells.Clin ExpImmunol. 2010;161(2):357-363. [34] Jiang XX, Zhang Y, Liu B, et al. Human mesenchymal stem cells inhibit differentiation and function of monocyte-derived dendritic cells.Blood. 2005;105(10):4120-4126. [35] Spaggiari GM, Abdelrazik H, Becchetti F, et al. MSCs inhibit monocyte-derived DC maturation and function by selectively interfering with the generation of immature DCs: central role of MSC-derived prostaglandin E2.Blood. 2009;113(26): 6576-6583. [36] Beyth S, Borovsky Z, Mevorach D, et al. Human mesenchymal stem cells alter antigen-presenting cell maturation and induce T-cell unresponsiveness.Blood. 2005; 105(5):2214-2219. [37] Németh K, Leelahavanichkul A, Yuen PS, et al. Bone marrow stromal cells attenuate sepsis via prostaglandin E(2)-dependent reprogramming of host macrophages to increase their interleukin-10 production.Nat Med. 2009; 15(1):42-49. [38] Choi H, Lee RH, Bazhanov N, et al. Anti-inflammatory protein TSG-6 secreted by activated MSCs attenuates zymosan-induced mouse peritonitis by decreasing TLR2/NF- κB signaling in resident macrophages.Blood. 2011; 118(2): 330-338. [39] François M, Romieu-Mourez R, Li M, et al. Human MSC suppression correlates with cytokine induction of indoleamine 2, 3-dioxygenase and bystander M2 macrophage differentiation. Mol Ther. 2012;20(1):187-195. [40] Bouffi C, Bony C, Courties G, et al. IL-6-dependent PGE2 secretion by mesenchymal stem cells inhibits local inflammation in experimental arthritis.PLoS One. 2010;5(12): e14247. [41] Liu XH, Kirschenbaum A, Yao S, et al. Interactive effect of interleukin-6 and prostaglandin E2 on osteoclastogenesis via the OPG/RANKL/RANK system.Ann N Y Acad Sci. 2006; 1068:225-233. [42] Gao S, Mao F, Zhang B, et al. Mouse bone marrow-derived mesenchymal stem cells induce macrophage M2 polarization through the nuclear factor-κB and signal transducer and activator of transcription 3 pathways.ExpBiol Med (Maywood). 2014;239(3):366-375. [43] Squillaro T, Peluso G, Galderisi U.Clinical Trials With Mesenchymal Stem Cells: An Update.Cell Transplant. 2016; 25(5):829-848. [44] Vanikar AV, Trivedi HL, Feroze A, et al. Effect of co-transplantation of mesenchymal stem cells and hematopoietic stem cells as compared to hematopoietic stem cell transplantation alone in renal transplantation to achieve donor hypo-responsiveness. Int Urol Nephrol. 2011; 43(1):225-232. [45] Tan J, Wu W, Xu X, et al. Induction therapy with autologous mesenchymal stem cells in living-related kidney transplants: a randomized controlled trial.JAMA. 2012;307(11):1169-1177. [46] Vanikar AV, Trivedi HL, Kumar A, et al. Co-infusion of donor adipose tissue-derived mesenchymal and hematopoietic stem cells helps safe minimization of immunosuppression in renal transplantation - single center experience.Ren Fail. 2014; 36(9):1376-1384. [47] Pan GH, Chen Z, Xu L, et al. Low-dose tacrolimus combined with donor-derived mesenchymal stem cells after renal transplantation: a prospective, non-randomized study. Oncotarget. 2016;7(11):12089-12101. [48] Obermajer N, Popp FC, Johnson CL, et al. Rationale and prospects of mesenchymal stem cell therapy for liver transplantation.CurrOpin Organ Transplant. 2014; 19(1):60-64. [49] Menard C, Pacelli L, Bassi G, et al. Clinical-grade mesenchymal stromal cells produced under various good manufacturing practice processes differ in their immunomodulatory properties: standardization of immune quality controls.Stem Cells Dev. 2013;22(12):1789-1801. [50] Haarer J, Johnson CL, Soeder Y, et al. Caveats of mesenchymal stem cell therapy in solid organ transplantation. Transpl Int. 2015;28(1):1-9. [51] Teo GS, Yang Z, Carman CV, et al. Intravital imaging of mesenchymal stem cell trafficking and association with platelets and neutrophils.Stem Cells. 2015;33(1):265-277. [52] Vandeputte C, Taymans JM, Casteels C, et al. Automated quantitative gait analysis in animal models of movement disorders.BMC Neurosci. 2010;11:92. [53] 徐璐,廖德怀,陈正,等.肾移植中经移植肾动脉输注骨髓间充质干细胞3例报告[J].中国组织工程研究,2010,14(53):10049-10052. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [6] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [7] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [8] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [9] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [10] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [11] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [12] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [13] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [14] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [15] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||